Abstract
Background:
Post-mastectomy pain syndrome (PMPS) has been reported to occur in 25–60% of patients following surgeries for breast cancer, the highest occurring cancer in women worldwide. There has been much research interest due to this high prevalence. However, there is still a lack of incorporation of PMPS prevention strategies in standard perioperative plans, and our understanding of this condition is still incomplete.
Objectives:
This narrative review discusses recent literature on modifiable risk factors, current approaches to prevention and treatment and potential directions for future treatment and research.
Methods:
A PubMed search with the relevant keywords was done for articles published in the last 10 years.
Results:
The incidence of PMPS can be reduced by early recognition and management of modifiable risk factors as well as the perioperative use of analgesics and regional nerve blocks. These also have a significant role in the management of established PMPS together with surgical interventions and physical therapy.
Conclusions:
PMPS is still poorly defined and hence underdiagnosed and undertreated at this point. Perioperative peripheral nerve blocks have a very promising role as preventive analgesia to reduce the risk of developing PMPS, but large-scale randomised controlled studies will need to be done to evaluate their comparative efficacy. There is a need to prioritise PMPS prevention as a standard inclusion into the perioperative plans of mastectomy patients.
Keywords
Introduction
Breast cancer is the most commonly occurring malignancy in women, with an incidence of more than two million new cases in 2016. It represents about 25% of cancers in females worldwide, outnumbering all other malignancies. 1 According to data from 59 countries in the CONCORD-2 study, the 5-year survival rates of patients diagnosed with breast cancer during 2005–2009 were ⩾85% in developed countries. 2
Despite the high survival rates, between 25% and 60% of women develop chronic pain following breast cancer procedures – a condition termed ‘post-mastectomy pain syndrome’ (PMPS) – which may in turn affect quality of life. The International Association for Study of Pain (IASP) describes PMPS as chronic pain (of more than 3 months in duration) in the anterior thorax, axilla and/or upper arm and is hypothesised to result from damage to major peripheral nerves during surgery. 3
However, different studies utilise different definitions for PMPS, which has resulted in the wide range of estimates of its occurrence. Studies considering only neuropathic symptoms report a lower incidence (23.9%) 4 compared to the ones that include other potential mechanisms such as lymphedema and musculoskeletal pain (42–47%).5,6 The term ‘persistent post-mastectomy pain’ (PPMP) has been used to encompass the latter broader definition and includes persistent pain after mastectomy, lumpectomy, lymph node dissection and reconstruction, as well as chemotherapy and radiation.7–9
As with many chronic pain conditions, PMPS has a negative impact on quality of recovery and patient satisfaction and can be severe, causing diminished quality of life, including poor sleep, long-term disability and interference with activities of daily living.10,11
Despite widespread recognition of PMPS, it is often untreated or undertreated. 12 Some possible reasons suggested for inadequate management of PMPS are the lack of quality information about optimal treatment12–14 and incomplete understanding of the mechanisms and risk factors for chronic pain development and prognosis. It is therefore timely to review the recent literature on modifiable risk factors for PMPS, current approaches to treatment and potential directions for future treatment and research.
Postulated mechanisms of PMPS
For many patients undergoing breast cancer treatment, there are multiple causes of pain such as being due to tumour growth, surgical nerve damage or adjuvant radiotherapy. Under such conditions, peripheral nociceptor stimulation is intense and prolonged while down-regulatory mechanisms are inhibited. Wind-up response results from progressive increase in the magnitude of C-fibre-evoked responses of dorsal horn neurons produced by repetitive activation of C-fibres. In addition, central sensitisation occurs via increased excitability and responsiveness, enlarged receptive field areas and physiological transformation in the central nervous system pain conduction pathways (e.g. dorsal horn neurons, spinal cord and brain).
Reducing the risk of developing PMPS
Many known risk factors for PMPS are not readily modifiable, including age, race, advanced stage at diagnosis and requirement for extensive surgery and adjuvant chemotherapy and radiotherapy.5,15–17
The recent research focus has been upon the identification of modifiable perioperative risk factors that increase the risk of transition from acute to chronic pain. These include preoperative factors such as addressing pre-existing psychosocial issues, prevention of thoracic wall nerve injury as well as reducing acute postoperative pain.10,18–20
Modifiable risk factors
Psychological aetiologies and intervention
Cancer patients report high rates of depression (10–25%), anxiety (10–30%) 21 and post-traumatic stress disorders (35%). 22 Anxiety and depression have been known to reduce pain thresholds and to produce anatomic changes that accentuate pain. 23
Multiple longitudinal observational studies and systematic reviews have shown that preoperative anxiety and depression are significantly associated with the subsequent development of moderate to severe PPMP.9,16,19,24,25 Management of psychosocial issues is multidisciplinary and involves psychiatrists, psychologists, counsellors, medical social workers and community support.
Prevention of thoracic wall nerve injury
One of the most commonly recognised causes of PMPS is neuropathic pain from nerve damage in the axilla and/or the chest wall during surgery.4,26 Current evidence demonstrates there is a significantly lower incidence of PMPS in patients who undergo sentinel lymph node biopsy compared to more extensive axillary lymph node dissection (ALND), whereby the intercostobrachial nerve (ICBN) is postulated to have higher incidence of injury.27–30 However, the mechanism by which ICBN damage causes neuropathic symptoms is poorly understood, and research findings have been conflicting. A recent prospective cohort trial found no difference in pain scores 1 week after surgery whether the ICBN was preserved, partially preserved or sectioned during ALND. 31 The outcomes were also similar 4 and 9 months after surgery. 32 ICBN preservation was even found to be a risk factor for continued pain at 1 year by one study. 15
Reduction of acute perioperative pain
Management of perioperative pain is significant clinically, as it is a readily modifiable risk factor. Multiple studies have shown that moderate to severe acute postoperative pain is a strong risk factor for PMPS (odds ratio 1.34–2.8).11,33 Although a recent meta-analysis of 30 observational studies showed similar results, there are no data from randomised controlled trials (RCTs). 34 Preventive analgesia is a perioperative intervention and encompasses pre-emptive analgesia (before surgical incision) and intraoperative and postoperative pain management. For breast surgery, multimodal preventive pain management strategies include the administration of local or regional (e.g. wound infiltration, pectoral nerve or paravertebral block (PVB) blocks) 15 anaesthetic creams or systemic non-opioid (e.g. ibuprofen, ketorolac and gabapentin) analgesics before and after the onset of noxious stimuli (e.g. surgical incision).
Current approaches to the prevention and treatment
The recently published PROSPECT 35 (procedure specific postoperative pain management) guideline for oncological breast surgery gave grades of recommendation according to overall levels of evidence for the various interventions in postoperative pain (Table 1).
Relationship between quality of study, levels of evidence (LoE) and grades of recommendation (GoR). 35 .
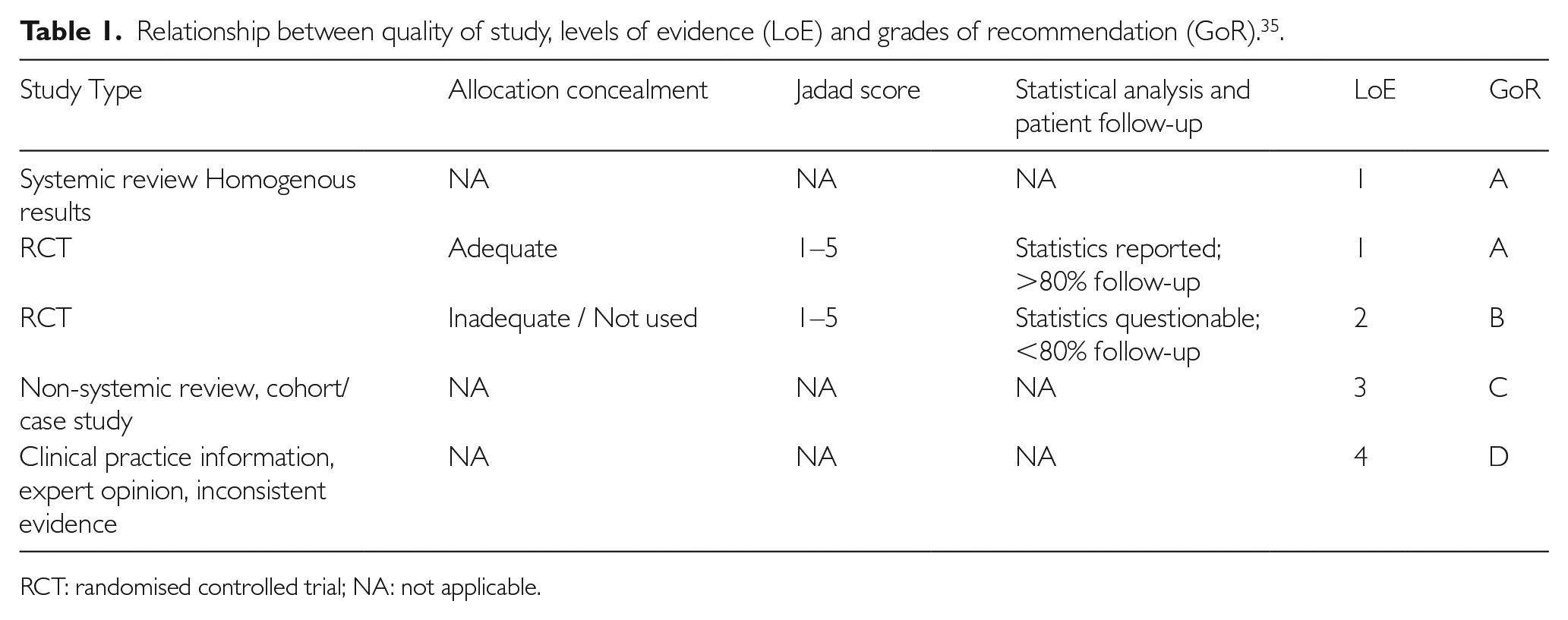
RCT: randomised controlled trial; NA: not applicable.
Perioperative systemic analgesics
The role of perioperative systemic analgesics in the treatment and prevention of PMPS are summarised in Table 2.
Pharmacotherapy agents used in management of PMPS.
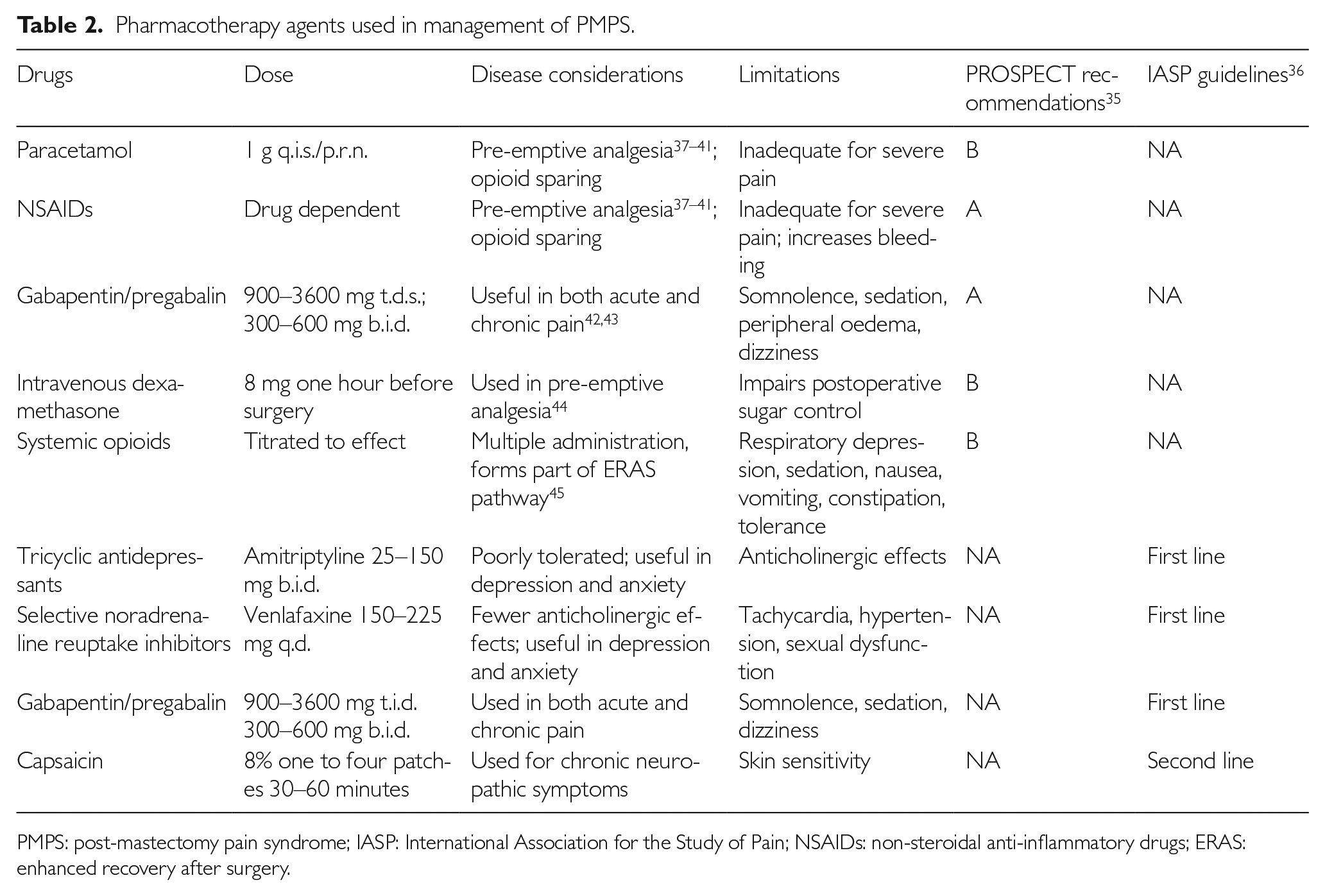
PMPS: post-mastectomy pain syndrome; IASP: International Association for the Study of Pain; NSAIDs: non-steroidal anti-inflammatory drugs; ERAS: enhanced recovery after surgery.
Regional nerve blocks and new drugs
Regional nerve blocks and new drugs are summarised in Table 3.
Regional nerve blocks used in prevention of PMPS.

PVB: paravertebral block; LA: local anaesthetic; SPB: serratus plane block.
Thoracic PVB
Local anaesthetic (LA) in the paravertebral space spreads over multiple levels to block thoracic spinal nerves as they exit the intervertebral foramina, extending laterally into the intercostal space and medially into the epidural space. This results in ipsilateral blockade of somatic and sympathetic nerves of the thoracic region. 46
The use of thoracic PVB (TPVB) in breast surgery reduces opioid use and postoperative pain and is highly substantiated in the literature.47–53 TPVB is effective as a perioperative analgesic and can even provide surgical anaesthesia. 54 The use of paravertebral catheters has not been reliably demonstrated to be superior to single-injection techniques at single or multiple levels. Moreover, single-injection TPVB requires a shorter time to perform and is less labour intensive compared to the multiple-injection technique or paravertebral catheters. 35
It should be noted that TPVB cannot provide analgesia to the axilla (i.e. T1 nerve distribution) reliably, 55 and supplemental LA infiltration to the area may be required.
PECS blocks
The PECS block and modified PECS block (also known as the PECS II block) are ultrasound-guided blocks targeting the inter-fascial plane between the pectoralis major muscle and the pectoralis minor muscle. 56 The PECS II block has an additional step of anesthetising the deeper space between the pectoralis minor muscle and serratus anterior muscle (SAM). 57 It has been reported to reduce opioid use in the first 12 hours, and patients had shorter post-anaesthesia care unit stays and overall hospitalisation time. 58
A further modification of the PECS II block proposes that the injection be performed deep instead of superficial to the SAM, thereby improving inter-fascial spread and sparing the long thoracic nerve. This will allow earlier assessment of nerve function in view of the risk of neuronal damage during surgical dissection. Recently published meta-analyses comparing the PECS II block with TPVB found there were no differences in pain scores or opioid consumption. PECS II blocks were not inferior to TPVB in reducing pain intensity and morphine consumption for the first 24 hours after surgery, and both were superior to systemic analgesia alone.59–61
In contrast to TPVB, the PECS II block is peripheral in nature and has less risk of sympathetic blockade and major bleeding. However, there is a lack of high-quality evidence and RCTs comparing the various technical approaches and their efficacy.
Serratus plane block
The serratus plane block (SPB) is performed more distal and lateral to the PECS II block. LA is injected in the two potential fascial planes superficial and deep to the serratus anterior muscle on the chest wall. This approach enables closer proximity to the intercostal nerves and results in anaesthesia of the hemithorax, although ICBN branches are also postulated to be involved.62–65
A recent meta-analysis found that SPBs lowered pain scores and opioid requirements in the postoperative period. There were no differences in pain scores and opioid requirements between patients who received SPB and TPVB. However, only five studies were included, and there was significant heterogeneity and deficiencies in blinding. 61
Pecto-intercostal fascia block
This ultrasound-guided block is performed at the medial aspect of the breast, 2–3 cm lateral to the sternal border at the level of the fourth rib. LA is deposited between the pectoralis major and external intercostal muscles. 66
Transversus thoracis muscle plane block
This is performed in the parasternal location. The LA is deposited in a deeper inter-fascial plane between the internal intercostal and transversus thoracis muscle. The anatomic disadvantages of this approach include proximity to the pleura and internal thoracic artery. 67 However, there have been no major complications reported. The extent of LA spread and subsequent analgesia following pecto-intercostal fascia block (PIFB) and transversus thoracis muscle plane (TTP) block require further study.
Combinations of peripheral blocks
In contrast to the thoracic paravertebral block, none of the inter-fascial approaches (PECS, serratus plane) block the supraclavicular nerves that contribute to the sensory innervation of the superior breast. Only the PIFB and TTP block anesthetise the anterior cutaneous branches of the intercostal nerves that supply the medial aspect of the breast. Thus, combinations of blocks are necessary to provide complete analgesia for many surgical procedures. 68
Liposomal bupivacaine
Liposomal bupivacaine is a newer drug used in regional blocks and infusions with a longer duration of action, ranging from 72 to 96 hours compared to bupivacaine which lasts between 8 and 12 hours. Preliminary evidence demonstrates efficacy, and it shows promise in use for longer reconstructive breast surgeries. Further studies comparing cost and outcomes between these drugs will need to be conducted. 69
LA wound infiltration
LA wound infiltration provides limited postoperative analgesia. A meta-analysis including eight RCTs comparing LA wound infiltration in oncological breast surgery found there was a reduction in pain scores observed in four trials and a reduction in rescue opioid consumption in two trials. However, the effect was limited to six hours and did not last more than 24 hours. 70
LA wound infiltration has a role in addition to regional analgesia techniques such as PECS and PVB, which may not provide appropriate analgesia to the axilla in the T1 nerve distribution.
EMLA
The preoperative application of EMLA (eutectic mixture of LA; AstraZeneca) over the axilla, supraclavicular and sternal area was studied in 46 patients undergoing breast cancer surgery, and its use delayed time to first analgesia requirement, even though there was no difference in pain scores and analgesic requirements in the first 24 hours. The incidence and intensity of chronic pain 3 months post operation was significantly lower. However, the study was small, and larger-scale studies will need to be performed to confirm these findings. 71
Management of patients with established PMPS
Systemic pharmacotherapy
Due to the heterogeneous clinical presentation of PMPS, effective pharmacotherapy will need to include a thorough patient assessment in order to identify the various factors associated with pain and dysfunction. It is often used in conjunction with non-pharmacological treatments. The choice of pharmacotherapy agents is usually dependent on contributing factors; for example, the use of antidepressants can afford relief for depression or anxiety on top of neuropathic pain.72–74
Antidepressants
Tricyclic antidepressants such as amitriptyline 25–150 mg q.d. or in two divided doses have been shown to be effective in managing PMPS as well as general neuropathic pain in RCTs. 75 However, their use is associated with significant side effects and lower tolerability. Venlafaxine, a serotonin norepinephrine reuptake inhibitor 150–225 mg q.d., has been shown to decrease average and maximum pain intensity significantly and had better compliance with fewer anticholinergic effects. 76
Antiepileptics
Gabapentin and pregabalin modify neurotransmitter release through their unique effects on voltage-gated calcium ion channels at the postsynaptic dorsal horns 77 and reduce excitability of neuronal cells, thus producing antiepileptic, analgesic and sedative effects. Gabapentinoids are the first-line treatment of neuropathic pain in patients with chronic post-herpetic neuralgia and peripheral neuropathy, although evidence for other types of neuropathic pain is limited.78,79 Levetiracetam does not appear to benefit PMPS symptoms at doses up to 3 g/day. 80
Topical capsaicin
Topical application of capsaicin (0.075% cream applied four times a day or a high-dose 8% skin patch for 30–60 minutes) was shown to offer potential symptomatic pain reduction for post-mastectomy patients after 6 weeks of treatment. However, significant site reaction, including a burning sensation, is often reported by patients.81,82
Regional anaesthesia techniques
Intercostobrachial fascial block
The intercostobrachial nerve arises from the lateral cutaneous branch of the second intercostal nerve and is vulnerable to damage during surgery for breast cancer, resulting in painful paraesthesias and chronic neuropathic pain. A pilot study conducted by Wijayasinghe et al. found that the second intercostal space, where the intercostobrachial nerve reliably passes through, could be consistently identified under ultrasound. This could then be a reliable approach for ultrasound-guided blocks of the intercostobrachial nerve. 83 A small study of six patients with chronic pain relating to treatment for breast cancer who underwent intercostobrachial nerve block found that all reported significantly reduced pain scores post procedure, with one patient reporting pain relief even at 3 months post injection, far beyond the expected duration of relief from LA. 84
Stellate ganglion block
The stellate ganglion block is mainly used for the treatment of painful disorders of the head, neck, upper limb and upper chest. It works by blocking the cervical sympathetic trunks, vertebral ganglia, lower cervical sympathetic ganglia and the upper thoracic sympathetic ganglia. A limited number of studies have shown that it is effective in the treatment of PMPS when given as a series of injections over time and reduces the need for analgesic medications as well as improves the range of motion of the shoulder.85,86 Other future considerations for studies should include prolonging duration of pain relief from the block, including options such as radiofrequency ablation and chemoablation. 86
Surgical interventions
Traumatic neuromas are a source of chronic neuropathic pain in the scar tissue post surgery. Nerves that are commonly involved include the intercostal, thoracodorsal, medial and lateral pectoral and long thoracic nerves. 87 Risk factors for their development include more extensive surgeries such as ALND and concurrent radiation therapy. 88 Neuromas contain nerves that have lower excitatory potential and cause allodynia and hyperalgesia. 89
Traumatic neuromas can be diagnosed with a positive Tinel’s sign over the intercostal nerves along the lateral chest wall whereby neuropathic symptoms are reproduced in the distribution of the patient’s pain. Further confirmation can be made with relief of symptoms after infiltration of the neuroma with 1% lignocaine. Surgery involves dissection along previous incisions and the identification of previously transected intercostal nerves along the scar tissues. The neuroma is then resected under traction, allowing the cut proximal end to retract deep into the intercostal muscles, and the overlying fascia is then closed over it in order to reduce recurrence.90–92
Autologous fat grafting
Fat injection has been used in patients with PMPS and severe scar retraction post mastectomy. Adipose tissue is first harvested from abdominal subcutaneous fat by liposuction under sedation and analgesia. It is then processed via Coleman’s technique and injected into painful scar areas at the dermohypodermal junction. Pain scores have been found to be lower post procedure compared to a control group (
Physical therapy
Chronic chest wall pain that persists beyond simple incisional pain can be the result of scarring of the incised tissues, leading to adherence to the underlying chest wall and decreased mobility. The use of physical therapy in post-mastectomy patients is summarised in Table 4.
Use of physical therapy in PMPS.

Patients who underwent physical therapy had improved upper-extremity function and quality of life 3 to 6 months after completing therapy compared to those who were only given information about exercise. 94 Patients who initiated physical therapy early had better 2-year outcomes for upper-extremity range of motion. 95 However, there are no universal guidelines on when to commence therapy, and studies have shown that there is increased wound drainage and seroma formation when therapy is started in the first week after surgery. 96
Physical therapy is under-utilised, with one study suggesting up to 90% of patients would benefit from some form of physical therapy, while only 30% received it. 97
Management of pain secondary to lymphoedema requires multimodal physical and occupational therapy, with the primary aim of reducing interstitial tissue swelling. This can be achieved with compression bandages or sleeve-and-glove compression garments, manual lymphatic drainage and exercises. 7 Skin hygiene and care reduces skin infection. Moisturisers, low-pH soaps and pH-neutral creams also prevent skin dryness and micro-tears. 98
Psychological therapies
Psychotherapy, including cognitive behavioural techniques, provide moderate benefits for depression, anxiety and coping skills in breast cancer patients. 99
Psychosocial interventions typically include group therapy, coping skills training and cognitive behavioural therapy. Relaxation skills also have been shown to have positive effects. A meta-analysis of 26 studies involving 1786 patients found a significant reduction in reported pain severity (Hedges’
A systematic review of RCTs using hypnotherapy found two which reported significantly greater reductions in pain relative to a comparison group. 100
Complementary and alternative medicine
Chronic cancer pain management requires a multipronged approach, and there is a significant role for complementary and alternative medicine in the management of pain symptoms and treatment side effects in cancer patients. Studies have found that a large proportion (25–60%) of breast cancer patients have used at least one form of complementary and alternative medicine to treat cancer pain and the side effects of chemotherapy/radiation to improve quality of life.101–103
Acupuncture has been found in many studies to be effective in managing some of the symptoms associated with breast cancer surgery, including pain, numbness, limited range of motion and lymphoedema. 104 It is also reported to have minimal complications or side effects. 105
Conclusions
The disease burden of PMPS is significant and yet underdiagnosed and under-treated at this point. However, this is set to change, with emerging data from current research in this condition, as evidenced by the number of recently published studies.
There is an urgent need for greater understanding of the mechanisms by which PMPS occurs and standardisation of its definition. Future directions of research include the use of predictive models to investigate the patient-specific risk of developing PMPS as well as a targeted risk-reduction strategy.
Regional anaesthetic techniques for preventive analgesia, and physical and complementary therapies have been recommended for the prevention and treatment of PMPS in various studies. A few recently concluded meta-analyses have demonstrated that many of the promising interventions used in the management of PMPS will require larger high-quality RCTs to be done before recommendations can be made as to their use in patients with PMPS.
There is a need to incorporate personalised biopsychosocial and multidisciplinary approaches in the management of PMPS individualised to each patient’s specific condition and experience. These would offer a multifaceted plan to patients at risk of or with PMPS. The incorporation of PMPS prevention strategies into standardised care plans such as enhanced recovery after surgery (ERAS) protocols will have the potential to help reduce disease occurrence and burden as well as greatly increase awareness and early treatment of this condition.
Footnotes
Acknowledgements
None.
Authors’ contributions
P.T. and D.C. researched the literature and conceived the study. P.T. wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Availability of data and materials
Data sharing is not applicable to this article, as no data sets were generated or analysed during the current study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Not applicable.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
Not applicable.
