Abstract
Fast and slow progressor phenotypes of infarct growth due to anterior circulation large vessel occlusion (ACLVO) remain poorly understood. We aimed to define clinical predictors of fast and slow progressors in a retrospective study of patients with ACLVO who underwent baseline advanced imaging within 24 hours of stroke onset. Fast progressors (ischemic core > 70 ml, < 6 hours after onset) and slow progressors (ischemic core ≤ 30 ml, 6 to 24 hours after onset) were identified amongst 185 patients. Clinical and laboratory variables were tested for association with fast or slow progressor status. In the early epoch, no significant differences were found between fast progressors and controls. In the delayed epoch, slow progressors had a median NIHSS of 14 versus 20 (p < 0.01) and MCA occlusion in 80% versus 63% (p < 0.05) relative to controls. In multivariate analyses, NIHSS (OR 0.83, 95% CI 0.73-0.95), hyperlipidemia (OR 4.24, 95% CI 1.01 – 19.3) and hemoglobin concentration (OR 0.75, 95% CI 0.57 – 0.99) were independently associated with slow progressor status. This study indicates that lower initial stroke symptom severity, a history of hyperlipidemia and mild anemia are associated with individual tolerance to ACLVO stroke.
Introduction
Anterior circulation large vessel occlusion (ACLVO) stroke is a neurological emergency that requires rapid consideration of endovascular reperfusion to achieve time-dependent, successful outcomes.1,2 Inter-individual variability of fast to slow progression of early infarct growth in ACLVO stroke is commonly observed in practice and may partly explain differences in patient response to successful reperfusion.3–6 Approximately 25% of patients with ACLVO stroke are fast progressors who present with large ischemic core volumes despite early hospital arrival due to rapid infarct growth after onset.3,7 On the other hand, approximately 55% of ACLVO patients are slow progressors who experience slow infarct growth and have small ischemic core volumes despite delayed presentation after onset.4,7,8 Therefore, early identification of fast and slow progressor phenotypes may help guide emergency triage across stroke systems of care and patient selection in future clinical trials of stroke therapies.
The spectrum of fast to slow progression of infarct growth in ACLVO stroke has been associated with vascular co-morbidities, leptomeningeal collateral grade and multimodal imaging features of cerebral hypoperfusion.5,9,10 However, collateral grading and advanced imaging are not widely available in the pre- or early hospital setting and the pathophysiological differences between fast and slow progressors remain incompletely understood. We aimed to determine whether commonly available demographic, clinical and laboratory variables can predict fast and slow progressor phenotypes of ACLVO stroke within 24 hours of onset.
Methods
This was a single-center retrospective study of patients admitted with acute occlusion of the M1 segment of the middle cerebral artery (MCA) or intracranial internal carotid artery (ICA) who underwent baseline CT perfusion or magnetic resonance imaging within 24 hours of stroke onset between 2014 and 2017. The study was approved by the University of Pittsburgh Institutional Review Board who waived the need for patient consent, in accordance with 45 CFR Part 46 and the guiding ethical principles of the Belmont Report. Determination of fast or slow progressor status was based on the time from last known well (estimated stroke onset) to qualifying imaging and ischemic core volume thresholds using previously established criteria. 7 Fast progressors were patients with ischemic core > 70 ml within 6 hours after estimated stroke onset. Slow progressors were patients with ischemic core ≤ 30 ml at 6 to 24 hours after estimated stroke onset.
We collected patient data on demographics, past medical history, statin use, admission National Institutes of Health Stroke Scale (NIHSS), BMI, complete blood count, glucose, hemoglobin A1c, cholesterol panel and ACLVO subtypes (intracranial ICA or MCA occlusion, with or without tandem cervical ICA occlusion). We used univariate comparisons and multivariate logistic regression analysis to test association of these variables with: 1. fast progressor status relative to all other patients (controls) within 6 hours after stroke onset; 2. slow progressor status relative to all other patients (controls) at > 6 to 24 hours after stroke onset; and 3. slow progressor status relative to fast progressor status (Figure 1). Continuous variables were tested for normality using the Shapiro-Wilk test and compared with the Mann-Whitney test. Categorical variables were tested using the Fisher’s exact test. Variables with p < 0.20 in univariate comparisons were included in respective multivariable regression models. An alpha-value < 0.05 was considered statistically significant.
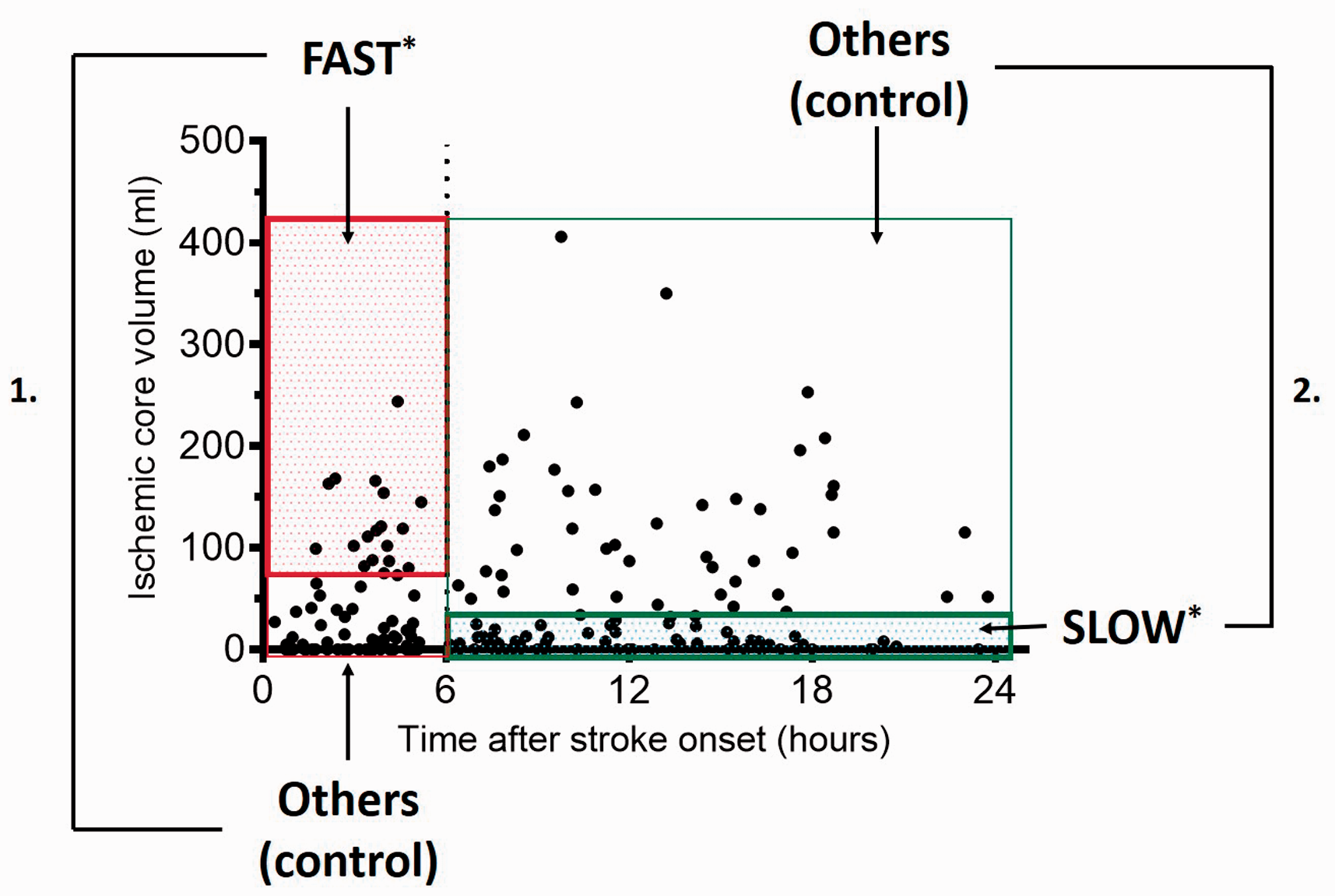
Study design overview. Scatterplot of baseline ischemic core volumes of ACLVO patients included in the study, adapted from Rocha et al. 7 Statistical analysis were setup for comparing: 1. fast progressors and all others (controls) within 0 – 6 hours after stroke onset; 2. slow progressors and all others (controls) at 6 – 24 hours after stroke onset; and 3. fast and slow progressor groups*.
Results
A total of 185 patients were included with medians (interquartile range) of age 72 (62 – 84), NIHSS 17 (12–21) and BMI 28 (24 – 32), of whom 60% were women. Patients had occlusion of the MCA in 72% or intracranial ICA in 28% of cases, and 20% had a tandem cervical ICA occlusion. Patients had a known history of diabetes in 27%, hyperlipidemia in 45% and statin use in 41% of cases. The median (interquartile range) serum glucose was 125 mg/dL (106 - 151), that for hemoglobin A1c was 5.8% (5.6 – 6.3) and low-density lipoprotein (LDL) was 90 mg/dL (63 - 115) (Table 1).
Clinical characteristics and univariate analysis.
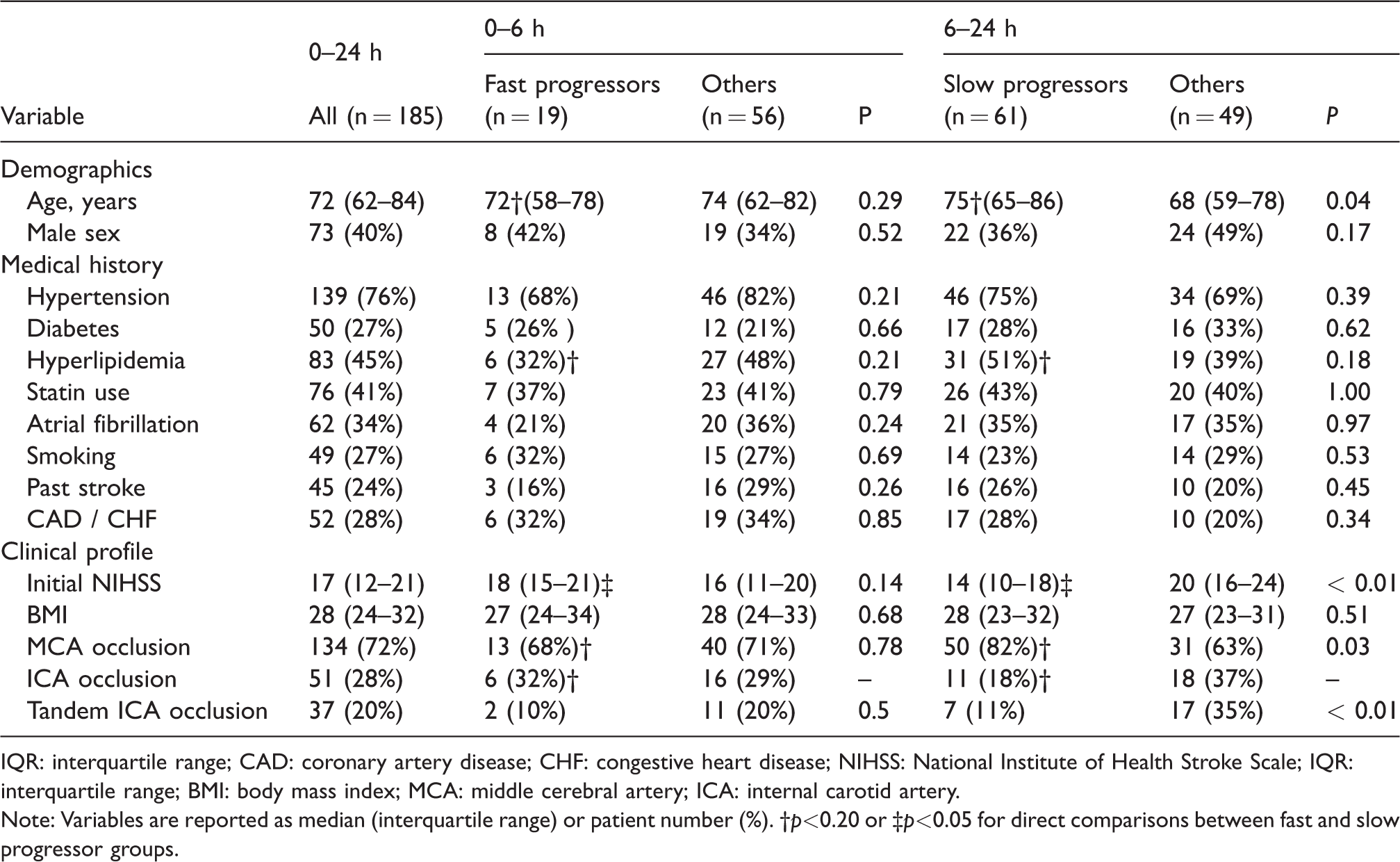
IQR: interquartile range; CAD: coronary artery disease; CHF: congestive heart disease; NIHSS: National Institute of Health Stroke Scale; IQR: interquartile range; BMI: body mass index; MCA: middle cerebral artery; ICA: internal carotid artery.
Note: Variables are reported as median (interquartile range) or patient number (%). †p<0.20 or ‡p<0.05 for direct comparisons between fast and slow progressor groups.
There were no statistically significant differences in age, sex, NIHSS, co-morbidities, laboratories or ACLVO subtype between fast progressors and controls within 6 hours after estimated stroke onset (Table 1). In the 6 – 24 hour epoch after estimated stroke onset, the median NIHSS was 14 (interquartile range, 10 - 18) for slow progressors and 20 (interquartile range, 16 - 24) for controls (p < 0.01). Slow progressors were older (median age 75 versus 68, p < 0.04), more frequently harbored an MCA occlusion (80% versus 63%, p < 0.05) and had lower rates of tandem cervical ICA occlusion (10% versus 35%, p < 0.01) relative to controls. Moreover, slow progressors had lower median hemoglobin (12.8 versus 13.7 g/dL, p < 0.05) and lower hematocrit (38.2 versus 41%, p < 0.05) relative to controls (Table 2). In multivariate logistic regression analysis, slow progressor status was negatively associated with NIHSS (OR 0.88, 95% CI 0.81-0.94) and hemoglobin concentration (OR 0.75, 95% CI 0.57 – 0.99) in the 6 – 24 hour epoch after stroke onset (Table 3).
Laboratory characteristics and univariate analysis.
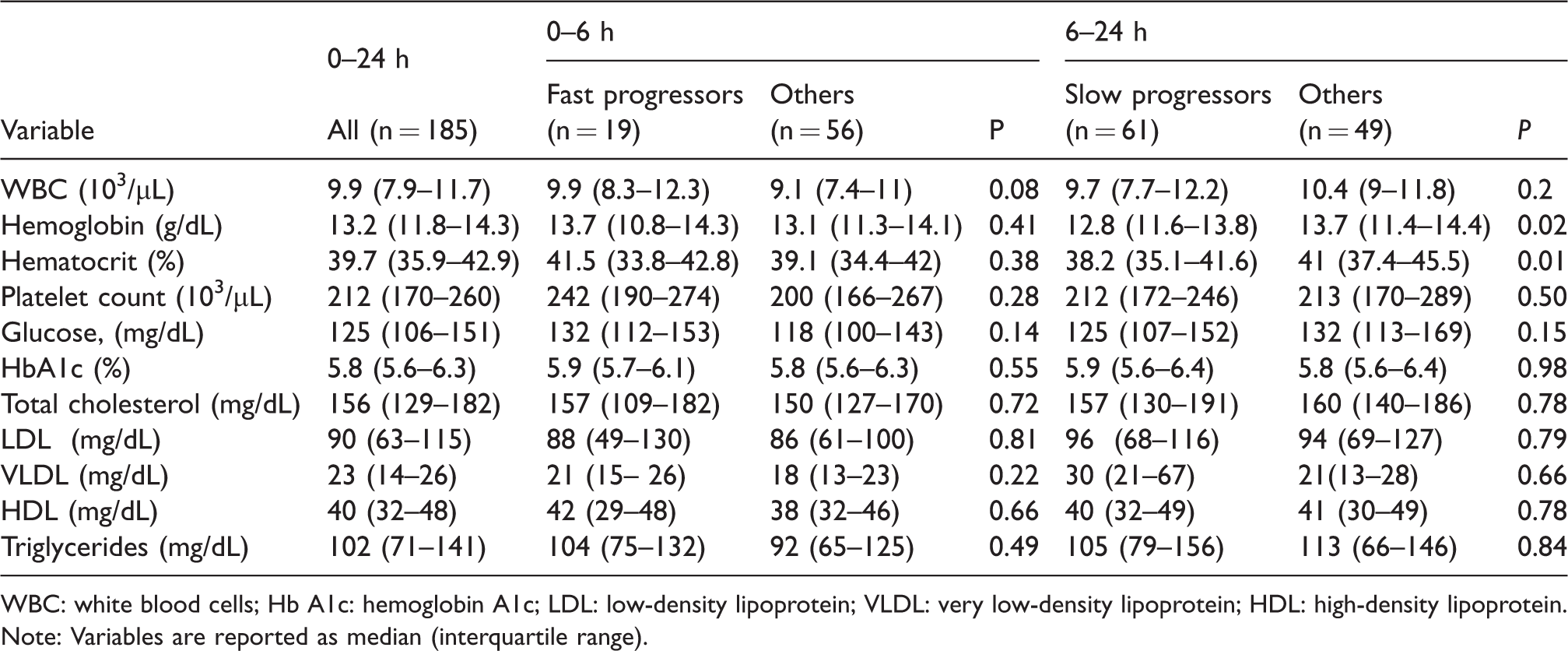
WBC: white blood cells; Hb A1c: hemoglobin A1c; LDL: low-density lipoprotein; VLDL: very low-density lipoprotein; HDL: high-density lipoprotein.
Note: Variables are reported as median (interquartile range).
Multivariate logistic regression for slow progressor status (> 6 to 24 h after stroke onset).
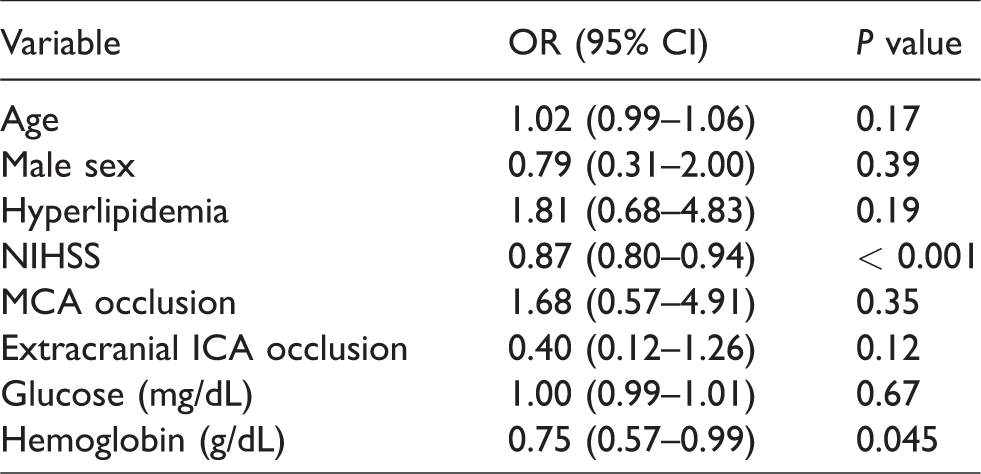
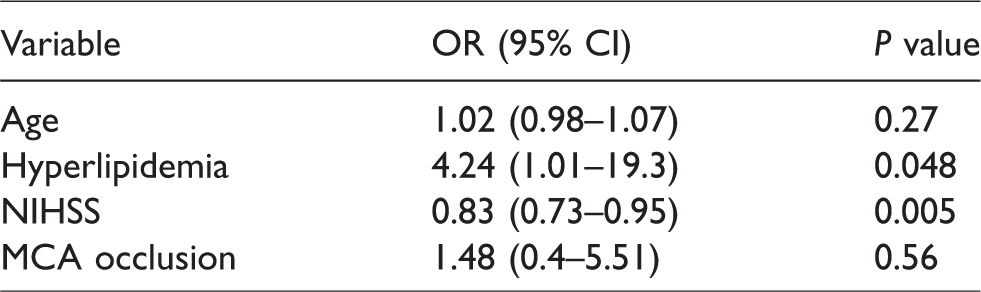
When slow and fast progressor groups were compared directly (Table 1), there was a trend for slow progressors having older age (p = 0.13) and history of hyperlipidemia (p = 0.13) but only NIHSS was found to be statistically different between the two groups (p = 0.01). On multivariate logistic regression analysis comparing the slow and fast progressor groups, hyperlipidemia (OR 4.24, 95% CI 1.03-19.3) and NIHSS score (OR 0.83, 95% CI 0.72-0.95) were independently associated with slow progressor status (Table 4). An odds ratio of 0.83 in this model indicates that every 1-point increase in the NIHSS decreases a patient’s likelihood of having the slow progressor phenotype by 17% during acute ACLVO stroke.
Multivariate logistic regression for slow progressor status (relative to fast progressor status).
Discussion
One of the main findings of this study is that the initial stroke severity measured with the bedside NIHSS had a strong negative independent association with the slow progressor phenotype of ACLVO stroke. After adjusting for covariates, we found that every 1-point increase in NIHSS was associated with up to 17% lower likelihood of slow progressor status within 24 hours after estimated stroke onset. A relatively lower NIHSS amongst slow progressors likely reflects increased ischemic tolerance to acute ACLVO in part due to better collaterals with lesser degree of hypoperfusion, 9 at least during initial presentation. This is consistent with a secondary analysis of pooled individual data from the MR CLEAN Trial and Registry which showed that ACLVO patients with good collaterals had a median NIHSS 15 relative to NIHSS 18 of those with poor collaterals. 11 Two recent studies also independently showed that slow progressors have lower NIHSS than fast progressors.5,6 While these prior studies noted univariate differences of NIHSS across collateral grade and early infarct growth rate, our multivariable analysis further establishes that the magnitude of NIHSS may help differentiate slow and fast progressors on first medical contact with ACLVO patients. Although most ACLVO patients will benefit from the earliest possible reperfusion,2,8,12 differentiating slow and fast progressors in the hyperacute setting is critical for prioritization of patient triage directly to the angiography suite or to bypass a primary stroke center.13–15 Moreover, fast and slow progressors of ACLVO stroke may respond differently to adjunctive neuroprotective interventions and early identification of these phenotypes will be important for designing future clinical trials of individualized stroke therapies. 16
The independent association of hyperlipidemia and lower hemoglobin concentration with the slow progressor phenotype raises several possible mechanisms for inducing ischemic tolerance to ACLVO stroke. First, patients with known hyperlipidemia may be more likely to take statins, which could lead to better collaterals with improved tolerance to ACLVO. 17 However, we found that rates of statin use were similar in fast and slow progressors, and across groups irrespective of time of presentation after stroke onset. Another possible explanation for the association of hyperlipidemia and slow progressor status may be that it predisposes to atherosclerotic disease which has been previously attributed to impart greater tolerance to acute ACLVO due to ischemic pre-conditioning.5,18 In our study, we found similar rates of previous stroke or coronary artery disease between fast and slow progressor groups. However, slow progressors were more likely to have mild anemia relative to controls. The latter is consistent with recent evidence that chronic hypoxemia promotes de novo collateral formation and limits subsequent infarct growth after MCA occlusion in adult mice. 19 Future studies are needed to define the role of hyperlipidemia, chronic anemia and ischemic preconditioning in the pathophysiology of fast and slow progressor phenotypes of ACLVO stroke.
In our study, slow progressors had more frequent MCA occlusion than intracranial ICA occlusion relative to fast progressors or when compared to non-slow progressors who presented beyond six hours after stroke onset. Although this trend did not reach statistical significance when adjusting for other covariates, these data support earlier findings that patients with intracranial ICA occlusion are more likely to have poor collateral status and suffer worse clinical outcomes similar to fast progressors.5,11 Slow progressors were also found to have lower rates of tandem cervical ICA occlusion relative to controls on delayed presentation. The latter group included patients with fast or intermediate progression of infarct growth. While earlier studies have found a possible association between cervical extracranial ICA stenosis and better collateral grade during acute ACLVO stroke,5,11 our data does not support this contention. Patients with tandem cervical ICA occlusion and intracranial ACLVO should be emergently considered for endovascular reperfusion given otherwise dismal clinical outcomes. 20 Future studies are needed to investigate the role of different ACLVO anatomical subtypes in determining fast and slow progressor status.
Our findings also point towards an interesting association between older age and slow progressor status relative to controls. Although these data may seem paradoxical, previous studies have demonstrated that increasing age is associated with reduced cerebral net water uptake, an imaging biomarker of ischemic edema after acute MCA occlusion.21,22 Therefore, one possible explanation for our observation is that older patients are less prone to acute cerebral edema in ACLVO 23 and experience slower progression of early infarct growth due to greater collateral capacity. 3 Prospective studies are needed to better define the role of age, early cerebral edema and collateral capacity in slow or fast progressor phenotypes of ACLVO stroke.
The main limitation of our study is its retrospective design and relatively lower number of fast progressors. This may have reduced the statistical power to find small yet physiologically important differences in tested variables between fast and slow progressors. Moreover, other putative physiological predictors of collateral status and infarct growth such as blood pressure or peripheral CO2 24 could not be tested in our cohort. Nonetheless, our data confirm and extend findings from other independent studies, therefore establishing a role for the bedside NIHSS and laboratory data in the assessment of individual ischemic tolerance to acute ACLVO. Future studies are needed with larger sample sizes to more definitely elucidate the relationship between vascular risk factors, anemia and ischemic preconditioning in fast and slow progressor phenotypes of ACLVO stroke.
In conclusion, we found that a history of hyperlipidemia, lower initial NIHSS and lower hemoglobin concentration are independent predictors of slow progressor status amongst ACLVO stroke patients, when considering commonly available clinical data in the acute setting. These findings have practical clinical implications and provide a framework for future research in the pathophysiology of fast and slow progressor phenotypes of ACLVO stroke.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TGJ: Consultant: Cerenovus (DSMB -modest), Biogen (modest). Stock (modest): Silk Road, Anaconda, Route92, Blockade Medical, Corindus, FreeOx Biotech.
Authors’ contributions
Conception, design: Rocha. Acquisition of data: Rocha, Desai, Son and Tonetti; Drafting the article: Rocha. Analysis and interpretation of data: Rocha, Desai, Son, Tonetti, Jovin, and Jadhav; Critically revising the article: Rocha, Desai, Son, Tonetti, Jovin, Jadhav; Administrative/technical/material support: Rocha, Desai, Son, Tonetti, Supervision: Rocha, Jadhav.
