Abstract
Introduction and objectives:
The rate of symptomatic lymphoceles requiring intervention after renal transplantation is reported to be only 5.6%. Controversies prevail in the current literature regarding the management of symptomatic lymphoceles post renal transplantation, with no established algorithm. The creation of a peritoneal window, frequently performed laparoscopically, is deemed the gold standard for management. We herin report the case of a lymphocele post renal transplant treated minimally invasively with intranodal lymphangio-embolisation, with a review of the current literature of this uncommon procedure.
Methods:
This was a retrospective review of this patient’s electronic medical records.
Results:
We present a 43-year-old male with end-stage kidney disease secondary to chronic glomerulonephritis, having been on hemodialysis for seven years. He underwent a deceased donor dual kidney transplant, complicated postoperatively by renal vein thrombosis in one of the grafts, resulting in early graft nephrectomy as well as a distal ureterovesical leak requiring reimplantation. On re-implantation on postoperative day 16, he was noted to have a persistent high drain ouput with a normal drain fluid creatinine. A right intranodal lymphagiogram was performed, and this demonstrated an active lymph leak around the transplanted kidney. A 33% glue (N-butyl cyanoacrylate)-Lipodiol infusion was then injected at a rate of 0.2 mL/min intranodally under fluoroscopic guidance. The patient underwent another repeat embolisation five days later for a residual lymph leak with satisfactory results. Drain output subsequently decreased, and the drain was removed.
Conclusions:
This case suggests that intranodal lymphangiography and embolisation may not only be a diagnostic tool but can be considered as an effective, minimally invasive and safe method for the treatment of lymphoceles after kidney transplantation.
Introduction
Lymphocele formation after kidney transplantation is common, with reported rates up to 26%. 1 They can develop at any time after surgery, but they occur most frequently within the first three months after surgery. Leakage of lymph can arise from the disrupted lymphatic channels around the recipient vessels and/or the allograft renal hilum. Various factors can contribute to lymphocele formation, such as surgical technique, recipient co-morbidities, immunosuppression and delayed graft function, among others.2,3 The majority of cases are asymptomatic and self-limiting. The rate of symptomatic lymphoceles requiring interventions has been reported to be around 5.6%. 4 The indications to intervene include the degree of mass effect exerted, graft dysfunction, septic complications or fistula formation. A wide range of treatment strategies are available, but no consensus exists on the optimal management of lymphoceles post kidney transplantation. 5 The creation of a peritoneal window is deemed as the gold standard treatment option. More recently, less invasive methods such as the instillation of various sclerosants have been described. We report the case of successful treatment of a post kidney transplant lymphocele using intranodal lymphangio-embolisation, and provide a literature review of this uncommon procedure.
Case report
We present a 43-year-old man with end-stage kidney disease secondary to chronic glomerulonephritis, having been on hemodialysis for seven years. He underwent a deceased donor dual kidney transplantation, with a stormy postoperative recovery, complicated by renal vein thrombosis of the inferiorly placed allograft, leading to graft rupture and requiring early graft nephrectomy. He subsequently had a ureterovesical anastomotic leak requiring ureteric reimplantation. By day 16 post ureteric reimplantation, the drain output remained persistently high – up to 1000 mL daily. Normal drain fluid creatinine excluded a urinary leak. Serum creatinine remained stable postoperatively at 90–125 µmol/L with preserved renal function.
A right intranodal lymphangiogram was performed, which demonstrated active lymph leakage around the transplanted kidney. Under ultrasound guidance and local anaesthesia, the largest right inguinal lymph node was accessed with a 21G needle, with the tip placed within the node and connected to an infusion pump (Figure 1). Lipiodol was infused at a rate of 12 mL/h (0.2 mL/min) under fluoroscopic monitoring. Spot images taken at intervals demonstrated contrast pooling around the allograft and discharging into the surgical drain tubing, indicating an active lymph leakage (Figure 2). Intranodal lymphangio-embolisation was performed, whereby a 33% N-butyl cyanoacrylate/lipiodol (0.7 ml) infusion was injected intranodally via the same needle under fluoroscopic guidance (Figure 3).

Ultrasound-guided inguinal lymph node (arrows) access for initiation of intranodal lymphagiogram using a 21G spinal needle (arrowheads).
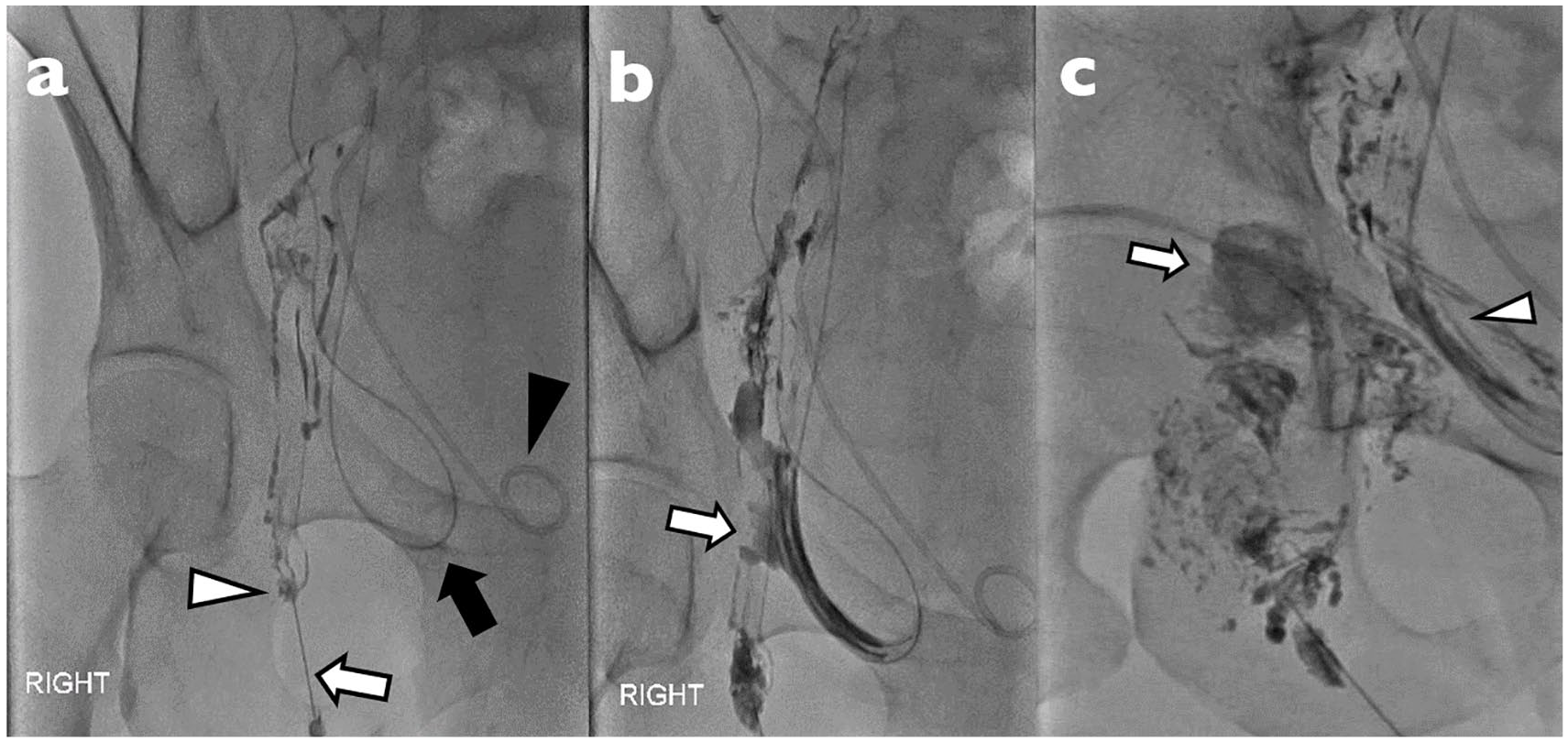
(a) Lipiodol lymphangiogram performed via injection through needle (white arrowhead) in lymph node (white arrow) with the presence of a double J stent indicated by the black arrowhead and an abdominal drain indicated by the dark arrow. (b) and (c) Subsequent follow-up spot images of continued flow of lymphatics with pooling around the graft, showing evidence of a lymphatic leak (white arrow) and flow outwards via the abdominal drain (white arrowhead).
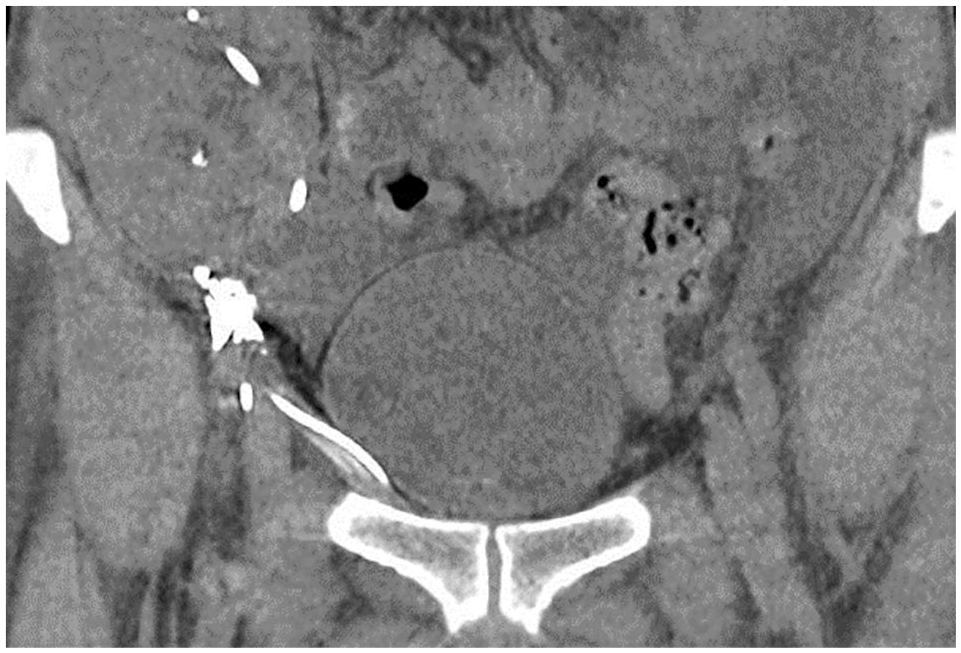
Computed tomography scan obtained one hour following Lipiodol injection shows effective glue embolisation with disrupted lymphatics.
Post procedure, although there was significant decrease in drain output, there was a suspicion of a small residual lymph leak as the drain output stabilised at 100–200 mL daily. A repeat intranodal lymphangiography was performed, which demonstrated a limited lymph leak, and lymphagio-embolisation was performed in the same sitting with good effect. There was no additional technical difficulty encountered during the repeat procedure, with an adjacent prominent lymph node accessed via the same technique. The drain output decreased further over the next three days, and the drain was removed. Thereafter, the recipient remained stable throughout the rest of his inpatient care, with no further sequelae. On discharge, his renal function was normal with an estimated glomerular filtration rate of 70–80 mL/min/1.73 m2 and normal serum creatinine of 70–80 µmol/L.
Discussion
Lymphorrhea is the abnormal flow of lymph that drains externally from disrupted lymphatic vessels or from the abdominal wall through the surgical wound. In kidney-transplanted patients, a lymphocele is formed where there is a collection of lymph in a pseudocystic entity lined by a hard fibrous capsule frequently localised around the graft. 6 There are specific forms of lymphatic leakage, such as chylorrhea and chylous ascites (chyloperitoneum), chylothorax and chyloretroperitoneum. This difference in classification is attributed to fluid appearance, content and location. While lymph fluid and chyle distribute to different lymphatic channel vessels, lymph fluid is clear or straw coloured, whereas chyle is milky due to its rich chylomicrons and triglyceride content.7,8
The aetiology of lymphocele formation following kidney transplantation is still under investigation. Proposed surgical risk factors include mode of procurement, grafts with multiple arteries and extensive dissection of lymphatics around the recipient iliac vessels and allograft kidney.9–11 In a review, Ranghino et al. showed that medical risk factors include obesity, diabetes, delayed graft function and use of particular immunosuppressive drugs. 12
Most commonly, lymphoceles are clinically asymptomatic and incidentally diagnosed during routine ultrasound examinations. Complications may manifest due to mass effect on surrounding structures such as the allograft ureter, leading to obstructive uropathy, which could result in graft loss if unresolved. In severe cases, loss of fluid, triglyceride, lymphocyte and immunoglobulin may lead to dehydration, nutritional deficiency and immunological dysfunction.13,14 As a result, wound dehiscence and infection may also occur. Before the diagnosis of lymphatic leakage, we must first rule out other postoperative complications such as ascites, inflammatory exudation from recent surgical intervention and urine leak with resultant urinoma. Physical signs suggestive of a lymphatic leak can be as subtle as swelling under the wound or comprising of clear or milky fluid from the wound or drain. Drainage amount increasing in relation to oral intake should also further raise suspicion of lymph leakage. Collection and biochemical analysis of drainage fluid is necessary. To distinguish lymphoceles from urinomas or seromas, biochemical analysis of the drain fluid is crucial (fat and protein content, pH, creatinine, triglycerides and specific gravity). Radiological evaluation is helpful to delineate the location and extent of the collection. This can be done through ultrasonography or cross-sectional imaging (computed tomography (CT) or magnetic resonance imaging (MRI)). Lymphangiography can be useful to assess the severity and location of active lymph leakage.
Lipiodol is an iodinated poppy seed oil which is used as a radio-opaque contrast agent. Lipiodol-based lymphangiography was widely utilised in the 1900s as a diagnostic procedure, but its popularity decreased with the advent of non-invasive imaging modalities such as MRI and CT. Nevertheless, lymphangiography still has a role in selected cases. While lymphangiography is a diagnostic procedure, it has shown to have therapeutic properties in cases of refractory lymph leakage, possibly from the inflammatory reaction to Lipiodol.15,16 Indeed, there has been a resurgence in the use of lymphangiography, possibly attributable to the increasing role of minimally invasive surgery in more complex cases, which might in turn result in a higher incidence of lymphatic leak. 9
There are two approaches to lymphangiography: the intranodal method and the pedal method. Bipedal lymphangiography is cumbersome to perform and requires incisions in the lower limbs to expose the fine lymphatics for cannulation, which itself is technically challenging. With its advent in imaging technology and image-guided techniques, the intranodal approach is now favoured over the pedal method as it is more consistent, less laborious and faster. It uses ultrasound guidance to locate and directly access an inguinal lymph node. Patients undergoing the procedure should be screened for cardiac and pulmonary diseases, particularly right-to-left cardiac shunts and pulmonary insufficiency, as the main complication of lipiodol-based lymphangiography is pulmonary embolisation. Additionally, there have been case reports describing lipid embolisation to the brain and subsequent neurological radiation changes.17,18 This phenomenon is poorly understood and is attributed to the disruption of pulmonary vasculature and disruption of filtering ability, allowing lipid emboli into the systemic circulation. 19 Other rare complications that have been described include hypersensitivity to the iodinated oil and hypothyroidism. 20
Lymphangiography has been shown to be effective for patients with chylothorax, chylous ascites and lymphatic fistulas. Alejandre-Lafont et al. suggested an increased therapeutic success rate with lower volumes of lymph leakage of <500 mL/day. 21
The choice of therapy for a post-kidney transplant lymphocele is dependent on various factors, such as the degree of symptoms, size of the lymphocele, extent of mass effect on surrounding structures, potential post-therapeutic complications and clinical status of the patient. Conservative management includes diet modification (a high-protein, low-fat, medium-chain triglyceride diet) and medical therapy (somatostatin analogue, vasoconstrictor pancreatic lipase inhibitors). Therapeutic options range from simple aspiration under direct imaging with or without sclerotherapy to more invasive options of laparoscopic or open surgery to fenestrate the lymphocele into the peritoneal cavity. For cases in which sclerosing therapy is utilised, an infusion of sclerosant is often infused after aspiration of the lymphocele cavity. Commonly used sclerosants include povidone iodine, ethanol and doxycycline. 22 Complications of sclerosing therapy consist of sclerosant allergy, spillage of the sclerosant causing surrounding inflammation and catheter-related infection. Principles of the traditional operative approach consist of creating drainage of the fluid collection into the abdominal cavity by connecting the cavity into the intraperitoneal space via either the open or laparoscopic approach. Lucewicz et al. evaluated the outcomes of 1113 cases of primary symptomatic lymphocele after kidney transplantation via their approach. Laparoscopic fenestration was associated with the lowest risk of recurrence at 8% compared to 16% and 51% for open surgery and aspiration therapy, respectively. 4 However, the laparoscopic approach was also noted to have risks, which included an overall 12% conversion rate to open surgery. Although typically successful, these surgical interventions are associated with a longer hospital stay and post-surgical complications. In addition, these patients must be physically able to tolerate further surgery, which might not be optimal in this group of already high-risk patients. This is where a minimally invasive technique such as lymphangiography and embolisation is an invaluable option as a single diagnostic and therapeutic procedure. The literature has also shown that repeat lymphangio-emoblisations, when indicated, can be done effectively with no additional complications.
We embraced a stepwise approach for our patient. In our recipient, the lymph leakage was significant, with an output of 800–1000 mL/day. Initially conservative treatment was adopted, with progression to consideration for therapeutic intervention as he became consistently fluid depleted due to high drain output. He was offered a laparoscopic approach for the creation of a peritoneal window as a definitive measure. However, in view of his stormy postoperative course, he was more inclined to explore non-operative modalities. On seeing significant leaks, we elected to perform direct glue embolisation. The time interval from first lymphangio-embolisation to resolution of lymph leakage was six days, which was shorter than the 8–17 days reported in other studies.23,24 Post procedure, his creatine remained stable at 95–110 µmol/L with preserved renal function. Our patient did not have to undergo additional general anaesthesia and its associated risks, with an equivalent comparable outcome.
The method of intranodal lymphangio-embolisation described is not novel. However, there is sparse evidence of the application of this technique as a treatment for lymphocele after kidney transplantation. The minimally invasive nature of the procedure with comparable outcomes renders it an attractive front-line option in comparison to sclerotherapy or surgery in consideration for approaching lymphocele after kidney transplantation. The choice of modality of treatment in this group of patients should be tailored and dependent on the clinical expertise available.
Conclusions
Our case demonstrates that intranodal lymphangio-embolisation can be an effective, minimally invasive and safe method for the treatment of lymphoceles or lymphorrhea after kidney transplantation.
Footnotes
Acknowledgements
None.
Authors’ contributions
L.E.J. and V.G. researched the literature and conceived the study. E.J. and E.A. were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. E.J. wrote the first draft of the manuscript. All authors reviewed and edited the manuscript, and approved the final version of the manuscript.
Availability of data and materials
Data sharing is not applicable to this article, as no data sets were generated or analysed during the current study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Singapore General Hospital does not require ethical approval for reporting individual cases or case series.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient for his anonymised information to be published in this article.
