Abstract
Introduction
This case of fenthion poisoning demonstrates the delayed, protracted and yet unpredictable nature of cholinergic effects which are unique to fenthion toxicity.
Case Presentation
A 36-year-old female presented to the emergency department with drowsiness and vomiting, 8 hours after an unknown pesticide ingestion. She had some cholinergic symptoms (miosis, incontinence, emesis) suspicious of organophosphate toxicity but was initially well with no bronchorrhea or bradycardia. During her admission, she developed increasing salivation and bronchorrhea resulting in respiratory failure requiring intubation 27 h’ post-ingestion. Possible aspiration pneumonitis was initially considered given the delayed nature of deterioration. However, blood toxicology results confirmed fenthion poisoning. Atropine and pralidoxime boluses were administered followed by continuous infusion. She was extubated after initial improvement and stabilization – only to require reintubation for recurring respiratory failure and seizures on Days 4 and 5, each time following apparent recovery. She was eventually weaned off mechanical ventilation only on Day 31.
Discussion and Conclusion
This case illustrates the importance of physician awareness to potentially innocuous presentations of fenthion poisoning as symptoms at the onset may appear benign and falsely reassuring. Serious toxic manifestations may be significantly delayed over a prolonged, waxing-and-waning clinical course because of the compound’s high lipid solubility and metabolism into potent secondary compounds.
Case report
A 36-year-old female with no other comorbidities was brought to the Emergency Department (ED) by the ambulance for drowsiness and vomiting. The patient called her friend threatening suicide and went to her friend’s house 1½ hours later where she was found to be drowsy and vomiting. She claimed to have ingested 500 mL of a mixture of pesticide and Dettol detergent. The patient complained of abdominal pain and headache upon her arrival at the ED. 8 hours post-ingestion.
On examination, the patient was drowsy (Glasgow Coma Score: E1V3M3) with some secretions and saliva around her mouth. Within an hour, she became more alert but still sleepy and able to answer questions and follow instructions. She was noted to have pinpoint pupils with vomitus over her clothes while her pants were also soiled with urine and faeces. She had no diaphoresis. Her lungs were clear and she was otherwise able to maintain oxygen saturations of 100% on room air. She was also afebrile and normotensive (blood pressure 98/54 mmHg) with normal heart rate (62 beats per minute). She underwent external decontamination to remove chemical contaminants on her body.
Initial lab investigations were unremarkable except for leukocytosis of 25.7 × 10 (9)/L. Her chest X-ray was normal. The patient was admitted to the general ward under the Gastroenterology service for inpatient endoscopic evaluation of her caustic ingestion. Her clinical course during her inpatient stay is summarized in Figure 1. Timeline of events. (a) Overall timeline of events and trend of cholinesterase (U/L over Days). (b) Initial events leading to diagnosis of fenthion poisoning.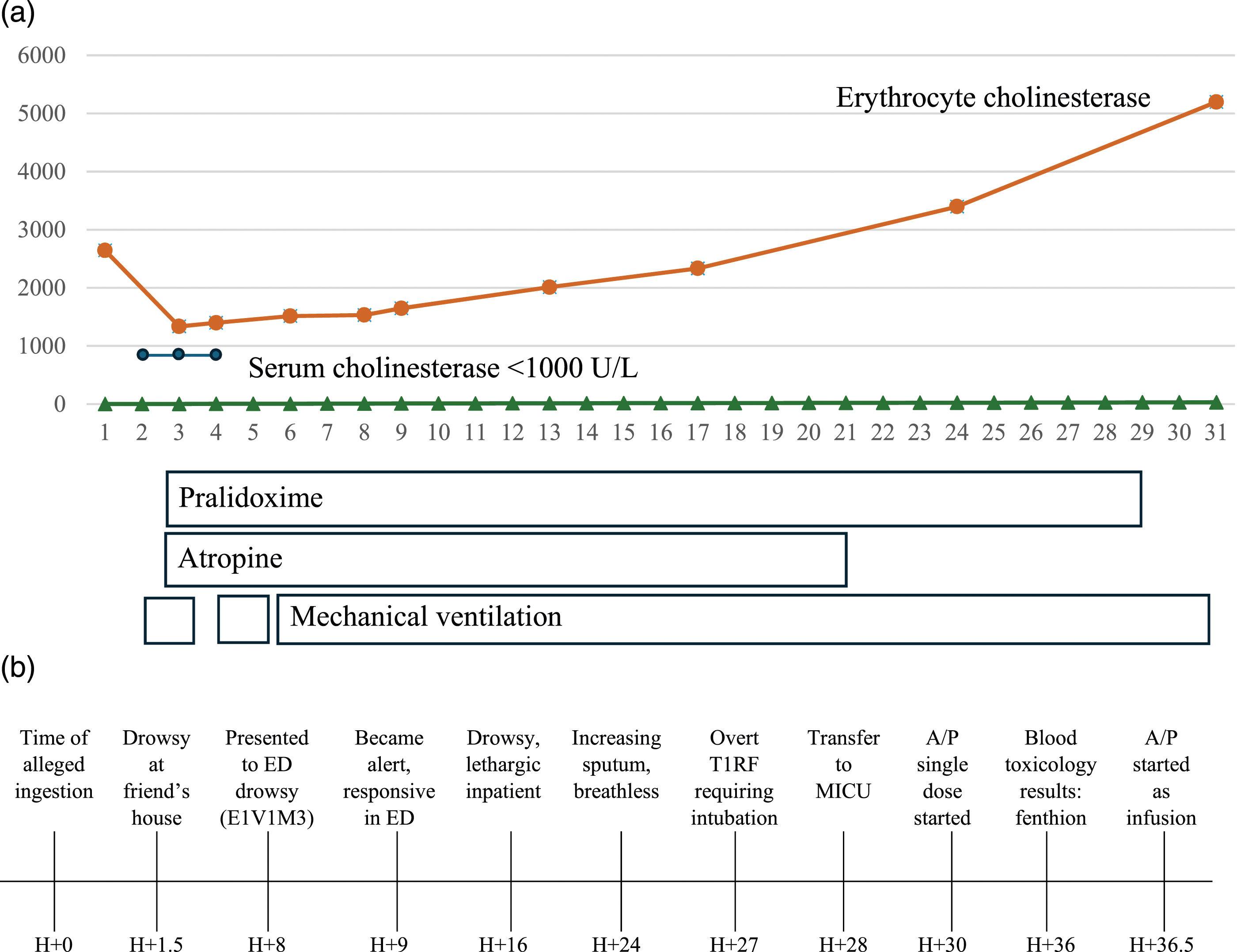
Despite an initial impression of organophosphate or carbamate poisoning, atropine and pralidoxime were held off because she was clinically not in cholinergic crisis and the agent was presumed to be a household agent with low potency and the amount ingested was likely to be small. Plasma and erythrocyte cholinesterase levels and blood toxicology specimen were dispatched.
She was found to have fluctuating drowsiness (GCS E2-4V4M5) and agitation 16 h later. She had also developed new-onset lower limb weakness and had soiled herself and the bed from faecal and urinary incontinence. Her lungs were still clear. In the early morning of the next day (24 h’ post-ingestion), she complained of breathlessness and had a lot more sputum and vomiting. She desaturated 3 hours later and required supplementary oxygen via a non-rebreather mask. She was intubated on Day 2 of admission for Type 1 Respiratory Failure, commenced on amoxicillin/clavulanic acid for anti-microbial prophylaxis and transferred to the Medical Intensive Care Unit (MICU) 28 h’ post-ingestion.
Given her initial recovery in the ED. and the extended time since ingestion (deterioration after at least 24 h), other differential diagnoses like chemical pneumonitis from aspiration of hydrocarbon or other co-ingestions like paraquat poisoning were deemed more likely until her erythrocyte cholinesterase levels drawn in the ED. returned as low (2644 U/L) 29 h’ post-ingestion. In view of her copious secretions requiring intubation and pinpoint pupils, she was started on atropine and was also given an intravenous loading dose of 1 g of pralidoxime. A single intravenous 0.4 mg bolus of atropine was adequate to achieve cessation of secretions and resulted in tachycardia (160 beats per minute) that persisted for 15 min. The decision was therefore made to hold off further boluses and maintenance infusion of atropine. Bronchoscopy revealed diffusely erythematous airway with thick secretions that were not typical of bronchorrhea but no obvious ulceration otherwise. Oesophago-gastroduodenoscopy was not performed in view of the bronchoscopy findings. Supplementary oxygen was tailed down to avoid hyperoxaemia which could worsen progression of lung failure in possible paraquat poisoning.
Blood toxicology report returned 36 h post-ingestion confirming fenthion poisoning while her urine toxicology did not detect presence of paraquat. This was a qualitative test with no laboratory reference range to trend the levels of fenthion in the blood. The patient was therefore, restarted on a pralidoxime infusion (500 mg/hour) and atropine infusion (0.02 mg/kilogram/hour; later increased to 0.08 mg/kilogram/hour) on Day 2 of admission. The patient self-extubated on Day 3 and was weaned to room air. Unfortunately, she developed Type 2 Respiratory Failure and seizures (manifesting as unresponsiveness, central cyanosis, small amplitude jerking of bilateral upper limbs lasting few minutes and left gaze deviation) on Day 4 and required re-intubation (2nd intubation).
By Day 5, there were no secretions in her endotracheal tube and her skin was dry. Her daily neurological examination revealed that her neck flexion strength was strong while her negative inspiratory force and slow vital capacity were also adequate. CT and MRI brain done in view of her seizures were essentially unremarkable. She was extubated on Day 5 as her mentation had returned to normal and her ventilator settings were minimal. However, following her extubation on Day 5, the patient again developed worsening Type 2 Respiratory Failure within 5 hours of her second extubation. Indeed, while preparing to re-intubate the patient, she suddenly turned unresponsive, started seizing again (manifesting as central cyanosis with flickering of the lips) and had to be re-intubated (3rd intubation).
Serum cholinesterase levels were <1000 U/L on Days 2-4 of admission. Her plasma and erythrocyte cholinesterase levels were trended to determine her suitability for extubation (Figure 1). Fenthion and its metabolites remained detected on a repeat blood toxicology sample sent on Day 5 but were subsequently undetected on a repeat sample sent on Day 17.
In view of prolonged intubation, a tracheostomy was performed on Day 12 of admission. She also underwent psychiatric evaluation in view of her near-fatal suicidal attempt. The patient was transferred from the Intensive Care Unit to an Intermediate Care Unit on Day 15 and subsequently to general ward on Day 18. Atropine infusion was weaned off gradually to monitor for reemergence of toxicity and finally stopped on Day 21 based on absence of cholinergic effects (secretions, diarrhea and bronchospasm) and after a repeat toxicology sample sent on Day 17 did not show any fenthion or its metabolites. Pralidoxime infusion was stopped only on Day 29, a week after atropine, to decrease the risk of a rebound following the prior multiple failed extubation attempts in view of the severe poisoning in this patient and the anticipated significant body load of fenthion. She was decannulated 2 days later (Day 31) and discharged on Day 36 once psychosocial issues had been addressed. She did not have any residual neurological complications upon discharge.
Discussion
Fenthion is a long-acting organophosphate which is used in agriculture as a flea-killer. Fenthion is extremely potent and a small spot applied to the back of an animal can exterminate fleas for months. 1 Reports of poisoning in humans is uncommon and often associated with only mild or minimal effects at onset which can often be falsely reassuring. The patient typically deteriorates and develops cholinergic symptoms late – symptoms peak only 30-96 h post-exposure – and may progress to severe, life-threatening respiratory compromise from cholinergic crises. 1 Moreover, clinical progression is non-linear and effects from fenthion toxicity may wax-and-wane over a course of anywhere between five to 48 days, rendering careful extended observation essential. This waxing-and-waning phenomenon is yet again noted in our case.
The prolonged toxicity of fenthion is due to its characteristics. Its high lipid solubility facilitates distribution in adipose stores and subsequent delayed redistribution into the systemic circulation. Moreover, fenthion is metabolized into secondary compounds that are even more potent cholinesterase inhibitors than the parent compound. 1 These protracted effects have serious clinical implications as they result in uncertainty over how long pralidoxime should be continued for optimal benefit, especially since overzealous oxime administration can tip the patient into cholinergic syndrome which mimics ongoing toxicity.
It was initially believed that pralidoxime is theoretically ineffective after the first 24–48 h from exposure due to the irreversible phenomenon of aging. However, recent studies have shown that delayed oxime administration may still confer benefit 2 if the metabolites are toxic or if the offending agent is highly lipid-soluble and prolonged absorption and subsequent redistribution from adipose depot are ongoing – such as in the case of fenthion, fenitrothion or clorfenthion. 3
Senanayake and Karalliedde (1987) reported three cases of fenthion poisoning and a fourth case of mixed fenthion and methamidophos poisoning. In this case series, pralidoxime was dosed at 1 g every 12 h for 24–48 h while atropine was continued. All patients developed “intermediate syndrome” which first manifested as respiratory insufficiency while two demised from respiratory failure. Interestingly, transient dystonic limb movements were also observed early in the course of fenthion poisoning. While there was no definite pattern to how neurologic manifestations develop, cranial palsies were the first to abate, followed by improvement in respiratory function and then proximal limb muscle strength, while neck flexion was last to return. However, this patient had preserved neck flexion strength even prior to her second deterioration on Day 5 which necessitated reintubation. Resolution of intermediate syndrome could take up to 18 days or even longer if confounded by delayed polyneuropathy as well. 4
Borowitz reported a two-year-old boy who swallowed flea-killer containing fenthion and had respiratory arrest 50 h after. 1 Pralidoxime was initially held off (due to concerns of reported ineffectiveness when given more than 48 h post-exposure) but was subsequently administered 18 h later. The child was extubated after 14 days but had to be reintubated 24 h later for another 3 days due to progressive hypoxemia and hypercapnia. Even then the patient still had increased oral and pulmonary secretions and bradycardia and required atropine. Pralidoxime was continued for 22 days in total, when erythrocyte and plasma cholinesterase levels recovered but were still only 80% of normal 1
A 1977 case series by Wadia et al described 32 cases of fenthion poisoning, of which 13 had respiratory paralysis and 10 died. 5 The authors highlighted that fenthion poisoning lacks marked muscarinic effects because none had overt pulmonary edema and only four had bradycardia – which can be falsely reassuring for clinicians taught to look out for the “Killer B’s” of bronchorrhea and bradycardia. 26 cases (81.3%) suffered paralysis with a mean time from ingestion to paralysis of 24 h (range: 4–72 h); among whom 68% had suppressed cholinesterase levels below 15% of normal mean value. This suppression from fenthion lasted much longer than with other agents – in two patients, the levels were virtually zero for 16 and 32 days respectively. In an editorial letter in 1988, 6 Wadia and Amin further reiterated that pralidoxime therefore needs to be given for a much longer duration, in some cases up to 6 days, but never as long as 22 days (as Borowitz had) and wondered about the clinical utility of pralidoxime in the later period.
Given the wide interindividual variations in cholinesterase levels, using cholinesterase levels to guide pralidoxime therapy might also be inaccurate and thus cannot be used as the sole guide to determine antidote treatment duration. Nonetheless, using clinical findings to assess recovery from fenthion poisoning appears grossly unreliable as well and deciding on what endpoint to use to determine safety for extubation and the optimal duration of oxime therapy remains unclear and a challenge.
Another possible method was to use the train-of-four, which was not used for this patient. Train-of-four repetitive stimulation refers to the use of a peripheral nerve stimulator to assess neuromuscular activity during neuromuscular blockade, typically under conditions of anesthesia. Although train-of-four may identify cases of severe poisoning, this technique has not been adequately validated in the clinical context of organophosphate poisoning. 7 Of note, Buckley et al. (2024) found that total block and rate-dependent block at 30 Hertz stimulation could achieve better discrimination of severe organophosphate poisoning compared to standard “train-of-four” monitoring at slower frequencies as the latter would be normal in most patients. 8
Yet another possible strategy was discussed by Tsatsakis et al (2002), 9 who presented two cases of fenthion poisoning. The authors added different concentrations of pralidoxime to the patient’s serum to assess the effects on cholinesterase reactivation in vitro and found substantial increase in activity from four mcg/ml (the assumed therapeutic concentration based on pharmacokinetic data) up to 10 mcg/ml, whereas 16 and 32 mcg/ml pralidoxime did not confer additional benefit. However, the authors did not translate these in vitro findings into clinical practice by adjusting the therapeutic dosage of pralidoxime to achieve target serum levels.
Conclusion
Fenthion poisoning can be tricky: the initial clinical presentation often appears benign at the onset; serious toxicity manifests late and over an extended period of days to weeks; while the waxing-and-waning clinical course may also mimic apparent recovery and fool the clinician into prematurely weaning the patient off atropine/pralidoxime treatment and mechanical ventilatory support. Using cholinesterase levels can help guide critical treatment decisions.
Footnotes
Author Contributions
K.R. and M.N. drafted the initial manuscript. Both contributed equally to the writing of this manuscript. Y.J.Y. provided critical interpretation of the blood toxicology results involved and case analysis. E.M.J.T., D.A. B.K.K.T. and R.P. provided critical review of the manuscript and were instrumental in providing guidance as subject matter content experts.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
