Abstract
Research on social status competition among women suggests that underlying hormonal shifts associated with the ovulatory cycle systematically drive alterations in preferences and behavior. Specifically, it is proposed that the fertile window, marked by heightened estradiol and lower progesterone levels, is related to increased psychological motivation for intrasexual social comparison, leading to increasing competitiveness and jealousy. In this pre-registered, longitudinal study, 257 women provided saliva samples for hormone assays, rated the attractiveness of other women, and self-reported intrasexual competitiveness and jealousy across four testing sessions. Multilevel analyses revealed no compelling evidence for (hormone-related) cycle shifts in intrasexual competitiveness, attractiveness ratings, or jealousy. Rather, women higher in intrasexual competitiveness seem to rate other women as more attractive in general. We discuss how our results contribute to a growing body of literature suggesting that women’s social attitudes and preferences are more stable and less hormonally influenced than previously assumed.
Introduction
According to social comparison theory, certain social judgments and attitudes toward others emerge from the need to reduce uncertainty regarding one’s own position in a social group (Garcia et al., 2013). These comparisons are directed toward those threatening one’s position from a lower social status or those occupying a desired higher social position, which can lead to hostile or derogatory attitudes (Chen Zeng et al., 2022; Cheng & Tracy, 2014). Further, social comparison is necessary for making adaptive decisions about whether or not to compete and with whom, judging probability of competition success, and the energetic or tactical investment required.
Within evolutionary frameworks, women 1 compete with other women for the attention, access, and commitment of desired mates (Fisher & Cox, 2011). Because men value physical beauty in potential partners (Walter et al., 2020), social comparisons to other women tend to focus on visual appearance and perceived attractiveness (Massar & Buunk, 2010; Vaillancourt & Sharma, 2011). As represented in Figure 1, competitive attitudes that arise from intrasexual social comparison in the mating context can be conceptualized as (1) competitive motivation, a desire to gain or maintain social status over other women, and (2) jealousy, the threat of losing social status or mating resources to other women. Resulting competitive behaviors can further be categorized into whether the behavior is self-focused (self-promotion), competitor focused (intrasexual competitiveness and derogation), or partner focused (mate-guarding; Arnocky et al., 2014; Vaillancourt, 2013).
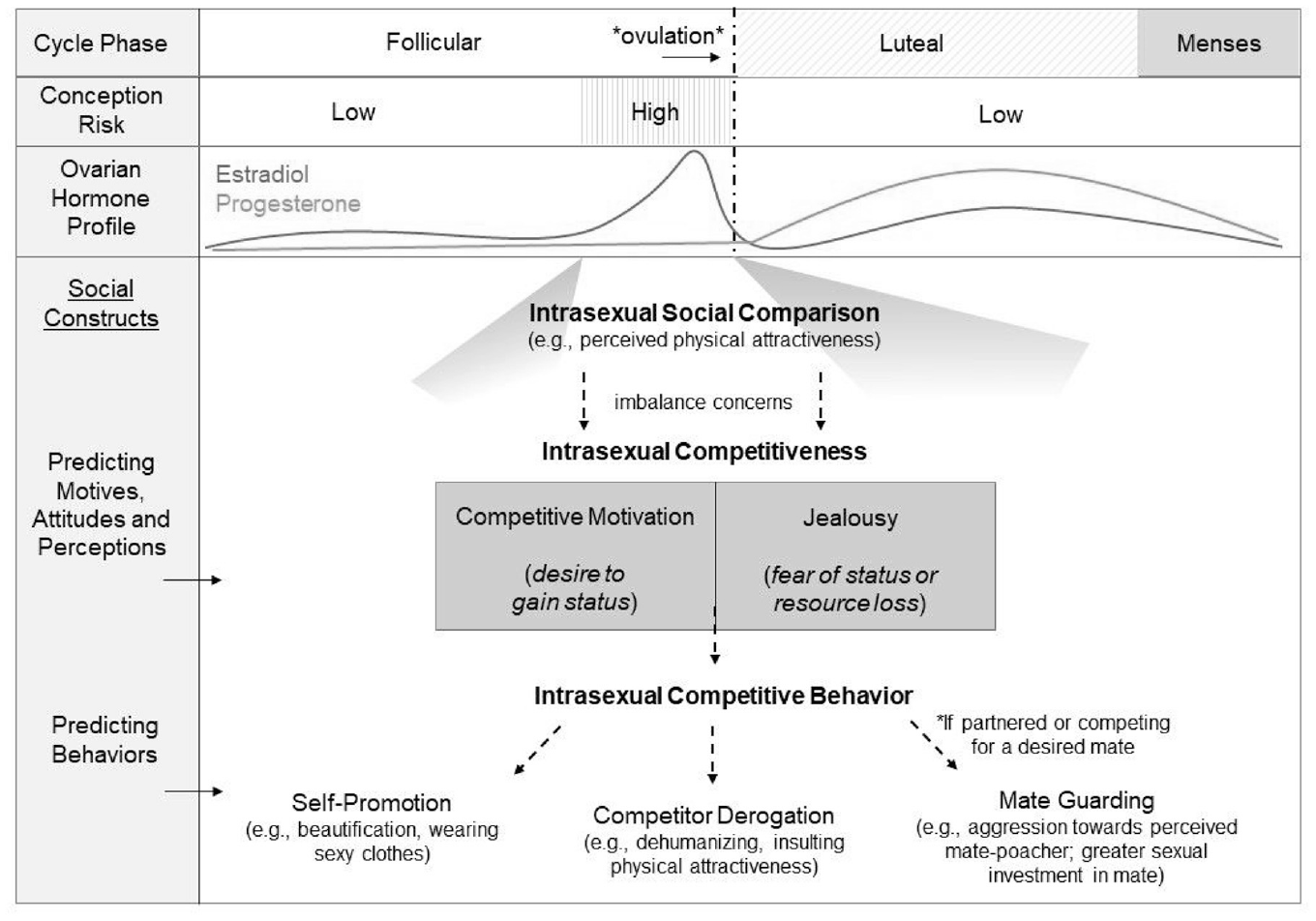
Overview of the Relationships Between the Menstrual Cycle and Intrasexual Social Comparison.
Central to the literature on intrasexual competition among women is the hypothesis that this behavior is affected by hormonal patterns underlying the ovulatory cycle, in that women derogate potential competitors when it matters most: when they are fertile and competing for mates (e.g., Fisher, 2004). Women are only fertile for up to 6 days per cycle and experience substantial hormonal changes across the cycle (see Figure 1): the fertile window is characterized by higher levels of estradiol and lower levels of progesterone. During the luteal phase (between ovulation and menstrual onset), levels of progesterone increase, whereas levels of estradiol decrease, except for a second smaller estradiol peak mid-luteal (Roney & Simmons, 2013). These hormonal changes across the cycle should regulate fertile phase shifts in intrasexual competition (Fisher, 2004).
Women’s Intrasexual Social Comparison Across the Ovulatory Cycle
Prior research has provided mixed support regarding cycle shifts in intrasexual competitive motivation (Table 1, for review see Arthur et al., 2022). When fertile, women perceive themselves as being more attractive (Arslan et al., 2021; Haselton & Gangestad, 2006; Schleifenbaum et al., 2021) and engage in more self-promoting behaviors, such as choosing to wear more sexy clothes (e.g., Durante et al., 2008; Haselton et al., 2007, but see Arslan et al., 2021). Further, fertile women seem to engage in more competitor derogation than non-fertile women, that is, in giving lower attractiveness ratings to other women (Fisher, 2004) or by higher dehumanization of other women by using more animal-related words to describe them (Piccoli et al., 2013). Further, there is evidence that fertile women self-report higher intrasexual competitiveness (Piccoli et al., 2013), which is suppressed by hormonal contraception (in partnered women, Cobey et al., 2013). Women also have shown more competitive behavior when fertile, by demanding more in ultimatum games (Eisenbruch & Roney, 2016), and by making lower offers in the dictator game, especially when the offer is made to an attractive woman (Lucas & Koff, 2013). However, results of two recent studies (Arslan et al., 2021; Hahn et al., 2016) show no evidence of fertility- or hormone-linked narcissistic rivalry or intrasexual competitiveness toward other women or any effects of relationship status or hormonal contraception thereon. Two other studies did not find significant links between cycle phase and ovarian hormone levels with behavior in economic games (Ranehill et al., 2018; Strojny et al., 2021). Using data from the same participants as the current study, Stern et al. (2021) did not find changes in perceived attractiveness of other women’s bodies across the cycle. Thus, recent research adds mixed findings and shows the need for replication studies to gain a better understanding of ovulatory cycle effects on intrasexual competition.
Overview of Methodological Characteristics and Results of Previous Studies Investigating Cycle- or Hormone-Related Shifts in Women’s Intrasexual Competitive Motivation.
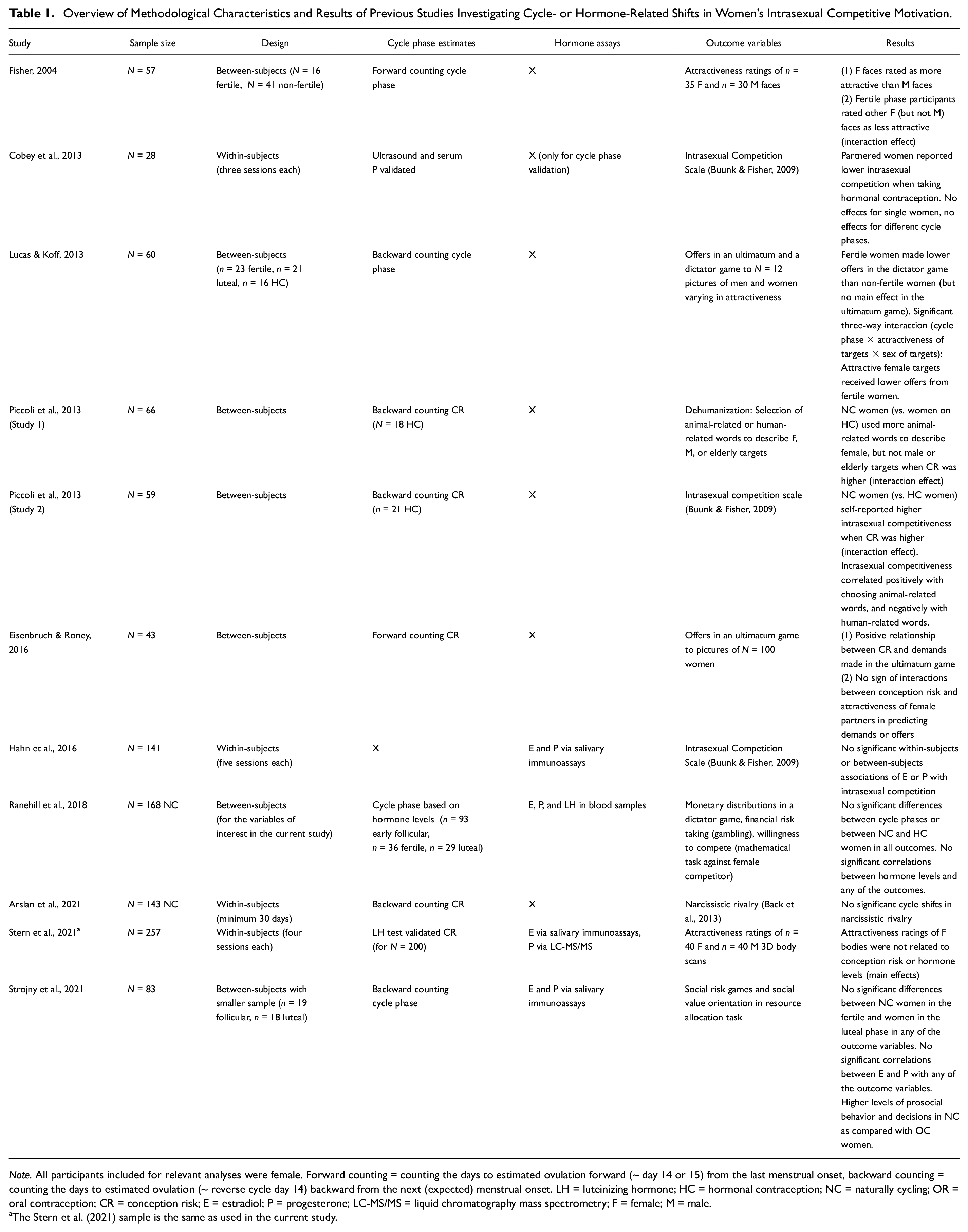
Note. All participants included for relevant analyses were female. Forward counting = counting the days to estimated ovulation forward (~ day 14 or 15) from the last menstrual onset, backward counting = counting the days to estimated ovulation (~ reverse cycle day 14) backward from the next (expected) menstrual onset. LH = luteinizing hormone; HC = hormonal contraception; NC = naturally cycling; OR = oral contraception; CR = conception risk; E = estradiol; P = progesterone; LC-MS/MS = liquid chromatography mass spectrometry; F = female; M = male.
The Stern et al. (2021) sample is the same as used in the current study.
Findings for shifting jealousy across the ovulatory cycle are even more mixed (Table 2). Three studies reported higher levels of jealousy when women were fertile or when estradiol levels were higher (Buunk & van Brummen-Girigori, 2016; Cobey et al., 2012; Geary et al., 2001). However, other studies did not find any compelling evidence that women’s experienced jealousy changes across the cycle (Arslan et al., 2021) or tracks changes in women’s estradiol and progesterone levels (Hahn et al., 2016, 2020). The association between jealousy and fertility might even go in the opposite direction as previously expected, as women may perceive higher attachment anxiety when progesterone levels are higher (in women’s luteal phase) to maintain the strength of romantic relationships while the body prepares for pregnancy (Reynolds, Makhanova, et al., 2018).
Overview of Methodological Characteristics and Results of Previous Studies Investigating Cycle- or Hormone-Related Shifts in Women’s Jealousy.
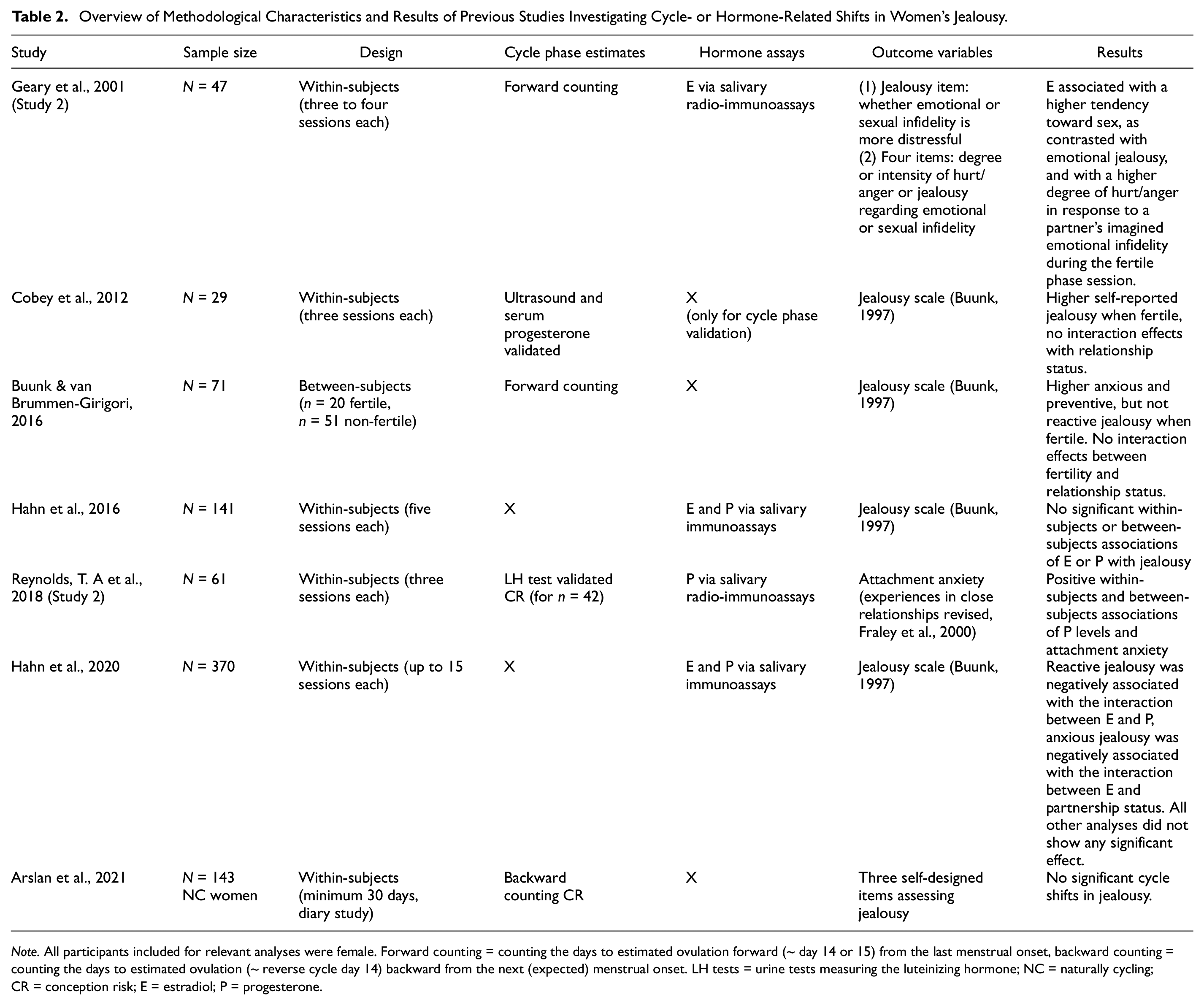
Note. All participants included for relevant analyses were female. Forward counting = counting the days to estimated ovulation forward (~ day 14 or 15) from the last menstrual onset, backward counting = counting the days to estimated ovulation (~ reverse cycle day 14) backward from the next (expected) menstrual onset. LH tests = urine tests measuring the luteinizing hormone; NC = naturally cycling;CR = conception risk; E = estradiol; P = progesterone.
Methodological Criticism of Previous Studies
Mixed results in prior research could be due to critical methodological variability in statistical power, cycle-phase determination, or hormone measure validity. During the recent years, ovulatory cycle research received criticism addressing methodological shortcomings. Most of the previous studies were potentially underpowered, and did not directly assess hormone levels, although hormones should regulate cycle shifts (Gangestad et al., 2016). Further, recent research casts doubt on how well salivary immunoassays (that are usually used to analyze hormone levels) reveal peaks that correspond to conception risk-estimated fertility windows, and suggests the use of liquid chromatography mass spectrometry (LCMS) as a potentially more valid alternative (Arslan et al., 2022; Schultheiss et al., 2019). Some studies relied on between-subjects designs (to measure a within-subjects effect) and on unvalidated self-reports of women’s position in their cycle to estimate the fertile window. Altogether, there is a need for large-scale studies in this research area that replicate and extend previous studies while overcoming methodological shortcomings.
In most of the previous studies summarized in Tables 1 and 2, intrasexual competitiveness and jealousy were measured through self-report inventories, initially capturing trait-level assessments (Buunk, 1997; Buunk & Fisher, 2009), that were adapted to capture state-level attitudes. Although adapting trait measures to capture cyclical variation is controversial, most previous studies reported positive findings for cycle shifts in these self-report measures. An additional way of measuring intrasexual competitiveness has been the assessment of attractiveness ratings of other women’s faces or bodies (Fisher, 2004; Stern et al., 2021, the latter using data from the same participants as the current study), as heightened competitive motivation would be marked by judging other women as less attractive (Fisher, 2004). Although the construct validity of that assumption has not been well established, same-sex facial attractiveness ratings as a measure of intrasexual competition are common practice (e.g., Fink et al., 2014; O’Connor & Feinberg, 2012; Sulikowski et al., 2022) and a direct replication on the influential pioneer study by Fisher (2004) has not been reported.
In the current study, we aim to conceptually replicate prior studies by directly addressing previous criticisms by employing a longitudinal design, validated fertility (conception risk) estimates, direct hormone assessments, and by recruiting a large sample size and employing Open Science practices. While cycle shifts in self-promotion seem to replicate in more recent studies, the evidence for intrasexual competitiveness and jealousy is mixed and uncertain. With this study, we aim to answer the research question whether women’s intrasexual social comparison systematically changes across the cycle, and in association with cycle-based intraindividual hormone fluctuations. By investigating these questions, we aim to gain a better understanding of (variation in) women’s social behavior.
Hypotheses
Given that most previous studies provided support for cycle shifts in women’s intrasexual social comparison (Tables 1 and 2), we expect cycle shifts in women’s intrasexual social status perceptions with the following preregistered hypotheses: 2
We define higher conception risk, the indicator of fertility, as (1) an estimated conception probability on a specific cycle day and (2) estradiol-to-progesterone ratio, and test our hypotheses for both predictor variables, as well as regulations of conception risk by assessed hormones. Unfortunately, we assessed estradiol levels via salivary immunoassays, as there was no available protocol for LCMS analyses (but progesterone levels were assessed using LCMS). We also preregistered hypotheses regarding potential mate attraction shifts that are not directly relevant to the current article. For reasons of transparency, we report all details and results in the Supplementary Material.
Method
This study was preregistered before data collection (https://osf.io/s7h93/). Open data, analysis script, and material are also provided (https://osf.io/pwy4a/). All participants signed a written consent form and the local ethics committee approved the study protocol.
Participants and Recruitment
We recruited 282 heterosexual female participants, of which 257 (aged 18–35 years, M = 23.2, SD = 3.3) finished all sessions and were therefore included in further analyses. 3 The dropouts were women who only attended one or two sessions (for details see Supplementary Material). Participants had to fulfill the following criteria: female, 18 to 35 years old, naturally cycling, and regular cycle length 25 to 35 days during the last 3 months (for details see Supplementary Material). All participants were recruited via flyers or a participant database at a German university and were of European origin. Our sample size largely exceeds the size required to achieve 80% power given a within-subjects design and anticipated main effects of moderate magnitude (Cohen’s d = 0.5; sessions = 96; minimum N = 48), as suggested by guidelines for sample sizes in ovulatory shift research (Gangestad et al., 2016). The current study has sufficient power to detect much smaller effect sizes with N = 257, four testing sessions per participant (=1,028 sessions) and using luteinizing hormone (LH) tests to validate scheduling, even if we restrict our sample regarding relationship status or women with validated conception risk estimates. Expected medium-sized two-way interaction effects will require about four times the suggested sample size (=384 sessions; Fleiss, 1986), which the current study still largely exceeds. Given the sample sizes and designs of previous studies (see Tables 1 and 2), our study should at least have sufficient test power to detect previously reported effect sizes.
Procedure
All participants took part in five individually scheduled sessions. In the first introductory session, participants received detailed information about the general procedure. Average cycle length as well as the dates of the last, the penultimate, and the next menstrual onset was assessed to plan the dates of the next sessions. Sessions two to five were computer-based testing sessions, scheduled based on backward counting and the observed LH test surge (for details see Supplementary Material). All participants completed two sessions in their expected fertile phase (late follicular) and two sessions in their expected luteal phase (mid-luteal and premenstrual). Scheduling was validated via LH test results and via following up to the day of the next menstrual onset. The starting session for each participant depended on their current cycle phase at the introductory session and their personal schedule (134 participants started testing when fertile, 123 in their luteal phase).
To control for effects of diurnal changes in hormone levels, all sessions were scheduled between 12 pm and 6 pm. Participants first completed a screening questionnaire, assessing their eligibility, control variables for saliva sampling (Schultheiss & Stanton, 2009), and the intrasexual competitiveness and jealousy items. Next, saliva samples were collected via passive drool. Then, participants rated all faces in a randomized order. Participants were instructed to evaluate all stimuli as they perceived them “in that moment,” independently of their own current relationship status, sexual orientation, general interest in other men, or ratings in previous sessions.
All data in the lab were conducted using the open source framework Alfred (Treffenstaedt & Wiemann, 2018). Participants also had to complete other tasks as part of a larger study that were randomized between participants and sessions (for details on all data see https://osf.io/dwcsm/). Upon completion of all sessions, participants received a payment of €60 or course credit.
Measures
Conception Risk
Participants’ conception risk (see Supplemental Table S1A) was assigned based on highly sensitive (10 mIU) LH urine tests from MedNet GmbH. Participants started LH testing after menstruation and continued until a positive result was followed by 2 days with negative tests (as suggested by Roney, 2018). Participants were provided with a minimum of 10 LH tests each and provided daily pictures of the tests to the investigators. Based on LH test results (e.g., negative tests only) or irregular cycles (>40 days, <20 days), 22% of all participants (n = 57) were excluded for all conception risk analyses (as conception risk cannot be reliably assigned). Details can be found in the Supplementary Material. All 257 women were included in the hormone analyses.
Hormone Measures
We collected four saliva samples from each participant (one per testing session). Contamination was minimized by asking participants to abstain from eating, drinking (except water), smoking, chewing gum, or brushing teeth for at least 1 hr before each session. The samples were stored at −80°C directly after collection until shipment on dry ice to the Kirschbaum Lab at Technical University of Dresden, Germany (one freeze-thaw cycle), where progesterone, testosterone, and cortisol (the latter two not relevant to the current study) were assessed via LCMS (Gao et al., 2015). Since the lab had no valid protocol for LCMS analysis of estradiol levels, the samples were reanalyzed for estradiol using the highly sensitive 17β-estradiol enzyme immunoassay kit (IBL International, Hamburg, Germany). The lab reported that their procedures yield Coefficients of Variation <11%. There was a significant large association between conception risk and estradiol/progesterone (E/P; β = 0.71, 95% CI = [0.41, 1.01], t = 4.24, p < .001), validating our primary hormone measure.
We centered all hormone values on their subject-specific means and scaled them afterward (i.e., divided them by a constant) to facilitate calculations in the linear mixed models (Figure S1). This is a common procedure to isolate effects of within-subject changes in hormones (e.g., Stern et al., 2021). This procedure did not change any findings compared to analyses with untransformed or log-transformed hormone values.
Attractiveness Ratings
Forty male and 40 female faces (front pictures with neutral facial expressions, all participants wearing white shirts) from the London faces database (DeBruine & Jones, 2017) were randomly selected (balanced across sex) and presented. Faces were rated on attractiveness (“How attractive is this person?”) on 11-point scales from −5 (extremely unattractive) to +5 (extremely attractive). Ratings from two sessions (from two different participants) are missing due to technical problems. For exploratory analyses, we took the independent attractiveness ratings on 7-point scales for female faces from n = 955 male raters that are provided with the dataset (α = .97, for details see Supplementary Material).
Intrasexual Competitiveness
Women’s intrasexual competitiveness was assessed using the Intrasexual Competition Scale (Buunk & Fisher, 2009), a self-report questionnaire that assesses attitudes of competitiveness to other women with 12 items. In an attempt to assess cycle-related shifts in these attitudes, we adapted the instructions so that participants were explicitly asked to report their feelings “for today.” Answers were reported on 7-point scales. Cronbach’s alpha was good (α = .88), all items were summed for a full-scale score and then z-scored.
Jealousy
Participant’s current levels of jealousy were assessed using the Jealousy Scale (Buunk, 1997), a self-report questionnaire assessing three dimensions of jealousy with five items each: preventive, reactive, and anxious jealousy. Again, we instructed participants to report their feelings on the particular testing day. Answers were provided on 5-point scales. Cronbach’s alphas were acceptable to excellent (jealousy full scale: α = .86, anxious: α = .91, preventive: α = .71, reactive: α = .83). The five items for each subscale were summed for the respective sub-scale score, all items were summed to obtain a total jealousy score. All (sub-)scale scores were z-scored.
Relationship Status and Partner Traits
Women’s self-reported relationship status was assessed in every testing session (“What is your current relationship status?”—“Single—in a committed relationship—in an open relationship—engaged—married”). Eleven participants (4%) changed their relationship status across different sessions; participant’s data were categorized in accordance with their relationship status on the particular testing day. Relationship status was effect coded with −1 = single and 1 = in a relationship (including all categories other than single). At the beginning of the study, 121 of the participants reported being partnered and 136 reported being single. We further assessed multiple traits of participant’s partners (e.g., attractiveness) that we used for exploratory analyses (details can be found in the Supplementary Material).
Statistical Analyses
All analyses were calculated with the statistic software R 4.1.0 (R Core Team, 2016; used packages are listed in the Supplementary Material). As we study a large number of different effects, we preregistered to adjust our alpha level to a significance threshold of p < .01, as a simple Bonferroni correction would be overly conservative. However, we later noticed that this alpha rate is in contrast to our power statement, which is based on an α = .05. Thus, we explicitly note when a finding should be considered as being significant based on our power statement (p < .05), but does not reach our predefined alpha level, potentially because of more restricted test power. All statistical tests are two-tailed. We computed multilevel regression models for all analyses; random slopes were modeled maximally following Barr et al. (2013). Reducing random slopes did not significantly improve model fit or change the results (for details see Supplemental Tables S29–S48).
Results
Preliminary Analyses
We first checked whether age and self-reported jealousy were significantly associated (Cobey et al., 2012). The correlations between age and self-reported jealousy (r = −0.03, p = .405, 95% CI = [−0.09, 0.04]), as well as age and self-reported intrasexual competitiveness (r = −0.06, p = .076, 95% CI = [−0.12, 0.01]), were non-significant. Consequentially, (as preregistered) age was not included as a covariate in further analyses.
Main Analyses
We tested whether participant’s attractiveness ratings of other women’s and men’s faces track changes in their conception risk or E/P ratio. As displayed in Table 3, there were large main effects for face sex, suggesting that women’s faces were generally rated as being more attractive than men’s faces. Contrary to our hypotheses, none of the other results were significant and all standardized effect sizes were close to zero. Neither conception risk scores, nor E/P ratio, nor their interaction with sex of the face were significantly related to attractiveness ratings. We found no compelling evidence that the relationship of conception risk and attractiveness rating is mediated by E/P ratio or intrasexual competitiveness, or that any of these effects are moderated by women’s relationship status (Supplemental Tables S2 and S4).
Multilevel Regression Analyses of Facial Attractiveness Ratings as a Function of Conception Risk or E/P Ratio and Face Sex.
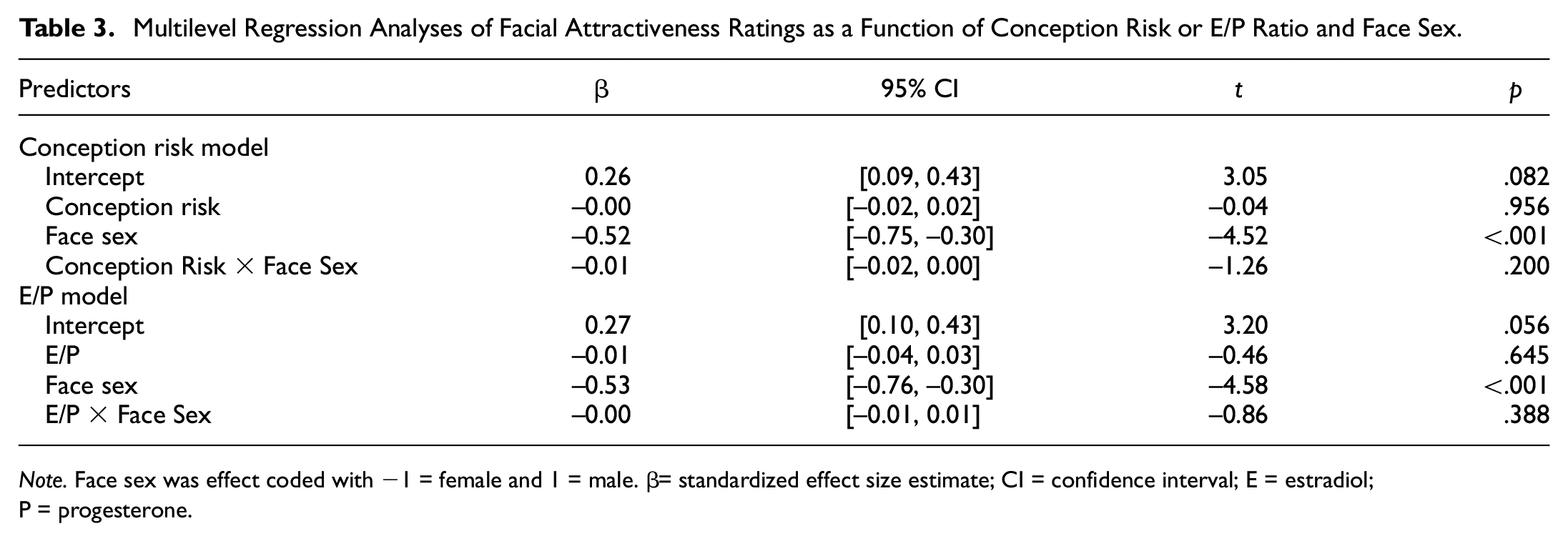
Note. Face sex was effect coded with −1 = female and 1 = male. β= standardized effect size estimate; CI = confidence interval; E = estradiol;P = progesterone.
Next, we tested whether participant’s self-reported intrasexual competitiveness tracked changes in their conception risk or E/P ratio. Conception risk or E/P ratio and their interaction with relationship status were not significant or meaningful predictors of intrasexual competitiveness scores (Table 4; Supplemental Tables S5 and S6). However, intrasexual competitiveness was significantly related to attractiveness ratings of female faces (β = 0.10, 95% CI = [0.04, 0.15], t = 3.49; p < .001), suggesting a small positive effect in that women, when self-reporting higher intrasexual competitiveness, rates other women’s faces as more attractive (independent of participant’s conception risk or E/P, Supplemental Table S3).
Multilevel Regression Analyses of Self-Reported Intrasexual Competitiveness as a Function of Conception Risk or E/P Ratio.
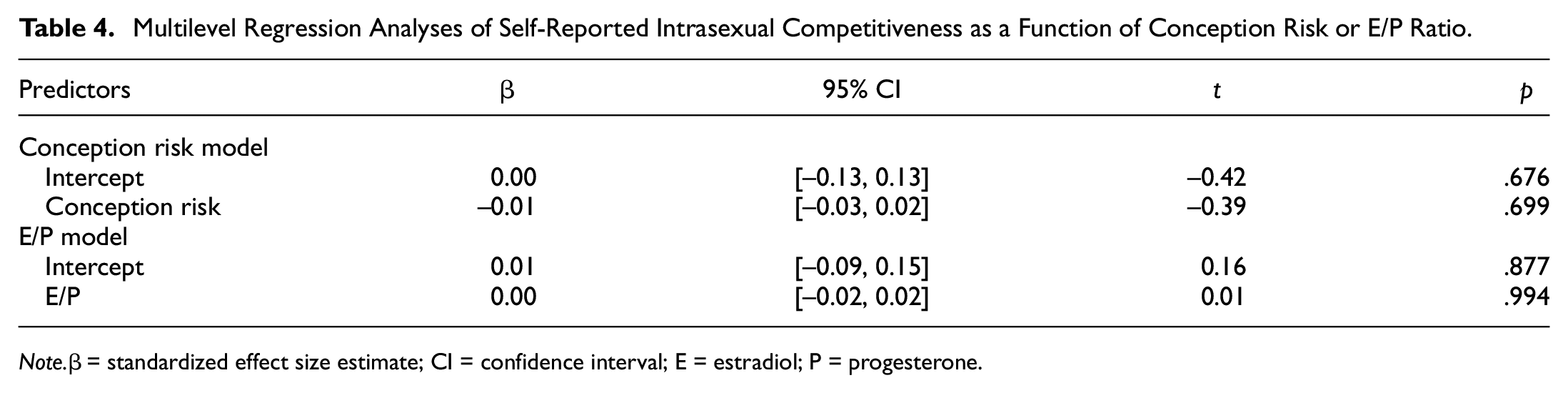
Note.β= standardized effect size estimate; CI = confidence interval; E = estradiol; P = progesterone.
For the final set of pre-registered hypotheses, we tested whether participant’s self-reported jealousy tracked changes in their conception risk or E/P ratio and whether associations are moderated by women’s relationship status. Conception risk or E/P ratio and their interaction with relationship status were not significant or meaningful predictors of jealousy (Table 5; Supplemental Table S7). However, we found a medium-sized positive correlation between intrasexual competitiveness and jealousy (r = .45, 95% CI = [0.40, 0.50], p < .001). We found comparable results when repeating all analyses with the three jealousy subscales as outcomes, rather than the full scale (Supplemental Tables S8–S13).
Multilevel Regression Analyses of Self-Reported Jealousy as a Function of Conception Risk or E/P Ratio.
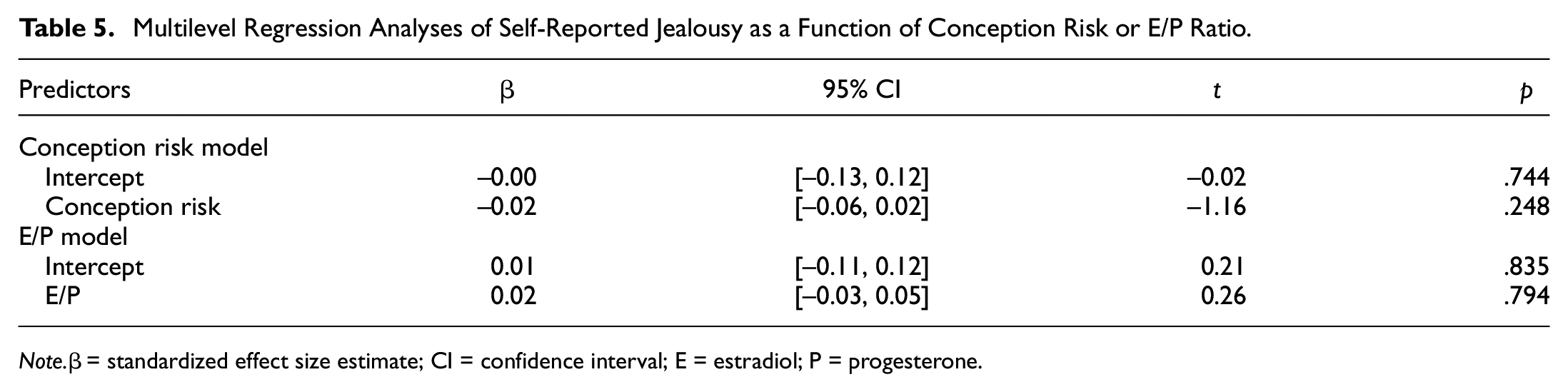
Note.β = standardized effect size estimate; CI = confidence interval; E = estradiol; P = progesterone.
Exploratory Analyses and Robustness Checks
To match other previous analyses reported in the literature, we investigated whether effects reported above change when modeling estradiol and progesterone as separate predictors, or their interaction, rather than E/P ratio. None of our investigated outcomes (attractiveness ratings, intrasexual competitiveness, or jealousy) were significantly associated with estradiol, progesterone, or their interaction. All effect sizes were close to zero. Detailed results can be found in the Supplementary Material (Supplemental Tables S17–S28). Further, all effects reported above were virtually identical when excluding outliers of hormone levels >3 SDs from the mean, and when log transforming hormone levels (see open script).
Next, we tested whether our null effects were due to trait-like rating scales by investigating between-subjects differences in mean self-reported intrasexual competitiveness and jealousy for participants who started testing in the fertile phase versus in the luteal phase, and by limiting our analyses to data from the first testing session only. None of these analyses revealed any significant effects, suggesting that our scales did not compromise within-person change across the cycle. Further, abovementioned results remained virtually identical when testing the effects of relationship status only for participants that did not change their relationship status across the study (Supplemental Tables S55–S58). Then, we tested whether partner and relationship-related attributes moderate our findings for partnered women. Partner height/weight or attractiveness did not significantly moderate any effects. Partner mate value negatively interacted with fertility indicators predicting male attractiveness ratings. Relationship quality positively interacted with E/P predicting female and male attractiveness ratings, and with self-reported intrasexual competitiveness predicting female attractiveness ratings. No other effects were significant, for details see Supplementary Material (Supplemental Tables S59–S72).
Finally, we investigated whether attractiveness ratings of other women’s faces track changes in participant’s fertility indicators when modeling an interaction effect with the face’s attractiveness rated by independent men (Supplemental Tables S73–S76). Results suggest neither significant main effects nor interaction effects of the fertility indicators. Male-rated attractiveness was strongly associated with participant’s attractiveness ratings and significantly positively interacted with self-reported intrasexual competitiveness.
Discussion
Popular theories in biosocial and evolutionary psychology suggest that women’s social status behaviors are regulated systematically by the hormonal and physiological processes underlying the ovulatory cycle. Specifically, the brief mid-cycle fertile window is thought to be associated with greater intrasexual social comparison and mate-seeking behavior. This study’s aim was to address whether or not women’s intrasexual social comparison changes across predicted phases of the menstrual cycle and in relation to phase-associated hormone levels. We found no compelling evidence that the fertile window of the cycle is related to increased derogation of other women as potential rivals, self-reported intrasexual competitiveness, or self-reported jealousy. Thus, attitudes and perceptions associated with status and competition with other females do not appear to vary systematically across the cycle. Further, we found no compelling evidence that the fertile window is associated with evaluating men’s faces as more attractive (supplement), in contrast to findings for men’s bodies, voices, or behaviors (Jünger, Kordsmeyer, et al., 2018; Jünger, Motta-Mena, et al., 2018; Stern et al., 2020). Rather, women higher in intrasexual competitiveness rated other women’s faces as being more attractive in general, regardless of conception risk or levels of ovarian hormones. This effect was stronger when the other women were rated as being more attractive by men. Further, women higher in intrasexual competitiveness also reported generally higher levels of jealousy.
Overall, we did not replicate previous studies reporting enhanced competitor derogation, self-reported intrasexual competitiveness, or jealousy in women’s fertile phase (see Tables 1 and 2). Nevertheless, our results are in line with a preregistered large diary study reporting that self-reported jealousy or narcissistic rivalry was unrelated to fertility (Arslan et al., 2021) and with results regarding evaluations of other women’s body attractiveness across the cycle from the same dataset (Stern et al., 2021). Our findings are further in line with two large-scale studies reporting no evidence for a link of ovarian hormones with self-reported intrasexual competitiveness or anxious jealousy (Hahn et al., 2016, 2020). Given that more recent studies reporting null effects (including the current study) employed larger sample sizes and used validated methods (Tables 1 and 2), the reported results cast doubt on previous findings.
Women might, contrary to prior theory, not provoke or derogate other women when they are fertile due to the high costs of victimization especially when at a higher risk for aggression from other females (Bröder & Hohmann, 2003; Hurst et al., 2017; Krems et al., 2016, 2021; Necka et al., 2018). Aggression can lead to social exclusion, decrease the desirability as a potential mate, negatively affect health and wellbeing, and might even affect the possibility to conceive (Archer, 2004; Campbell, 1999; Vaillancourt, 2013). Rather than systematically fluctuating in coordination with fertility, the present study showed that women higher in intrasexual competitiveness reported higher levels of jealousy and rated other women’s faces as being more attractive in general. These findings suggest that intrasexual competitiveness increases the salience of other women’s physical attractiveness and are in line with the suggestion that women should direct their attention and competitive efforts toward their most formidable same-sex competitors (Reynolds, Baumeister, & Maner, 2018). Comparative evaluations of physical attractiveness would be a necessary precursor. Further, in the current study, (heterosexual) women rated other women’s faces as being more attractive than men’s faces in general, which suggests that the facial pictures were evaluated based on generalized perceptions of a feminized conceptualization of beauty rather than on sexual desire, per se. Thus, context-independent pictures of faces may be less likely to capture state fluctuations in mating psychology associated with underlying hormonal shifts. Indeed, previous studies report that trait-like, stable or genetic factors account for more variation in face perceptions than contextual factors (e.g., menstrual cycle or self-perceived attractiveness; Zietsch et al., 2015).
In summary, our results contribute to a growing line of research suggesting that women do not have hormone-dependent and strongly fluctuating social preferences across their cycle. These results are similar to other recent research that has shown no cycle-relevant changes in women’s emotion recognition ability (Shirazi et al., 2010) and political orientation (Harris et al., 2014), although there is debate on mating preferences (e.g., Gangestad et al., 2019; Jones et al., 2019; Stern et al., 2019). Important cycle-related patterns of cognitions and behavior have been observed and are necessary to uncover. However, it is possible that such effects are specific to a narrower set of directly reproductively related cognitions and behaviors, such as higher sexual desire and feeling more attractive (e.g., Arslan et al., 2021; Jones et al., 2018; Schleifenbaum et al., 2021). Thus, perhaps women’s social attitudes and preferences are more stable and less hormonally influenced than previously assumed.
Limitations
There are several limitations of this study. First, we used facial photographs from a database that did not include information about the target women’s position in the cycle. Previous research suggests that women are more attentive to or respond with higher levels of jealousy to other fertile women (Hurst et al., 2017; Krems et al., 2016, 2021; Necka et al., 2018). Thus, results may differ when explicitly investigating shifts in attractiveness ratings across the rater’s and target’s cycle phases. Second, we did not investigate real social interactions. The fact that women did not derogate other women’s attractiveness when evaluating facial pictures does not necessarily mean that they would not try to derogate other women in direct social interactions, that is, during a real competition. Third, the measures we used to assess self-reported intrasexual competitiveness and jealousy were originally designed to assess traits rather than states, which might have led to little variation in these measures across the cycle (although previous studies reported effects using the exact same measures and between-subjects analyses do not suggest limited ability to detect cycle effects). Fourth, we collected our data in a WEIRD country (Germany) and are not able to generalize our results to other countries or investigate cultural differences. Fifth, recent work has pointed out that estradiol levels assessed with salivary immunoassays may not correspond to conception risk or show the expected peak in the fertile phase (Arslan et al., 2022). Thus, we cannot rule out that analyzing estradiol with a different method (e.g., potentially more valid analyses via LCMS that were not available when our hormone assays were analyzed) would have led to different results.
Conclusion
Our results suggest that attitudes and perceptions associated with social comparison to other females do not appear to systematically or meaningful vary across the cycle. Rather, women higher in self-reported intrasexual competitiveness might generally be more attentive to other women’s attractiveness. Findings from this study corroborate other recent larger sample studies to provide collectively strong evidence that women’s social behaviors and preferences are more stable and trait-like than is suggested by broad theories about cycle-related “hormonal” variability among women. Sweeping claims that hormonally mediated fluctuating preferences and “instability” of women have the potential to propagate harmful stereotypes. As such, future research should conduct specific hypothesis-guided studies using rigorous and transparent methodology.
Supplemental Material
sj-docx-1-spp-10.1177_19485506221117712 – Supplemental material for Women’s Intrasexual Competitivenessand Jealousy Across the Ovulatory Cycle: A Hormone-Based Study
Supplemental material, sj-docx-1-spp-10.1177_19485506221117712 for Women’s Intrasexual Competitivenessand Jealousy Across the Ovulatory Cycle: A Hormone-Based Study by Julia Stern, Tabea Hildebrand and Kathleen Casto in Social Psychological and Personality Science
Footnotes
Acknowledgements
We thank Paula Bange, Laura Botzet, Sarah Forsthoff, Isa Garbisch, Kim Gloystein, Salome Johannigmann, Indra Kirchberg, Laila Knapp, and Sabine Ostermann for data collection.
Handling Editor: Lowell Gaertner
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation)—Project number 254142454/GRK 2070.
Supplemental Material
Supplemental material for this article is available online.
Notes
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
