Abstract
Background
The emergence of the Coronavirus Disease 2019 (COVID-19) Pandemic threatened living kidney donor transplantation services. Data on ABO-incompatible kidney transplants (ABOi KT) performed during the COVID-19 Pandemic is scarce.
Objectives
This study sought to compare characteristics and outcomes of ABOi KT performed before and during the COVID-19 Pandemic at an academic medical centre in Singapore.
Methods
A retrospective cohort study was performed on ABOi KT from 1st December 2009 to 31st May 2023. Data was compared between patients who received ABOi KT before the COVID-19 Pandemic (1st December 2009 to 10th March 2020 or pre-COVID-19 era) and during the COVID-19 Pandemic (11th March 2020 to 31st May 2023 or COVID-19 era).
Results
The study population consisted of 28 ABOi KT, 19 during the pre-COVID-19 era and nine during the COVID-19 era. There were no significant differences in baseline characteristics between the two groups and notably waiting time for transplantation was not significantly increased during the COVID-19 era. The median dose of Rituximab given for desensitization was low at 135 (range 0-500) mg and 100 (range 100-200) mg for both pre-COVID-19 and COVID-19 era patients. There were no significant differences in post-operative outcomes. COVID-19 infected 57.1% (
Conclusion
Despite greater complexity and immunosuppression in ABOi KT, ABOi KT can be performed safely during the Pandemic, provided patients are adequately vaccinated and healthcare system and process are in place to minimize the risks of COVID-19.
Introduction
Singapore ranks third with respect to the incidence of treated end-stage kidney disease (ESKD) (366 per million population) but has one of the lowest kidney transplants (KT) rates (11 per million population). 1 This is despite many initiatives to improve deceased kidney donation such as an opting out legislation for deceased kidney donation, explant biopsy assessment of expanded criteria deceased donors and donation after cardiac death programs. 2 As a result, living kidney donation have increased in recent years to meet the demands of an ESKD epidemic and in 2019 accounted for 63% of all kidney donations in Singapore (unpublished data). Apart from paired kidney donor exchanges, the availability of plasmapheresis and immunoadsorption technologies have allowed patients to receive KT from incompatible kidney donors. Singapore was the first country in South-East Asia to perform ABOi KT in 2008 which has since become an established option for KT.
However, the emergence of the Coronavirus Disease 2019 (COVID-19) Pandemic (2020-2023) threatened living kidney donor transplantation services in Singapore. There were concerns that KT could not be performed safely due to the perceived risks of donor-transmitted COVID-19, early fatal post-transplant infections and utilization of hospital resources that were required for COVID-19 care. However, Singapore adopted a risk-stratified approach to continuing different levels of living kidney donor transplantation according to alert levels (green, yellow, orange, and red) of the national Disease Outbreak Response System Condition (DORSCON). 3 Furthermore, transplant candidates were keen to proceed with transplantation when they were asked about their response to a transplant offer during the Pandemic. 4 During the Pandemic, Singapore was under orange alert status of the DORSCON system (signifying significant local transmission of COVID-19 with severe cases reported) but KT programs were permitted to continue living kidney donor transplantation as permitted by the availability of hospital resources.
There are few studies of the impact of the COVID-19 Pandemic on KT in South-East Asia (SEA), a region where COVID-19 mortality was higher than other regions in the world. 5 However, mortality rates in KT recipients from SEA during the Pandemic was low.6–8 COVID-19 related mortality was observed to be significantly lower in Asia than in Europe (2.7 vs 197 deaths per million population) and also the United States where 16% of deaths amongst active kidney transplant recipients in was attributed to COVID-19. This observation could be attributed to a younger population with lesser comorbidities in kidney transplantation patients in Asia.9,10 There are no studies on the impact of the Pandemic on high immunological risk KT like those using ABOi kidney donors from SEA. Given that our experience of performing kidney transplantation during the Pandemic was favorable and that ABOi KT follow similar COVID-19 related protocols like other transplants, we hypothesize that the outcomes of ABOi KT performed during the Pandemic were also good. As a result, the objectives of this paper are to compare the characteristics and clinical outcomes of ABOi kidney transplant performed at the Singapore General Hospital (SGH) before and during the Pandemic. 11
Methods
Study design
During the COVID-19 Pandemic, the KT program at SGH established a COVID-19 KT registry to prospectively collect data about KT recipients presenting with COVID-19. The registry was approved by the SingHealth Centralized Institutional Review Board (No. 2021/2823) to collect data as part of a clinical audit to monitor outcomes of clinical care protocols implemented for KT recipients and donors during the Pandemic. All transplantation procedures were performed under the framework of the Declaration of Helsinki and Declaration of Istanbul.
The study was a retrospective cohort study of patients undergoing ABOi KT at SGH between the period of 1st December 2009 to 31st May 2023. The inclusion criteria included patients who were aged 21 years or older and had completed at least 1 year of follow-up at SGH after KT. Patients who had received ABOi KT at other centres were excluded from the analysis.
Clinical data collection
Data was retrieved from the electronic medical records and stored on the encrypted transplant registry. Patients were followed-up from the date of transplant to the end of their first-year transplant anniversary. Demographic characteristics and details of desensitization were collected. Primary outcome included graft loss which was defined as return to dialysis and also patient death with a functioning graft. Secondary outcomes included length of hospital stay, post-operative infections, delayed graft function, graft function at 1 year and important complications such as major cardiovascular events (MACE) occurring within 1 year of follow-up. All episodes of rejection were confirmed on biopsy and defined by the Banff classification of renal allograft histopathology. Delayed graft function was defined as the need for dialysis during the first week of transplant surgery. MACE was defined as the occurrence of myocardial infarction, cardiac failure, or sudden cardiac death. COVID-19 infections were defined as those confirmed by a positive polymerase chain reaction test of a nasopharyngeal sample.
COVID-19 mitigation measures
The COVID-19 mitigation and management measures implemented to ensure safe living kidney donor transplantation was previously described. 8 Briefly, both recipients and living kidney donors were vaccinated with at least 2 COVID-19 vaccines and screened for COVID-19 before transplant surgery. In addition, staff were vaccinated and regularly swabbed for asymptomatic COVID-19. After discharge from hospital, patients were advised on safe distancing measures. KT recipients were also given a hotline to inform the transplant coordinators if they were infected. If infected, patients were triaged by the transplant team either for hospital admission with IV Remdesivir or home-based monitoring and treatment with telemedicine and oral Molnupiravir. 9 Monoclonal antibodies against COVID-19 such as Tixagevimab and Cilgavimab or Sotrovimab were also given early during the Pandemic but discontinued later with the emergence of Omicron subvariants where these antibodies were not effective against.
ABO-incompatible kidney transplantation protocol
All except 2 patients received Rituximab at 21 to 30 days before surgery but the doses were reduced over time from an initial 375 mg/kg/m2 to a current fixed low dose of 100-200 mg according to baseline CD20 cell count. Patients were usually admitted 2 weeks before their transplant surgery to undergo alternate day plasmapheresis or immunoadsorption. On admission, patients were started on tacrolimus at a starting dose of 0.075 mg/kg twice daily but in the last 2 years, the dose has been based on CYP3A5 genotyping test results. Perioperatively, patients were given Basiliximab or Thymoglobulin 1 mg/kg for 3 days, the latter given to those with positive crossmatch and/or donor-specific antibody results. IV methylprednisolone at a dose of 500 mg was also given for the first 2 days of transplant surgery and switched to oral prednisolone 20 mg with weekly taper till a nadir dose of 5 mg was reached and subsequently maintained. Mycophenolate Mofetil was initially given post-operatively at a dose of 1g twice daily but since 2023, the dose was routinely reduced to 750 mg twice daily at 1 month unless there was early post-transplant rejection. This protocol reduction was introduced to reduce the incidence of BKV infections. For antibody removal, alternate day double filtration plasmapheresis (DFPP) was used in most cases except those with very high anti-ABO titers (e.g., 3 256) where immunoadsorption (IA) columns were employed. In some cases, a hybrid approach alternating between DFPP and IA was used to minimize the costs of an IA alone approach. On the day before surgery, therapeutic plasma exchange (TPE) with full plasma replacement instead of DFPP was used to reduce the risk of hemorrhagic complications. Daily monitoring of anti-ABO titers was performed, and, in most patients, 4 to 5 antibody removal sessions were given before KT to achieve a target titer of 1:32 or less. During the first few cases of ABOi KT, post-operative TPE with full plasma replacement was routinely given in the first week of surgery but this was subsequently switched to an on-demand strategy where post-operative treatments were only given if the anti-ABO titer rose by 1 step (during the first week of surgery) or 2 steps (during the second week of surgery). The discontinuation of routine post-operative TPE was triggered by the observation that anti-ABO titer remained low and stable after transplant surgery. Post-plasmapheresis intravenous immunoglobulin (IVIG) was given for some cases but was subsequently discontinued.
Anti-microbial prophylaxis
All ABOi KT recipients were given
Statistical analysis
Analysis of the data was performed using IBM SPSS Statistic software (IBM Corp). Data for non-normally distributed data was presented as median with their respective ranges. Categorical data was evaluated using the Fisher-Exact test while continuous data underwent analysis through the Mann-Whitney U test. The tests were performed at a 95% confidence interval and significance threshold was set at
Results
Baseline characteristics
Baseline characteristics of study population.

Abbreviation: ABOi. ABO-incompatible; COVID-19, coronavirus disease 2019; CMV, cytomegalovirus; D, donor serostatus; DSA, donor-specific antibodies; R, recipient serostatus.
Desensitization
Desensitization and immunosuppression data on ABO-incompatible kidney transplants.
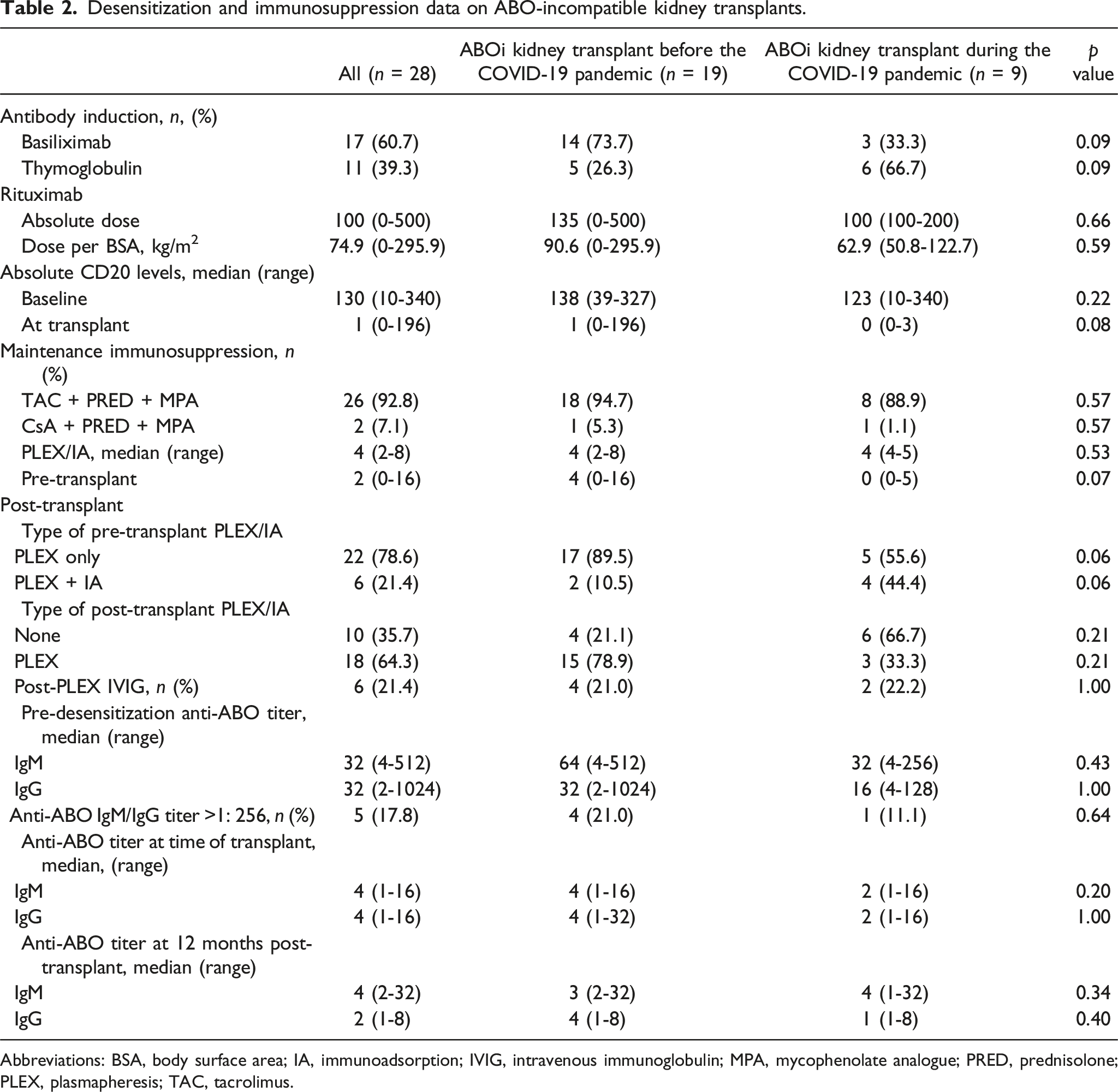
Abbreviations: BSA, body surface area; IA, immunoadsorption; IVIG, intravenous immunoglobulin; MPA, mycophenolate analogue; PRED, prednisolone; PLEX, plasmapheresis; TAC, tacrolimus.
Prior to the initiation of plasmapheresis, the median baseline titer of anti-ABO IgM and IgG for the entire study population was both 1:32 and patients with high anti-ABO titers of 1:256 or greater represented a small proportion (Table 2). The median number of pre-operative plasmapheresis and/or immunoadsorption used were 4 sessions with 78.6% being plasmapheresis (Table 2). Target titers less than 1:32 were achieved in all patients. Post-transplant plasmapheresis was given in 64.3% of patients but this was less frequent among patients transplanted during the COVID-19 era. During the Pandemic, patients only received post-transplant plasmapheresis only if their anti-ABO titers rose significantly or if they had DSAs that needed to be suppressed. IVIG was given in 21.4% of patients but by the time the Pandemic began, IVIG was reserved only for those with positive crossmatch associated with significant level of DSA. Median anti-ABO IgM and IgG titers remained stable thereafter (Table 2). No significant differences in anti-ABO titers were observed between eras.
Transplant-related outcomes
One-year clinical outcomes of ABO-incompatible kidney transplantation.

Abbreviations: BKV, BK virus; CMV, cytomegalovirus; eGFR, estimated glomerular filtration rate; HCV, hepatitis C virus; HEV, hepatitis E virus; MACE, major cardiovascular event, OT, operating theatre; UTI, urinary tract infection; URTI, upper respiratory tract infection.
Outcomes of COVID-19 infections among patients with ABO-incompatible kidney transplantation
Clinical outcomes of COVID-19 infections in ABO-incompatible kidney transplant recipients.

Abbreviations: COVID-19, coronavirus disease 2019; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
Discussion
The objective of this paper was to compare the characteristics and clinical outcomes of ABOi KT performed before and during the Pandemic, an area which has not been examined in the literature before. In SEA, ABOi KT is performed infrequently where Singapore was the first country to perform ABOi KT (2008) followed by Thailand (2010), Malaysia (2011), Indonesia (2018) and Vietnam (2022).12,13 However, only this study and another from Malaysia have reported their experience of ABOi KT in detail. In Malaysia, the ABMR rate and 1-year death-censored graft survival rate was 8.3% and 96% respectively while the ABMR rate and 1-year death censored graft survival rate in this study was 7.1% and 96.4% respectively. 13 Protocols from both countries were similar but with different permutations of antibody removal technologies, use of IVIG and doses of Rituximab. There was a higher rate of IVIG treatment in Malaysia at 76.9% compared to 21.4% in this study. 13 The lower use of IVIG in our centre arose from concerns that anti-ABO antibodies contained in IVIG formulations may trigger antibody mediated rejection or interfere with the interpretation of antibody titres.14,15
When the Pandemic started, there were concerns that performing kidney transplantation may be risky when profound immunosuppression increased the risk of complications from COVID-19. 16 However, our centre adopted a risk-stratified approach modulated by the COVID-19 incidence in the general population, the availability of healthcare resources and the ability to detect or treat COVID-19.3,8 Precautions against COVID-19 was adopted which included protocol SARS-CoV-2 screening for staff and inpatients, use of N95 masks, strict hand hygiene, social distancing and early access to vaccines. 17 In addition, all patients had access to a COVID-19 hotline where they could notify the transplant team of being tested positive for the infection and receive prompt treatment. 11 Finally, patient knowledge and positive attitudes towards COVID-19 precautions and vaccination were high among kidney transplant recipients, empowering patients to better care for themselves during the Pandemic. 18 These factors were effective as the frequency of severe COVID-19 infections among our KT recipients in another study was only 2.6%. 19 Notably, the mortality rate was even lower at 0.5%, an observation unique in SEA where higher mortality rates of 5.6% to 42.1% were observed in other countries from the region.7,20,21 When our program compared outcomes of living kidney donor transplantation performed before and during the COVID-19 Pandemic, there were no significant differences in perioperative outcomes except for a longer cold ischemia time attributed to the pre-operative COVID-19 screening tests required for both donor and recipient. 8 COVID-19 infections among patients transplanted at SGH during the Pandemic were also associated with good outcomes as there was no cases of pneumonia, admissions to intensive care unit and death. 8 Notably, 95% of patients transplanted during the Pandemic were vaccinated, an important factor in generating these favourable outcomes. 8 In this study, all except 1 patient undergoing ABOi KT during the Pandemic was vaccinated with at least three doses of COVID-19 vaccine. 88.9% of patients received at least three doses of the primary vaccination against SARS-CoV-2 pre-transplant while the remaining 11.1% of patients received 2 doses pre-transplant. This minimal number of doses of COVID-19 vaccine is important since it is associated with less severe infections and death.22,23
However, this earlier study from Singapore did not examine outcomes of ABOi KT recipients. Some would argue that such types of KT requiring Rituximab should not be performed due to observations that B-cell depletion is associated with poor response to vaccination, severe disease and mortality from COVID-19.24,25 COVID-19 infection may also be protracted in such patients, requiring reduction of immunosuppression which carries the risk of rejection. 26 As a result, some centres have deliberately modified their immunosuppression protocol to avoid over immunosuppression during the Pandemic.27,28 Nevertheless, real world observations suggest that ABOi KT could be safely performed without a significant increase in adverse post-transplant outcomes. In this study, the waiting time for transplantation was not significantly affected by the Pandemic whereas others have shown that in areas with a high case rate of COVID-19, the waiting time for transplantation was longer. 29 Notably, during the COVID-19 Pandemic, we did not modify selection or immunosuppression protocol for ABOi KT, which accounted for the lack of differences in the immunosuppression regimens used between the 2 periods. As a result, no patients suffered rejection or graft loss during the Pandemic while the rejection and graft survival rate of ABOi KT performed prior to the Pandemic was 10.5% and 94.7% respectively. These observations are similar to those from a larger multicentre study in India of 38 patients undergoing ABOi KT during the Pandemic. In this study also involving unmodified ABOi KT protocols, the reported biopsy proven rejection and graft loss rates was acceptable at 13.1% and 2.6% respectively. 30
Outcomes from COVID-19 infections among ABOi KT recipients were also good in this study. Regardless of the period they were transplanted, no patient required admission to intensive care unit or lost their graft or died. Factors that contributed to these excellent outcomes included prompt access to specialized transplant care and over 90% of patients had received at least three doses of COVID-19 vaccine. The low dose of Rituximab given to patients undergoing ABOi KT during the Pandemic could have been another favorable factor as it did not significantly suppress the humoral response to COVID-19 vaccine, with 62.5% of vaccinated patients with COVID-19 infection exhibiting detectable levels of SARS-CoV-2 antibodies at the time of COVID-19.
In conclusion, this study demonstrated that good post-transplant outcomes could still be achieved during a Pandemic for ABOi KT. As the world transits into an endemic COVID-19 world, the findings of this study are reassuring and do not deter the continuation of high immunological risk transplantation activities. However, such high immunological risk transplantation procedures are likely only to be doable safely in countries with adequate resources and well established regimental screening for COVID-19 infection. Programs and patients would need to maintain vigilance as well as adopt an active maintenance COVID-19 vaccination strategy to mitigate the risks of severe COVID-19. This is especially so when mortality from COVID-19 remains higher than the common influenza and other endemic respiratory viruses. 31
Footnotes
Acknowledgments
The authors would like to acknowledge the contributions of the infectious disease physicians and the COVID-19 isolation ward teams at the Singapore General Hospital who worked tirelessly and professionally during the COVID-19 Pandemic.
Author contributions
DLZL and TK collated and analyzed data to prepare the manuscript. HQY, LYT, CT, YJH, CL, HX, NCS, SLT, NK, EN, SO, TJQ and LCY contributed data and reviewed the manuscript for revisions. SDT performed the final review and endorse the paper for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
