Abstract
Background:
Patients with chronic liver disease often suffer from poor quality of life. The Chronic Liver Disease Questionnaire (CLDQ) is a validated tool to assess health-related quality of life in these patients. It has been translated and validated for use in many countries and languages globally.
Objectives:
We aimed to translate Mainland Chinese Mandarin to Singapore Mandarin and perform cross-cultural adaption of CLDQ for the Mandarin-speaking population in Singapore (CLDQ-SG).
Methods:
This study was conducted based on the International Society for Pharmacoeconomics and Outcomes Research Principles of Good Practice. The study consisted of two parts: part one involved cognitive debriefing and cultural adaption of CLDQ, and part two was a pilot study on the first version of CLDQ-SG among adult patients with chronic liver disease in a tertiary hospital.
Results:
During the cognitive debriefing process of part one, questions beginning with “recent” (最近) two weeks in Mandarin were changed to “last” (在过去) two weeks. Eighteen patients were recruited for part two of the study (50% male, mean age 49±13 years). Time taken to complete CLDQ-SG was 15±8 minutes, and the mean score was 5.1±0.5. The reliability of measurements for all domains was good, with an intra-class correlation coefficient ≥0.8. Items one and four needed further restructuring. There were no discrepancies between CLDQ and CLDQ-SG.
Conclusion:
This study showed that CLDQ-SG was culturally acceptable by the Mandarin-speaking population in Singapore. There were only two items that needed revision in the finalized CLDQ-SG.
Keywords
Introduction
Worldwide, chronic liver disease (CLD) is increasingly a major health burden to society. It is estimated to be the fifth most common cause of death after heart disease, stroke, lung disease and cancer. 1 A similar trend is seen in Singapore, where liver cancer is the third and fourth most common cause of cancer deaths in our male and female population. 2 Locally, the most common cause of liver cirrhosis is chronic hepatitis B, followed by alcohol and cryptogenic liver cirrhosis. 3 The majority of patients with decompensated cirrhosis develop ascites, and this is associated with poorer prognosis. These patients are also at risk of developing hepatocellular carcinoma, requiring frequent hospital attendance for surveillance. 4
Fatigue and psychological conditions are often associated with CLD. Studies have shown that fatigue and psychological disturbances in patients with chronic hepatitis C are more prevalent than those with other chronic diseases.5,6 The consequences of cirrhosis and other CLDs negatively impact all aspects of wellbeing and health-related quality of life (HRQOL), especially those with ascites and lower limb edema.7-9
The chronic liver disease questionnaire (CLDQ) is a disease-specific HRQOL instrument designed for patients with CLD, regardless of etiology or severity of disease. 10 It contains 29 items divided into six domains: “fatigue,” “emotional function,” “worry,” “activity,” “abdominal symptoms” and “systemic symptoms.” All items in the questionnaire are pertinent to symptoms experienced during the last two weeks on a seven-point Likert scale of 1–7, indicating none to maximum frequency. Domain scores are the means of the items and overall score is the mean of all domains (Figure 1).

Original CLDQ in English.
CLDQ is easy to administer with good correlation to disease severity. It has been cross-culturally adapted and validated into several different languages,11-15 including Mandarin for Chinese in Southern China. This pilot study seeks to adapt and translate the Mainland Chinese Mandarin CLDQ (CLDQ-MC) to Singapore Mandarin (CLDQ-SG) through the process of cognitive debriefing so as the reduce respondent burden and improve reliability of the tool for a proper validation study. The actual validation study has since been completed and will be reported separately.
Methods
There were two parts to this study; the first was the adaption process of CLDQ-MC to CLDQ-SG and the second was a pilot test of CLDQ-SG in patients. Written informed consent was obtained from all study participants in part two of the study.
Part one
We used the International Society for Pharmacoeconomics and Outcomes Research Principles of Good Practice as a guide in the translation process. 16 The cross-cultural adaption process for patient-reported outcomes measures consists of (1) preparation for translation; (2) forward translation; (3) reconciliation; (4) back translation; (5) harmonization; and (6) cognitive debriefing.
Prior to translation and conducting the pilot study, we obtained permission to adapt and translate CLDQ from Yonoussi et al. 10 and Zhou et al., 15 the original authors of the English and Mandarin versions of the CLDQ, respectively. Forward translation from CLDQ-MC to CLDQ-SG was performed independently by two local bilingual professionals (a Mandarin language teacher and a Singaporean Chinese nurse). The translated CLDQ-SG was compared with the original English CLDQ during the reconciliation phase to ensure that word meanings were consistent with the original English CLDQ. During the back-translation phase, CLDQ-SG was independently translated back into English by another bilingual Singaporean Chinese teacher. Finally, in the harmonization stage both CLDQ-SG and CLDQ-MC were compared with the original CLDQ in order to arrive at the first consensual version of CLDQ-SG.
The first consensual version of CLDQ-SG underwent cognitive debriefing in a small group of medical and non-medical personnel. They were asked to review the CLDQ-SG questionnaire word by word to check if they understood the meaning of each question and whether the choice of words was culturally appropriate. They were also asked to identify any inappropriate or ambiguous expressions and the comprehensiveness of the instructions. Our decision rule was: if more than 25% of participants found an item to be problematic, i.e. difficult to understand or meaning is ambiguous, the problematic word will be changed according to their recommendations. All their suggestions were recorded and changes were made before conducting the pilot test of CLDQ-SG.
Part two
The pilot test was conducted in the Gastroenterology outpatient clinics of Singapore General Hospital on adult patients with CLD who were at the clinic from May 2014 to July 2015. Inclusion criteria were Mandarin-speaking adults ≥21 years old, diagnosed with any CLD and ability to read both English and Mandarin. Exclusion criteria were patients with active hepatic encephalopathy or cognitive impairment, extrahepatic organ failures, active malignancy or psychiatric diseases. Patients were encouraged to give feedback on any difficulty completing the questionnaire and to provide any recommendation to the choice of words or phrases. The decision rule for part two of the study was: if the same question is identified as problematic, i.e. difficult to understand by at least 50% of the participants, CLDQ-SG will be revised and re-piloted. During the pilot test, patients had to complete both the original CLDQ and CLDQ-SG consecutively. In order to prevent participants from remembering the order of answers, all items in the original CLDQ were rearranged in a random manner.
Results
Cognitive debriefing, part one
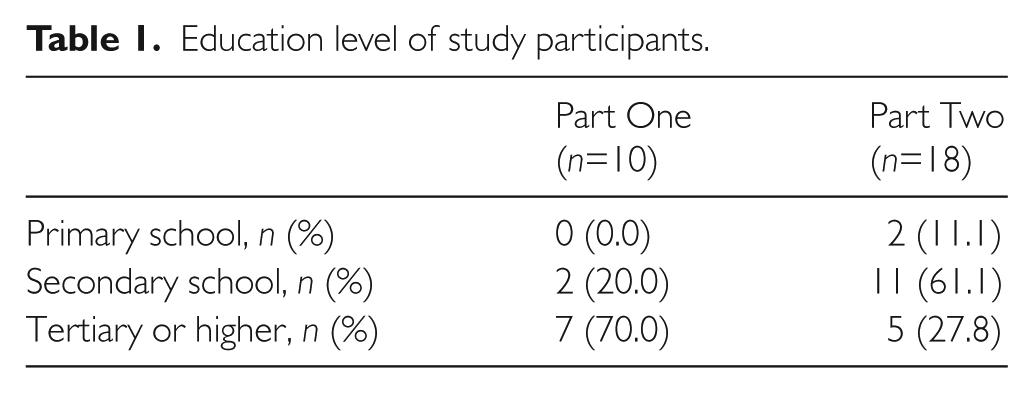
Ten Singaporean Chinese medical and non-medical personnel (age: 25–50 years; three males) were asked to rate the equivalence between the first consensual version and the original CLDQ. Of these participants, 70% had tertiary education (Table 1). From the cognitive debriefing, three changes were recommended. First, all participants agreed that the word “recent” (最近) two weeks in CLDQ-MC had to be changed to the “last” (在过去) two weeks, which has the same literal translation to the original CLDQ. Second, it was recommended that a comma (,) be inserted before all sentences that began with “During the last two weeks…” (在过去的十四天里). The insertion of comma (,) was deemed necessary to break down the long sentences so that it would be easier for the lay person to understand. Finally, the phrases “how much of the time” and “how often during” which meant either “有多少时间 or 时间有多少” in Mandarin were recommended as this better described the questions. Therefore, to describe frequency in Mandarin that is commonly understood by the Chinese population in Singapore, this was changed to: “频率有多高 and 次数有多少.”
Education level of study participants.
Part two
In total, 18 patients were recruited for this study. Half of the study participants were male and mean age was 49±13 years. Almost 90% of study participants had at least secondary level education, with the remaining participants having completed primary level education only (Table 1). The questionnaire was self-administered but study participants were allowed ask for clarification if they were unsure of the items. The majority of the study participants had chronic viral hepatitis (44.4%), and the remaining had non-cirrhotic portal hypertension, autoimmune hepatitis, primary biliary cholangitis, Wilson’s disease and cryptogenic liver cirrhosis. Time taken to complete CLDQ-SG was 15±8 minutes. The reliability of measurements for all domains was good, with intra-class correlation coefficient (ICC) ≥0.8 (Table 2). Cronbach’s alpha of the CLDQ-SG scale was 1 (Table 3).
Comparison of intra-class of original CLDQ vs. CLDQ-SG by domains.

CLDQ: Chronic Liver Disease Questionnaire; SG: Singapore.
Scores and internal consistency of various domains and scales of CLDQ-SG and CLDQ.

α: Cronbach’s alpha; CLDQ: Chronic Liver Disease Questionnaire; min: minimum; max: maximum; SD: standard deviation; SG: Singapore.
Some 22.4% of study participants identified items one and four of CLDQ-SG as potentially difficult to understand. Participants commented that “困扰” in item one and “困乏” in item four were not commonly used in Singapore. Instead, “烦恼” and “困” were suggested as replacements in items one and four, respectively (Table 4). None of the questions were offensive and there were no major discrepancies in the answers between English CLDQ and CLDQ-SG questionnaires. All the recommended changes from the pilot were reviewed and changes made to the final version of the CLDQ-SG (Figure 2).
Revision of the CLDQ-SG sentence structure following Part Two of study.

CLDQ: Chronic Liver Disease Questionnaire; MC: Mainland China; SG: Singapore.

Final version of the CLDQ-SG after Part Two of study.
Discussion
CLDQ is a validated questionnaire, specifically assessing HRQOL of patients with CLD. This study was undertaken to adapt the CLDQ questionnaire validated in China for use in Mandarin-speaking Singaporeans. There have been several studies evaluating survival and prognosis of patients with CLD in Singapore3,17 but none evaluating HRQOL. This study reports the process of adapting the CLDQ-SG questionnaire for future studies.
The cognitive debriefing aspect of this study serves to elicit cognitive response from study participants on their understanding, appropriateness and complexity of the questionnaire. Studies have shown that incomplete or incorrect self-administration of HRQOL questionnaires is often due to lack of patient understanding.18,19 This may be influenced by psychosocial factors and education levels. During the adaption process, it was found that the Mandarin word “recent” used in Mainland China was not commonly used in Singapore. Therefore, the word “recent” was changed to “last” which had the same literal meaning as the word used by Younossi et al. 10 This study emphasized the importance of local adaption of the CLDQ as Singapore Mandarin differs from China Mandarin. 20 In fact, several versions of the Mandarin CLDQ have been adapted locally for use in different provinces in China because of differences in language and culture,15,21,22 but none is suitable for use in Singapore.
During the pilot, the format and instructions were found to be acceptable to patients. It took them 15 minutes to complete the questionnaire, which was similar to another CLDQ validation study among Serbian patients. 23 ICC for all domains was ≥0.8 in CLDQ-SG and this exceeds the minimum acceptable value of an ICC >0.75. 24 However, there are several limitations to our study. Firstly, the number of study participants in this study was very small. This was only an initial pilot study with the aim of translation and cultural adaption, focusing on the process of cognitive debriefing. Secondly, Part Two of this study required study participants who were bilingual, thereby potentially introducing selection bias for participants who were more highly educated, although the majority of our study participants had only secondary level education. However, the validity of CLDQ-SG for assessment of HRQOL in the Mandarin-speaking Chinese population with CLD in Singapore will need to be separately assessed in a larger study.
Conclusion
The CLDQ-SG is culturally acceptable by our local Chinese population. A few modifications were necessary to suit the Singaporean Chinese population. CLDQ-SG has been validated in another larger study and will be reported separately.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Availability of data and materials
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Authors’ contributions
Hiang Keat Tan: data analysis, drafting of manuscript and formulation of research idea; Yean Lee: data collection, formulation of research idea; Pei Yuh Chia, Shaik Hussain Nurshifa and Travis Kim Chye Tan: data collection and review of manuscript; Stephanie Fook-Chong: advanced statistical analysis and critical review of manuscript, Pik Eu Chang: drafting of manuscript, critical review of manuscript and formulation of research idea; Aloweni Fazila: formulation of research idea/protocol, supervision of data extraction/analysis, writing of manuscript.
All authors read and approved the manuscript.
Conflict of Interest
The authors declare that there is no conflict of interest.
Informed consent
Informed consent was obtained from all study participants from Part Two of study
Ethical approval
This study was approved by Singhealth Centralised Institutional Review Board (reference number: 2014/359/A)
