Abstract
Sulcal arteries perfuse the anterior two-thirds of the spinal cord, and spinal cord infarction as a result of sulcal artery occlusion is rare. Most reported cases are associated with vertebral artery dissection, and commonly involve the cervical spinal cord. A 74-year-old man presented with sudden onset weakness and numbness after a brief bout of abdominal pain. Further investigations concluded that this was sulcal artery syndrome. We report a case of sulcal artery syndrome affecting the thoracic spinal cord presenting as Brown–Sequard syndrome. Sulcal artery syndrome usually has good prognoses, unlike anterior spinal artery infarction.
Introduction
Sulcal (central or sulco-commissural) arteries are perforating branches of the anterior spinal artery, which perfuse the anterior two-thirds of the spinal hemicord (Figure 1). Sulcal artery strokes are uncommon.1–3 Most reported cases have been associated with vertebral artery dissection, 4 and appear to involve the upper cervical cord. 4 In general, sulcal artery syndrome has a good prognosis, compared with anterior spinal artery syndrome. 2 We report a rare case of sulcal artery infarct of the thoracic spinal cord, presenting as an incomplete, acute Brown–Sequard syndrome.

Cross-section of cervical spinal cord, showing the anterior and posterior spinal arteries with their branches and territories.
Case description
A 74-year-old gentleman, with a history of diabetes, hypertension, ischemic heart disease, chronic kidney disease, and an incidentally noted abdominal aortic dissection, presented with acute onset of abdominal pain. Pain was localized to the epigastrium, right hypochondrium, and right iliac fossa, with radiation to the back. Over the next few hours, he developed right hip pain and weakness affecting the right leg, associated with numbness of the left lower limb. At nadir, he was unable to walk independently. Neurological examination revealed right lower limb weakness (Medical Research Council (MRC) grade 3/5) and impaired pain and temperature sensation below the left thoracic (T4) dermatomal segments. Vibration and proprioception were intact bilaterally, and the results of the rest of the neurological examination were normal. No clinical progression was noted after the first few hours. Investigations included full blood count, serum electrolytes, liver function tests, and C-reactive protein level, which produced results within normal limits. Given the acute onset of neurological deficits, the possibility of spinal cord infarction was considered. Contrast magnetic resonance imaging of the thoracic spine showed a single, focal, discrete, short segment, intramedullary T2W hyperintense signal at the T4–T5 level, involving the right hemicord (Figure 2). There was an absence of cord expansion or lesion enhancement. Diffusion-weighted images were degraded by motion artifacts and could not be interpreted. No abnormal flow voids were noted within the dural space. No abnormalities were noted in the thoracic vertebral bodies or intervertebral disks.
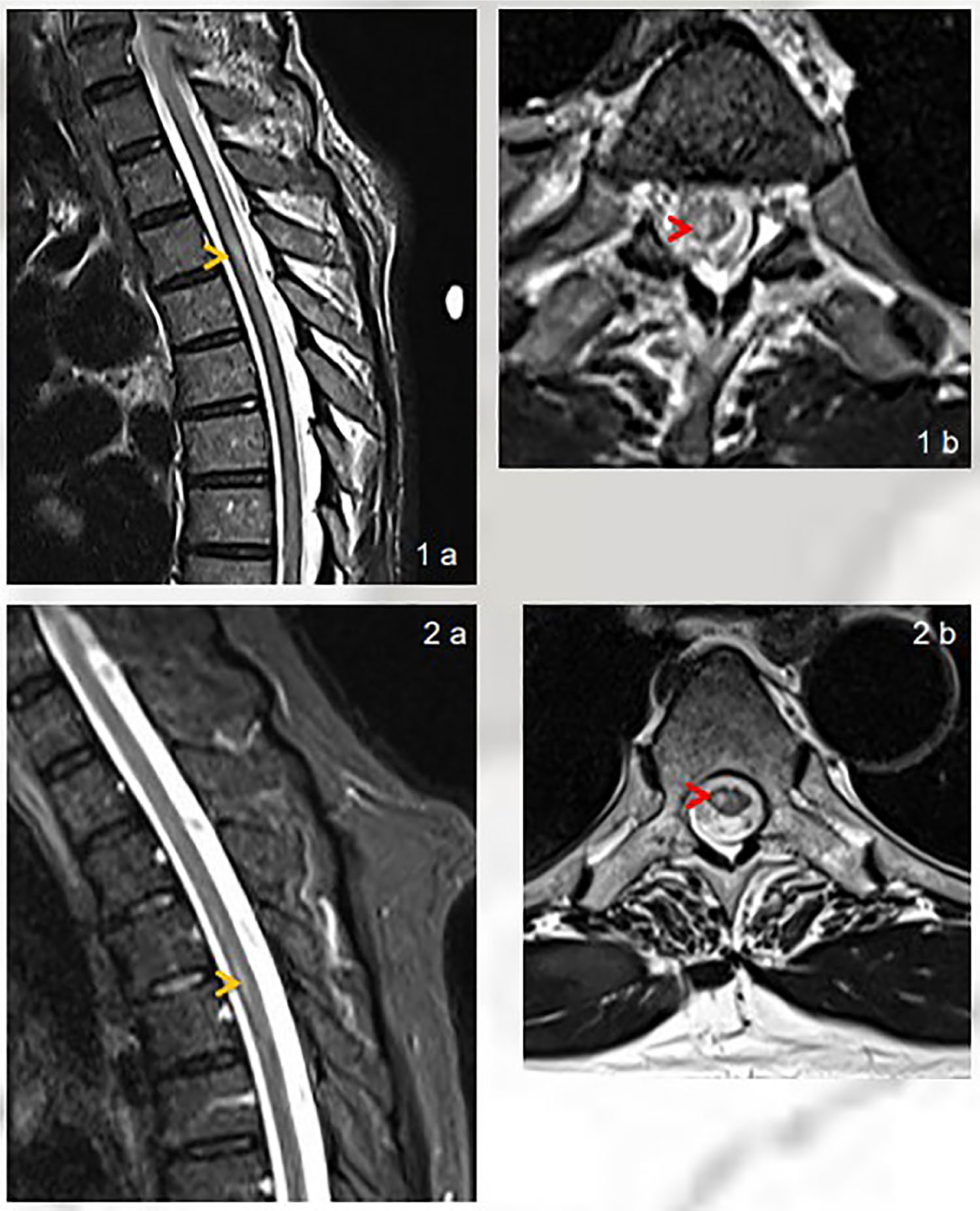
Focal, T2 weighted hyperintensities seen on sagittal (1a, 2a; orange arrowheads) and axial (1b, 2b; red arrowheads) sections of the thoracic spinal cord.
Overall, the clinico-radiological picture was consistent with a diagnosis of spinal cord infarction, involving the T4 sulcal artery. Computed tomographic (CT) imaging of the aorta was performed, in order to delineate an embolic source. The CT aortogram (Figure 3) showed a short segment of chronic dissection flap at the level of the aortic hiatus and mid descending aorta, with a number of intramural thrombi and atherosclerotic plaques. The artery of Adamkiewicz was not well visualized. The patient was managed conservatively. With rehabilitative measures, improvement in motor power was noted over several weeks. At the last clinical follow-up, power in the right lower limb was noted to be normal (MRC grade 5/5), with persistent residual loss of pain and temperature sensation (L3–S2) on the left lower limb.

CT aortogram showing evidence of chronic aortic dissection and calcification. A dissection flap (yellow arrows) is seen in the upper panel, while the lower panel shows evidence of significant calcification (white square). No evidence of acute aortic dissection was noted.
Differential diagnosis
The differential diagnosis for an acute ischemic myelopathy includes traumatic, compressive, inflammatory or infective, and demyelinating etiologies. Our patient had no recent falls or traumatic injury. While non-traumatic compressive myelopathies may occasionally present acutely (for example, epidural hematoma and spinal abscesses), selectivity in long-tract involvement is not notable. Localized back pain may be prominent with such pathologies. Most compressive etiologies, however, tend to be sub-acute to chronic in evolution.
In patients presenting with an acute myelopathy with recent infection or concurrent fever, or if there is clinical or laboratory evidence for sepsis or raised inflammatory markers, infection (septic myelitis) or inflammatory (post-infection or para-infectious transverse myelitis) etiologies must be excluded. In such individuals, lumbar puncture is mandatory, unless otherwise contraindicated. Our patient did not report recent infections or immunization, and laboratory investigations did not show evidence of raised inflammatory markers.
Multiple sclerosis may present with acute myelopathy and, owing to discrete lesions, is commonly known to show selective long-tract involvement. Our patient did not have previous history suggestive of central nervous system demyelination, including optic neuritis. While an isolated attack of multiple sclerosis is a reasonable differential diagnosis in younger patients, such presentations in elderly patients would be rather unusual.
Venous infarction of the spinal cord and venous congestion secondary to underlying dural arterio-venous fistula may present with acute myelopathy. Venous congestion or infarction, however, does not respect “arterial territories”; thus, selectivity of long-tract involvement, such as that seen in our patient, is unlikely to occur with venous spinal pathologies. Infiltrative lesions of the spinal cord, with acute intra-lesional hemorrhage, may also be considered as a differential for acute myelopathies. Though most such patients present with acute or chronic neurological deficits, spinal imaging with contrast is crucial for exclusion of these lesions.
Discussion
Spinal cord infarction is a rare but devastating disorder, caused by a wide array of etiologies. The vascular supply of the spinal cord consists of one anterior spinal artery, which supplies the anterior two-thirds of the spinal cord, and a pair of posterior spinal arteries, which supply the rest of the spinal cord. The anterior spinal artery arises from the vertebral arteries, and is augmented by 31 pairs of radicular arteries, arising from the segmental intercostal arteries and the aorta. The artery of Adamkiewicz most commonly arises at the level of the T9–12 intercostal artery (branch of the aorta), and supplies the lower two-thirds of the spinal cord via the anterior spinal artery. From the anterior spinal artery, sulcal arteries (Figure 1) travel into the anterior cord, dividing to supply either the right or the left anterior cord. Figure 1 shows the anterior and posterior spinal arteries with their respective branches and territories. There are numerous known variations in the vascular supply. 5
Etiologies of spinal cord infarction include pathologies affecting the thoraco-abdominal aorta (occlusion, secondary to arteriosclerosis, vasculitis, infection, emboli, and thrombosis), iatrogenic emboli, hypoperfusion, and venous infarction. 4 Most spinal cord infarctions involve the anterior two-thirds of the spinal cord, in an anterior spinal artery distribution. 4 As both anterior horns are affected by an anterior spinal artery infarct, patients typically present with bilateral weakness and dissociated sensory loss secondary to the involvement of the bilateral corticospinal and spinothalamic tracts; vibration and proprioception are preserved, as there is sparing of posterior columns. Occlusion of the artery of Adamkiewicz is expected to produce deficits akin to an anterior spinal artery infarct, as it is the main contributor to anterior spinal artery supply in the lower two-thirds of the spinal cord. Our patient did not have symptoms in keeping with holo- anterior spinal artery involvement.
Rarely, cord infarction may occur secondary to emboli in the occlusion of a sulcal artery or its branch. Unlike anterior spinal artery infarcts, where bilateral anterior hemicords are involved, unilateral sulcal artery infarctions involve only one-half of the anterior spinal cord. Thus, patients present with an acute unilateral weakness, with contralateral loss of pain and temperature sensation. Unlike the typical Brown–Sequard syndrome, vibration, proprioception and light touch sensation are normal, as the posterior columns are not involved. Our patient’s deficits were thus concordant with sulcal artery syndrome, rather than Brown–Sequard syndrome.
Sulcal artery syndrome appears to involve largely the upper cervical spinal cord, which may be a consequence of unilateral occlusion of anterior spinal artery duplication. 4 Radiological features of sulcal artery syndrome include swelling of the cord, or T2-weighted hyperintensities in the central, para-median, and unilateral anterior spinal cord, corresponding to the spinal sulcal artery territory. 1 Diffusion-weighted imaging is useful to demonstrate acute spinal cord infarcts, but may not be performed routinely. Thus, in patients with acute myelopathies where spinal cord infarction is suspected, diffusion-weighted imaging must be requested.
Most cases of sulcal artery infarction reported in the literature were secondary to vertebral artery dissection, 2 and involved the cervical spinal cord.2, 3 Very rarely, direct compression of the artery of Adamkiewicz by disk herniation 6 has been reported to cause spinal cord infarction; however, as stated earlier, our patient had deficits concordant with sulcal artery syndrome rather than anterior spinal artery or complete Brown–Sequard syndrome. Moreover, no abnormalities were noted in the thoracic vertebral bodies or intervertebral disks.
In our patient, a CT aortogram showed multiple atheromatous lesions and mural thrombi, involving extensive areas of the thoracic and abdominal aorta. The artery of Adamkiewicz could not be reliably traced. Visualization of the artery of Adamkiewicz on a CT aortogram remains challenging. Even with a slice thickness of 0.25 mm, sensitivity for identification is about 90%; for demonstration of full vascular continuity of the anterior spinal artery with the aorta (important for differentiation from radiculomedullary veins), sensitivity is reported to be 70%. 7 For a standard 0.5 mm slice thickness on a multidetector CT aortogram, the identification rate is less than 50%. 7 In our patient, given the acute presentation with abdominal pain, an embolus from the aorta was deemed to be the most likely etiology of the spinal cord infarct. All reported cases of sulcal artery syndrome, including our patient, presented with unilateral, incomplete Brown–Sequard syndrome-like presentation. The prognosis is better in sulcal artery syndrome than in anterior spinal artery syndrome. 2 This is because anterior spinal artery infarction is viewed as a watershed or terminal zone infarction. 2
Conclusion
Sulcal artery syndrome must be considered as a differential diagnosis in acute-onset incomplete Brown–Sequard syndrome. While the cervical spinal cord is most commonly involved in sulcal artery syndrome, rare involvement of the thoracic cord may occur. Sulcal artery syndrome usually has a good prognosis. Emboli, secondary to underlying atheromatous disease or vascular dissection, are the commonest mechanism of sulcal artery syndrome. Thus, vascular imaging must be performed in patients where there is clinical suspicion of sulcal artery syndrome.
Footnotes
Acknowledgements
None
Authors’ contributions
May Myat Win wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript
Availability of data and materials
Not applicable.
Ethical approval
Changi General Hospital (Singhealth) prescribes that no IRB ethical approval is needed for individual case reports as long as consent taken from the patient.
Informed consent
Written and informed consent was obtained from the patient for their anonymised patient information to be published in this article.
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
