Abstract
Purpose
After stent-assisted treatment for intracranial diseases, three-dimensional time-of-flight magnetic resonance angiography is a noninvasive follow-up method, but susceptibility artifacts prevent accurate evaluations of stented arteries. Sampling perfection with application-optimized contrast using different flip angle evolution (SPACE) sequence often used for vessel wall imaging is less susceptible to susceptibility artifacts, since it is a spin-echo sequence. Hence, we evaluated the feasibility of black-blood magnetic resonance angiography generated from vessel wall imaging data obtained using the SPACE sequence in the depiction of stented arteries by comparing with three-dimensional time-of-flight magnetic resonance angiography and digital subtraction angiography.
Methods
Our study group comprised 11 consecutive patients. For both three-dimensional time-of-flight magnetic resonance angiography and black-blood magnetic resonance angiography, the contrast ratio obtained from the stented artery and the normal artery proximal to the stent were calculated. And the depiction of stented arteries was visually evaluated. Additionally, the relative diameter index obtained from the stented artery and the normal artery proximal to the stent were calculated for three-dimensional time-of-flight magnetic resonance angiography, black-blood magnetic resonance angiography and digital subtraction angiography.
Results
The contrast ratio of the stented artery was significantly lower than that of the normal artery on three-dimensional time-of-flight magnetic resonance angiography, but no significant difference was seen using black-blood magnetic resonance angiography. Regarding both the diameter index and the visual assessment score, black-blood magnetic resonance angiography was significantly better than three-dimensional time-of-flight magnetic resonance angiography. On black-blood magnetic resonance angiography, the diameter index was equal to that of digital subtraction angiography, and the flow signal was homogeneous and continuous in most the cases.
Keywords
Introduction
Stent-assisted treatment for intracranial diseases is frequently employed for the transcatheter embolization of wide-necked aneurysms using the neck-remodeling technique1,2 as well as for that of the anatomically difficult aneurysm. 3 Although long-term benefits have been denied, stenting may still be beneficial in some patients with severe intracranial artery stenosis in combination with percutaneous transluminal angioplasty (PTA).4,5 Stents can cause endothelial hyperplasia or thrombosis.6–8 The gold standard for assessment stented intracranial arteries is digital subtraction angiography (DSA). But DSA is invasive, and there is a risk of adverse events due to the administration of contrast materials. On the other hand, three-dimensional time-of-flight magnetic resonance angiography (3D-TOF MRA) is non-invasive and widely used for screening of intracranial arterial diseases. However, the clinical value of 3D-TOF MRA is limited because of artifacts, including susceptibility artifacts and turbulent or laminar flows.9–11 Meanwhile, for intracranial vessel wall imaging (VWI), we have used the sequence of sampling perfection with application-optimized contrast using different flip angle evolution (SPACE; Siemens Healthineers, Erlangen, Germany), which is based on a fast spin-echo technique with variable flip angle refocusing pulses. As the VWI generated by the SPACE is spin-echo-based, it can produce images less affected by susceptibility artifacts causing reduced signal losses.12,13 A previous study of VWI using the same method reported that an in-stent thrombus could be visualized. 14 However, there is no report of quantitative evaluation of the accuracy of stented arteries between 3D-TOF MRA and VWI. Therefore, in the present study, we compared the depiction of stented arteries between black-blood MRA (BB-MRA) generated from VWI data obtained using the SPACE sequence and conventional 3D-TOF MRA as well as DSA.
Materials and methods
Patients
Our study group comprised 11 consecutive patients, including three men and eight women. Patient age ranged from 56–77 years with a mean of 68.8 years. All the patients underwent 3D-TOF MRA and VWI in one magnetic resonance (MR) examination session after undergoing stent-assisted treatment between January 2018–May 2019. The interval between DSA and MR imaging was a minimum of 1 day, a maximum of 10 days, and an average of 2.8 days. Ten patients underwent stent-assisted coil embolization for a cerebral aneurysm. The locations of the cerebral aneurysms were as follows: intracranial artery (ICA), 5; middle cerebral artery (MCA), 1; anterior communicating artery (A-com), 1; vertebral artery (VA), 2; and basilar artery (BA), 1. The remaining patient underwent PTA and stenting for symptomatic MCA stenosis. The stents used for the treatments were as follows: Neuroform Atlas (Stryker Neurovascular Fremont, California, USA), 4; LVIS (MicroVention, California, USA), 5; LVIS Jr (MicroVention, California, USA), 1; and Enterprise (Codman and Shurtleff, Miami, USA), 1. This study is a retrospective study using MR images that were obtained in routine examinations, and there is no information included in the submitted material that allows identification of the patients. So, informed consent was not obtained. All research activities were performed in accordance with the Declaration of Helsinki.
MR imaging protocol
All patients underwent an MR examination using a 3-T MR imaging scanner (Verio, Siemens Healthineers, Erlangen, Germany) with a 32-channel head coil. The SPACE sequence used for VWI was based on a fast spin-echo technique with variable flip-angle refocusing pulses. The imaging parameters were as follows: TR, 550 ms; TE, 23 ms; bandwidth, 501 kHz; FOV, 140 × 140 mm; matrix, 160 × 256; in-plane resolution, 0.9 × 0.5 mm; slice thickness, 0.5 mm; number of slices, 160; average, 1; scan time, 5 min and 52 s. 3D-TOF MRA was performed using the following parameters: TR, 23 ms; TE, 3.95 ms; flip angle, 18°; bandwidth, 177 kHz; FOV, 167 × 200 mm; matrix, 318 × 448; in-plane resolution, 0.5 × 0.4 mm; slice thickness, 0.6 mm; number of slices, 120; slabs using multiple overlapping thin-slab acquisition; scan time, 6 min and 30 s. The BB-MRA was generated by minimum intensity projection (MinIP) with a slice thickness of 30 mm and a slice interval of 2 mm on a console of the MR imager using the VWI data.
Image analysis
Contrast ratio
For both 3D-TOF MRA and BB-MRA, one observer manually placed a region of interest of 0.03 cm2 at the central part of the stented artery, the normal artery proximal to the stent, and the normal white matter on the source images and measured the signal intensity (SI) for each area. The SI of the stented arteries were measured at five points from the proximal end to the distal end of the stent with approximately equal intervals for each patient, and the average value was calculated. The SI of both the normal artery proximal to the stent and the normal white matter were measured five times for each patient, and the average value was calculated. The SI was measured at the same location for both 3D-TOF MRA and BB-MRA using the image fusion function of ZIO station (Ziosoft, Tokyo, Japan). Afterwards, the contrast ratio (CR) of the stented artery and the normal artery proximal to the stent were calculated using the following formula: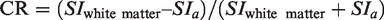

The statistical difference was assessed by comparing the CR of the stented artery with that of the normal artery proximal to the stent using both 3D-TOF MRA and BB-MRA.
Visual assessment
We visually assessed the depiction of the artery at the post-stenting site between the maximum intensity projection (MIP) image generated from 3D-TOF MRA and BB-MRA generated from VWI using the four-grade visual assessment score: 0 = complete signal loss, 1 = discontinuous flow signal, 2 = continuous and heterogeneous flow signal, 3 = continuous and homogeneous flow signal. Two neuroradiologists with over 15 years of experience (MG, KT) independently performed the image reviews, and 3D-TOF MRA, BB-MRA, and DSA were evaluated separately without knowledge of the 3D-TOF MRA or DSA results. When there was a difference in the results between the two observers, a final decision was made by consensus.
Vessel diameter measurement
For DSA, 3D-TOF MRA, and BB-MRA, one observer measured the diameter of the stented artery and the normal artery proximal to the stent in the cross section for each artery. For DSA, the vessel diameter was measured on the viewer, and both for 3D-TOF MRA and BB-MRA, the vessel diameter was measured at the same location using the fusion function of the ZIO station by manual plotting, and the full width at half maximum was regarded as the diameter. The diameter of the stented artery was measured at five points from the proximal end to the distal end of the stent with approximately equal intervals, and the average value was calculated. The diameter of the normal artery proximal to the stent was measured at five points closer to the proximal end of the stent, and the average value was calculated. And the relative diameter index (DI) was calculated as follows: diameter of the stented artery/diameter of the normal artery proximal to the stent.
Statistical analysis
We performed the statistical analyses using commercially available software (IBM SPSS Statistics version 23; IBM, Armonk, New York, USA). The difference between two groups was assessed by Mann-Whitney U test. And the differences among three groups was assessed by Kruskal-Wallis. Each values were indicated mean value ± standard deviation (SD), and
Results
Contrast ratio
On 3D-TOF MRA, the CR of the stented arteries and normal arteries proximal to the stent was 0.39 ± 0.19 and 0.66 ± 0.03, respectively, and the CR of the stented arteries was significantly lower than that of the normal arteries (
Visual assessment
The interobserver agreement was good for 3D-TOF MRA (κ = 0.73) and very good for BB-MRA (κ = 1). The visual assessment scores of the stented arteries were 1.27 ± 0.79 for 3D-TOF MRA and 2.9 ± 0.3 for BB-MRA, respectively. A significant difference between 3D-TOF MRA and BB-MRA was seen (
Relative diameter index
The DI was 0.53 ± 0.13 for 3D-TOF MRA, 0.90 ± 0.05 for BB-MRA, and 0.92 ± 0.08 for DSA. The DI of 3D-TOF MRA was statistically significantly different from that of BB-MRA (
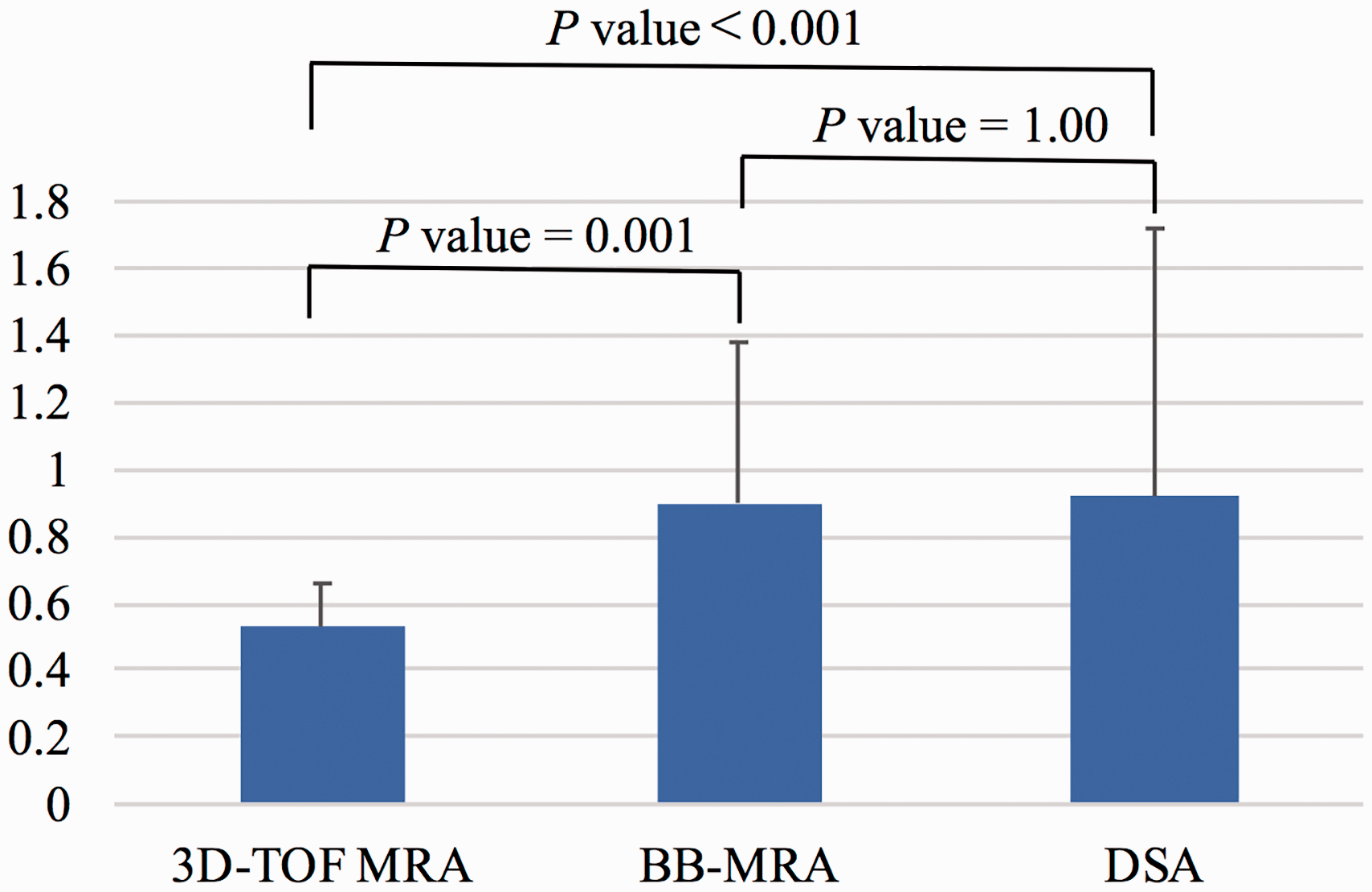
Comparison of relative diameter index among three-dimensional time-of-flight magnetic resonance angiography (3D-TOF MRA), black-blood magnetic resonance angiography (BB-MRA) and digital subtraction angiography (DSA). The relative diameter index (DI) was 0.53 ± 0.13 for 3D-TOF MRA, 0.90 ± 0.05 for BB-MRA and 0.92 ± 0.08 for DSA. The DI of 3D-TOF MRA was statistically significantly smaller from that of BB-MRA (
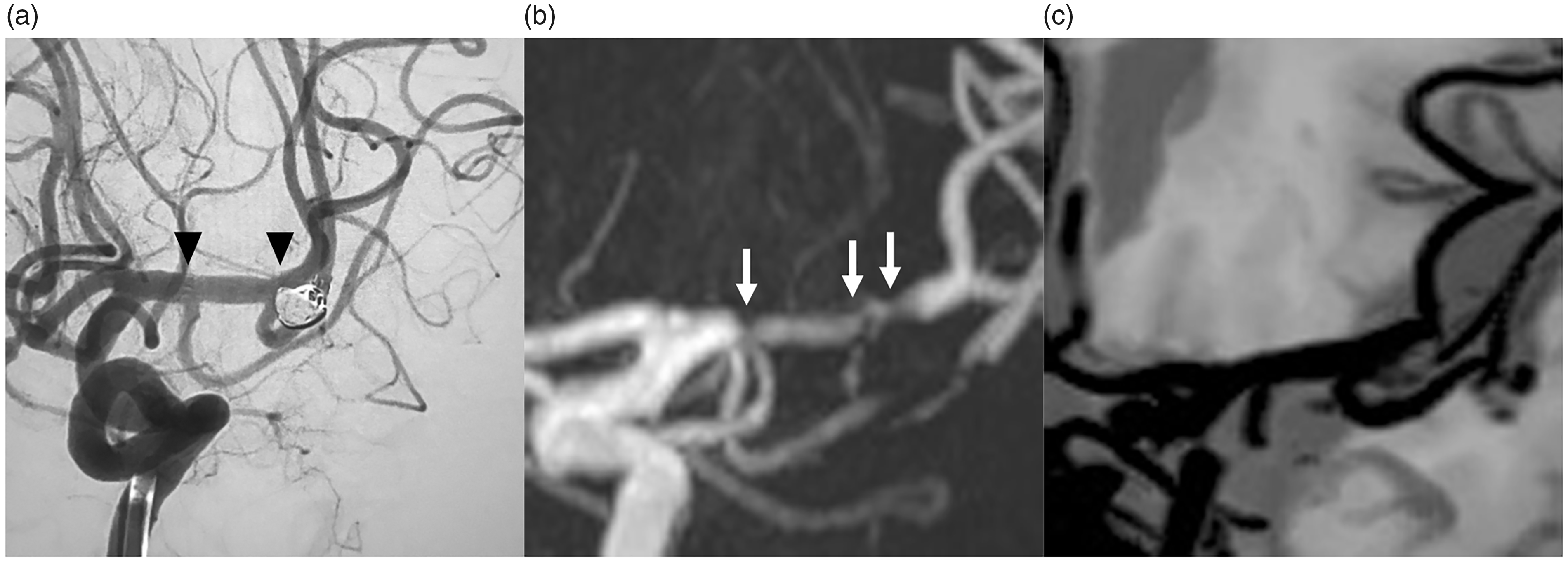
A 68-year-old female. (a) Digital subtraction angiography (DSA), (b) three-dimensional time-of-flight magnetic resonance angiography (3D-TOF MRA), and (c) black-blood magnetic resonance angiography (BB-MRA). (a) Stent-assisted coil embolization was performed for a left middle cerebral artery aneurysm using a Neuroform Atlas stent. Arrowheads indicate stent edges. (b) On 3D-TOF MRA, the flow signal within the stent is discontinuous and heterogeneous (visual assessment score = 2) and there seems to be stenosis (arrows) (the relative diameter index = 0.52). (c) On BB-MRA obtained 2 days after DSA, however, the flow signal in the stent is continuous and homogeneous (visual assessment score = 3), indicating no stenosis (the diameter index = 0.93) and consistent with DSA findings (the diameter index = 0.94).
Discussion
Our study reveals that BB-MRA based on VWI can be a useful follow-up method after stent-assisted treatment for intracranial arterial diseases because it can evaluate the lumen of the stented artery more accurately.
As for the result of the CR of the stented artery, on 3D-TOF MRA the CR of the stented artery was lower than that of the normal artery proximal to the stent in all the cases. On the other hand, on BB-MRA no significant difference in the CR was seen between the stented artery and the normal artery proximal to the stent. This is presumably be due to the fact that 3D-TOF MRA is susceptible to susceptibility artifacts produced by the stent because the modality is based on a gradient sequence, whereas BB-MRA from VWI is less affected by such artifacts because it is based on a spin-echo sequence.12,13
In addition, on MIP images obtained from 3D-TOF MRA, the blood signal in the stented artery was often discontinuous or heterogeneous. On the other hand, BB-MRA from VWI depicted a continuous blood signal in the stented artery in almost all the cases, and the visual assessment score was significantly different between 3D-TOF MRA and BB-MRA. On 3D-TOF MRA, the CR of the stented artery was not satisfactory, which also affected the visual evaluation. Whereas, on BB-MRA the CR of the stented artery was good, and visual assessment of stented artery was excellent. As this result was also likely to have been caused by reduction in susceptibility artifacts, BB-MRA from VWI seems to depict post-stented arteries more clearly.
Furthermore, as for the DI, we also calculated that of DSA in addition to that of 3D-TOF MRA and BB-MRA and evaluated the accuracy of diameter of the stented artery by comparing them. Results showed that the DI of 3D-TOF MRA was statistically significantly different from that of BB-MRA and DSA, and the DI of 3D-TOF MRA was smaller than that of DSA in all the cases. Meanwhile, there was no statistically significant difference in the DI between BB-MRA and DSA. We consider that these results are, like that of CR, due to the fact that 3D-TOF MRA is susceptible to susceptibility artifacts produced by the stent because the modality is based on a gradient sequence, whereas BB-MRA is less affected by such artifacts because it is based on a spin-echo sequence.12,13 In this study, MRI was performed from 1–10 days after DSA, and both DSA and BB-MRA results were considered to depict the same clinical status. So, DIs of 0.90 ± 0.05 for BB-MRA and 0.92 ± 0.08 for DSA showed no difference between BB-MRA and DSA. In the present study, we revealed that BB-MRA can be more accurate for assessing the vessel diameter of the stented artery. Similar comparison of the stented artery among 3D-TOF MRA, BB-MRA, and DSA has not previously been described.
Our study has several limitations. First, as the number of cases was regrettably small, we admit that the power of our statistical analyses is significantly weak. Additionally, again due to the small number of cases, we could not compare the results among several types of stent. Reasons why we could not obtain a sufficient number of cases were as follows. Our study was a single-center study. Furthermore, not all cases of cerebral aneurysm were indicated for stent-assisted coil embolization. Recently, a flow-diverter stent (Covidien, Irvine, California, USA) that enables to treat wide-necked and large aneurysms without using a coil has become available. 15 However, as a flow-diverter stent has a high metal coverage, assessments of arteries after its placement might be more difficult, compared with other stents. Anyway, a comparison of various types of stents, including a flow-diverter stent, is needed in the future. Second, the causes of metallic stent artifacts on MRI can include not only magnetic susceptibility artifacts, but also radiofrequency (RF) shielding artifacts.9–11 When a stent is placed in a continuously changing gradient of magnetic field, an eddy current is generated in the wire of the stent, and a minute magnetic field in the direction opposite to the gradient magnetic field is generated. The RF shielding artifact is a phenomenon in which such minute magnetic fields reduce the sensitivity of receiving and emitting signals within the stent. For 3D-TOF MRA, shielding artifacts reportedly have a larger effect on the signal of the stented artery than susceptibility artifacts. 16 However, since a previous study of VWI using the same method as that used in the present study reported that an in-stent thrombus could be visualized, 14 we consider that our BB-MRA findings generated from VWI correspond to the black-blood imaging of blood flow, and not a loss in signal arising from RF shielding artifacts. Additionally, our literature search revealed no reports on RF shielding artifacts in VWI data. Third, as previously mentioned, DSA is the gold standard for assessing stenosis of stented artery. But DSA is invasive and has a risk of adverse events due to the administration of contrast materials. In addition, as DSA causes radiation exposure, it cannot be performed frequently for the evaluation of the stented arteries.
A previous study of follow-up with 3D-TOF MRA for intracranial aneurysm with stenting or stenting and coiling reported that contrast-enhanced 3D-TOF MRA improves vessel lumen visualization compared to non-contrast 3D-TOF MRA. 17 But the ability of contrast-enhanced 3D-TOF MRA to depict a stented artery is reportedly inferior to that of DSA. 18 Furthermore, the use of contrast material has a risk of causing some adverse events. To avoid the risk and increased cost, we did not compare our method with contrast-enhanced MRA in this study.
To the best of our knowledge, there is no guideline describing the optimal follow-up timing after endovascular treatment. In general, follow-up after stent-assisted treatment for intracranial arterial disease is performed with a protocol of each institute. Kim et al. 19 reported that in-stent stenosis occurred in 22 cases among 123 cases of aneurysm, and the mean timing of identification of in-stent stenosis was 8.90 months (range, 6–17 months). On the other hand, Soize et al. 18 reported that the first imaging follow-up of intracranial aneurysm treated by endovascular embolization is performed 3–6 months after with further follow-up at various intervals depending on each institute. Then, classically, follow-up study was performed at 12–24 months and at 3–5 years after. Furthermore, long-term follow-up of 10 or more years may be required depending on the size of aneurysm and the condition of the residual neck. Similarly, follow-up study after stent-assist treatment for intracranial arterial disease seems to require a long-term approach. A previous study has reported the diagnostic accuracy of 3D-TOF MRA for aneurysm after coil embolization in comparison with DSA. 18 Based on the results of our current study, performing BB-MRA in addition to 3D-TOF MRA after stent-assisted treatment makes it possible to accurately evaluate not only a treated aneurysm but also the diameter and lumen of the stented artery.
For follow-up after stent-assisted treatment for intracranial arterial aneurysms, it is important to evaluate in-stent stenosis and recurrent of aneurysm in clinically. However, our study did not include these cases. Kim et al. 14 reported that an in-stent thrombus could be visualized using VWI. Therefore, we consider that our study of BB-MRA generated VWI would have been more convincing with the addition of cases of in-stent stenosis and thrombosis. So, as the number of cases increases in the future, we hope that we will have the opportunity to examine these issues in detail.
Conclusion
BB-MRA based on VWI can be a useful follow-up method after stent-assisted treatment for intracranial arterial diseases.
Footnotes
Acknowledgement
The authors wish to thank Masaaki Shojima, Department of Neurosurgery, Saitama Medical Center, Saitama Medical University, for his assistance.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
