Abstract
Objectives:
Copeptin, myeloperoxidase and pro-adrenomedullin have emerged as potential biomarkers for diagnosis and prognosis of acute coronary syndrome (ACS). However, their applicability in patients with chronic kidney disease (CKD) remains unknown as these patients were excluded from previous studies. Our objective was to determine the superior novel cardiac marker to predict 30-day and six-month adverse cardiac events (ACEs) defined as cardiac-related death, myocardial infarction and ventricular fibrillation.
Methods:
A prospective observational study was carried out. Patients were included if they presented to the emergency department with symptoms suggestive of ACS and had CKD as defined as a serum creatinine of more than 130 µmol/l. Copeptin, myeloperoxidase and pro-adrenomedulin assays were performed. Occurrence of ACE was traced from review of the patients’ case records and the registry of deaths.
Results:
A total of 724 patients were recruited: 60.6% were male and 68.6% were Chinese. The median age was 67 years. Among those recruited, 88.3% had CKD stages 4 and 5, with 33.5% on dialysis. The rates of ACE at 30 days and six months were 15.1% and 21.7%, respectively. All readings of the three biomarkers were not significantly different in patients with ACE compared with those without both at 30 days and six months. The areas under the curve for copeptin, myeloperoxidase and pro-adrenomedullin were 0.53, 0.50 and 0.45, respectively (
Conclusions:
The poor performance of the biomarkers may be attributable to lack of specificity for ACS, as elevated levels could be from other causes in CKD patients. Routine testing cannot be recommended.
Introduction
Cardiac markers aid in the diagnosis and prognostication of myocardial infarction in patients with chronic kidney disease (CKD) who present with clinical, electrocardiogram, or imaging findings suggestive of cardiac ischaemia.
In addition to traditional risk factors such as hypertension, diabetes, hyperlipidaemia, smoking, and elderly age, patients with CKD have an increased likelihood of cardiovascular events due to CKD-related physiological derangements such as uraemia, anaemia, increased cytokine and calcium load, and abnormalities in bone mineral metabolism.1,2 In a meta-analysis by van der Velde et al., 3 hazard ratios for cardiovascular events for patients with estimated glomerular filtration rates (eGFRs) of 60, 45, 15 ml/min per 1.73 m2 were 1.03, 1.38 and 3.11, respectively, compared with an eGFR of 95. Patients with CKD also have a worse prognosis after an acute coronary syndrome or coronary interventions. In the Valsartan in Acute Myocardial Infarction (VALIANT) trial, 4 a reduction of eGFR was associated with an increase in mortality at three years after an acute myocardial infarction. Similarly, operative mortality for CKD patients undergoing coronary artery bypass grafting was higher with a reduction in eGFR. 5 Consequently, patients with renal dysfunction may benefit from early diagnosis and treatment of cardiac disease, which may translate into improved survival.
Serum creatine kinase MB isoenzyme (CKMB) was once considered to be the gold standard in diagnosing acute myocardial infarction. Cardiac-specific troponins (troponins T and I) were subsequently introduced in the early 1990s and demonstrated superiority over CKMB. However, there are problems with the use of CKMB and cardiac-specific troponins in patients with CKD. Elevations in CKMB and troponins T and I are often observed in asymptomatic patients with CKD because of left ventricular wall tension, acute or chronic volume overload, silent plaque rupture in the presence of diffuse coronary atherosclerosis, uraemia-induced apoptosis of cardiomyocytes, and impaired renal clearance of free troponins T or I.6–9 There are also different assays for troponin I by different manufacturers because of lack of standardisation and hence heterogeneity in antibody reactivity.10,11
Novel cardiac markers such as copeptin, myeloperoxidase and pro-adrenomedullin (pro-ADM) have surfaced in recent years to aid clinicians in the diagnosis and prognostication of acute coronary syndrome (ACS). Copeptin is the C-terminal part of pro-arginine vasopressin (pro-AVP), released together with AVP during the processing of the precursor peptide. 12 It is released by the neurons of the hypothalamo-neurohypophysial system for osmoregulation and cardiovascular homeostasis. The function of copeptin upon release remains unknown at present. Nonetheless, copeptin has been reported as an excellent diagnostic biomarker – a negative level at time of presentation in the emergency department (ED) has a negative predictive value for ACS of up to 99.7%.13–15 Copeptin is also a strong marker for predicting mortality and morbidity: Elevated levels were found in patients who died or were readmitted with heart failure after an acute myocardial infarction, compared with survivors. 16
Leucocytes play an important role in the transformation of a stable coronary artery plaque to an unstable atherosclerotic plaque rupture.17–21 The pathogenesis is due to oxidative stress and inflammation. Myeloperoxidase is a pro-inflammatory enzyme stored in polymorphonuclear neutrophils and macrophages. By amplifying the reactivity of hydrogen peroxide through the generation of hypochlorous acid, free radicals and reactive nitrogen species, 22 myeloperoxidase results in the oxidation of lipids contained with low-density lipoprotein cholesterol and consumption of endothelial-derived nitrous oxide, leading to vasoconstriction and endothelial dysfunction. Levels are elevated in patients with chronic angina, with substantial decrease after an ACS. 23 Myeloperoxidase is also useful in predicting mortality and myocardial infarction at one year following an ACS.24–26
ADM causes vasodilation 27 via the production of nitric oxide, 28 increases cardiac output 29 and increases GFR leading to diuresis and natrituresis. 30 It has been isolated in adrenal medulla, heart, brain, lung, kidney, gastrointestinal organs, endothelium and vascular smooth muscle cells, with its secretion controlled by cytokines, hormones and physical stimuli. Pro-ADM is the more stable midregional fragment of ADM, allowing for reliable quantification. Other than being a powerful independent predictor of death and heart failure following acute myocardial infarction, 31 baseline levels of pro-ADM were also independently associated with future cardiovascular events across the entire spectrum of coronary artery disease in symptomatic patients. 32
Despite the emergence of these three novel cardiac markers and the subsequent surge in studies on them in current literature, their applicability for diagnosis and prognostication of ACS in patients with CKD remains unclear as these patients were excluded from previous studies. Therefore, there is a need to address this gap by evaluating the diagnostic and prognostic values of these novel cardiac markers in patients with CKD. The objective of our study was to assess the utility of these novel cardiac markers in predicting 30-day and six-month adverse cardiac events (ACEs) in CKD patients presenting to the ED with chest pain or symptoms suggestive of ACS.
Methods
Setting
This study was conducted in the ED of a tertiary hospital in Singapore with an ED attendance of about 135,000 patients a year.
Design
A prospective observational study was carried out from January 2010 to June 2012. Consecutive patients were included if they presented to the ED with a chief complaint of chest pain or symptoms suggestive of ACS and had preexisting CKD, defined as a serum creatinine of more than 130 umol/l. Patients were excluded if it was a reattendance for the same episode of complaint, or if consent could not be obtained or was denied.
In addition to troponin T, which was routinely performed, copeptin, myeloperoxidase and pro-ADM assays were also performed on the blood samples obtained at presentation to the ED. The analytical methods were: (1) copeptin (BRAHMS, Hennigsdorf, Germany) with a detection limit 4.8 to 500 pmol/l and a 20% coefficient of variation (CV) of 12 pmol/l via automated immunofluorescence assays on the Brahms Kryptor analyser; (2) myeloperoxidase (Abbott Laboratories, IL, USA) with a detection limit 20.0 pmol/l to 10,000.0 pmol/l and a 20% CV 50.0 pmol/l via chemiluminescent microparticle immunoassay on the ARCHITECT i1000SR analyser; and (3) pro-ADM (BRAHMS) with a detection limit 0.05 to 10 nmol/l and a 20% CV of 0.25 nmol/l via automated immunofluorescence assays on the Brahms Kryptor analyser. Performance verification was not carried out by the hospital laboratory as these tests were brought in specifically for this study and are not available as routine tests performed in the hospital. Table 1 shows the reference ranges in healthy individuals and recommended cut-offs based on international data. The results of the biomarkers were collected for the sole purpose of analysis in this study and not made known to physicians caring for the patients or used in deciding their clinical management.
Reference ranges and recommended cut-offs of biomarkers.
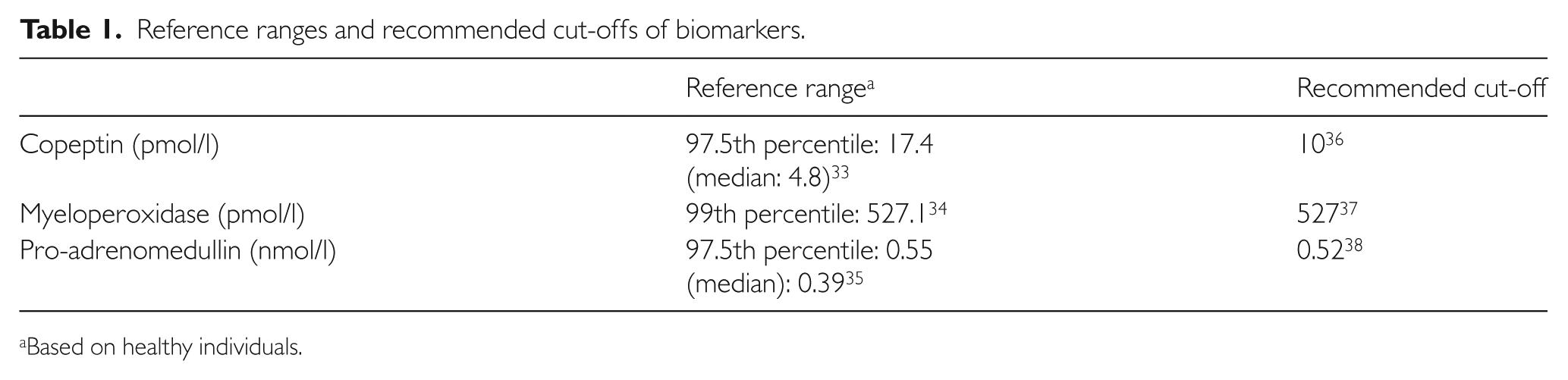
Based on healthy individuals.
All patients were subsequently followed up for the occurrence of ACE, a composite end-point defined as cardiac-related deaths, myocardial infarction and/or ventricular fibrillation, which looks primarily at safety as a clinical goal. 39 The occurrence of ACEs was traced by a trained research coordinator who either reviewed the patients’ case records, performed a phone follow-up or sent out letters if they could not be contacted by phone. If the patients remained unreachable, the registry of deaths was checked.
This study was approved by the institutional review board and funded by the Venerable Yen Pei-National Kidney Foundation Research Fund.
Statistical methods
Sample size was determined for a two-tailed
Statistical analysis was performed using IBM SPSS version 16. Comparisons of the distribution of concentrations of each cardiac marker between participants with ACE and those without were analysed with the Mann-Whitney
Results
Patient characteristics
A total of 724 patients were recruited, of whom 60.6% were male and 68.6% were Chinese. The median age was 67 years. Within our cohort, 373 (51.5%) patients had previous ischaemic heart disease, 639 (88.3%) patients had CKD stage 4 and 5, and 243 (33.5%) patients were on dialysis (Table 2). Copeptin, myeloperoxidase and pro-ADM assays were performed for 277 (38.3%), 688 (95.0%) and 587 (81.1%) patients, respectively.
Patient characteristics.
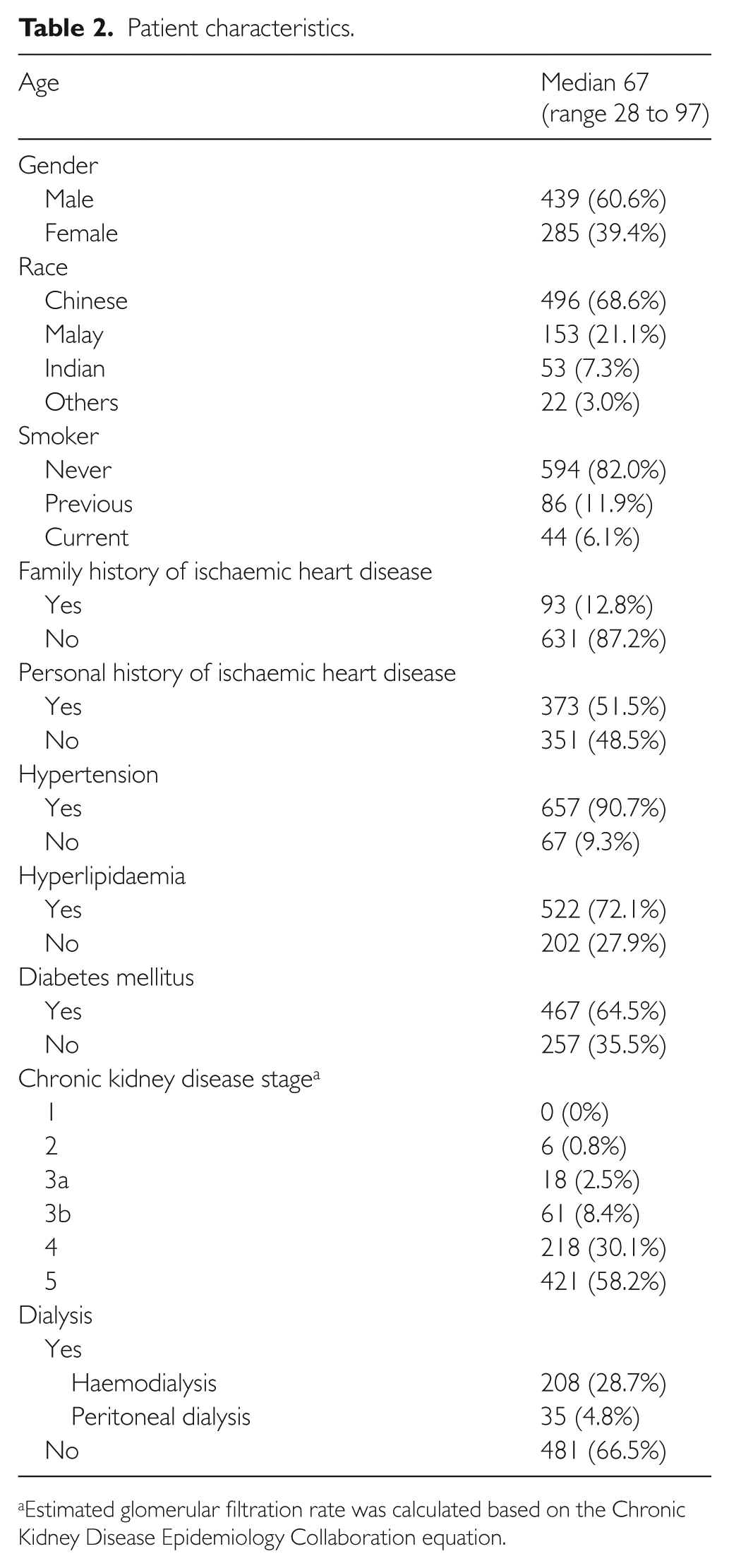
Estimated glomerular filtration rate was calculated based on the Chronic Kidney Disease Epidemiology Collaboration equation.
ACEs and all-cause death
The most common ACE was myocardial infarction at 14.5% and 20.7% at the 30-day and six-month time points, respectively, followed by cardiac-related death and ventricular fibrillation. The 30-day and six-month incidence of any ACE was 15.1% and 21.7%, respectively. The overall mortality rate was 5.9% at 30 days and 14.1% at six months, with cardiac-related death accounting for about 40% of all deaths (Tables 3 and 4).
Adverse cardiac events and all-cause death.

Occurrence of adverse cardiac events (ACEs) by cardiac marker.

Performance of cardiac markers
When analysed according to CKD stages, all three cardiac markers did not prognosticate for 30-day and six-month ACE, with areas under the curve (AUC) ranging from 0.4 to 0.6. Additionally, Figures 1 to 6 show that within each homogeneous stratum of CKD stage patients, the distribution of values were not different between the ACE and non-ACE groups for all three cardiac markers (
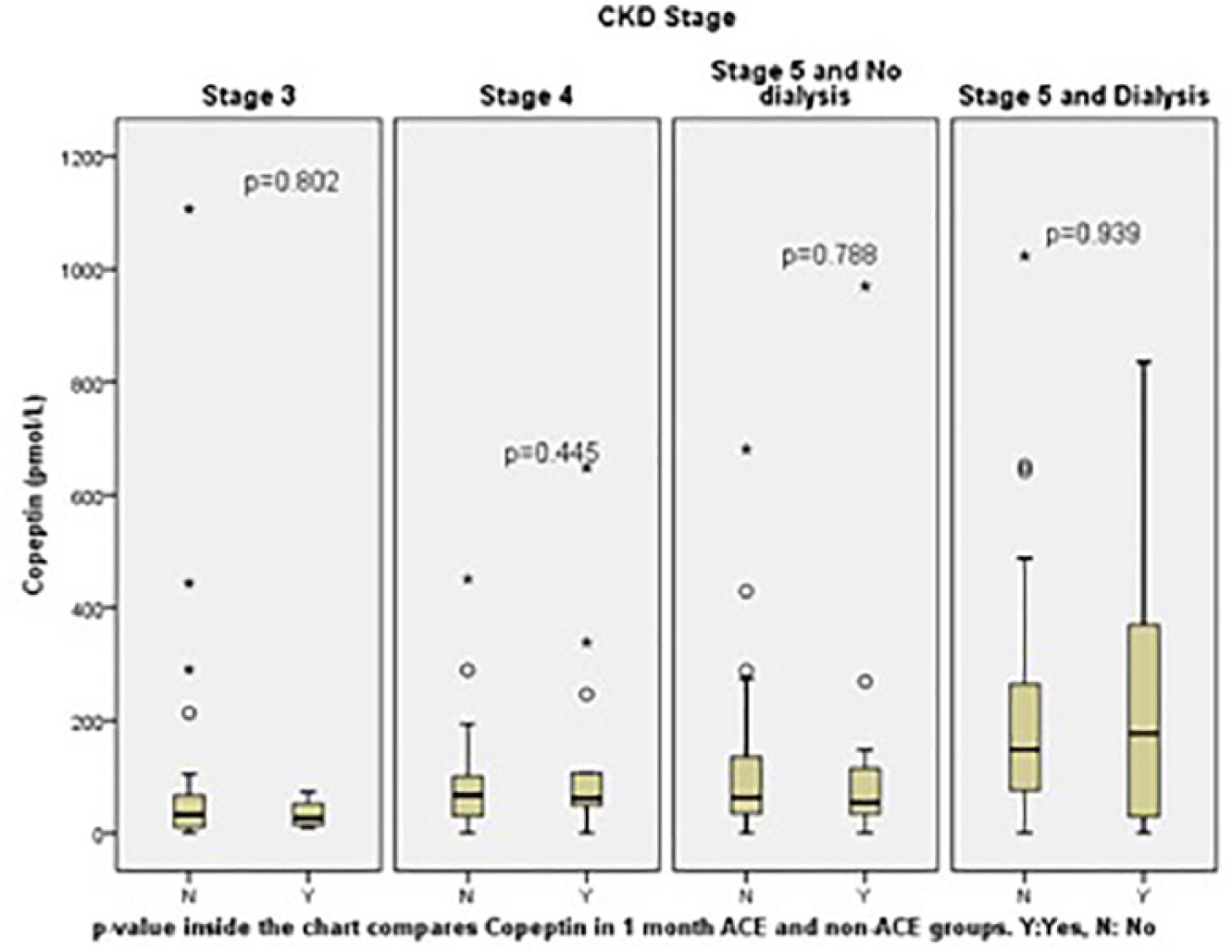
Copeptin level for 30-day adverse cardiac events (ACE) by chronic kidney disease (CKD) stage. Patients with stages 1 and 2 CKD are not presented as sample size was too small (

Copeptin level for six-month adverse cardiac events (ACE) by chronic kidney disease (CKD) stage. Patients with stages 1 and 2 CKD are not presented as sample size was too small (

Myeloperoxidase (MPO) level for 30-day adverse cardiac events (ACE) by chronic kidney disease (CKD) stage. Patients with stages 1 and 2 CKD are not presented as sample size was too small (

Myeloperoxidase (MPO) level for six-month adverse cardiac events (ACE) by chronic kidney disease (CKD) stage. Patients with stages 1 and 2 CKD are not presented as sample size was too small (

Pro-adrenomedullin (ADM) level for 30-day adverse cardiac events (ACE) by chronic kidney disease (CKD) stage. Patients with stages 1 and 2 CKD are not presented as sample size was too small (
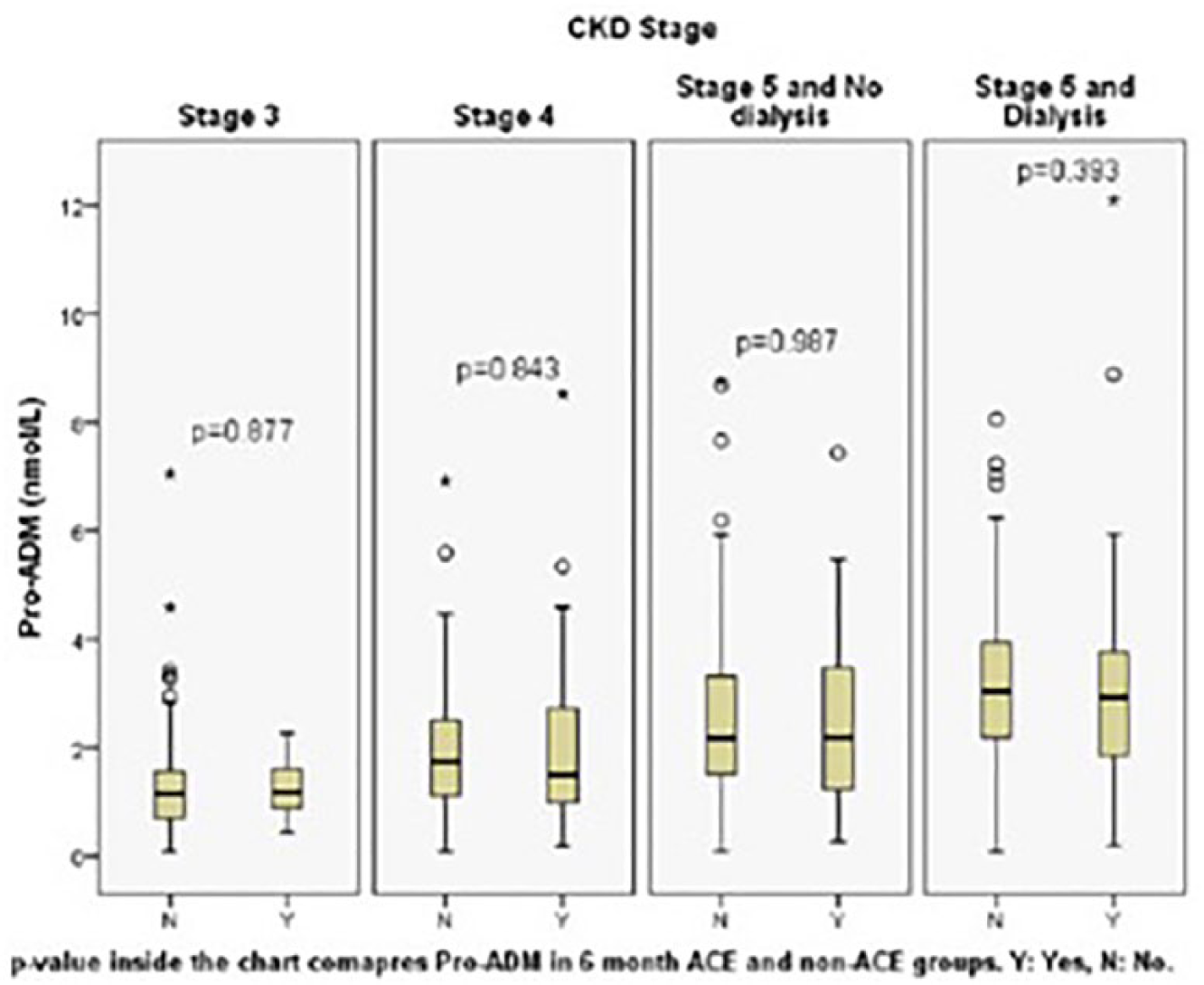
Pro-adrenomedullin (ADM) level for six-month adverse cardiac events (ACE) by chronic kidney disease (CKD) stage. Patients with stages 1 and 2 CKD are not presented as sample size was too small (
In contrast, when analysed according to CKD stages, troponin T was a good prognostic marker for 30-day and six-month ACE, with AUC ranging from 0.6 to 0.8. Figures 7 and 8 further show the distribution of values being different between ACE and non-ACE groups (

Troponin T level for 30-day adverse cardiac events (ACE) by chronic kidney disease (CKD) stage. Patients with stages 1 and 2 CKD are not presented as sample size was too small (

Troponin T level for six-month adverse cardiac events (ACE) by chronic kidney disease (CKD) stage. Patients with stages 1 and 2 CKD are not presented as sample size was too small (
Discussion
In patients with CKD, the prevalence of coronary artery disease is as high as 73%. 40 As this group of patients presenting with chest pain to the ED will have higher pretest likelihood for ACS, 41 clinicians need to be able to accurately diagnose and prognosticate these patients by further risk stratification to guide disposition from the ED and further therapy. However, like CKMB and cardiac troponins in patients with CKD, copeptin, myeloperoxidase and pro-ADM have not been found to be useful in prognosticating ACEs such as cardiac-related death, myocardial infarction and ventricular fibrillation in patients with CKD who present to the ED with chest pain or symptoms suggestive of ACS. Therefore, we do not recommend its routine use for this purpose as all three novel cardiac markers were not associated with 30-day or six-month ACE.
Copeptin is a marker of neurohormonal stress. The body responds to acute and life-threatening diseases by releasing AVP and copeptin, with levels reflecting the individual stress level of the body. 42 Copeptin has a longer half-life of days in contrast to the short half-life of 5–20 minutes for AVP, and is preferred for detection over AVP. 43 After a myocardial infarction, there is a rapid rise of copeptin levels to peak values before decline over the next two to five days. 16 Copeptin has been found to be useful in the early ruling out of myocardial infarction when used in combination with cardiac troponin, potentially negating the need for serial tests and observation.13,44 However, it is not specific to the cardiovascular system, and is elevated in a wide array of diseases, including sepsis, 45 chronic obstructive pulmonary disease, 46 lower respiratory tract infection, 47 stroke48,49 and traumatic brain injury. 50 Furthermore, specific to patients with CKD, levels are increased because of reduced GFR, leading to decreased clearance 51 and may vary because of changes in fluid status. 52 Therefore, it is not a useful biomarker in these patients.
The activation of neutrophils and macrophages leads to a rapid release of myeloperoxidase, which has a half-life between four and five hours. 53 Myeloperoxidase levels have been shown to be useful in patients with acute myocardial infarction, presenting less than two hours from onset of symptoms. 54 However, this has not been shown to be consistent, with other studies showing that myeloperoxidase is inferior to high-sensitivity cardiac troponin in diagnosing acute myocardial infarction even in patients presenting up to three hours from symptom onset. 55 Elevated levels of myeloperoxidase have also been shown to be prognostic of poorer outcomes among patients with ST-elevation myocardial infarction up to two years post-event. 56 Once again, myeloperoxidase can be elevated in any infective, inflammatory or infiltrative process, and is not limited to the cardiovascular system. Furthermore, patients with CKD are at an increased risk of developing oxidative stress because of metabolic disorders, immune deficiency and persistent inflammation. 57 The state of persistent inflammation is highly prevalent in this group of patients, 58 attributable to the inverse correlation between GFR and biomarkers of inflammation (interleukin-1β, interleukin-1 receptor antagonist, interleukin-6, tumour necrosis factor-α, C-reactive protein and fibrinogen). 59 All these factors contribute to the low utility of this biomarker in patients with CKD.
Pro-ADM, with a half-life of a few hours, peaks between 24 to 48 hours after onset of symptoms of myocardial infarction.60–62 While it has not been shown to have utility in the early diagnosis of myocardial infarction, it may be prognostic of adverse outcomes and all-cause mortality among patients presenting with acute chest pain.31,63 Elevated levels are seen in other cardiovascular conditions, including congestive heart failure64,65 and hypertension, 66 as well as noncardiovascular conditions such as systemic inflammatory states 67 and type 2 diabetes mellitus. 68 Once again, increased levels seen in patients with CKD69,70 further confound its interpretation in the setting of ACS, reducing its relevance for clinical use.
Levels of copeptin, myeloperoxidase and pro-ADM also vary across CKD stages. With progression of CKD, copeptin levels increase because of its decreased renal clearance. 71 Pro-ADM levels also increase with declining eGFR because of the increase levels of ADM, which is renoprotective and has important biological functions within the kidneys.72,73 On the other hand, myeloperoxidase levels decline steadily, possibly because of the inhibitory effect of uraemic toxins on the enzyme. 74 Despite accounting for these variations with renal function in our analyses, all three cardiac markers were not shown to be prognostic for ACE.
The cost of test kits for CKMB and troponins T and I are S$4, S$5 and S$12, respectively. In comparison, the cost of test kits for copeptin, myeloperoxidase and pro-ADM are S$56, S$5 and S$32, respectively. Given the higher cost and inability to prognosticate for ACEs, it would not be cost effective or appropriate for clinicians to use these three novel cardiac markers in patients with CKD.
Cardiovascular disease is the leading cause of morbidity and mortality in patients with CKD, accounting for more than 50% of all deaths, 75 with a two-year post-myocardial infarction mortality rate of up to 73%. 76 In our study, cardiac-related death accounted for about 40% of all deaths occurring at the 30-day and six-month time points. The lower mortality rate could be accounted for by the inclusion criteria of our study population: patients with CKD presenting to the ED with chest pain or symptoms suggestive of angina. Only a subset of these patients eventually had acute myocardial infarction, resulting in a lower mortality rate among our patients. The lower proportion of cardiac-related death in our study also bears testimony to the advances in medical therapy. With developments in diagnostics and therapeutics, patients with cardiac and renal diseases now have improving outcomes and prolonged survival. Lastly, the inclusion of patients with early stages of CKD without dialysis may have resulted in lower all-cause and cardiac-related mortalities in our study population.
Limitations
The number of tests performed for each cardiac marker was lower than the number of patients recruited because of the unavailability of test kits and laboratory personnel throughout the day. However, this did not affect the power of our study as the target sample size for each cardiac marker was adequately met.
Variability of levels with onset of symptoms
In our study, time of onset of symptoms and blood sampling was not accounted for. However, the stability of the biomarkers and identical treatment of all samples should minimise effects on the validity of results.
Single testing vs serial testing
Serial testing of cardiac markers, over a period of several hours, has been reported to improve diagnostic accuracy. For instance, an increase in troponin T of as little as 0.10 ug/l over a period of several hours has an increased likelihood of ACEs. 77 Our study used a single testing at presentation to the ED as there was no previous study to prove the utility of copeptin, myeloperoxidase and pro-ADM in patients with CKD. We did not proceed to carry out serial testing of these novel cardiac markers as their role as cardiac markers were not clear. Further studies may be performed to establish the relation of the variability of the novel cardiac markers to myocardial injury, as this may allow us to determine whether changes in the novel cardiac marker levels over time are significant in helping us risk-stratify patients.
In conclusion, in patients with CKD presenting to the ED with symptoms suggestive of angina, the performance of single measurements of copeptin, myeloperoxidase and pro-ADM as novel markers to prognosticate 30-day and six-month ACE was poor. This observation was largely due to the lack of specificity as elevated levels could be due to other causes in patients with CKD. Further studies should be conducted before determining their utility in clinical practice to assist the clinician in risk stratification, resource allocation and disposition planning.
Footnotes
Authors’ contributions
Jen Heng Pek carried out data gathering, statistical analysis and paper writing.
Stephanie Fook-Chong provided input for statistical analysis and paper writing.
Jason Chon Jun Choo provided input on the research idea and protocol.
Carol Hui Chen Tan was involved in carrying out the research.
Ziwei Lin carried out the research protocol, data gathering and paper writing.
Choong Meng Chan provided input on the research idea and protocol.
Chin Pin Yeo was involved in carrying out the research protocol.
Swee Han Lim generated the research idea and provided guidance on the research protocol.
Availability of data and materials
The datasets generated and/or analysed during the current study are available from corresponding author.
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical approval
Ethical approval was granted by the institutional review board at SingHealth, Singapore.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Venerable Yen Pei-National Kidney Foundation Research Fund.
Informed consent
Written informed consent was obtained from the patients for their anonymised information to be published in this article.
