Abstract
Background:
Calcineurin inhibitors are the cornerstone of maintenance immunosuppression after kidney transplant. While studies on predominantly Caucasian populations recommend tacrolimus over cyclosporine, the effects on Singapore’s local population remain unclear.
Objectives:
This study aimed to compare the impact of tacrolimus against cyclosporine on post-transplant outcomes in our local kidney transplant population.
Methods:
A single-centre retrospective chart review was conducted on ABO- and human leucocyte antigen (HLA)-compatible kidney transplantations between 1 January 2011 and 15 August 2018. Patients who received basiliximab induction, prednisolone, mycophenolate and either tacrolimus or cyclosporine were included and followed up for at least one year. Recipients of transplantations at other institutions or other immunosuppressive regimens were excluded. Patient and graft outcomes and adverse effects were collected.
Results:
Overall, 120 patients on tacrolimus and 49 on cyclosporine were included. Patients on tacrolimus were older. This group had more deceased donor transplants, a higher proportion with donor-specific antibodies (DSAs) present and more HLA mismatches. There were no differences in patient and graft survival, graft function and acute rejections at one year, despite adjusting for age, transplant type, presence of DSAs and total HLA mismatches. The tacrolimus group had more infectious admissions (odds ratio=0.27, 95% confidence interval 0.098–0.73, p=0.01) after adjusting for age, transplant type, HLA mismatches, presence of DSAs and acute rejections, with increased severity and more opportunistic infections. More patients on cyclosporine required a change to alternative immunosuppressants (p=0.003).
Conclusion:
Our study demonstrated comparable short-term post-transplant outcomes between cyclosporine and tacrolimus. Tacrolimus appears more tolerable but may be associated with infection risks.
Introduction
Calcineurin inhibitors (CNIs) such as cyclosporine and tacrolimus are the mainstay of immunosuppression in the modern era of kidney transplantation. Tacrolimus in particular was shown to improve allograft function, with fewer rejections compared to other regimens in the landmark Efficacy Limiting Toxicity Elimination-Symphony (ELITE-Symphony) trial. 1 Previous studies showed tacrolimus to be superior to cyclosporine in reducing graft loss and acute rejection, with less dyslipidaemia 2 and increased cost-effectiveness.3,4 However, tacrolimus may be associated with a higher incidence of complications such as diabetes mellitus. 5 In 2009, Kidney Disease: Improving Global Outcomes 6 revised their guidelines to suggest that tacrolimus be the first-line CNI for maintenance immunosuppressive therapy post kidney transplant.
In alignment with international recommendations, Singapore General Hospital (SGH) revised its protocol for tacrolimus to be used as the first-line CNI since June 2016. However, the impact of different CNIs on post-transplant outcomes in our local multi-ethnic population is unclear, since most studies were performed on predominantly Caucasian populations. While previous local studies have shown improvement of one-year graft survival following the introduction of cyclosporine in 1985 7 and tacrolimus in 2000, 8 these patients did not receive antibody induction therapies which are today regarded as the standard of care. The impact of different CNIs has not been studied in more recent local studies. 9
As such, a single-centre retrospective chart review was performed to compare the one-year post-transplant outcomes of kidney transplant recipients at SGH receiving cyclosporine or tacrolimus-based immunosuppressive regimens prescribed at discharge.
Methods
Study design
This was a single-centre retrospective chart review of kidney transplant recipients at SGH with standard immunological risk to evaluate the impact of cyclosporine versus tacrolimus on post-transplant outcomes. Data were retrieved from the SGH Renal Transplant Registry and electronic and hardcopy records.
Inclusion and exclusion criteria
A total of 287 patients who received a deceased or living kidney donor transplant between 1 January 2011 and 15 August 2018 were screened for inclusion into the study. Patients were included if they received basilixmab induction and were discharged from their transplant surgery admission with corticosteroids, mycophenolate and either cyclosporine or tacrolimus but excluded if they received (a) lymphocyte-depleting agents (e.g. thymoglobulin), (b) ABO-incompatible or cytotoxic and flow cross-match positive transplants, (c) transplants performed at other institutions, (d) other maintenance immunosuppressive regimens (e.g. mTOR inhibitors) or (e) of they were <18 years of age. Patients were then allocated to study groups based on CNI used on discharge (tacrolimus or cyclosporine).
Baseline characteristics and outcomes
Data on immunosuppressive regimens at post-transplant discharge and one year post transplant, and baseline characteristics on demographics, transplant details and immunological data were obtained.
Outcomes were measured over a period of at least one year. Primary outcomes included patient survival, graft survival and graft function at discharge, 6 months and 12 months post transplant (determined with estimated glomerular filtration rate (eGFR) calculated using the Modification of Diet in Renal Disease equation). Secondary outcomes included biopsy-proven allograft rejection (including treated borderline rejections) occurring after discharge from transplant admission, hospital admissions with a primary diagnosis of infection, admissions for opportunistic infections1,10 (OIs; BK virus (BKV),
Statistical analysis
Analysis was performed with IBM SPSS Statistics for Windows v25 (IBM Corp., Armonk, NY). Descriptive statistics such as median and interquartile range (IQR) were obtained. Categorical and continuous data were analysed with the chi-square test and Mann–Whitney
Results
Baseline characteristics
The study included 169 patients: 120 received tacrolimus, and 49 received cyclosporine at discharge. The tacrolimus group was older, had more deceased transplants, a higher proportion with donor-specific antibodies (DSAs) present and more total human leucocyte antigen (HLA) mismatches (Table 1).
Baseline characteristics according to regimen at post-transplant discharge.

DSA information was available for 102 tacrolimus cases and 36 cyclosporine cases.
IQR: interquartile range; ESRF: end-stage renal failure; CGN: chronic glomerulonephritis; NA: not applicable; DSA: donor-specific antibody; PRA: panel reactive antibody;
Primary outcomes
Patient and graft survival rates at one-year follow-up were similar between both groups (Table 2). The eGFR was significantly higher in patients on cyclosporine versus tacrolimus at discharge after transplant (53.4 vs. 42.6 mL/min/1.73 m2,
Outcome measures at one year post renal transplant.
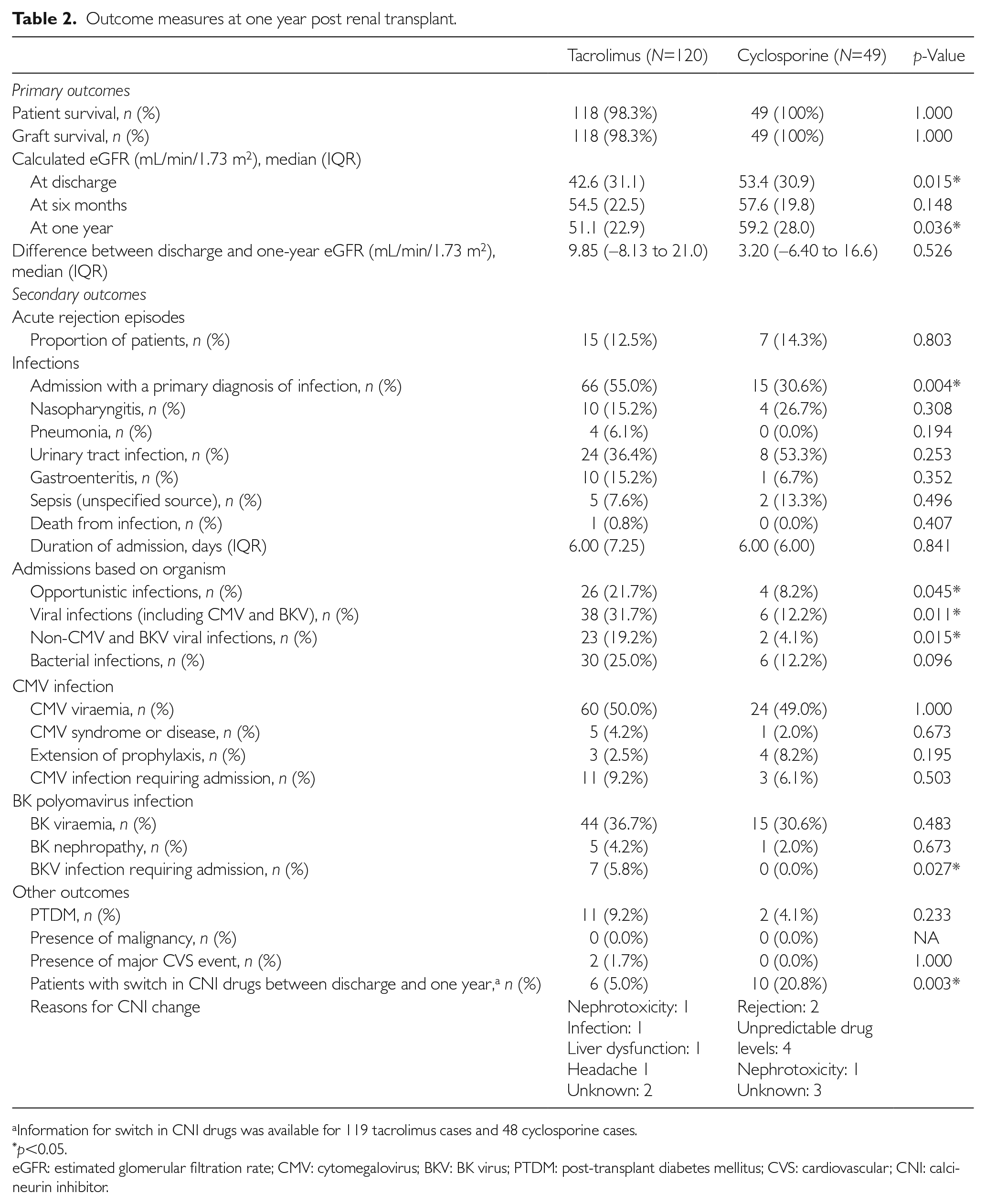
Information for switch in CNI drugs was available for 119 tacrolimus cases and 48 cyclosporine cases.
eGFR: estimated glomerular filtration rate; CMV: cytomegalovirus; BKV: BK virus; PTDM: post-transplant diabetes mellitus; CVS: cardiovascular; CNI: calcineurin inhibitor.
Secondary outcomes
There were no significant differences in the frequency of acute rejection episodes after discharge from transplant surgery (Table 2) in both groups (12.5% vs. 14.3%,

Kaplan–Meier analysis of time to first admission with a primary diagnosis of infection. Mean time to admission for tacrolimus group was 37.3 months (95% confidence interval (CI) 28.9–45.6) compared to 53.7 months (95% CI 41.5–65.9) in the cyclosporine group. Log rank test showed
The average durations of admissions for infections were similar, but admission rates for OIs, non-BKV and CMV viral infections such as parvovirus and influenza were higher in the tacrolimus group (Table 2). The tacrolimus group also appeared to have more severe infections, having the only infection-related death and more BKV infections requiring admission, despite both groups having similar BKV infection rates.
Switching of CNIs
A higher proportion of patients on cyclosporine required crossover to tacrolimus between discharge and one-year follow-up compared to the tacrolimus group (20.8% vs. 5.0%,
Discussion
Our study demonstrated comparable outcomes between tacrolimus and cyclosporine, with similar patient and graft survival. Allograft function at one year post transplant was similar after adjusting for possible confounders in contrast to the ELITE-Symphony trial 1 and a Cochrane systematic review. 14 However, the improvement in the eGFR was numerically higher on tacrolimus, similar to Cheung et al. 15 Thus, benefits of tacrolimus on graft function could be observed, despite a less than ideal donor and recipient profile.
Unlike previous studies5,16 involving predominantly Caucasian populations, our study observed similar rejection rates between tacrolimus and cyclosporine regardless of the type of kidney transplant. This is similar to studies on a Saudi 17 and Chinese population 15 which found no significant difference in rejection rates, suggesting possible differences in the effect of tacrolimus on different ethnicities.
Our study demonstrated more infection-related admissions, more severe infections and susceptibility to OIs on tacrolimus versus cyclosporine, in contrast to previous studies.2,16 One reason may be genetic variations in CYP3A5 18 involved in tacrolimus metabolism. 19 Frequencies of CYP3A5 expression 20 vary in different ethnicities 21 (15–35% in Asians vs. 5–15% in Caucasians), and studies on liver transplantation have suggested that CYP3A5 expressers experience more infections on tacrolimus, possibly due to higher dose requirements, 22 a lower immune response 23 and the accumulation of active metabolites. 24
Tacrolimus is also noted to be a stronger immunosuppressant than cyclosporine,25,26 possibly contributing to more admissions for OIs and more severe infections in our study. There were also more BKV infections requiring admission in the tacrolimus group, consistent with earlier studies showing increased severity 27 and persistence 28 of BKV infections on tacrolimus.
More patients on cyclosporine required crossover to tacrolimus in our study, mainly due to graft rejection and unpredictable drug levels, similar to findings by Vincenti et al. 29 Tacrolimus may also be more tolerable 30 with fewer cosmetic side effects, 31 and some adverse effects (e.g. tremors) may be reversible with a dose reduction. 16
Limitations
Being a retrospective study in a single institution, this study is prone to confounders and bias. The sample size was small, with a more adverse donor–recipient profile in the tacrolimus group (e.g. higher immunological risk,32,33 older recipients 34 and more deceased donors 35 ). To address potential confounders, patients were matched pharmacologically in immunosuppressive and induction agents, and multivariate analysis was performed.
This study also focused on short-term (one-year) outcomes. Risks of adverse outcomes (e.g. PTDM 36 and malignancy 37 ) increase with duration of immunosuppression, and comparisons of long-term outcomes are needed, since local transplant recipients have good survival at five years and beyond. 9
To evaluate the efficacy and adverse effects of a tacrolimus immunosuppressive regimen better in Singaporean kidney transplant recipients, studies involving other institutions and prospective studies could be conducted.
Conclusion
While our study demonstrated comparable short-term outcomes on patient survival, graft survival and function, and rejection rates between tacrolimus and cyclosporine, the results are uncertain due to confounding by different baseline characteristics. Tacrolimus may be more tolerable but may be associated with more OIs and more severe infections. Findings of previous studies may not be wholly applicable to our local population, and more studies are needed to clarify the efficacy and safety profile of tacrolimus.
Supplemental Material
Supplementary_Appendix – Supplemental material for Impact of tacrolimus versus cyclosporine on one-year renal transplant outcomes: A single-centre retrospective cohort study
Supplemental material, Supplementary_Appendix for Impact of tacrolimus versus cyclosporine on one-year renal transplant outcomes: A single-centre retrospective cohort study by Pei Wen Ong, Terence Kee and Quan Yao Ho in Proceedings of Singapore Healthcare
Footnotes
Acknowledgements
O.P.W. would like to express her thanks to T.K. and H.Q.Y. for their guidance and valuable feedback during the course of this project. The authors would like to express their thanks to the transplant coordinators for their help in data collection.
Authors’ contributions
T.K. and H.Q.Y. researched literature and conceived the study. O.P.W. was involved in protocol development, data collection and data analysis. O.P.W. wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Availability of data and materials
The data sets generated and/or analysed during the current study are available from the authors.
Ethical approval
Further ethical deliberation for this audit was not required by the SingHealth Centralised Institutional Review Board (CIRB Ref: 2019/2682), as this was a clinical audit of routine clinical care, where participants were not subjected to additional risks or burdens beyond usual clinical practice.
Informed consent
Waiver of informed consent for this audit was approved by the SingHealth Centralised Institutional Review Board (CIRB Ref: 2019/2682), as this was a clinical audit of routine clinical care, where participants were not subjected to additional risks or burdens beyond usual clinical practice.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
