Abstract
Cardiac amyloidosis is a severely underdiagnosed cause of heart failure with preserved ejection fraction. We report a case of highly probable transthyretin (ATTR) cardiac amyloidosis (ATTR-CA) diagnosed through the assistance of non-invasive multimodality imaging. An 81-year-old man presented with worsening dyspnoea, reduced effort tolerance and limb swelling. Examination and bedside investigations demonstrated congestive cardiac failure. On arrival, N-terminal-pro B-type natriuretic peptide was 2400 ng/L, and high-sensitivity troponin T was 78 mmol/L. Echocardiography showed severe left and right ventricular hypertrophy, and a Doppler study revealed diastolic dysfunction. Cardiac magnetic resonance imaging revealed on non-conventional dark blood sequence an abnormal inversion time for nulling myocardium suggestive of infiltrative disease, including amyloidosis. The patient was referred for nuclear-based studies involving technetium-99m pyrophosphate which demonstrated changes highly diagnostic of ATTR-CA. Early diagnosis of ATTR-CA remains paramount due to the increasing availability of disease-modifying therapies. Current guidelines recognise the role of multimodality imaging in confidently recognising the disease without the need for histological evidence in the appropriate context, providing an alternative means of diagnosis.
Introduction
Cardiac amyloidosis (CA) is a severely underdiagnosed cause of heart failure with preserved ejection fraction (HFpEF) and restrictive cardiomyopathy in Malaysia. 1 We present a unique case of highly probable transthyretin CA (ATTR-CA) diagnosed entirely through the assistance of non-invasive multimodality imaging.
Case presentation
An 81-year-old man presented following a year of worsening dyspnoea, reduced effort tolerance and peripheral limb swelling, which had worsened over the past three weeks. He had no known medical illnesses prior to presentation and was a non-smoker. His vitals on arrival include a blood pressure of 124/78 mmHg, pulse rate of 64 bpm, respiratory rate of 20 breaths per minute and oxygen saturation of 88% on room air which improved with oxygen supplementation. He appeared cachectic, and an examination revealed a dullness to percussion in the right lower zone with reduced air entry on auscultation. There was also bibasal crepitations. Electrocardiogram revealed atrial arrhythmia with variable atrio-ventricular block, wide QRS complexes suggestive of bi-fascicular block and intraventricular conduction delay, without overt changes suggestive of ischaemia (Figure 1). Chest radiography confirmed evidence of cardiomegaly, bilateral pleural effusion and pulmonary oedema (Figure 1). Initial blood investigations include a N-terminal-pro B-type natriuretic peptide of 2400 ng/L and high-sensitivity troponin T of 78 mmol/L (Table 1).

Clinical investigations including an (a) electrocardiogram revealing atrial arrhythmia with variable atrio-ventricular block, wide QRS complexes suggestive of bi-fascicular block and intraventricular conduction delay, without overt changes suggestive of ischaemia and (b) chest radiography suggestive of pulmonary oedema with evidence of bilateral pleural effusion and cardiomegaly.
Blood investigations, serum and urine electrophoresis analysis.

Reference ranges are shown in parentheses.
Transthoracic echocardiography demonstrated an ejection fraction of 65%, pleural effusion and evidence of increased left and right ventricular wall thickness, alongside bi-atrial enlargement. Doppler study for diastolic function revealed abnormally reduced medial (0.02 m/s) and lateral e′ (0.03 m/s), and elevated E/e′ (28 m/s) values, with elevated left atrial volume (36 mL/m2), indicative of a restrictive physiology and supporting a diagnosis of HFpEF (Figure 2). 2 Furthermore, echocardiography did not show any significant valvular lesions. Cardiac magnetic resonance imaging was also performed, which revealed on non-conventional dark blood sequence an abnormal inversion time for nulling myocardium suggestive of infiltrative disease, including amyloidosis (Figure 3). Coronary angiography was performed which revealed normal coronaries. Serum and urine protein plain gel electrophoresis, via separation of agarose gel with amido black, and acid violet for immunofixation to monoclonal light chains and paraprotein did not reveal evidence of immune paresis (Table 1).
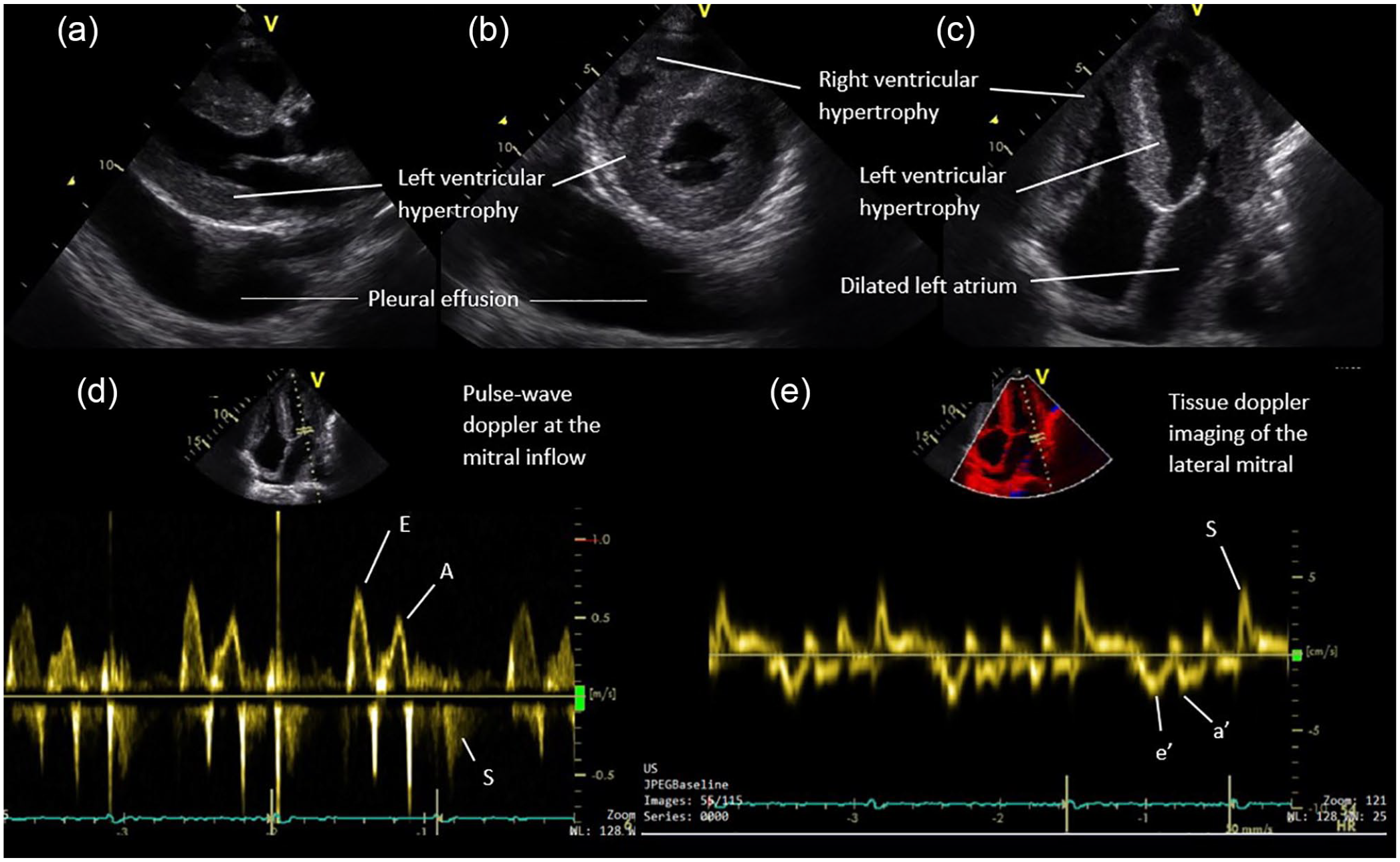
Transthoracic echocardiography in the (a) parasternal long-axis plane, (b) parasternal short-axis plane and (c) apical four-chamber plane demonstrating pleural effusion, left and right ventricular hypertrophy and dilated left atrium. Doppler study including (d) pulse-wave Doppler to assess mitral inflow and (e) tissue Doppler imaging of the lateral mitral annulus and medial mitral annulus (not shown) revealed values suggestive of diastolic dysfunction.

Cardiac magnetic resonance using non-conventional dark blood at the short-axis view demonstrating (a) an abnormal inversion time for nulling myocardium indicating abnormal myocardial tissue, as well as (b)–(e) at the short-axis view moving from the base to apex and using non-conventional dark blood late gadolinium enhancement sequence where there was diffuse, heterogenous transmural enhancement. Both were suggestive of infiltrative disease, including possible amyloidosis.
Due to this, a decision was made to pursue an imaging-based route of diagnosis. The patient was referred for nuclear-based studies, specifically involving technetium-99m pyrophosphate (Tc-99m-PYP; Figure 4). Following administration of 15.67 mCi Tc-99m-PYP, moderately diffused increase tracer uptake in the heart with attenuated uptake in the ribs and whole skeletal system was seen. There were no other sites with radiotracer uptake. A heart-to-contralateral ratio of 1.69 and Perugini grade 3 were highly diagnostic of ATTR-CA. Unfortunately, single photon emission computed tomography (SPECT), which would have been useful to confirm PYP uptake was in the myocardium rather than the blood pool, was not performed, as there was difficulty in sourcing an institution which could provide such a test. Nevertheless, the available investigations were sufficient to suspect a diagnosis of ATTR-CA. As for his atrial arrhythmia and atrioventricular dissociation, the patient and his family were not keen for further invasive management, but he remains on close monitoring for complications.

Nuclear imaging using technetium-99m pyrophosphate (a)–(c) demonstrating moderately diffused increase tracer uptake in the heart with attenuated uptake in the ribs and whole skeletal system. There were no other sites with radiotracer uptake. (c) A heart-to-contralateral ratio of 1.69 and Perugini grade 3 were highly diagnostic of transthyretin cardiac amyloidosis.
Discussion
Interest surrounding cardiac amyloidosis, specifically the ATTR-CA phenotype, has largely been driven by increasing numbers of effective disease-modifying therapies, such as TTR stabilisers, including tafamidis and diflunisal. 3 These treatments often confer benefit when prescribed during the early stages of the disease, and therefore prompt diagnosis is paramount. The introduction of a parallel pathway for the diagnosis of ATTR-CA through non-invasive means provides a useful alternative to diagnose this condition with very high specificity without the need for histology through endomyocardial biopsy, after light chain deposition disease has been ruled out by urine and serum immunofixation. 4
Currently, the role of nuclear medicine is emerging in the diagnosis of ATTR-CA. Not only is it non-invasive, but it is also easy to perform, with the ability to distinguissh the AL subtype from the ATTR subtype. Bone-seeking agents such as Tc-99m labelled 3,3-diphosphono-1,2-propanodicarboxylic acid, PYP and even methylene diphosphonates have been shown to be taken up by the myocardium in patients with ATTR-CA. 5 Preferential binding of Tc-99m-PYP to the myocardium in ATTR-CA patients may be attributed to the fact that ATTR fibrils in the myocardium have a higher calcium content. 6
We were fortunate in being able to pursue the non-invasive route, following a cascade of testing which was supportive of the diagnosis of ATTR-CA. However, clinicians should be aware that this is rarely the case in reality, and an endomyocardial biopsy may still be essential. This is often the case when the Tc-99m-PYP scan reveals a Perugini score <2, or the urine and/or serum immunofixation shows the presence of monoclonal gammopathy. 3 Another important lesson from our case study is the need for SPECT imaging in diagnosing ATTR-CA. SPECT identifies myocardial retention of technetium-based isotopes, and is useful in discriminating the blood pool on planar scans that result in false-positive testing from myocardial uptake of the isotope.3,7
Conclusion
ATTR-CA remains a rare and progressive disease with non-specific clinical manifestations, causing delays in clinically recognising this entity. Current guidelines recognise the role of multimodality imaging in confidently recognising the disease without the need for histological evidence in the appropriate context, providing an alternative means of diagnosis.
Footnotes
Acknowledgements
The authors would like to acknowledge Universiti Teknologi MARA, Sungai Buloh, for supporting the submission of this case report.
Authors’ contributions
R.E.F.R.S. undertook the data collection and analysis and drafted the manuscript. H.A.Z.A. and S.S. drafted and revised the manuscript.
Availability of data and materials
The data that support the findings of this study are available from UiTM, Sungai Buloh, but restrictions apply to the availability of these data, which were used under licence for the current study and so are not publicly available. Data are, however, available from the authors upon reasonable request and with permission of UiTM, Sungai Buloh.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from the Universiti Teknologi MARA (UiTM) Sungai Buloh Ethics Committee. The manuscript does not report on any animal data or tissue.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient for his anonymised information to be published in this article.
