Abstract
Recent subcortical infarction (RSI) in the lenticulostriate artery (LSA) territory with a non-stenotic middle cerebral artery is a heterogeneous entity. We aimed to investigate the role of LSA combined with neuroimaging markers of cerebral small vessel disease (CSVD) in differentiating the pathogenic subtypes of RSI by whole-brain vessel-wall magnetic resonance imaging (WB-VWI). Fifty-two RSI patients without relevant middle cerebral artery (MCA) stenosis on magnetic resonance angiography were prospectively enrolled. RSI was dichotomized as branch atheromatous disease (BAD; a culprit plaque located adjacent to the LSA origin) (n = 34) and CSVD-related lacunar infarction (CSVD-related LI; without plaque or plaque located distal to the LSA origin) (n = 18). Logistic regression analysis showed lacunes (odds ratio [OR] 9.68, 95% confidence interval [CI] 1.71–54.72; P = 0.010) and smaller number of LSA branches (OR 0.59, 95% CI 0.36–0.96; P = 0.034) were associated with of BAD, whereas severe deep white matter hyperintensities (DWMH) (OR 0.11, 95% CI 0.02–0.71; P = 0.021) was associated with CSVD-related LI. In conclusion, the LSA branches combined with lacunes and severe DWMH may delineate subtypes of SSI. The WB-VWI technique could be a credible tool for delineating the heterogeneous entity of SSI in the LSA territory.
Keywords
Introduction
Recent subcortical infarction (RSI) in the lenticulostriate artery (LSA) territory with a non-stenotic middle cerebral artery (MCA) is a heterogeneous entity. The seminal work of Fisher and Caplan categorized this special type of acute ischemic stroke (AIS) into two main etiological subtypes: lacunar infarction (LI) from intrinsic cerebral small vessel disease (CSVD) pathologically characterized by lipohyalinosis and fibrinoid degeneration (CSVD-related LI); and branch atheromatous disease (BAD) due to a macro or micro atheromatous plaque in the parent artery occluding the orifice of the perforating vessel(s).1–3 However, despite advances in neuroimaging, there is inconsistency in the radiologic definition of RSI covering size, location, form and shape;4,5 and no new understanding of pathology, such that RSI without MCA stenosis being erroneously classified as cryptogenic or as part of broader CSVD in current stroke classification systems.6–8
The recent development of intracranial vessel wall imaging (VWI) now allows detailed evaluation of the vessel wall and atherosclerotic plaques within intracranial arteries, even when conventional magnetic resonance angiography (MRA) is normal.9,10 Recent studies including our own, have demonstrated that novel whole-brain vessel-wall imaging (WB-VWI) is capable of imaging both the MCA wall and LSA lumen in a single image setting,11–13 providing a powerful tool for differentiating the pathogenesis of RSI and offering potential in guiding therapeutic intervention.
Herein, we present the results of our investigation to determine differences in clinical features, LSA morphology, and CSVD neuroimaging markers (lacunes, white matter hyperintensities, cerebral microbleeds, and enlarged perivascular spaces) according to two different etiological subtypes RSI, defined prospectively according to designated BAD and CSVD-related LI, based on the spatial relationship between MCA plaques and LSA origin on WB-VWI.
Methods
Patients
Between November 2018 and March 2020, we prospectively recruited patients admitted to the Department of Neurology, West China Hospital, with the following inclusion criteria: (i) a first-ever single subcortical AIS (no lesion diameter limit) in the LSA territory (basal ganglia, corona radiata, and internal capsule) 14 identified on diffusion-weighted imaging (DWI) performed within 7 days of symptom onset; and (ii) no relevant MCA stenosis confirmed by MRA. Exclusion criteria were: a history of stroke or transient ischemic attack; coexistent ≥50% stenosis at the ipsilateral intracranial internal carotid artery or relevant extracranial arteries detected by CTA or MRA; multiple acute subcortical infarcts or cortical infarcts on DWI; non-atherosclerotic vasculopathies, such as dissection, vasculitis, or Moyamoya disease; evidence of cardioembolism (e.g. recent myocardial infarction within three weeks, atrial fibrillation, dilated cardiomyopathy, valvular heart disease, infective endocarditis, and patent foramen ovale) identified on 24-h electrocardiographic or Holter monitoring, and transthoracic echocardiography.
Data were recorded on demographic characteristics and vascular risk factors that included age, sex, height and weight for calculation of body mass index, history of hypertension, diabetes mellitus, hyperlipidemia, cardiac arrhythmia, and coronary artery disease, current smoking status and time of symptom onset. Baseline neurological severity was assessed on the National Institutes of Health Stroke Scale (NIHSS). Functional outcome was assessed by trained staff using the modified Rankin scale (mRS) at 90 days, either by telephone or in-person interviews. The study was approved by the ethics committee of West China Hospital (2018521) and was conducted according to the Declaration of Helsinki with each patient providing written informed consent.
MRI protocols
All enrolled patients underwent WB-VWI within 2 weeks of symptom onset on a research-dedicated 3.0-T MRI (MAGNETOM Trio, Siemens, Erlangen, Germany) equipped with a 32-channel head coil. The protocol included diffusion-weighted imaging (DWI, TR/TE of 4000/91 ms; field of view 220 × 220mm2; 20 slices with slice thickness of 5 mm; voxel size = 1.1 ×1.1 × 5mm3; 2b values of 0 and 1000 s/mm2; scan time of 1:53 min), three-dimensional time-of-flight MRA (3D TOF-MRA, TR/TE = 22/4 ms; acceleration factor (iPAT)=2; matrix size = 320 × 320; field of view = 170 × 170 mm2; 155 slices with slice thickness of 0.53 mm; voxel size = 0.53 × 0.53 × 0.53mm3; scan time = 6 min), susceptibility-weighted image (SWI, TR/TE, 28/20 ms; 72 slices with slice thickness 2 mm, voxel size = 0.6 × 0.6 × 2mm3; scan time = 6 min), and conventional T1-weighted, T2-weighted, fluid-attenuated inversion recovery (FLAIR) images. WB-VWI sequence was performed based on inversion-recovery (IR) prepared SPACE (Sampling Perfection with Application-optimized Contrast using different flip angle Evolutions)15,16 with the following parameters: TR/TE = 900/14 ms; field of view =170 × 170 mm2; 240 slices with slice thickness of 0.53 mm; voxel size = 0.53 × 0.53 × 0.53 mm3; scan time = 8 min.
Image analysis
All images were reviewed and analyzed with commercial software (OsiriX MD, Pixmeo SARL, Bernex, Switzerland) by two experienced readers (SJ and JS) blind to clinical information. WB-VWI data were analyzed before conventional MRI images. In cases of discrepancies, a consensus read was performed with both readers and a senior and more experienced reviewer (BW) who was not involved in the initial grading. The details of interrater agreement of WB-VWI imaging parameters were described previously. 13
Classification of BAD and CSVD-related LI by WB-VWI
The diagnosis of RSI in the LSA territory was based on published templates, 14 regardless of the lesion size. Multi-planar reconstruction (MPR) and coronal minimum intensity projection (MinIP) were generated in one image setting with WB-VWI images, which enabled discrimination between BAD and CSVD-related LI in RSI patients by simultaneous direct imaging of the MCA lumen and LSA morphology. Based on the spatial relationship between MCA plaque location and the LSA origin, RSI was dichotomized as BAD (a culprit plaque located adjacent to the LSA origin; Figure 1a and c) and CSVD-related LI (without plaque or plaque located distal to the LSA origin; Figure 2a and c).
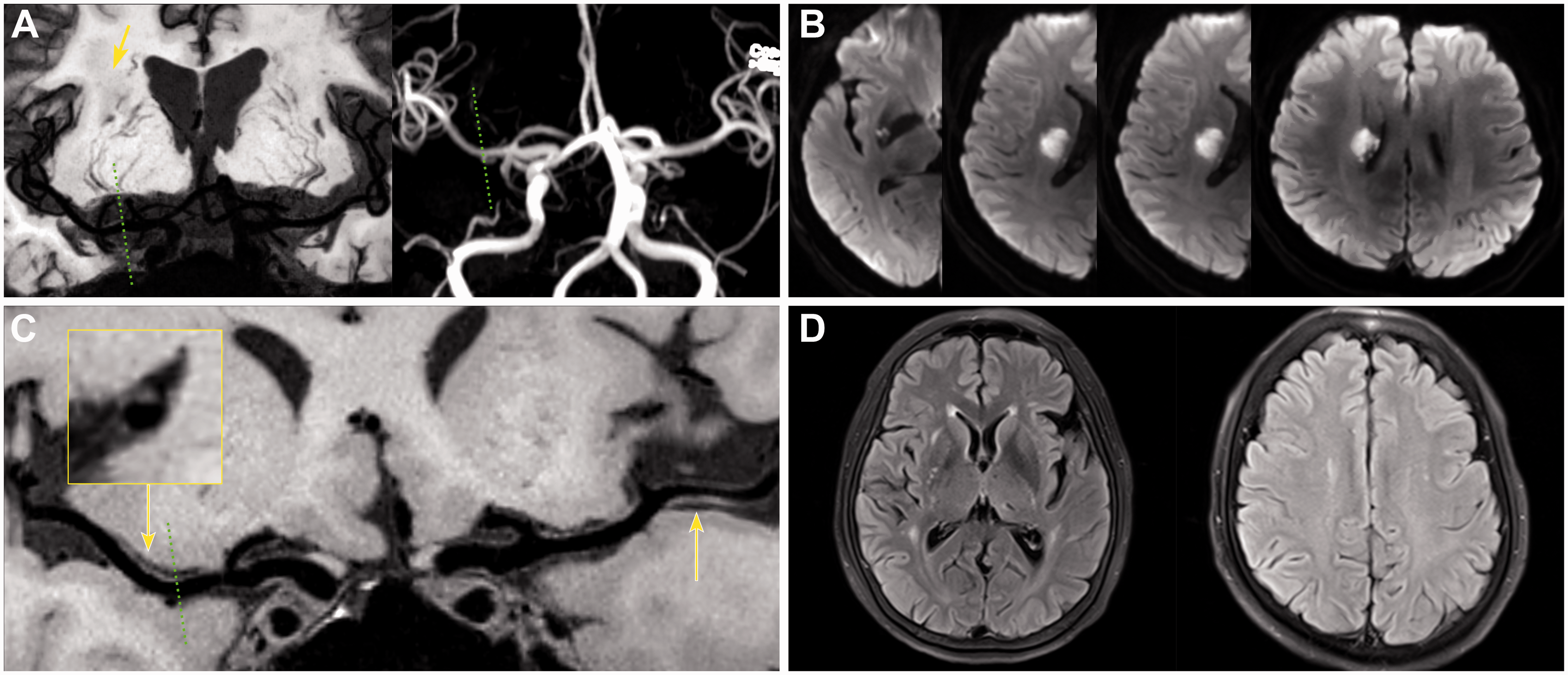
Representative case of BAD based on WB-VWI. (a) Coronal MinIP showed RSI in the right LSA territory (arrow) and revealed impairment of the right LSAs compared to the left side; Magnetic resonance angiography (MRA) showed no stenosis on the relevant MCA; (b) DWI showed proximal RSI involvement 4 consecutive axial slices. (c) Curved multi-planar reconstruction (curved-MPR) showed a culprit plaque adjacent to the corresponding LSA origins (dashed lines), demonstrated by the magnified image on cross-section view; a nonculprit plaque was also detected on left MCA (left arrow); (d) FLAIR displayed silent lacunes in the basal ganglia and mild WMH.
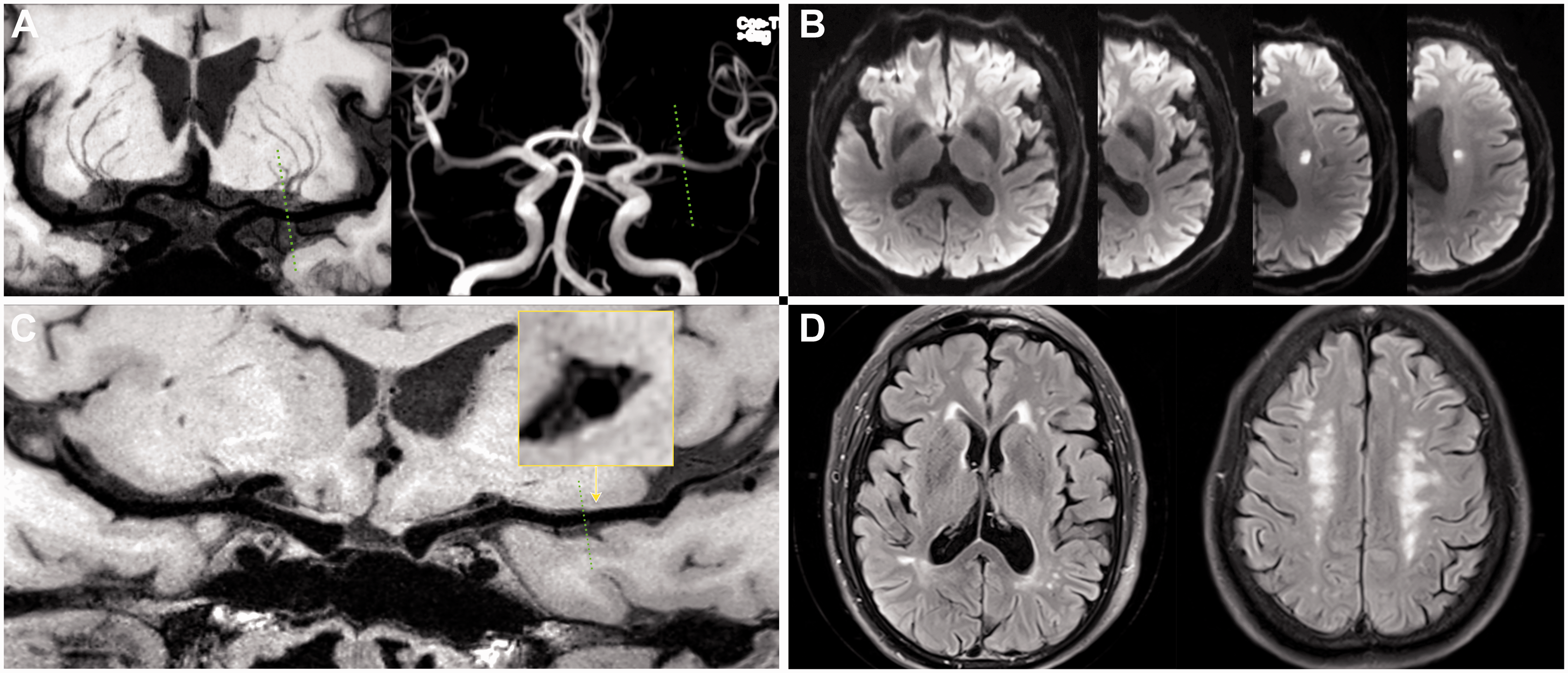
Representative case of CSVD-related LI based on WB-VWI. (a) Coronal MinIP showed almost symmetrical LSAs between the symptomatic and asymptomatic sides; Magnetic resonance angiography (MRA) showed no stenosis on the relevant MCA; (b) DWI showed distal RSI involvement 2 consecutive axial slices; (c) Curved multi-planar reconstruction (curved-MPR) showed without culprit plaque on relevant MCA (dashed lines), demonstrated by the magnified image on cross-section view; (d) FLAIR displayed severe deep WMH but without silent lacunes.
Analysis of lenticulostriate arteries
LSA images were generated using coronal MinIP with 10–20 mm thickness on WB-VWI. All visible LSAs originating from the M1 and M2 segments of the MCA were included in analysis. The morphological characteristics of visible LSAs (number and length of stems and branches) were quantitatively analyzed on the coronal slab MinIP image between BAD and CSVD-related LI patients; only blood vessels pointing toward the anterior perforated substance were counted. In order to delineate the distributions of LSAs, manually tracing of the full length of the visualized vascular skeleton was performed on signal measurements of consecutive MinIP images, as described previously. 13 Stems were defined as the portion of the LSA that originated directly from the MCA. Branches are defined as daughter vessels originating from parent LSA stems or as stems with no branches (single vessel), including LSA stems >5mm; when an artery branched <5 mm of the origin of the MCA, each stem was counted separately 17 as >70% of branches were found to originate from common trunks. 18 When an artery branched at a more distal site, the stem was counted and measured using the longest branch. Figure 3 illustrates the method used for counting stems and branches, and measuring length.
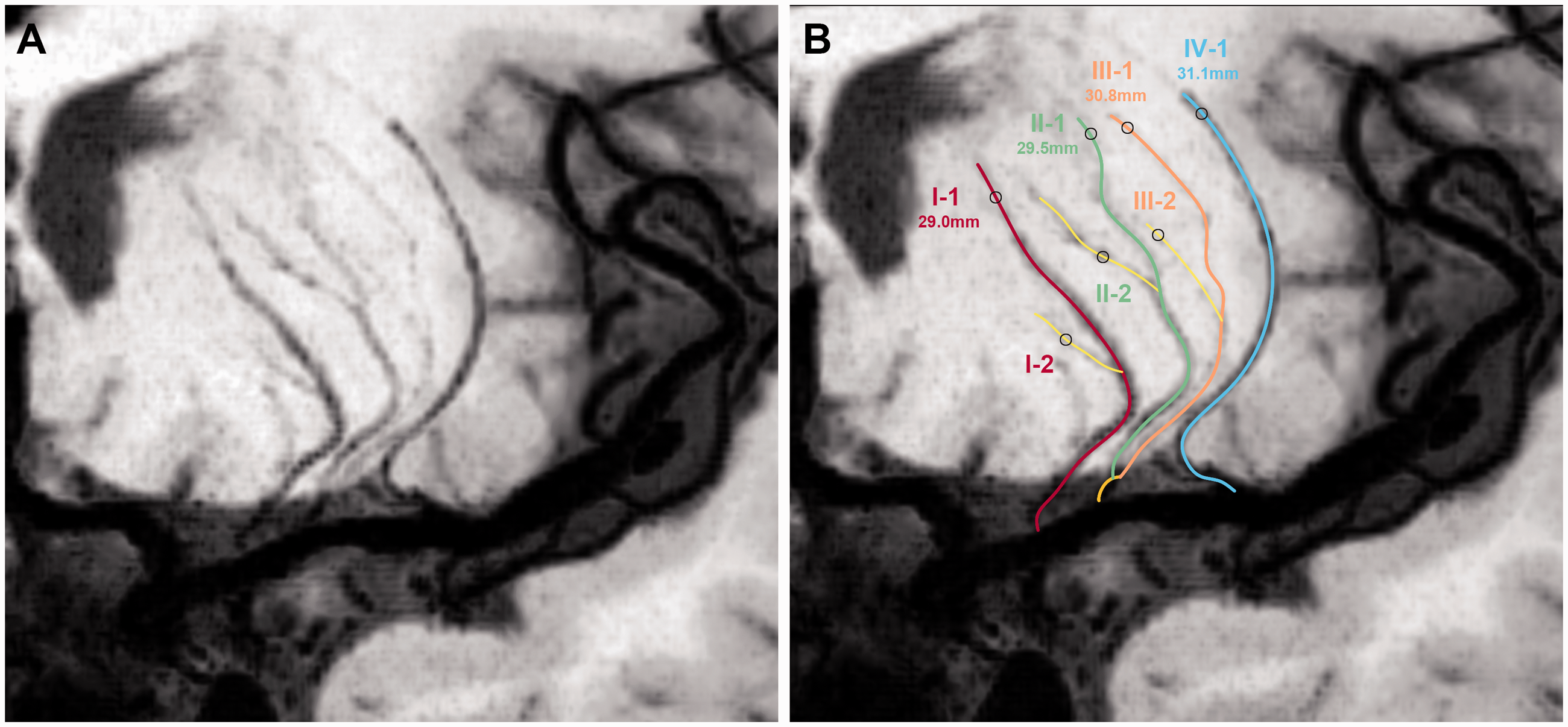
A schematic illustration of the measurement of LSAs. (a) The reconstructed Coronal MinIP image of LSAs. (b) Corresponding line tracings of the LSAs. Stems are labeled with Roman numerals and branches are labeled with Arabic numerals. The measurements are the lengths of each stem, respectively. II and III of the LSAs originated from the common stem.
Assessment of cerebral small vessel disease
We defined a lacune of presumed vascular origin as a small (3–15 mm) round or ovoid lesion in the basal ganglia, internal capsule, centrum semiovale, or brainstem, of CSF signal intensity on T2 and FLAIR, generally with a hyperintense rim on FLAIR and no increased signal on DWI (Figure 1d), and not compatible with clinical findings.19–21 The extent of deep white matter hyperintensities (DWMH) and periventricular white matter hyperintensities (PWMH) on the FLAIR images were rated using the Fazekas scale (Figure 2d) 22 where a score ≥ 2 was regarded as severe WMH.
Cerebral microbleeds (CMBs) were rated on susceptibility-weighted imaging as homogeneous rounded lesions of signal loss, within a diameter of 2–10 mm.19,23 Enlarged perivascular spaces (EPVS) were defined as small (<3 mm) punctate and linear hyperintensities on T2-weighted imaging in the basal ganglia or centrum semiovale, and rated as 0 to 4 on a validated semiquantitative scale. 24 We only counted EPVS in the basal ganglia as specifically associated with CSVD.24,25 A CSVD compound score ranging from 0 to 4 was constructed to express the total burden of CSVD, as previously described. 26
Assessment of infarct dimensions
The DWI lesion volumes were measured using a semi-automated volumetric assessment by OsiriX software. The lesion area was manually outlined slice by slice, and the total volume was calculated for all regions of interest. The maximum infarct diameter was measured by the axial slice of the DWI and classified as large if ≥15 mm in axial diameter. The number of lesion axial slices visible on consecutive axial DWI slices were recorded. Lesion location was assessed by DWI and categorized as proximal and distal lesions. Proximal lesion was defined as an infarction located adjacent to the MCA and extending to the basal surface of the MCA (Figure 1b); distal lesion was defined as an infarction located in a distal area but not extending to the basal surface of the MCA (Figure 2b). The infarct involvement of the lowest portion of the basal ganglia was considered as an extension to the basal surface. 27
The presence of concomitant asymptomatic cerebral arterial atherosclerosis (ACAS) was defined as any extracranial and intracranial cerebral artery stenosis except for those related to current lesions on the 3D-TOF MRA or CTA images.
Statistical analysis
The normality of the data was analyzed using the Kolmogorov–Smirnov test. All quantitative data were expressed as means ± standard deviation (SD) or as medians with interquartile range (IQR), and qualitative data were summarized as count (percentage). Categorical variables were analyzed using a Chi-square test or Fisher exact test as appropriate, and continuous variables were compared using t-test (for continuous normally distributed data) or Mann–Whitney U test (for continuous, non-normally distributed data) between the two groups. Multivariable logistic regression models were used to identify independent predictors of the two infarction patterns, including all variables with P < 0.05 on univariate analysis. All the statistical tests were two-sided with a P value < 0.05 considered significant. All statistical analyses were performed using SPSS version 25.0 (SPSS Inc., Chicago, IL, USA).
Results
Patient characteristics
Of the 52 RSI patients (mean age 55.8 ± 10.3 years; female 25.0%) with no relevant MCA disease, 34 (65.4%) and 18 (34.6%) were classified as BAD and CSVD-related LI, respectively, according to WB-VWI. The median interval between symptom onset and WB-VWI was 6.6 ± 3.4 days (range 1 to 14 days). Table 1 shows there were no significant differences in the demographics, vascular risk factors, concomitant ACAS, and laboratory findings of the BAD and CSVD-related LI groups.
Comparison of baseline characteristics between BAD and CSVD-related LI groups.
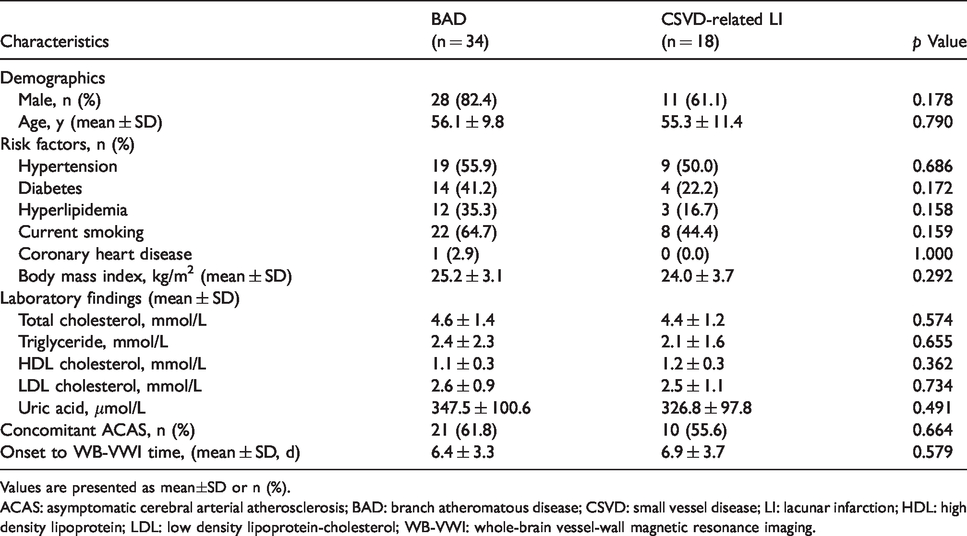
Values are presented as mean±SD or n (%).
ACAS: asymptomatic cerebral arterial atherosclerosis; BAD: branch atheromatous disease; CSVD: small vessel disease; LI: lacunar infarction; HDL: high density lipoprotein; LDL: low density lipoprotein-cholesterol; WB-VWI: whole-brain vessel-wall magnetic resonance imaging.
Comparison of BAD and CSVD-related LI according to infarcts characteristics and CSVD MRI markers
The number of lesion slices tended to be larger in patients with BAD compared to those with CSVD-related LI, with borderline significance (3 [3–4] vs 2.5 [2–3]; P = 0.065), as well as the number of patients with lesion slices ≥3 (76.5% vs 50.0%; P = 0.053; Table 2 and Figure 4). However, the proportion of proximal lesions in the BAD (85.3%) was similar to the CSVD-related LI (77.8%) groups (P = 0.702). Although the average axial lesion diameter (1.72 ± 0.65 cm vs 1.46 ± 0.68 cm) and lesion volume (2.56 ± 2.25 cm3 vs 1.98 ± 2.02 cm3) were larger in the BAD group than in the CSVD-related LI group, these were not significant (P > 0.05), and there were comparable baseline NIHSS scores and functional outcome between the groups.
Univariate and multivariate logistic regression analysis of relative factors for subtypes of RSI.
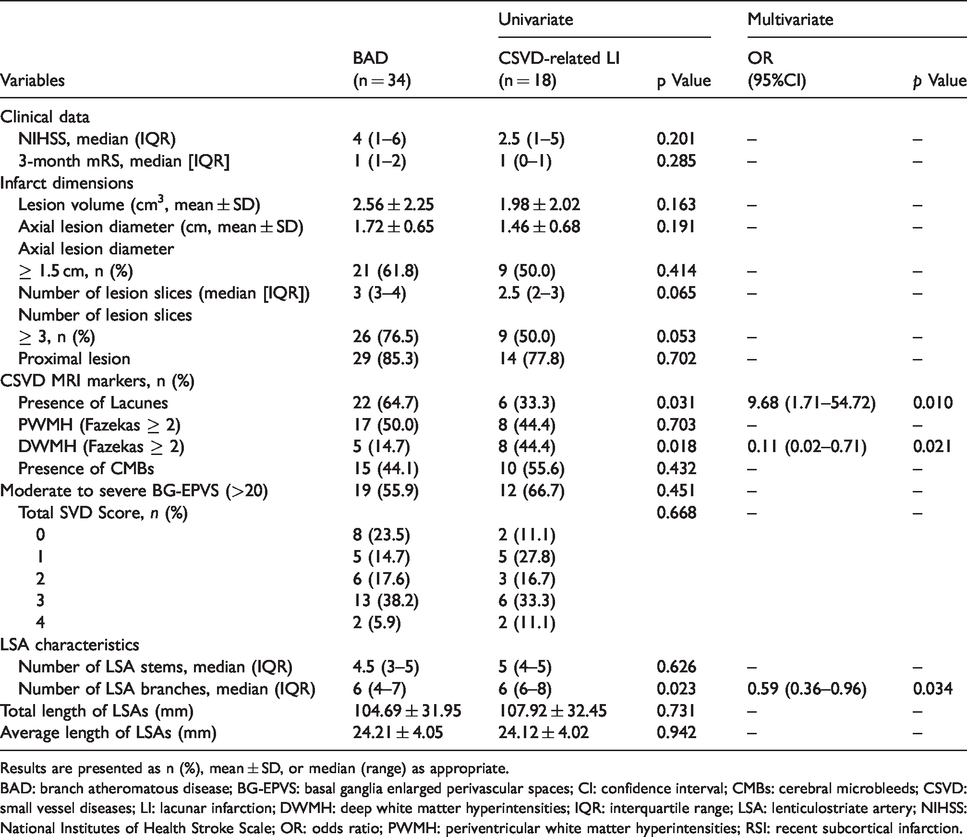
Results are presented as n (%), mean ± SD, or median (range) as appropriate.
BAD: branch atheromatous disease; BG‐EPVS: basal ganglia enlarged perivascular spaces; CI: confidence interval; CMBs: cerebral microbleeds; CSVD: small vessel diseases; LI: lacunar infarction; DWMH: deep white matter hyperintensities; IQR: interquartile range; LSA: lenticulostriate artery; NIHSS: National Institutes of Health Stroke Scale; OR: odds ratio; PWMH: periventricular white matter hyperintensities; RSI: recent subcortical infarction.
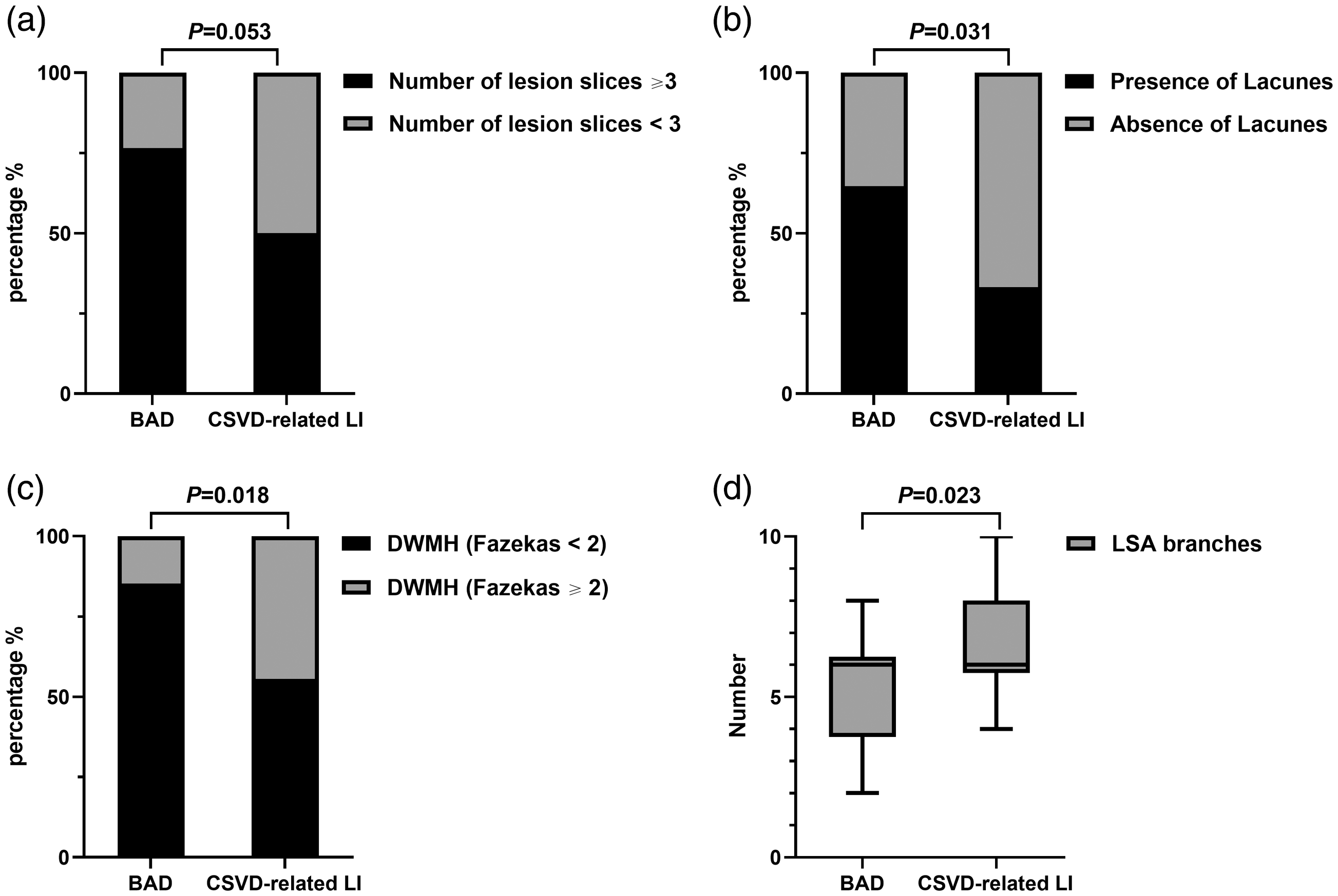
Comparison of underlying predictors between BAD and CSVD-related LI groups. Number of lesion slices (≥ 3) (a), presence of Lacunes (b), severe deep white matter hyperintensities (DWMH) (Fazekas ≥ 2) (c), and number of LSA branches (d) in BAD and CSVD-related LI groups.
Of the four CSVD MRI markers, patients with BAD (64.7%) had significantly more lacunes compared to those with CSVD-related LI (33.3%) (P = 0.031), whereas severe DWMH (Fazekas ≥ 2) were more common in patients with CSVD-related LI (44.4%) than in those with BAD (14.7%) (P = 0.018), as shown in Table 2 and Figure 4. However, there were no differences with respect to CMBs, moderate-severe EPVS in the basal ganglia (>20), and total burden score of CSVD between groups.
Comparison of BAD and CSVD-related LI according to LSA characteristics
The number of LSA branches was larger in patients with CSVD-related LI (6-8) than those with BAD (4–7; P = 0.023; Table 2 and Figure 4), whereas the number of stems did not differ in CSVD-related LI (4–5) and BAD (3–5) groups (P = 0.626). The total length and average length of LSAs were also similar between groups (Table 2).
The relative factors for subtypes of RSI by multivariate analysis
In a logistic regression model, the presence of lacunes (odds ratio [OR] 9.68, 95% confidence interval [CI] 1.71–54.72; P = 0.010) and smaller number of LSA branches (OR 0.59, 95% CI 0.36–0.96; P = 0.034) were significantly associated with BAD, whereas severe DWMH (OR 0.11, 95% CI 0.02–0.71; P = 0.021) was independently associated with CSVD-related LI (Table 2).
Discussion
In our study using a novel WB-VWI modality for direct in vivo visualization of the spatial relationship between MCA plaques and LSA origin we were successful in distinguishing BAD and CSVD-related LI within the heterogeneous entity of RSI in the LSA territory. Of 52 RSI patients with normal-appearing relevant MCA, we found a higher proportion with BAD than CSVD-related LI. Furthermore, we also found that a smaller number of LSA branches and presence of lacunes were associated with BAD, while severe DWMH was associated with CSVD-related LI. However, conventional risk factors and infarct size were not different between the two groups. These results support the hypothesis for two distinct underlying pathogenic subtypes (BAD and CSVD-related LI) for RSI in the LSA territory, despite similar clinical features and infarct appearances.
The term BAD was originally coined in 1989 as an alternative mechanism to the pathological finding of lipohyalinosis and to emphasize it being a major and neglected aspect of RSI.3,28 However, there have been very limited further pathological studies, and it has been difficult to establish a direct relationship between parental plaques and relevant perforating arteries with contemporary imaging modalities. Although previous studies using VWI have indicated the orifices of LSAs are vulnerable to nearby atherosclerotic plaques on the parent MCA, resulting in RSI,29,30 such a mechanism is only presumed because the morphology of LSAs cannot be reliably detected at the same time by usual VWI technique. Now that WB-VWI is feasible for simultaneously visualizing the LSAs and MCA wall, our study would appear to be the first in allowing in vivo categorization of RSI into BAD and CSVD-related LI according to the original pathological descriptions. In line with previous findings,10,30 a relatively high frequency (65.4%) of BAD was observed in our prospective cohort, suggesting this is the most common cause of RSI even with a normal MRA.
To date, there has been little consistency over the dimensional cutoff used to define the two distinct types of RSI.4,28 Based on presumed vascular pathologies, BAD are usually inferred clinically if images show infarcts involving the basal surface, while more distal infarcts are regarded as CSVD-related LI.27,30 In the majority of neuroimaging studies, BAD in the LSA territory is considered if with a large lesion in a single slice (diameter ≥ 15 mm) or across multiple (≥3) axial slices, whereas a small lesion (<15 mm; <3 slices) is defined as CSVD-related LI.31–33 Our findings suggest that the number of axial lesion slices (≥3), although with marginal significance (P = 0.053) provides better appreciation of the discrepancy of infarct rather than axial lesion diameter (P = 0.191), as indicated in previous studies for predicting the mechanism of RSI and anatomical distribution of LSAs.34,35 In agreement with most other studies,5,30 there were no other differences in terms of vascular risk factors, neurological severity and prognosis between the groups.
As LSAs are the major cerebral small arteries related to RSI in the MCA territory, delineating their morphological characteristics could provide crucial insights into etiologic mechanisms and tailored therapies. In our study, we found the smaller number of LSA branches was independently associated with BAD, which appears consistent with pathological studies indicating the cause to be a plaque in the parent artery blocking the orifice of penetrating artery. The occlusion occurring at the origin of LSAs can reduce blood flow in the microvessels and lead to a decrease in imaging signal intensity, thus result in the affected LSA branches go undistinguished and manifest as a decrease of the absolute number of LSA branches on WB-VWI images. Although LI is often considered to be caused by intrinsic small artery disease pathologically characterized by lipohyalinosis, the greater number of underlying LSA branches suggests alternative explanations from BAD. First, although the presence of an occluded perforating artery related to the LI was proposed by Fisher several decades ago, this so-called lacunar hypothesis remains unproven.36,37 Conversely, evidence has been accumulating to suggest that early endothelial dysfunction rather than simply arteriolar occlusion plays an important role in the pathogenesis of CSVD-related LI. 38 Second, luminal occlusion might be a late-stage phenomenon in most pathological studies and does not explain the early pathology of CSVD-related LI, 39 as supported in our study where the inclusion of those with first-ever lacunar AIS and a relatively lower rating of CSVD burden, which may represent the early-stage of CSVD-related LI, thus maintaining relatively high patency of LSA branches. The comparable stems and length of LSAs between BAD and CSVD-related LI could be explained by incomplete obstruction of LSA stems by MCA atherosclerotic plaques in BAD. In addition, since the current imaging technique cannot visualize proximal microatheroma in the orifice of LSAs itself, the CSVD-related LI groups may also have a proximal occlusion of LSA stems even without junctional plaque on relevant MCA. Furthermore, the large size of stems compared with branches could enable them to be less susceptible to resolution on WB-VWI.
It is interesting that presence of lacunes was independently associated with BAD, whereas severe DWMH was independently associated with CSVD-related LI. Because lacunes, WMH, CMBs, and EPVS are all indicators of CSVD, 19 those four MRI markers are generally thought to be more closely related to CSVD-related LI than BAD.40–42 However, different underlying pathogenetic mechanisms could account for variable CSVD features. 39 It was quite intriguing that the presence of lacunes was more prevalent in BAD groups than that of CSVD-related LI groups, which implied that there might be heterogeneous pathogenesis associated with the presence of silent lacunes. An epidemiological study has shown that the presence of previous lacunes is more frequent in patients with potential mechanisms other than CSVD. 43 The higher detection of silent lacunes in those with BAD could be due to their relatively larger lesion size, in agreement with the previous autopsy findings,1–3 making them easier to detect on images. Conversely, severe DWMH rather than PWMH, was associated with CSVD-related LI, suggesting that DWMH might be a sensitive marker of CSVD. Similar findings have been reported in another study which showed that the progression of WMH volume was associated with increased risk of new LIs in the deep white matter. 44 Our findings confirmed the hypothesis of different mechanism of region-specific WMH that PWMH is more likely determined by chronic hemodynamic insufficiency (hypoperfusion) or cerebrospinal fluid leakage, whereas DWMH is more linked to CSVD.45,46
Differentiating different underlying pathogenic mechanisms of RSI may be clinically important. According to current classification systems,6–8 clinicians can mistakenly classify a heterogeneous entity of RSI as either cryptogenic by the degree of stenosis or as CSVD by lesion size; whereas WB-VWI can be explicitly classified the condition as being due to large artery atherosclerosis (BAD, a culprit plaque located adjacent to the orifices with impaired LSAs) or small-vessel disease (CSVD-related LI, no culprit plaque found on relevant MCA with relative normal LSAs combined with severe DWMH). Pinpointing well-defined classification of RSI subtypes based upon etiologic mechanism is crucial for guiding optimal treatment and assessing prognosis in individual patients. Thus, future trials and observational studies should aim to RSI according to underlying mechanisms (e.g. CSVD-related LI vs BAD), so that the tailored strategies can be applied in clinical practice. Where possible, WB-VWI should be used to identify the different sub-group of RSI.
We recognize our study has several limitations. First, the sample size was relatively small, largely due to our narrow inclusion criterion for RSI whereby patients with proximal embolic sources were purposefully excluded. Our goal was to only include patients without relevant MCA disease, in order to evaluate the different underling mechanisms between so-called pure in situ RSI in the LSA territory, which is often classified as being due to CSVD in practice. Second, we cannot entirely exclude the possibility that some CSVD-related LI were caused by penetrating artery atheroma, as the current method of WB-VWI cannot visualize microatheroma in the proximal proportion of LSAs, although this putative mechanism of RSI is far from being pathologically proven. Third, the metrics of the LSA length were derived from manual tracing, which is time-consuming and somewhat subjective, which may be addressed through machine learning algorithms. Finally, it should be noted that the anatomical variation of LSAs in different individuals, but we considered the distribution of LSA anatomy between patients to be random.
In conclusion, LSA branches combined with lacunes and severe DWMH may delineate subtypes of RSI. Our study has shown the WB-VWI technique can differentiate the heterogeneous nature of RSI in the LSA territory without MCA disease on MRA. As well as having a useful role for AIS subtype classification, the approach has potential for risk stratification of RSI for prevention and therapeutic treatments.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Key Development Plan for Precision Medicine Research (2017YFC0910004), National Key R&D Program of China, Ministry of Science and Technology of China (2016YFC1300500-505) and National Natural Science Foundation of China (81671146; 81870937).
Authors’ contributions
Study design, study conception: SJ, BW, JYS, TC; initial manuscript draft: SJ; data interpretation and image analysis: SJ, BW, TC, YYY, ZLH; acquisition of data and clinical analysis: JYS, TY, YY, QD, TW, SMW; revising the manuscript for important intellectual content: BW, ZLH, CSA.
