Abstract
Keywords
Introduction
Primary anti-phospholipid syndrome (APLS) is a rare autoimmune thrombophilia that can present with cerebrovascular disease. Hypercoagulability resulting in vascular thrombosis is the primary pathogenic mechanism leading to the most common cerebrovascular manifestation, ischemic stroke. 1 Intracranial vessel wall MRI (VWI) is useful for distinguishing vasculopathies. 2 Little is known about the intracranial VWI features of patients with APLS-related cerebrovascular events. We report VWI findings of a woman with cerebral infarcts and subarachnoid hemorrhage due to Moyamoya syndrome (MMS) attributable to primary APLS.
Case description
A 37-year-old woman presented with acute dizziness and left hemiparesis. A day prior, she noted a transient sensation of falling to the left. The next morning, she noticed decreased strength in her left arm after which she presented to the emergency department. On physical exam, she had a left facial droop and mild left hemiparesis. She had no significant past medical history and was a nonsmoker.
Laboratory testing showed triple positivity for anti-phospholipid antibodies (APLAs) with an elevated dilute Russell viper venom time ratio (DRVVT) to 1.5, elevated beta-2 glycoprotein IgM (36U/mL; normal 0–12.9U/mL), and elevated anti-cardiolipin IgM (15U/mL; normal 0–12U/mL) titers, with the upper limit of the reference range in the testing laboratory being the 99th percentile. Partial thromboplastin time was elevated to 57.7 s (normal 25.1–36.5 s). The rapid plasma reagin (RPR) was positive with a negative treponemal antibody test, consistent with a false-positive RPR. Her low-density lipoprotein was 136 mg/dL and lipoprotein(a) was 46 mg/dL. Serum Lyme and HIV were negative. Erythrocyte sedimentation rate was 41 mm/hr with negative anti-nuclear antibody, anti-neutrophil cytoplasmic antibodies, rheumatoid factor, and anti-Ro/anti-La. A lumbar puncture was unremarkable with negative varicella zoster IgM/IgG.
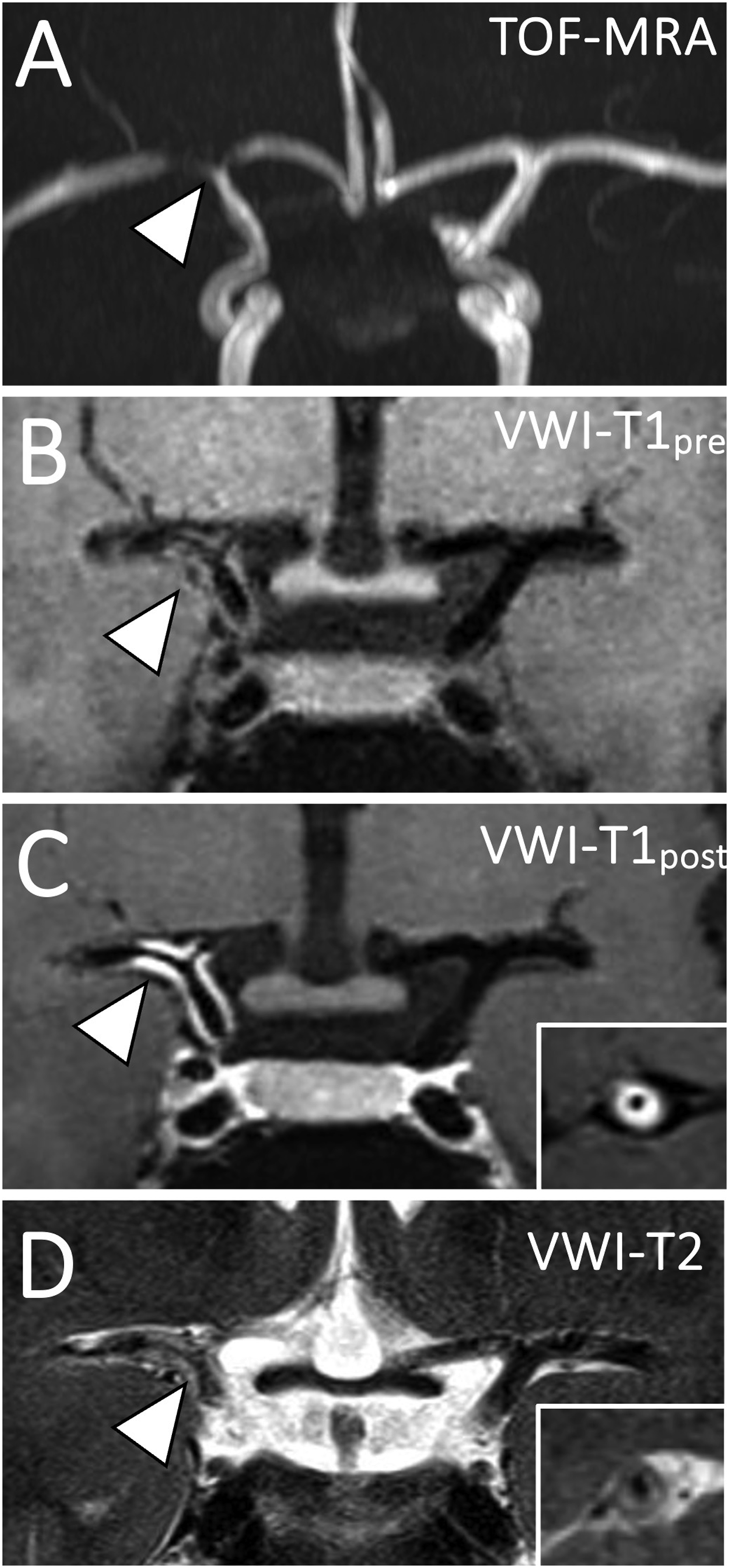
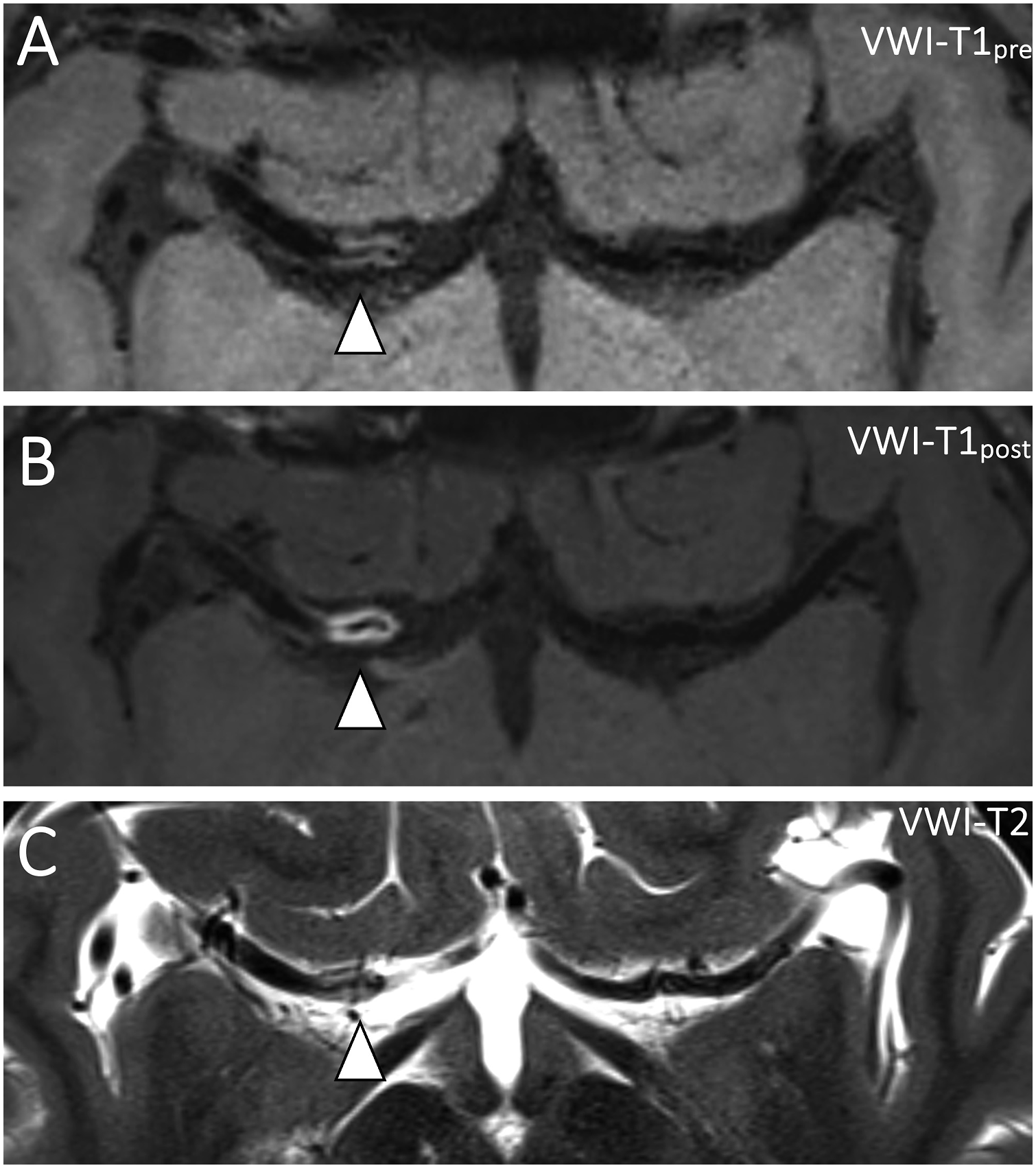
Head CT showed an acute right basal ganglia infarction and right cerebral convexity subarachnoid hemorrhage. CT angiogram showed severe narrowing of the right internal carotid artery (ICA) terminus and proximal middle cerebral artery (MCA) and anterior cerebral artery (ACA). MRI confirmed acute ischemia on diffusion-weighted imaging of the right basal ganglia (Figure 1(a)). Postcontrast fluid-attenuated inversion recovery (FLAIR) imaging showed sulcal FLAIR hyperintense subarachnoid hemorrhage (Figure 1(b)) and a characteristic “ivy sign” of slow flow in the ipsilateral cortical vessels (Figure 1(c)). Postcontrast FLAIR imaging also showed vessel wall edema of the stenotic right ICA/MCA/ACA vessels (Figure 1(d)). Flow-limiting stenosis on time-of-flight MRA images of the ICA terminus (Figure 2(a)) was present. Intracranial VWI showed contiguous long-segment concentric vessel wall thickening with homogeneous signal on precontrast T1w-VWI (Figures 2(b) and 3(a)) and homogeneous, concentric vessel wall enhancement on postcontrast-VWI (Figures 2(c) and 3(b)). High-resolution T2-weighted VWI showed concentric, homogeneous T2 hyperintensity of the vessel walls, suggestive of wall edema (Figures 2(d) and 3(c)). No intraluminal signal or filling defect was present to suggest an intraluminal subocclusive thrombus. The juxtaluminal surface of the stenotic vessel walls was smooth without surface irregularity. Susceptibility weighted imaging showed no linear hypointense signal corresponding to the stenotic vessel to suggest thrombus. MRI brain in stroke evaluation. (a) Diffusion-weighted imaging shows acute ischemia in the right basal ganglia (arrowhead). (b) Axial postcontrast fluid-attenuated inversion recovery (FLAIR) sequence shows sulcal FLAIR hyperintensity along the right frontoparietal convexities (arrowheads) and (c) FLAIR hyperintense slow flow in right temporo-occipital cortical veins (“ivy sign”; arrowheads). (d) In addition to right temporo-occipital subarachnoid hemorrhage (arrowheads), FLAIR hyperintensity of the vessel walls of the stenotic proximal right middle cerebral artery and internal carotid artery terminus (arrow) are also noted. Coronal T1- and T2-weighted vessel wall MR imaging of APLS-associated vasculopathy. (a) Coronal time-of-flight magnetic resonance angiography (TOF MRA) shows flow-limiting stenosis of the right internal carotid artery terminus and proximal middle and anterior cerebral arteries. (b) Coronal pre- and (c) postcontrast T1-weighted vessel wall MR imaging shows avid, homogeneous, and smooth concentric vessel wall enhancement of the stenotic segments (arrowhead). Inset shows right middle cerebral artery in cross-section with concentric vessel wall thickening and enhancement but patent lumen. (d) High-resolution vessel wall T2-weighted imaging shows concentric T2 hyperintensity of the vessel walls of the stenotic cerebral arteries (arrowhead). Inset shows donut-like homogeneous T2w hyperintensity of the middle cerebral artery vessel wall in cross-section. Axial T1 and T2-weighted vessel wall MR imaging of APLS-associated vasculopathy. (a) Axial pre- and (b) postcontrast T1-weighted vessel wall MR imaging shows homogeneous and smooth concentric vessel wall enhancement of the visualized internal carotid artery terminus and proximal M1 middle cerebral artery (arrowhead). (c) Axial high-resolution vessel wall T2-weighted imaging shows edematous changes based on T2 hyperintensity of the vessel walls (arrowhead).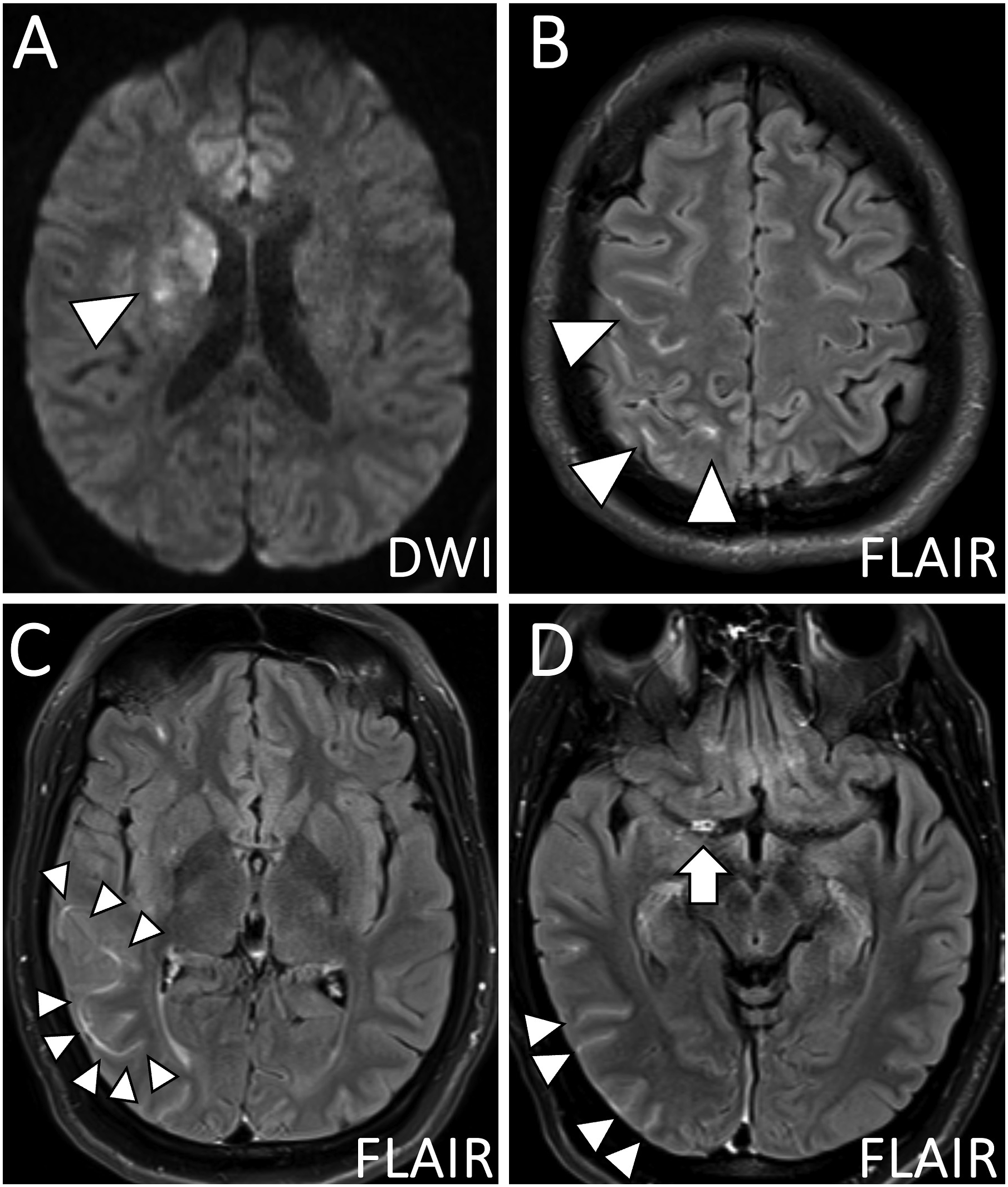
The rest of the intracranial arteries appeared normal without intracranial or cervical atherosclerosis. A cerebral digital subtraction angiography showed severe stenosis of the right ICA terminus, collateral vessel formation from the right posterior cerebral artery to the right basal temporal lobe, and lenticulostriate hypertrophy, suggestive of early Moyamoya vasculopathy. Transthoracic echocardiogram showed no valvular disease, a mildly reduced ejection fraction to 45%, and global hypokinesis, which resolved 2 days later upon imaging with cardiac MRI.
Consistent with the accepted diagnostic criteria for APLS, 3 repeat laboratory testing 3 months later showed persistently elevated beta-2 glycoprotein IgM antibodies and positive lupus anticoagulant, confirming a diagnosis of primary APLS presenting with MMS. She was started on enoxaparin, bridged to warfarin, and discharged to an inpatient rehabilitation facility for 2 weeks. One week later, she underwent a right superficial temporal artery to MCA bypass with dural inversion. At 8-week follow-up post-surgery, she had no additional cerebrovascular events. She endorsed greatly improved strength in the left hand and demonstrated only minimal left upper extremity and facial weakness on exam.
Discussion
Primary APLS is an autoimmune thrombophilia that can present with cerebrovascular pathology. Ischemic stroke is the most commonly reported CNS manifestation with stroke occurring in 19.8% of patients in a cohort of 1000 patients. 1 Other reported cerebrovascular manifestations include cerebral vein thrombosis/venous infarctions, 1 white matter T2 hyperintensities, 4 cortical atrophy, 4 arterial dissections, 5 and Moyamoya syndrome. 6 Rare associations between APLS and reversible cerebral vasoconstriction syndrome have also been reported and may be due to activated endothelial cells causing vasoconstrictor release.7,8
The most common mechanism of APLS-associated stroke is thrombotic occlusion of major arteries/arterioles or venules in the brain. APLAs disrupt coagulation inhibitors and activate immune cells to create a proinflammatory and hypercoagulable state. 9 APLA may also cause vessel occlusion through a proliferative vasculopathy, characterized by smooth muscle cell proliferation of the medial and intimal layers resulting in progressive concentric stenosis.9,10 The proliferative mechanism is poorly understood but may be a distinct cellular mechanism from both thrombosis and vasculitis. 10 As shown in our case, it may present as long segmental vascular stenosis in young patients without evidence of atherosclerosis or systemic inflammation.10,11
Little is known about VWI manifestations of APLS-associated vasculopathy. The largest case series with VWI findings included six patients with APLS vasculopathy and cerebral ischemia. Two patients showed concentric vessel wall thickening/enhancement of the ICA, two patients showed nonenhancing concentric vessel wall thickening of the MCA and ICA, one patient showed eccentric vessel wall thickening/enhancement of the ICA, and one patient showed eccentric thickening but concentric enhancement of the basilar artery. 11 Our case showed steno-occlusive disease of the right ICA terminus in a characteristic Moyamoya pattern with long-segment concentric smooth vessel wall thickening and homogeneous vessel wall enhancement. Our VWI exam also included high-resolution T2w VWI, which showed concentric T2w hyperintense wall edema expanding the vessel walls. These findings favor a focal inflammatory vasculopathy.
In light of the patient’s confirmed laboratory diagnosis of APLS, we favor the VWI findings to represent APLS-associated proliferative vasculopathy resulting in MMS.10,11 A thromboembolic complication of APLS resulting in a subocclusive thrombus in the ICA terminus was felt to be less likely in the (1) absence of an intraluminal filling defect on VWI, (2) lack of heterogeneous precontrast T1w or postcontrast vessel wall signal, and (3) lack of corresponding hypointense susceptibility weighted imaging signal.12–15 Furthermore, the patient did not have any other systemic thromboembolic complications.
Given the affected vascular anatomy of the ICA terminus, idiopathic Moyamoya disease (MMD) could also be considered. However, many studies show a strong association of negative wall remodeling on VWI in patients with idiopathic MMD, characterized by a decreased outer vessel wall diameter of the terminal ICA from thinning of the tunica media.16–19 In contrast, our case showed outward concentric expansion of the vessel wall likely due to inflammation and edema as seen on the postcontrast T1w-VWI and T2w acquisitions, respectively.
Other diagnostic considerations include atherosclerosis or primary angiitis of the central nervous system (PACNS). However, VWI features of atherosclerosis include eccentric vessel wall thickening with enhancement, intrinsic T1 hyperintensity, or luminal surface irregularity,20,21 all of which were absent in our case. The patient’s age, lack of cardiovascular risk factors, and absence of atherosclerosis in other vascular beds also suggested intracranial atherosclerosis was less likely. PACNS was not favored due to both clinical and imaging features, namely, a normal cerebrospinal fluid analysis, paucity of other vessel involvement, no white matter FLAIR hyperintensities, and lack of disseminated acute or chronic infarcts. 22
The standard treatment for initial thrombosis and secondary prophylaxis in APLS is warfarin or another vitamin K antagonist.23,24 Higher rates of recurrent thrombosis occur when using direct oral anticoagulants. 23 To establish reperfusion in patients with MMS and ischemic stroke, surgical revascularization is frequently used. 25 Cases of MMS due to APLS require careful consideration of anticoagulation decisions given the risk of hemorrhage from collateral vessel formation. 25 Wang et al., report five cases of APLS-related MMS, among which two were successfully treated with antiplatelet drugs, one died of cerebral ischemia despite surgery and antiplatelet therapy, and two died of hemorrhage following either dual antiplatelet or anticoagulation therapy. 6 In this case, a superficial temporal artery-MCA bypass was performed with a plan to continue therapeutic anticoagulation indefinitely. Periodic surveillance imaging will be done to monitor for cerebrovascular events and involvement of vasculature in the contralateral hemisphere.
In summary, we report the VWI imaging findings of a case of Moyamoya syndrome due to APLS vasculopathy. VWI can be a useful adjunct to guide the work-up of possible APLS-associated stroke.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
