Abstract
An 81-year-old man presented to the Emergency Department with shortness of breath, generalised weakness, numbness, giddiness, nausea and vomiting after consuming an inadequately prepared Traditional Chinese Medicine preparation that contained herbal aconitum (Chuanwu and Caowu). His electrocardiogram (ECG) and rhythm strips showed multiple runs of non-sustained ventricular tachycardia monomorphic ventricular tachycardia and slowed polymorphic ventricular tachycardia. He was treated with intravenous (IV) amiodarone, magnesium and lignocaine, and was started on IV noradrenaline after developing haemodynamic compromise. There was no digoxin detected in the blood and urine. At 12 h, aconitine was not detected in the blood (cut off at <1 ng/mL) but aconitine and hypaconitine was detected in the urine qualitatively. He underwent a coronary angiogram at 12 h post-admission which showed minor coronary artery disease. A formal echocardiogram showed left ventricular ejection fraction 50–55% with no regional wall motion abnormalities of the left ventricle. He made an uneventful recovery and reverted to normal sinus rhythm at 29 h of admission. He was discharged well on Day 4 of admission with a diagnosis of polymorphic ventricular tachycardia secondary to Aconitum poisoning.
Keywords
Introduction
Aconitum is a common herb used in TCM 1 for its analgesic and anti-inflammatory properties. It is used for conditions such as rheumatism, 2 joint pains, 3 abdominal pains and gynaecological disorders such as menstrual irregularities and dysmenorrhea. Despite the common use, they have been multiple Aconitine toxicity cases reported in China, Hong Kong, Taiwan and India. 4 This is a case of polymorphic ventricular tachycardia from aconitine poisoning after ingestion. It details and discusses the presentation, treatment and progress of the patient and how body fluid testing can be performed to confirm the diagnosis.
Case Presentation
An 81-year-old man presented to the ED as an urgent ambulance case with shortness of breath, generalised weakness, numbness, giddiness, nausea and vomiting. He denied experiencing any chest pain. He had significant cardiovascular risk factors of diabetes mellitus, hypertension, hyperlipidaemia and chronic kidney disease.
His vital signs on arrival in the ED were a temperature of 36.2, heart rate of 125 beats per min, blood pressure of 103/62 mmHg, respiratory rate of 18 and pulse oxygen saturation of 97% on supplementary oxygen of 2 L/min. His capillary blood glucose level was 11.6 mmol/L.
His pre-hospital 12-lead Electrocardiogram (ECG) showed multiple polymorphic pre-mature ventricular complexes (PVC) with non-sustained ventricular tachycardia (NSVT) – Figure 1. Multiple polymorphic pre-mature ventricular complexes (PVC) with non-sustained ventricular tachycardia (NSVT).
On examination, patient was alert but restless, complaining that he felt extremely uncomfortable. His pupils were 4 mm in size and equally reactive to light. He was not diaphoretic and an examination of his cardiac, respiratory, neurological and gastrointestinal systems was unremarkable.
Three days prior, the patient had complaints of headache and left eye pain. He had visited a general practitioner and had a normal neurological assessment with normal eye pressures. Due to persistent pain, the patient subsequently went to a TCM practitioner and he was prescribed three packets of medical herbs to help improve his blood circulation. Patient had prepared one packet of the medical herbs himself and drank it 2 hours before his presentation at the ED. He developed symptoms 30–60 min after taking the concoction, and vomited the mixture once at home and once in the ED.
Patient subsequently developed a stable monomorphic ventricular tachycardia (Figure 2) in the ED. An intravenous (IV) Amiodarone infusion of 300 mg over 30 min was commenced. Monomorphic ventricular tachycardia.
The ventricular tachycardia terminated into a sinus rhythm with multifocal PVC 5 min into the infusion, which was continued. The patient subsequently turned hypotensive 15 min into the infusion and became drowsy. The amiodarone infusion was stopped. As there was no shockable rhythm, a peripheral IV adrenaline infusion of 1mcg/ml was started and patient stabilised. A repeat 12-lead ECG showed a slowed polymorphic ventricular tachycardia – Figure 3. Slowed polymorphic ventricular tachycardia.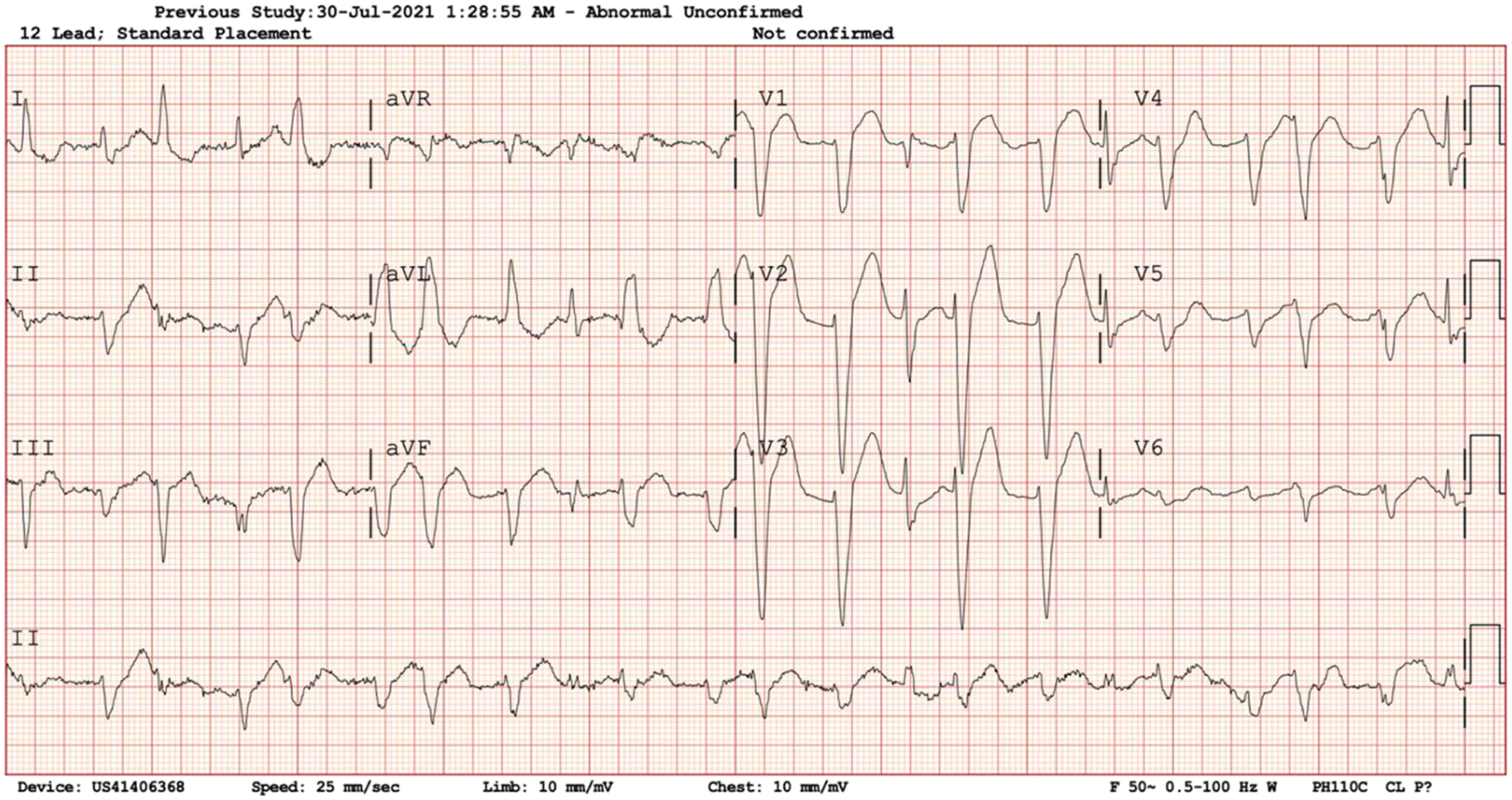
Initial investigation results.

A bedside ultrasound showed a visually estimated left ventricular ejection fraction (LVEF) of 60%, a normal sized right ventricle with no pericardial effusion. Significant inferior vena cava respiratory variation was noted.
The patient was started on IV crystalloids and magnesium replacement. The cardiology service was consulted for consideration of cardiac catheterisation to rule out an ischemic cause for the ventricular dysrhythmias. A left radial intra-arterial line was placed for haemodynamic monitoring. A left femoral vein central venous catheter was inserted and a noradrenaline drip was started in lieu of adrenaline for vasopressor support.
When the patient stabilised, IV amiodarone was continued and a total of 300 mg was given. The patient developed another run of slowed polymorphic ventricular tachycardia during treatment – Figure 4. Slowed polymorphic ventricular tachycardia.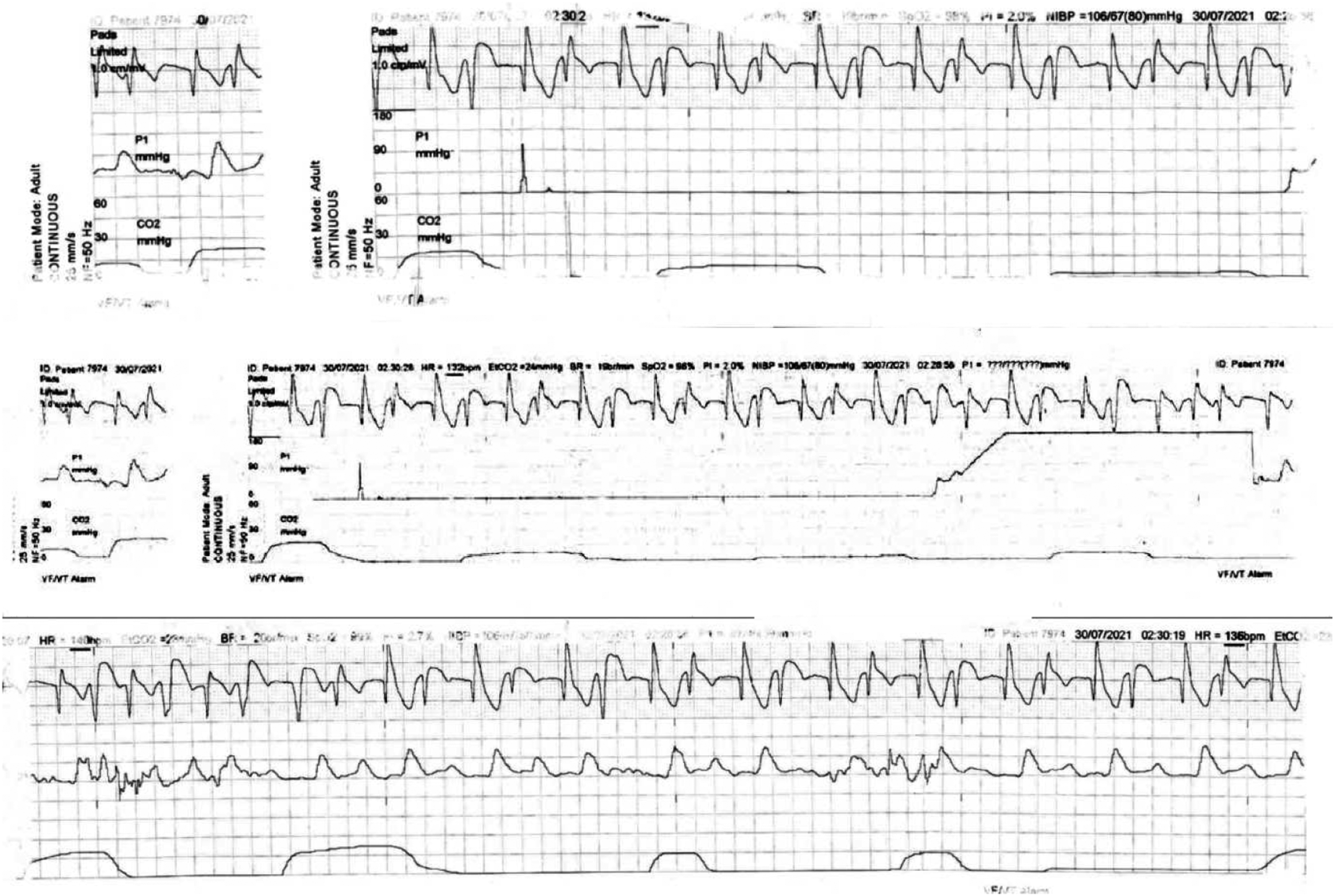
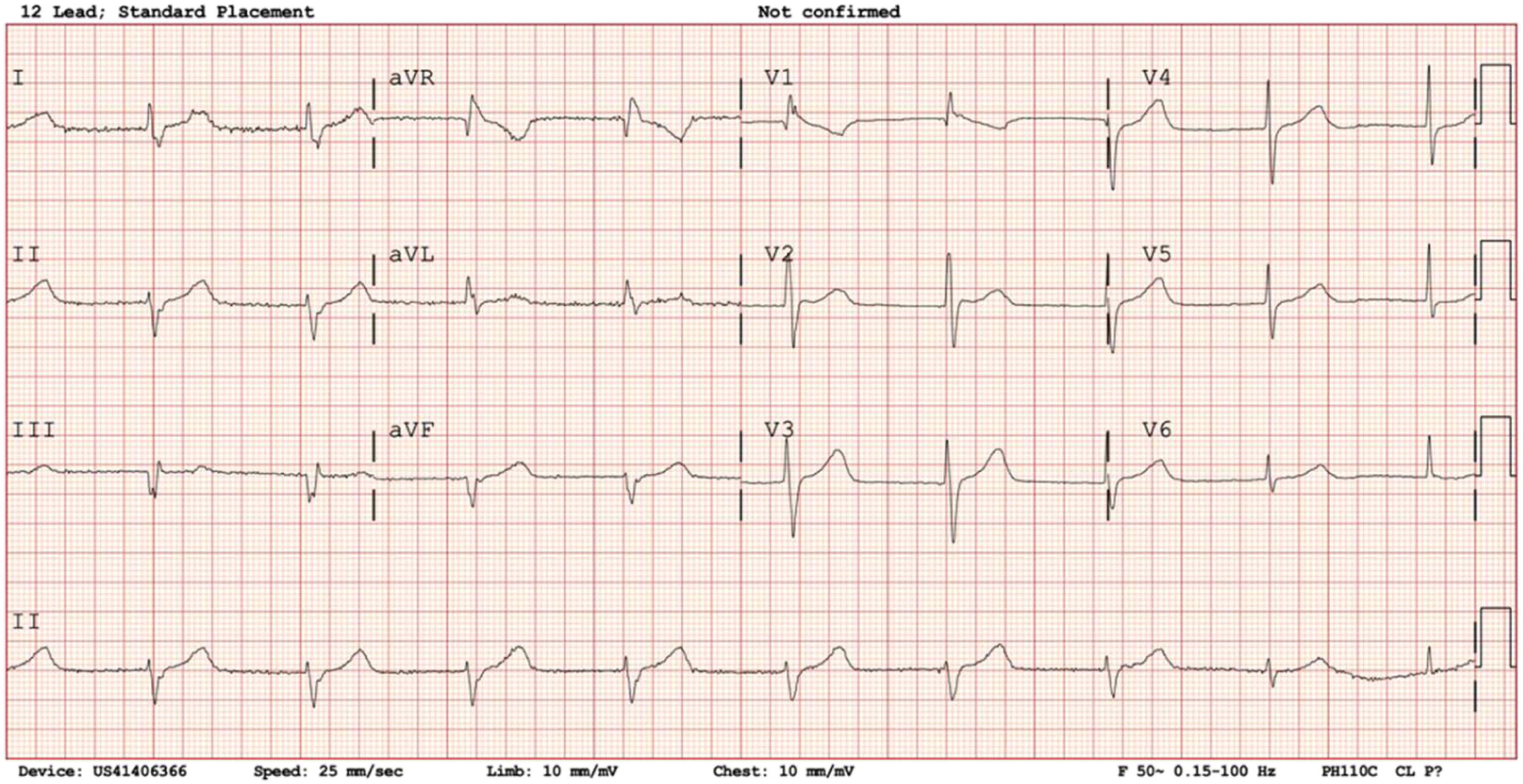
A further 100 mg bolus of IV lignocaine was given. ECGs over the next few hours showed alternating left bundle branch (Figure 5) and right bundle branch blocks (Figure 6) on a background accelerated idioventricular rhythm. There was no further VT run noted. Accelerated idioventricular rhythm, LBBB (11 h post-ingestion). Junctional rhythm, incomplete RBBB (15 h post-ingestion).
The patient underwent a coronary angiogram at 12 h post-admission which showed minor coronary artery disease. A formal echocardiographic exam showed LVEF 50–55% with no regional wall motion abnormalities of the left ventricle. A mild posterior mitral valve prolapse with moderate mitral regurgitation was noted. The patient made an uneventful recovery with Noradrenaline discontinued at 16 h of admission and amiodarone (total dose 1.2 g) stopped at 24 h of admission. He reverted to normal sinus rhythm at 29 h of admission (Figure 7). Normal sinus rhythm (31 h post-ingestion).
The patient was discharged well on Day 4 of admission with a diagnosis of polymorphic ventricular tachycardia secondary to Aconitum poisoning. Blood and urine was sent at 12 h post-ingestion for toxicological analysis. There was no digoxin detected in the blood and urine. Aconitine was not detected in the blood (cut off at <1 ng/mL). Aconitine and hypaconitine was detected in the urine as a qualitative result.
A TCM prescription (Figure 8) obtained from the patient’s family post-admission showed the presence of aconitum-containing compounds, namely, Chuanwu/Radix Aconiti (川乌) and Caowu/Radix Aconiti Kusnezoffii (草乌), at 3g each. In particular, Banxia (半夏), a compound known to be incompatible with Aconitum, was also noted in the prescription. Picture of prescription formula and medical herbs obtained from the patient.
Further history from the patient revealed that he was given instructions to brew the medical herbs and reduce the volume by boiling from 3 bowls to 0.8 bowls. He was not given specific instructions on how long he should brew the medical herbs for. He had brewed his first packet of medical herbs for 30 min, reducing it from a 3-bowl to a 1.5-bowl volume, thinking that drinking more would be better for him. He left it to cool for 1 h prior to consumption and developed symptoms 30–60 min later.
Discussion
Aconitine Toxicity And Use Of Chuanwu And Caowu In TCM
Chuanwu and Caowu are two examples of Chinese herbal drugs that are derived from Aconitum carmichaelii. 5 One of the active alkaloid compounds in Aconitum, diester diterpene alkaloids (DDAs), also known as Aconitine, affects the cardiac system, the central nervous system (CNS) and the gastrointestinal system (GIS). 4 It acts on the voltage-sensitive sodium channels in the cell membranes of excitable tissues of the myocardium, nerves and muscles. It works by inhibiting the inactivation of the voltage-dependent sodium channel, by substitution into a binding site. This results in a disruption of the neurotransmitter release and hence the neural signal transmitting pathway. 6 The nerves in the cardiac system are affected by the prolonged excitation phase of the sodium channel, resulting in a delay of the repolarization phase of the action potential and therefore initiating pre-mature excitation. The CNS and GIS are affected in a similar way as well.
In cases of Aconitine toxicity, patients present with a combination of neurological, gastrointestinal and cardiovascular features. Typical signs and symptoms include weakness, numbness, abdominal cramps, nausea, vomiting and cardiac dysrhythmia with haemodynamic instability. In our case report, our patient presented with very similar neurologic and gastrointestinal symptoms. He also had clear evidence of increased cardiac automaticity. 4
In TCM, there are 18 herbs incompatible with each other described in a verse named ‘18 incompatible medicaments’. In this verse, the herbs Beimu (贝母), Gualou (瓜蒌), Banxia (半夏), Bailian (白蔹) and Baiji (白芨) were regarded to be incompatible with Aconitum. In our patient’s prescription, we noted the presence of Banxia which is incompatible with Chuanwu and Caowu. 7
The toxicity of Aconitum can be greatly reduced by simple boiling. This detoxification process involves the hydrolysis of the DDAs into less toxic monoesters. 8 People from the rural villages of Shaanxi Province in China used to cook and consume Aconitum in winter to keep themselves warm. They would cut the root into slices and boil them in a soup till dry, add water and re-boil them for hours to several days. 9 Similar preparation methods have been observed among the Indians as well. 10 In more recent times, autoclaving is the newest technique to detoxify the Aconitum. It involves steaming the crude herbs at 127oC under a pressure of 0.15 mPa for 60–90 min. This method has been shown to reduce most of the toxic compounds while preserving the efficacy of the medication. 11
In our case report, our patient had not boiled the medical herbs from a 3-bowl to 0.8 bowl volume as instructed, as he had thought that drinking more of the herbs would be beneficial for him. The patient was also not given clear instructions with regard to the preparation volumes and the duration of boiling.
Clinical Management Of Tachyarrhythmias Induced By Aconitine Toxicity
As there is no specific antidote for Aconitine toxicity, supportive management is the mainstay. Our patient had initially presented with polymorphic PVCs which subsequently degenerated into a malignant ventricular tachyarrhythmia with haemodynamic compromise. Amiodarone and Flecainide have been reported as the preferred first-line drugs in treating ventricular tachyarrhythmia induced by Aconitine toxicity.4,12,13 In a recent case series of 63 human aconitine toxicities, flecainide or amiodarone appear to be more associated with a return to sinus rhythm than lidocaine and/or cardioversion, although it is not established whether the administration of treatment caused reversion to normal sinus rhythm. 14 Magnesium sulphate has also been reported to be used to treat cardiac arrhythmia in Aconitine toxicity.14,15
As we had very little prior information, our initial approach to the patient’s dysrhythmia was based on Advanced Cardiac Life Support principles. Our patient had more than one dysrhythmia on presentation. ECGs and telemetry strips captured multiple runs of non-sustained ventricular tachycardia (NSVT), monomorphic ventricular tachycardia (VT) and slowed polymorphic ventricular tachycardia. We gave IV Magnesium, targeting >1 mmol/L as one of our rhythm control strategies. When our patient turned hypotensive without a shockable rhythm during his IV Amiodarone infusion, peripheral Adrenaline was administered as a vasopressor and eventually replaced with Noradrenaline. We noted that there was no apparent detrimental arrhythmogenic effect on his cardiac rhythm when these two agents were used. The use of inotropic therapy in haemodynamically unstable patients due to refractory cardiac dysrhythmia has been supported in the literature. 4 Early use of cardiopulmonary bypass to maintain systemic perfusion and oxygenation has also been recommended. 4
Identification of Toxicants in Aconitine Intoxication
In animal studies, aconitine absorption is noted to be very fast (approximately 30–58 min), with rapid elimination and a very short half-life of approximately 80 min as it has a low protein binding (24–32%) . 16 Aconitine level in blood appears to be short-lived as observed in previous studies.4,17 Aconitine was not detected in a 12-h blood sample (cut off at <1 ng/mL) from our patient. Aconitine and hypaconitine was detected in his urine as a qualitative result 12 h post-ingestion. Urine aconitine has been shown to persist for days and can be useful as a qualitative test to confirm the diagnosis. 17
Clinically, our patient converted to sinus rhythm by Day 2 of admission and was discharged on Day 4 of admission without any permanent sequelae.
Conclusions
Aconitum herbs are commonly used in Traditional Chinese Medicine. The toxicity of Aconitum can be greatly reduced by simple boiling. Adequate preparation instructions should be given to patient when Aconitum is being prescribed by the practitioner. Amiodarone has anecdotally been the anti-arrhythmic agent of choice to treat the cardiac dysrhythmia associated with Aconitum poisoning. We noted that there was no apparent detrimental arrhythmogenic effect on our patient’s cardiac rhythm when we added Adrenaline and subsequently Noradrenaline for blood pressure support. The presence of Aconitine in blood appears to be short-lived and urine aconitine has been shown to persist for days as seen in our patient.
Footnotes
Author Contributions
All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
Changi General Hospital does not require ethical approval for reporting individual case report because informed consent has been obtained from patient.
Informed Consent
Written informed consent was obtained from the patient for his anonymised information to be published in this article.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
