Abstract
Background:
Laparoscopic living-donor nephrectomy is the current epitome of living kidney donation surgery. We review our experience in living-donor nephrectomies over the last 19 years, transitioning from open surgery to hand-assisted laparoscopy to full laparoscopic techniques.
Methods:
We retrospectively identified all living-donor nephrectomies performed at our institution from 1976 to 2018. The donors were categorised according to surgical techniques: open (ODN), hand-assisted laparoscopy (HALDN) and full laparoscopy (LDN). We reviewed changes in donor demographics over the years. Surgical outcomes between groups were compared from 2000 to 2018. We also compared the outcomes of LDN between different time periods to evaluate our learning curve.
Results:
A total of 214 living-donor nephrectomies were performed between 2000 and 2018. The majority were left sided (93%) and had single renal artery anatomy (90%). There were 22 ODN, 20 HALDN and 163 LDN cases. The mean operating time was 84±43, 151±32 and 179±37 minutes for ODN, HALDN and LDN, respectively (p<0.001). There were no statistically significant differences in mean warm ischaemia times (p=0.921) and length of hospital stay (p=0.114) between groups. The overall 30-day surgical morbidity rate was 9.3%, with a major complications rate of 0.9%. The mean warm ischaemia time for LDN was significantly different (p<0.001) between time periods: 281±260, 184±94 and 140±42 seconds for the periods between 2005–2009, 2010–2014 and 2015–2018, respectively.
Conclusion:
This study confirms the safety of living-donor nephrectomies performed at our institution, a centre with a modest volume of kidney transplants.
Keywords
Introduction
Living-donor kidney transplantation (LDKT) is currently the best treatment option for end-stage kidney disease (ESKD). In Singapore, the outcomes of kidney transplantation have been excellent, with 5- and 10-year graft survival rates of 96.5% and 79.4%, respectively, for LDKT performed at our institution.1,2 With the chronic shortage of cadaveric organs, combined with good short- and long-term donor and recipient outcomes, LDKT is an attractive option for ESKD patients.3,4 Living kidney donation is generally safe; the surgical mortality has remained stable over the years at 3.1/10,000 donors, with no increased risk of death and only a small absolute risk increase of ESKD in the long term.5,6 The traditional open donor nephrectomy (ODN), performed through a long flank incision with rib resection, is associated with extended convalescence periods and long-term wound issues. 7 This has led to the adoption of minimal incision approaches, culminating in laparoscopic donor nephrectomy (LDN). In this study, we review our experience and evolving practice in living-donor nephrectomies over the past two decades.
Methods
In this Institutional Review Board–approved study (CIRB 2018/3172), we retrospectively reviewed the electronic medical records to retrieve the surgical outcomes of all living-donor nephrectomies performed at the Singapore General Hospital during the period from 2000 to 2018. The hard-copy surgical records for transplants performed in the period before 2000 were not accessible for this study. From the available transplant registry data, we also reviewed the demographic trends of the living kidney donors from 1976 to 2018.
The donors were grouped according to the surgical techniques: ODN, hand-assisted LDN (HALDN) and LDN. At our institution, living kidney donation surgeries were performed by a team of urologists with core interests in kidney transplantation. ODN was performed through the traditional flank incision without rib resection. Organ retrieval in HALDN was through the hand-port incision (midline or paramedian). All LDNs were performed transperitoneally, except for one done via the retroperitoneal approach. The kidney was extracted through a lower abdominal skin crease incision on the ipsilateral side for all laparoscopic cases.
Operative outcomes, which were warm ischaemia time (WIT), operating time (OT), length of hospital stay (LOS) and surgical complications, were compared between the groups. Operative blood loss and analgesia requirements were not reviewed due to the lack of documentation. WIT was defined as the duration from the start of renal artery clamping to cold organ perfusion with histidine–tryptophan–ketoglutarate solution on ice. Length of hospital stay was calculated from the date of transplant until hospital discharge. Surgical complications within 30 days of transplant operation were graded according to the Clavien–Dindo system. 8 Cases with missing data were excluded from the analysis. Within the LDN group, we also compared the WIT and OT between different time periods (2005–2009 vs. 2010–2014 vs. 2015–2018) to evaluate our experience over time.
Standard descriptive statistics were used to summarise the data. Continuous data comparisons between two groups were done using a two-tailed Student’s t-test, and between three groups using one-way analysis of variance. Categorical variables were compared using the chi-square test. We defined statistical significance as p<0.05. The statistical software used was MedCalc v18.2 (MedCalc Software, Ostend, Belgium).
Results
Donor demographics
A total of 470 living-donor nephrectomies were performed from 1976 to 2018, of which 214 were done between 2000 and 2018. Figure 1 shows the trends for the donor types and age of donation over the years. While the number of LDKT performed annually was stable over the decades, there was a shift in the donor demographics. In our early experience, all the donor–recipient pairs were biologically related. Biologically unrelated kidney transplantations were performed from the 1990s onwards, and the numbers have been rising ever since. In a similar trend, the mean age at kidney donation has increased in recent years.

Donor demographics across the decades. Timeline since the first living-donor transplantation performed at our institution in 1976, showing the trends in (a) donor types and (b) age at donation.
The donor and kidney characteristics in the period from 2000 to 2018 are summarised in Table 1. The mean age at donation was 44.4 years, with almost equal gender distribution. There was a trend for older donors in the LDN group compared to the ODN or HALDN groups, although this was not statistically significant. Most cases (62%) were biologically related. The kidneys were procured predominantly from the left side (93%), and most had single renal artery anatomy (90%). While significantly more left-sided nephrectomies were performed using the minimally invasive techniques compared to the open technique, there were no differences in the complexity of vascular anatomy.
Donor characteristics (2000–2018).

p-Value for comparisons between ODN, HALDN and LDN.
SD: standard deviation; LDN: laparoscopic donor nephrectomy; HALDN: hand-assisted LDN; ODN: open donor nephrectomy.
Surgical outcomes
There were 22 ODN, 29 HALDN and 163 LDN performed within the study period. Over the years, our practice evolved from exclusively ODN to HALDN and finally to LDN (Figure 2). The mean OT was longer with the more minimally invasive surgical (MIS) approach: 84 minutes for ODN, 151 minutes for HALDN and 179 minutes for LDN (p<0.001). There were no significant differences in WIT (188–211 seconds, p=0.921) and LOS (3.2–3.9 days, p=0.114) between the three groups. A summary of the surgical outcomes is shown in Table 2.
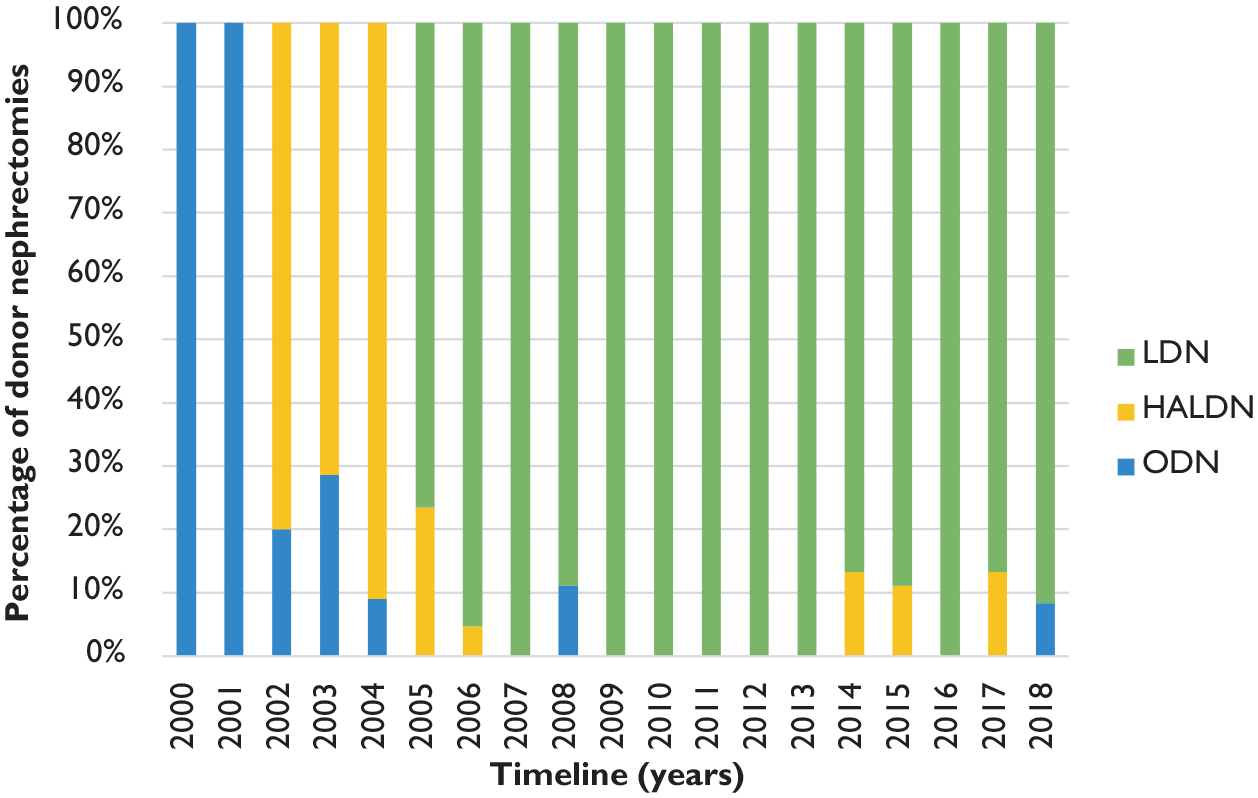
Evolving practice of donor nephrectomies of more than 19 years. Each bar represents the total number of cases for that year, and the methods of surgery expressed in percentages. LDN: laparoscopic donor nephrectomy; HALDN: hand-assisted LDN; ODN: open donor nephrectomy.
Surgical outcomes.

Excluding open conversion cases.
n.m.: not meaningful; SGF: slow graft function, defined as serum creatinine ⩾3 mg/dL at postoperative day 5; DGF – delayed graft function, defined as requiring dialysis in the first week after transplantation.
Surgical complications within 30 days
The overall perioperative morbidity rate for donor nephrectomies was 9.3% (20/214). The incidence of major (grade 3) complications was 0.9% (2/214), with the rest being minor (grade 1–2) events. Both grade 3 events occurred in the LDN group, prior to 2012, giving it a major morbidity rate of 1.2%. One was a case of pneumothorax requiring chest tube insertion, and the other was a readmission for symptomatic chylous ascites requiring percutaneous insertion of abdominal drain. There were no life-threatening complications or mortalities in our series.
Surgical timings of LDNs
Comparing between time periods, the mean OT in minutes for LDN was 173±38 for the 2005–2009 period, 178±33 for the 2010–2014 period and 192±38 for the 2015–2018 period. While the differences were statistically significant (p=0.047), they were not meaningful. The mean WIT was 281±260 seconds for the 2005–2009 period, 184±94 seconds for the 2010–2014 period and 140±42 seconds for the 2015–2018 period; these differences were statistically significant (p<0.001). The WIT also became more consistent with the passage of time, as reflected by the narrower spread of data (Figure 3). This suggests a learning curve to the technique of LDN. The mean WIT was longer for multiple renal arteries compared to a single renal artery (301±426 vs. 205±132 seconds, p=0.05).
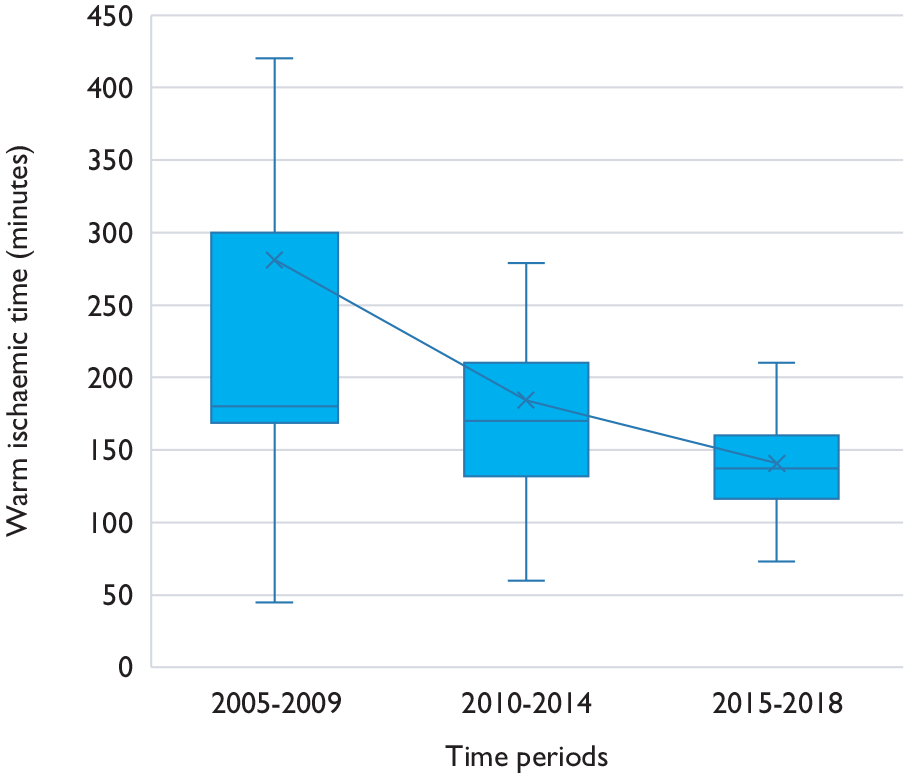
Trend in warm ischaemia time for laparoscopic donor nephrectomies. Timeline grouped into five-year time periods. Crosses represent mean values for the respective time periods.
Recipient outcomes
There were 24 (11.2%) recipients who had reduced graft function, of whom 11 (5.1%) had slow graft function and 13 (6.1%) had delayed graft function. One recipient from the HALDN group had graft nephrectomy for a non-perfused allograft which was not salvageable. Various factors could have influenced the recipient outcomes, but these were beyond the scope of this study.
Discussion
Since the first LDKT in Singapore was performed in 1976 at the Singapore General Hospital, we have observed a shift in donor demographics. Living kidney donors are now older and more frequently biologically unrelated to the recipients compared to the preceding decades. Despite a significant proportion of donors being >50 years old, our overall surgical morbidity rate of 9.1% was comparable to those reported in larger series. 9 Indeed, one study found no difference in complication rates in older donors compared to the younger controls (17% vs. 18%). 10 In our series, major complications were rare (0.9%) and all fully resolved without long-term sequelae. There were no mortalities.
Over the last 19 years, our practice of living kidney procurements has evolved from open surgery to minimal incision techniques. Hand-assisted laparoscopy (HAL) was a transitional step prior to the adoption of full laparoscopy. While others have touted the advantages of HAL techniques over full laparoscopy, especially the superior surgical manipulation and blunt dissection using a hand, 11 we have not experienced significant advantages over full laparoscopy. Furthermore, LDN confers better cosmesis, as our kidney extraction scar in the lower abdomen can be easily hidden by underwear. Comparing the three approaches, others have reported shorter OT and WIT for open followed by HAL and laparoscopic procurement techniques, and longer LOS for ODN.12,13 Our findings are similar in that OT and WIT were shortest (although the latter was not statistically significant due to missing data from the earlier time periods) in the ODN group, followed by HALDN and LDN. Unlike those studies, the LOS was not significantly different between our study groups; our mean LOS of 3.9 days was shorter than those (6–9.6 days) reported in other studies.12,13 One confounder would be that we were comparing the last two years of ODN, when the surgeons’ experience was at their ‘peak’ versus 14 years of LDN, which would include the early learning curve for this technique. Open nephrectomies performed before 2000 were not analysed due to the difficulty of accessing the hardcopy records, which is a major limitation to this study. Another explanation could be that we did not resect the 12th rib during open nephrectomy, resulting in less postoperative morbidity.
Comparing the outcomes of LDN between the different time periods, the mean OT was similar. This could be explained by the fact that many of the operative steps during LDN are the same as non-donor laparoscopic nephrectomies, which are familiar to urologists. The main differences are in the time-sensitive sequence of vascular control and organ extraction. To minimise the cold ischaemia time, our institution’s practice is to prepare the recipient concurrently. This may sometimes prolong the donor surgery to delay vascular clamping until the recipient is ready, making the OT less accurate in reflecting the actual surgical duration. The learning curve for LDN could be seen in the mean WIT, which was halved over time, reflecting our experience accrual as we refined our methods and became more proficient. This reduction in WIT was observed despite performing more complex cases with multiple renal arteries in recent years. Other authors have also reported on the learning curve for LDN. 14 Within the last decade, we started using vascular staplers to secure the donor renal vessels, rather than Hem-o-lok and metal clips. This change in practice could have contributed to shortening the WIT and narrowing its range.
The overwhelming majority of the kidneys were procured from the left side, consistent with the practice in most transplant centres worldwide in view of the longer left renal vein, and most had a single renal artery anatomy. Wedge excision and repair of the inferior vena, as well as gaining adequate renal artery length, in right donor nephrectomy can be challenging to perform laparoscopically. We prefer to do right donor nephrectomy using the HAL approach, reserving full laparoscopic techniques for selected cases (i.e. inherently long vessels). A small proportion of the donor kidneys in our study had multiple renal arteries, which have been shown by other investigators to be linked to longer operative times and higher donor complications. 9 While complex renal artery anatomy has been associated with poorer recipient outcomes, a Singapore study demonstrated that good results could be achieved. 15 In our experience, meticulous dissection of complex left renal vein anatomy, such as the retro-aortic and circum-aortic configurations, can still be done laparoscopically with little impact on WIT.
Many newer MIS techniques have been reported, but most of these are variations to LDN, such as robotic-assisted laparoscopy, retroperitoneal laparoscopy, single-site laparoscopy and natural orifice transluminal endoscopic surgery (transvaginal route of organ retrieval). A novel technique of ‘finger-assisted’ ODN by a team in the UK has reported smaller combined length of incisions, shorter OT, comparable WIT and complication rates, while costing less than LDN 16 The cost-effectiveness of these methods will need to be investigated before they are adopted. Generally, MIS techniques tend to have higher costs than open surgery.12,17 Until a newer approach has been proven to be better, safe and cost-effective, LDN and traditional ODN will remain our core surgical techniques of donor nephrectomy.
Conclusions
The best-in-class outcomes in a centre with modest volume of kidney transplants can be achieved by maintaining a high standard of surgical discipline and proficiency. This is accomplished by concentrating the caseloads to a dedicated team of transplant surgeons. This ensures that donor safety is paramount, as a healthy individual undergoes a major surgery entirely for the benefit of another.
Footnotes
Acknowledgements
We would like to thank the renal transplant coordinators at the Singapore General Hospital for their unrelenting support in completing this study.
Authors’ contributions
E.J.A. and V.H.L.G. researched the literature, conceived the study and gained ethical approval. Y.L.L., X.Y. and G.Y.H.N. were involved in data collection. C.W.L. was involved in data collection and access to renal transplant registry. E.J.A. analysed the data and wrote the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Availability of data and materials
The data sets generated and/or analysed for the current study are not available for review.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
SingHealth Central Institutional Review Board (2018/3172).
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
Informed consent was not sought for the present study because waiver of consent was obtained.
