Abstract
Background
Brain stroke is a rare, life-threatening condition associated with pituitary apoplexy (PA), resulting from direct arterial occlusion due to mechanical compression secondary to the sudden enlargement of the pituitary adenoma, or to vessel vasospasm, induced by tumor hemorrhage.
Case report
We report the case of a 64-year-old woman with PA complicated by bilateral anterior circulation stroke due to critical stenosis of both anterior cerebral arteries (ACA). Despite the quick surgical decompression and consequent blood flow restoration, the neurological conditions of the patient did not improve and she died 18 days later. Ten other cases of anterior circulation stroke due to PA were retrieved in a systematic review of literature. Clinical and neuroradiological features of these patients and treatment outcome were assessed to suggest the most proper management.
Conclusion
The onset of neurological symptoms suggestive for brain stroke in patients with PA requires performing an emergency Magnetic Resonance Imaging (MRI), including Diffusion-weighted and angiographic MR-sequences. The role of surgery in these cases is debated, however, transsphenoidal adenomectomy would permit us to decompress the ACA and restore blood flow in their territories. Although the prognosis of PA-induced anterior circulation stroke is generally poor, a timely diagnosis and treatment would be paramount for improving patient outcome.
Keywords
Introduction
The potential life threats associated with pituitary apoplexy (PA) have been reported by several studies.1,2 Among them, the acute hemorrhage/infarction of a pituitary adenoma may lead to the abrupt compression of the surrounding structures, with the consequent sudden development of hypopituitarism, visual deficits up to blindness, and neurological deficits, typically oculomotor palsies with palpebral ptosis.1,2
Rarely, PA may induce ischemic brain stroke, responsible for a significant increase in mortality and permanent neurological sequelae.3,4 Because of the rarity of this association, the exact etiopathogenesis, the most appropriate diagnostic and therapeutic management, and patients’ prognosis remain to be clarified.3,4
We report a case of brain stroke involving the bilateral anterior circulation territories due to PA. Systematic literature search has also been performed with the aim of identifying associated risk factors, type, and timing of diagnosis and treatment, and of assessing the relative patients’ outcomes, in order to suggest the most proper clinical and neuroradiological management of this rare condition.
Case report
A 64-year-old woman presented to the emergency room for the abrupt onset of headache and bilateral visual disturbance. Patient history was remarkable only for end-stage kidney disease in IgA nephropathy, treated with hemodialysis. Brain Computerized tomography (CT) scan showed a large inhomogeneous and hyperdense round sellar mass with suprasellar extension and sporadically parietal calcifications, highly suggestive for PA, and no other brain alterations (Figure 1). Bio-humoral assays demonstrated panhypopituitarism, promptly treated with hydrocortisone, and acute renal failure (creatinine 10.7 ng/mL). Immediately after these examinations, urgent dialysis session was started. However, few hours after the PA symptoms onset, a rapid deterioration of consciousness was observed, followed by right hemiparesis and hemodynamic instability, which required resuscitation maneuvers and patient intubation. CT scan and MRI. (a) CT scan demonstrating the presence of a round inhomogeneous hyperdense intrasellar formation with focal and peripheral calcifications. (b) MRI. T1-w non-contrast sequence in the sagittal plane demonstrating the presence of a midline tumor in the sellar-suprasellar region, compatible with large pituitary apoplectic adenoma. (c). Contrast T1-w sequence in the sagittal plane showing linear enhancement of the pituitary gland surrounding the hemorrhage. (d) and (e). T2-w (d) and T1-w (e) in the axial plane demonstrating the presence of a portion with signal intensity compatible with fluid in the anterior portion of the apoplectic PA (white arrow), compatible with acute hemorrhage (1–7 days) due to the signal intensities features. The apoplectic PA, and in particular the hemorrhagic portion of the tumor, was located immediately inferior to both A1 segments of the ACA (red arrows). (f). Time of flight (TOF) MRA demonstrating the superior displacement and narrowing (red arrow) of both A1 segments of the ACAs due to the mass effect of the PA. PA: pituitary apoplexy; ACA: anterior cerebral arteries.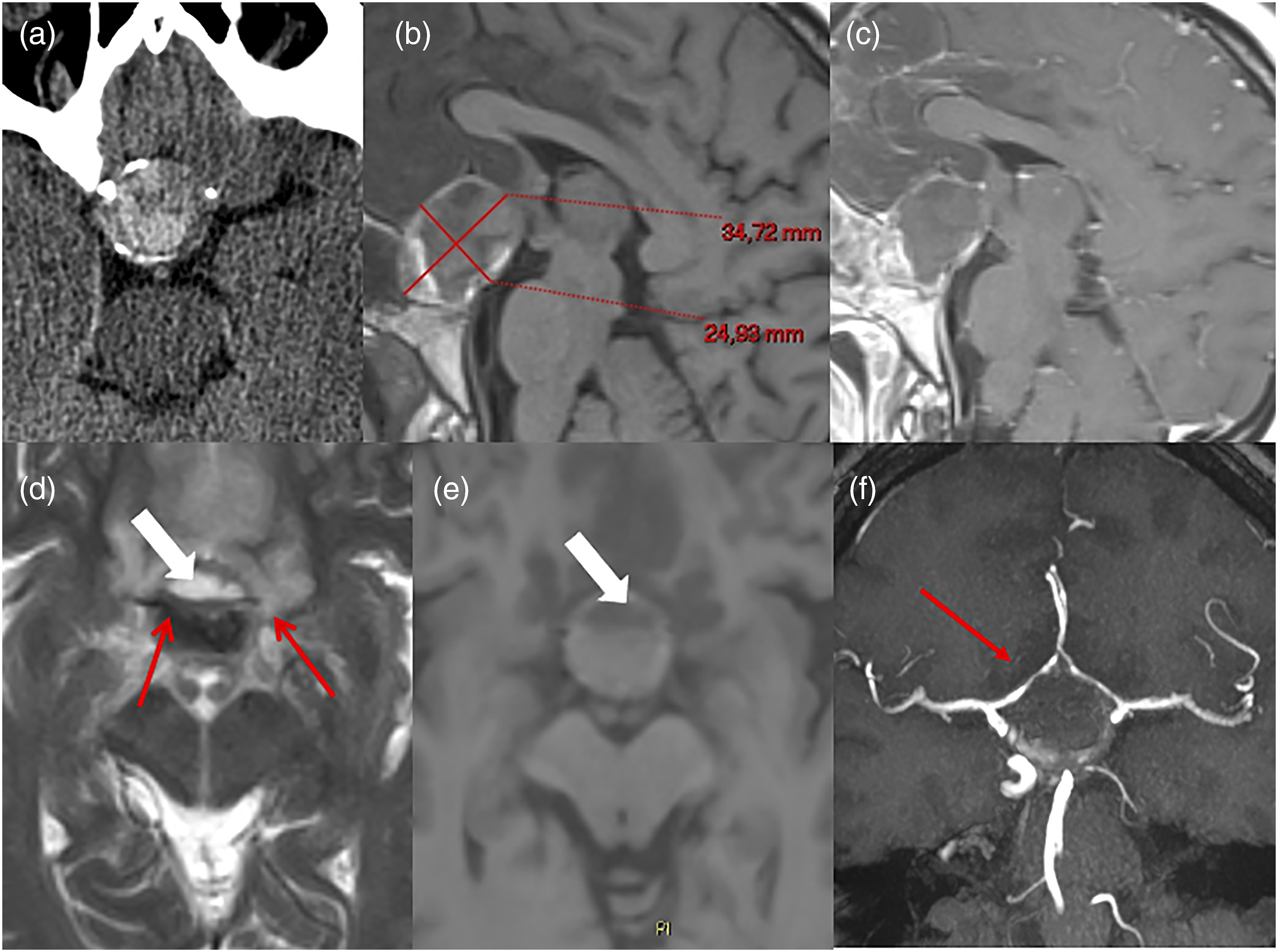
After achieving hemodynamic stability, urgent brain Magnetic Resonance Imaging (MRI) was performed at 12 h from symptoms onset, confirming the presence of a large tumor with endosuprasellar extension, with areas of mild hypointensity in T1-w and marked hyperintensity in T2-w sequences, strongly suggestive for recent intra-tumoral bleeding, compatible with the clinical suspicion of apoplectic pituitary adenoma (Figure 1). Moreover, the MRI showed bilateral anterior-circulation ischemic strokes, mainly extending on the left side, with features typical of acute (6–24 h) onset (Figure 2). On the right side, the ischemic lesion involved the region of the basal ganglia supplied by the perforating vessels of the A1 segment (head of the caudate and anterior portion of the lenticular nucleus) and the proximal arteries deriving from the callosomarginal artery. Conversely, on the left side the ischemic lesion involved all the distal territories supplied by the arteries deriving from the callosomarginal artery (Figure 2). The MR-angiography (MRA) showed the bilateral superior displacement and critical narrowing of the A1 segments of both anterior cerebral arteries (ACAs), and the increase of the internal carotid artery (ICA) A1 angle on both sides, caused by the mass effect exerted by the PA (Figure 1). MRI. Demonstration of ischemic lesions in the right basal ganglia (head of the caudate and anterior portion of the lenticular nucleus), in the anterior portion of the left caudate and in the bilateral frontal lobes, majorly extending on the left side. On the right side the involved territory is represented by territories supplied by perforators of the A1 segment and of the proximal group of the callosomarginal artery (infraorbital, frontopolar and anterior frontal arteries), while on the left side also by the distal branches of the callosomarginal artery (middle-posterior frontal, paracentral and superior parietal arteries). The presence of gyriform high signal and sulcal effacement in the T2-w sequence (a, c), mild hyperintensity in the T2-w Fluid Attenuated Inversion Recovery (FLAIR) sequence (b, d) and marked restriction of the diffusion in the DWI sequence (e, g) with consensual elevated reduction in the related Apparent Diffusion Coefficient (ADC) map (f, h) is indicative of acute (6–24 h) ischemic lesions.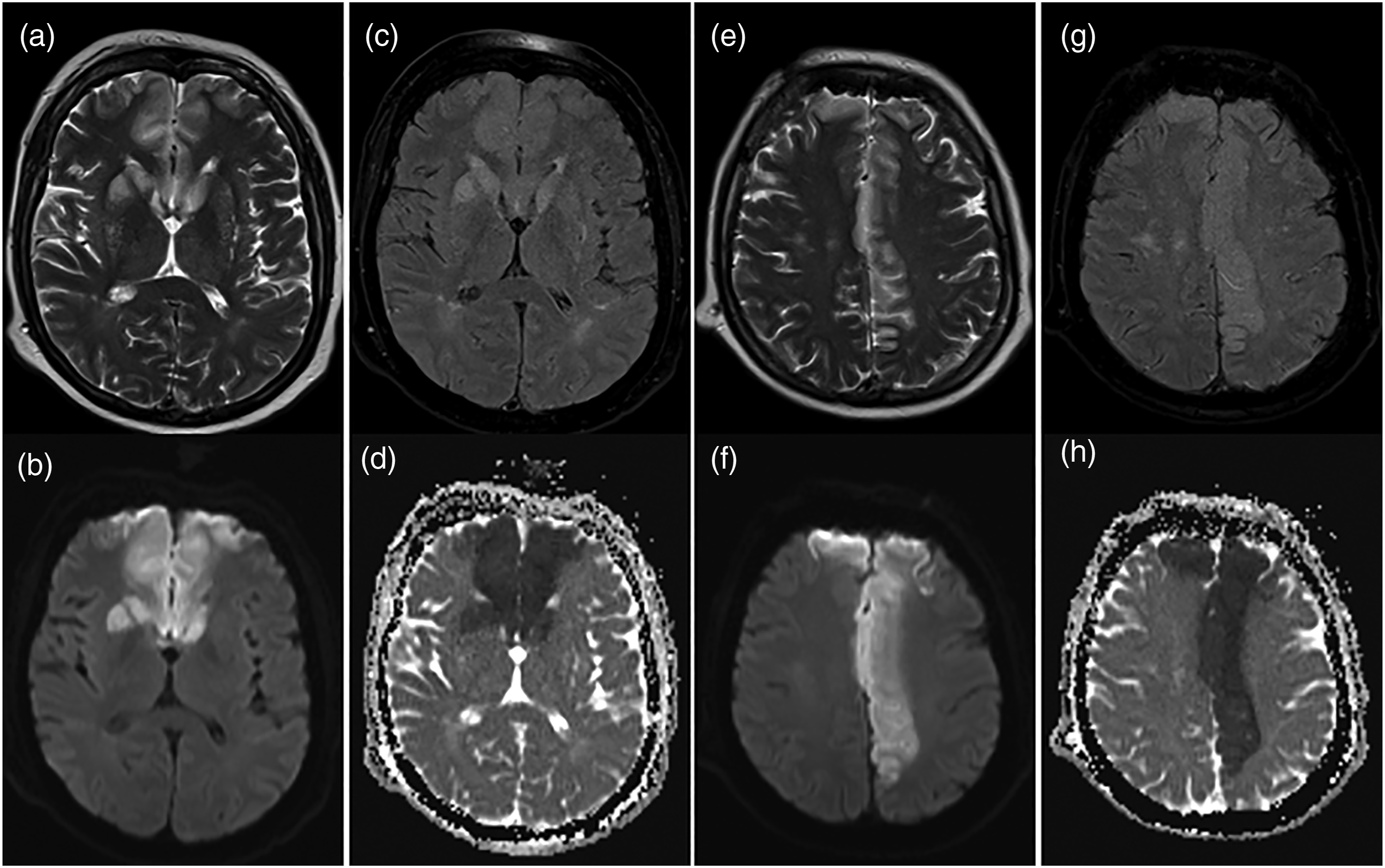
The patient underwent emergency endoscopic endonasal adenomectomy immediately after the MRI. A largely hemorrhagic lesion was identified, confirming MRI findings, and it was completely removed with consequent decompression of ACAs. On post-operative MRI, both A1 segments recovered their normal diameter and anatomical position, but no improvement of the ischemic lesions was observed (Figure 3). The histological examination identified a gonadotroph adenoma with large areas of hemorrhagic infarction (Figure 3). The post-operative course was unfavorable. The patient did not recover consciousness and died 18 days later for respiratory complications. Post-operative MRI and histopathological examination. (a) T2-w FLAIR sequence in the sagittal plane after surgery demonstrating the increase in hyperintensity due to an irreversible ischemic lesion in the anterior portion of the frontal lobes. (b). T1-w non-contrast sequence in the sagittal plane demonstrating the complete removal of the PA and the reduction of the surrounding structures, with preservation of the pituitary stalk (white arrow). (c). TOF MRA demonstrating the reduction of the superior displacement and narrowing (red arrows) of both A1 segments of the anterior cerebral arteries with a reduction of the angles formed between the internal carotid artery and the A1 segment as well (arrowheads), due to the removal of the PA. (d). Histopathological hematoxylin/eosin staining, showing a focus of residual tumor cells, surrounded by extensive necrotic tumor and large hemorrhagic areas. PA: pituitary apoplexy.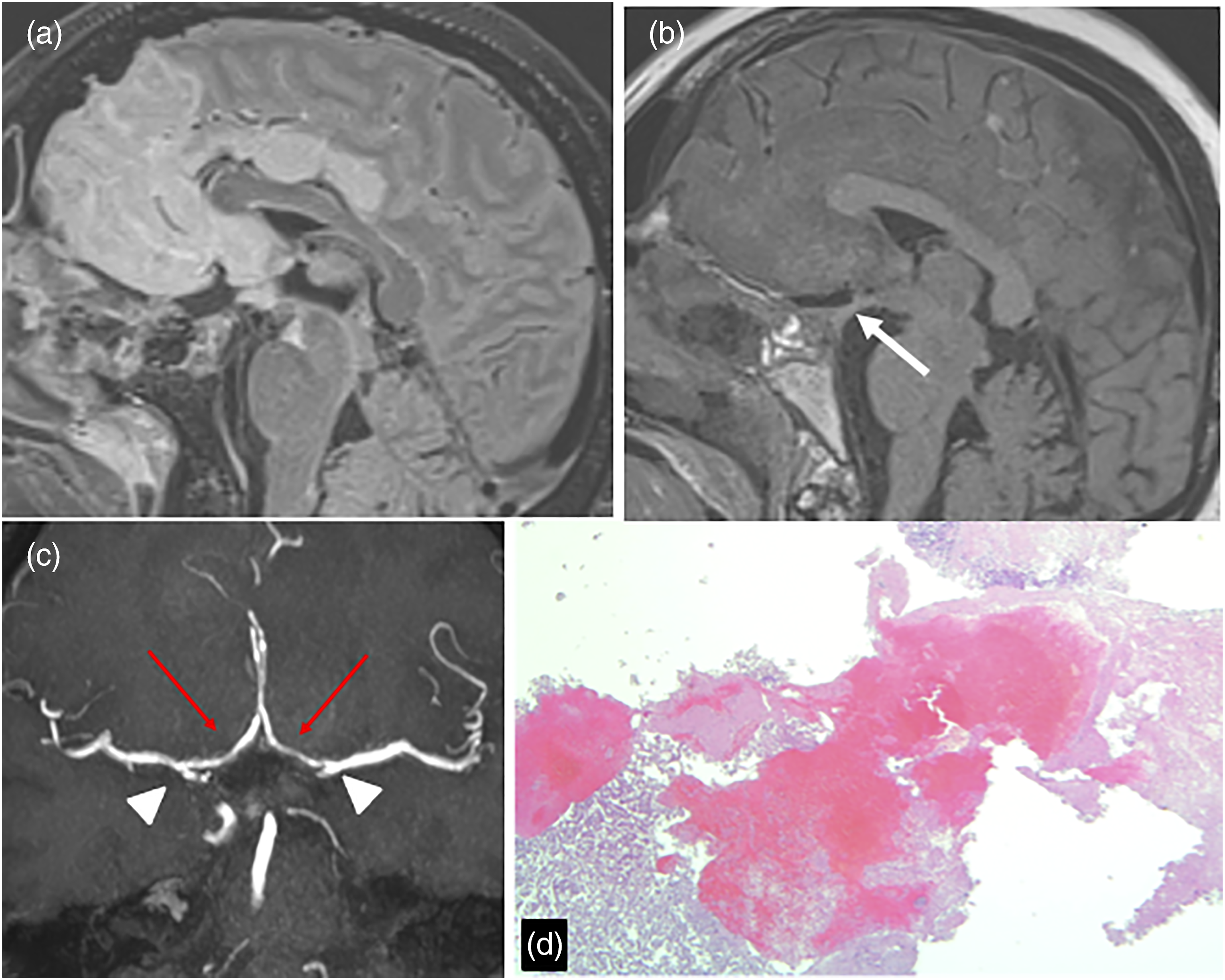
Discussion
We presented a very rare case of bilateral anterior circulation stroke caused by PA, manifesting with abrupt loss of consciousness and right hemiparesis. For the following hemodynamic instability, the proper neuroradiological investigations and surgical treatment were postponed of few hours and despite the blood flow restoration no clinical improvement was observed.
PA defines the acute ischemic or hemorrhagic pituitary infarction. It mainly occurs in patients with pre-existing pituitary adenomas or, very rarely, in normal pituitary glands.1,2 Various predisposing factors have been reported, including heterogeneous endogenous conditions, as well as medical treatments, causing the alteration of the physiological arterial brain flow and coagulation.1,2 The ischemic and hemorrhagic changes found in PA could be caused by the compression of the hypophyseal arteries, or of their branches, against the sellar diaphragm, as well as by an excessive demand of vascular supply from a rapidly growing adenoma.5,6 Most of the patients complain of no or only moderate symptoms, typically headache, visual defects, or manifestations of partial hypopituitarism that can be managed conservatively.5,6 However, in a minority of cases, PA can present with sudden onset of severe headache, vision loss, cranial nerve deficits and, even more rarely, intracranial hypertension and consciousness impairment.5,6 Brain stroke represents one of the most threatening consequences of PA, significantly increasing the risk of long-term disability and death (overall mortality has been assessed at 26.8%).3,4
In literature, 47 patients with brain ischemic stroke secondary to PA have been reported, including one from our Institution in a 15-year-old girl.3,4,7 The occlusion/critical stenosis of ICA in its supraclinoid or intracavernous tracts, secondary to the parasellar expansion of the apoplectic adenoma, was the most common cause of brain infarction associated with PA; the involvement of other vessels was much rarer.
3
Brain strokes involving the anterior-circulation territories are even rarer consequences of PA, indeed they are described only by 10 studies, for a total of 11 cases reported in literature8–17 (Figure 4). Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow-chart of the systematic review of literature performed to collect all cases of brain stroke due to anterior cerebral circulation impairment caused by PA. Literature review was performed quering MEDLINE database using keywords and MeSH terms in different combinations using the Boolean operators “AND” or “OR.” The string ((“pituitary apoplexy”) OR (“pituitary tumor”) OR (“pituitary tumour”) OR (“pituitary adenoma")) AND ((stroke) OR (infarction) OR (infarct) OR (ischemia) OR (ischemia)) AND ((“anterior circulation”) OR (“anterior cerebral artery”) OR (“anterior communicating artery”) OR (A1) OR (A2) OR (ACoM)) was entered (date last search: 2 July 2021). Search was limited to original studies written in English, performed in human subjects. After duplicate removal, articles were screened on the basis of the title and the abstract; for those deemed appropriate, the full text was obtained and reviewed to extract the data of interest. The reference list of the selected articles was examined to identify other potentially relevant studies. Only original studies or case reports describing a brain stroke due to PA involving the cerebral circulation were considered. PA: pituitary apoplexy.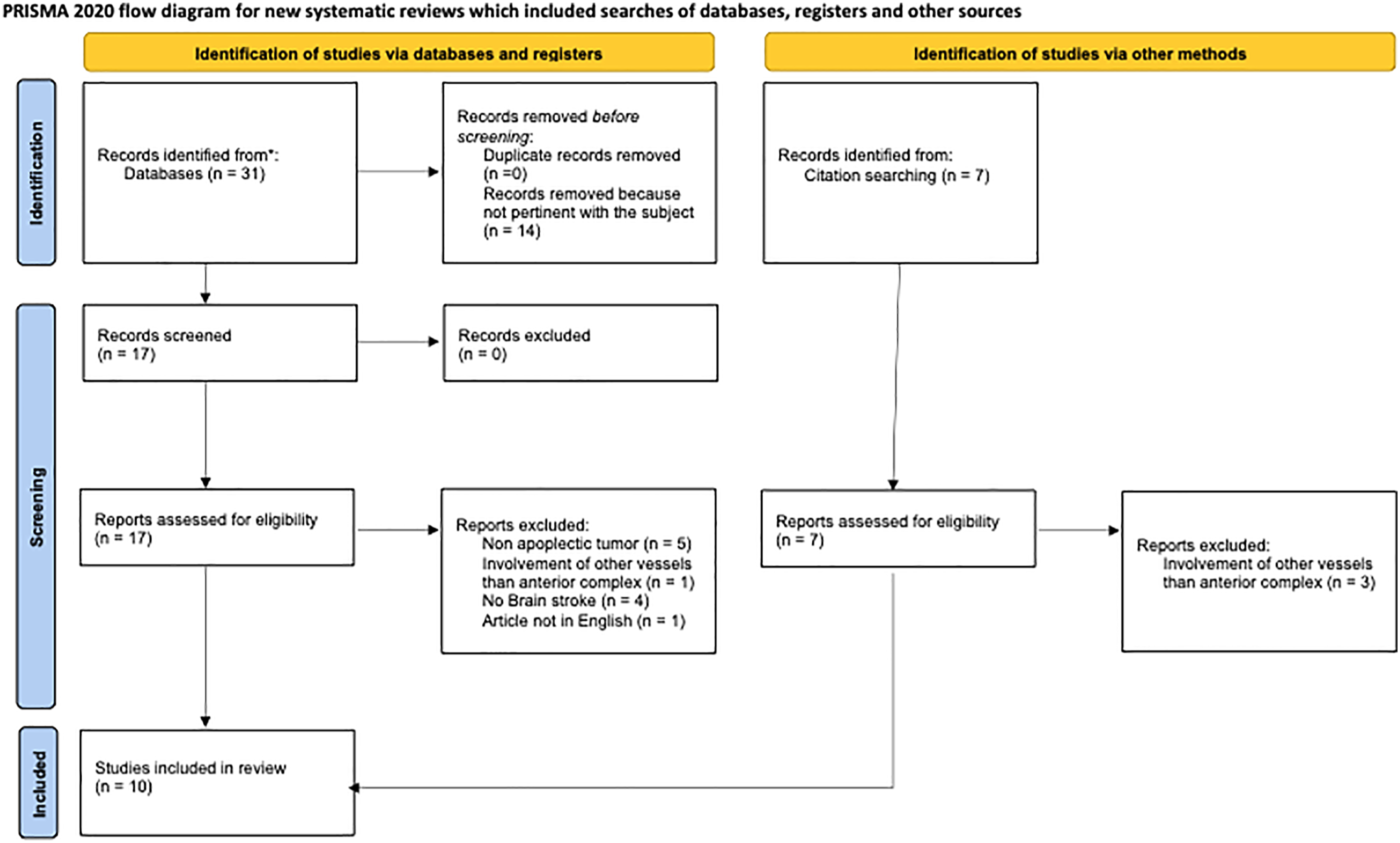
Main features of patients presenting with anterior circulation stroke and pituitary apoplexy according to systematic literature review.
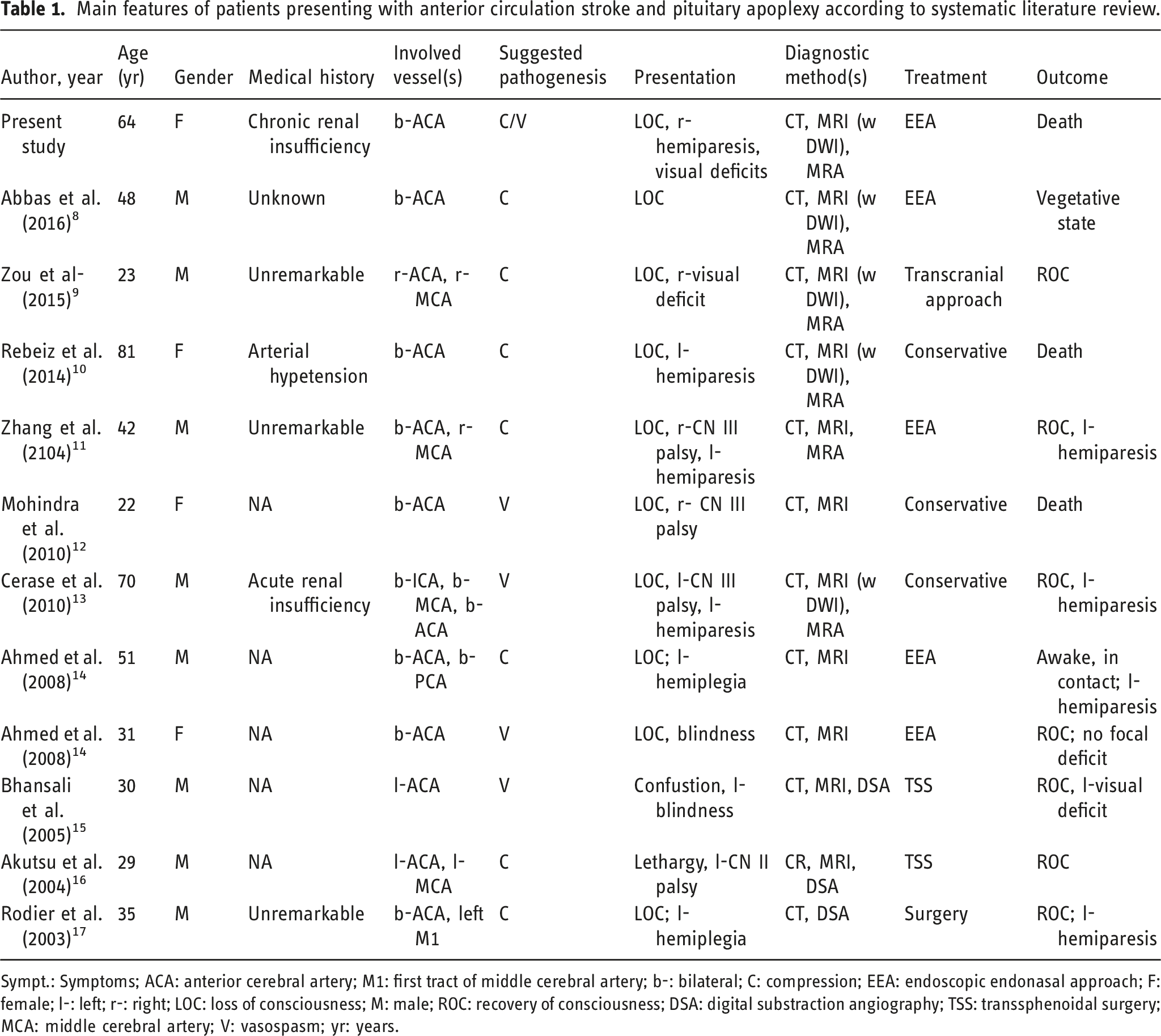
Sympt.: Symptoms; ACA: anterior cerebral artery; M1: first tract of middle cerebral artery; b-: bilateral; C: compression; EEA: endoscopic endonasal approach; F: female; l-: left; r-: right; LOC: loss of consciousness; M: male; ROC: recovery of consciousness; DSA: digital substraction angiography; TSS: transsphenoidal surgery; MCA: middle cerebral artery; V: vasospasm; yr: years.
Two physiopathological mechanisms underlying the sudden reduction in blood flow responsible for the occurrence of ischemic stroke as a consequence of PA have been proposed: (1) The direct mechanical compression of one or multiple brain vessels by the rapidly expanding pituitary apoplectic adenoma; (2) The indirect effect of vasospasm. The first hypothesis seems to explain the majority of the cases. It typically involves the ICA(s) and presents with cranial nerve deficits due to the expansion into the parasellar region of the adenoma.3,4 The second is less frequent. It can potentially occur at any level of the Willis’ circle, commonly involves multiple vessels simultaneously, and it is usually associated with a more favorable outcome.3,4 Based on our systemic review of literature of anterior circulation infarcts due to PA, the stroke was due to the direct compression with the consequent critical stenosis of the ACA-ACoA complex in seven cases, and to vasopasm affecting these vessels in the remaining four.
In our patient, we suppose that several factors could have contributed to the development of ischemic stroke. In fact, the rapid superior displacement of both A1 segments of the ACAs, demonstrated by the increased ICA-A1 angle in both sides due to the expansion of the apoplectic tumor, could have impaired the brain tissue blood supply in these territories. Moreover, the mechanical compression and the related local vasospasm could have caused the critical narrowing of these vessels, which, in conjunction with hemodynamic instability for the metabolic alterations of the patients, would have resulted in transient cerebral hypoperfusion in these territories, determining an irreversible ischemic stroke.
The most appropriate management of ischemic stroke secondary to PA remains debated. An early diagnosis is crucial, as suggested by Cerase et al., who proposed to perform an urgent MRI with specific DWI sequences in all cases of PA developing neurological symptoms suggestive for stroke. 13 Some studies advise for urgent adenomectomy to decompress the involved arteries, while others prefer a conservative management, since the post-operative risk of long-term disability and death remains significant.9,18 Despite the negative outcome of our patient, which could have been affected by the time required for hemodynamic stability restoration, based on personal experience and a vast body of literature on brain stroke, we consider that the patients’ outcome could be strongly dependent on the surgical timing from symptom onset.3,4 Therefore, we suggest that transsphenoidal adenomectomy aimed at decompression of ACAs should be performed as early as possible.
The main study limitation is the reduced number of available cases in literature, some of which do not report clinical data (i.e., patient medical history, comorbidities, and timing between symptom onset and treatment) that could be crucial in determining outcomes. The main strengths are the systemic literature review and analysis of cases of anterior circulation stroke due to a PA, and the addition of a case described in detail for clinical presenting features, timing, and course of treatment.
Conclusions
Although rare, brain ischemic stroke could be a possible risk in patients with PA. In case of sudden onset of neurological deficits and/or loss of consciousness secondary to vascular involvement by an apoplectic tumor, emergency neuroradiological imaging MRI including DWI and MRA is strongly recommended. Although the role of transsphenoidal adenomectomy in decompressing the anterior circulation vessels is debated, we suggest performing the surgery as quick as possible to increase the chance of early revascularization of ischemic brain areas, and, thus, the chance of recovery of patients’ neurological and conscious state.
Footnotes
Acknowledgments
We thank Dr. Cecilia Baroncini for her English revision of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Authors’ note
The authors state that the content of the submitting manuscript, in part or in full, has not been published previously and has not been submitted elsewhere for review. The Authors certify that this manuscript is a unique submission and is not being considered for publication with any other source in any medium.
Informed consent
Patient consent was not obtained because of patient death.
