Abstract
Purpose/Aim
This study aimed to assess the effectiveness of joint lavage in managing knee osteoarthritis (OA) by evaluating its effect on pain relief, inflammatory markers, cartilage-degrading enzymes, and oxidative stress.
Methods
Seventy patients with Kellgren–Lawrence grade 2 or 3 knee OA were selected for this single-center study. Joint lavage was performed, and pain and function were measured using the visual analog scale (VAS) and Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores at baseline and 24 weeks postintervention. Synovial fluid samples were collected at baseline, before lavage, and 24 weeks postintervention. Samples were stored at −80°C and analyzed in batches to minimize variability. At the time of analysis, the samples were thawed and evaluated for levels of proinflammatory cytokines, interleukin-1β (IL-1β), interleukin-6 (IL-6), and tumor necrosis factor alpha (TNF-α), matrix metalloproteinase-3 (MMP-3), and total oxidant status (TOS), and oxidative stress index (OSI).
Results
Postintervention, VAS, and WOMAC scores significantly decreased (P < 0.001), with 100% achieving the minimal clinically important difference (MCID). Patient acceptable symptom state (PASS) rates varied: VAS (80%), WOMAC pain (50%), function (81.4%), and total (84.3%). Cytokine levels (IL-1β, IL-6, TNF-α) and MMP-3 significantly decreased (P < 0.001), along with TOS and OSI. Baseline TNF-α, IL-6, and IL-1β levels were significantly correlated with improvements in VAS and WOMAC scores. Moderate correlations were observed between reductions in IL-6/TNF-α and improvements in VAS/WOMAC. No significant associations were found between confounders and outcomes.
Conclusions
Joint lavage resulted in marked pain relief and functional improvement while significantly reducing inflammatory markers, cartilage-degrading enzymes, and oxidative stress.
Keywords
Introduction
Knee osteoarthritis (OA) is a common degenerative joint disease characterized by cartilage breakdown, joint pain, stiffness, and reduced mobility.1,2
The progression of OA is driven by both mechanical wear and biochemical factors, including inflammation. Proinflammatory cytokines, such as interleukin-1β (IL-1β), interleukin-6 (IL-6), and tumor necrosis factor alpha (TNF-α), contribute to cartilage degradation by stimulating enzymes like matrix metalloproteinases (MMPs), particularly MMP-3. Oxidative stress also plays a critical role, further damaging chondrocytes and contributing to joint inflammation and degeneration.3-7
Joint lavage is a minimally invasive procedure that aims to flush the joint cavity to remove harmful debris, inflammatory byproducts like cytokines, and oxidative molecules, potentially reducing pain and improving joint function.7-9 While previous studies have demonstrated short-term symptomatic relief,9-11 the long-term effects of joint lavage on disease progression, particularly in reducing inflammatory markers, degrading enzymes, and oxidative stress, have not been well studied.
This study aims to assess the efficacy of joint lavage in modifying these local biochemical processes, offering a potential multi-targeted treatment for OA beyond symptom management.
Materials and Methods
This was a retrospective single-center study. All interventions and data were handled within the Orthopedics and Traumatology Clinics of the Bursa City Hospital. The study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. Approval for the study was obtained from the Institutional Ethics Committee on May 29, 2024 (decision number: 2024-9/5).
Patient Selection
Patients who underwent joint lavage between February 1, 2021, and September 30, 2023, were enrolled and followed up for the next 6 months. These data were retrospectively obtained from clinical records for patients. The data extraction was followed by a quality verification process, where 2 researchers independently reviewed the records to ensure accuracy in applying inclusion/exclusion criteria and completeness of the dataset.
Joint lavage was indicated for patients with persistent knee pain unresponsive to conservative treatments, such as NSAIDs and physical therapy, for at least 6 months, as well as for those who had previously received intra-articular applications such as steroids, hyaluronic acid, or PRP without experiencing adequate improvement for at least 1 year.
Eligible patients were identified based on clinical records according to the following inclusion criteria: (1) Individuals aged 50 years or older with primary knee OA, in line with the American College of Rheumatology (ACR) guidelines.12,13 (2) Kellgren–Lawrence (KL) grade 2 or 3 knees, as observed through bilateral weight-bearing radiographs (anteroposterior and lateral views); (3) visual analog scale (VAS) >40; (4) Patients experienced persistent knee pain for at least 6 months prior to enrollment; (5) Ambulatory without assistance or assistive devices; (6) No marked deformity of the knee, such as valgus or varus deformities; (7) Patients without instability associated with knee ligament dysfunction; (8) In cases of bilateral symptomatic KOA, the more painful side was selected for evaluation.
Exclusion criteria included (1) Medication use: Use of painkillers, muscle relaxants, or non-steroidal anti-inflammatory drugs (NSAIDs) within the last 6 weeks; Use of oral, intravenous, or intramuscular steroids within the past 6 months; (2) Prior interventions: intra-articular injections or physical therapy treatments within a year; knee surgery or trauma in the last 6 months; (3) Contraindications to invasive treatments, such as disorders of blood clotting, cardiopulmonary insufficiency, or local or systemic infection; (4) Neurological deficits in the lower limbs; (5) History of systemic disease, such as rheumatoid arthritis, immunologic diseases, malignancy, pseudogout, septic arthritis, or other collagen diseases; (6) Pain in the ipsilateral lower limb caused by other conditions, such as lumbar disk herniation, diabetic peripheral neuropathy, arteriosclerosis obliterans, or saphenous nerve entrapment. Following this process, patients who met all criteria and had complete variables recorded at baseline and 24 weeks post-intervention were included. After accounting for missing data and patients who did not complete the 24-week follow-up, a total of 70 patients were included in the final analysis. These patients had a median age of 58 years. The sex distribution showed that 15.7% of the patients were male (n = 11), while 84.3% were female (n = 59). The average weight of the study group was 77.9 kg with a median height of 1.72 m. The median body mass index (BMI) was recorded as 26.46 kg/m². Based on the Kellgren–Lawrence classification, 21.4% of the patients (n = 15) were categorized as grade 2, and 78.6% (n = 55) were classified as grade 3.
Study Protocol
All participants provided informed consent, which included comprehensive information about potential risks and side effects as part of routine clinical practice for invasive procedures. Upon arrival at the outpatient clinic, demographic data, such as age, sex, height, weight, and body mass index (BMI), were recorded. VAS pain scores, Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores, and other clinical data were collected as part of routine clinical practice, following standardized procedures during patient visits. Baseline clinical assessments were conducted, including the VAS, WOMAC scores, and radiographic evaluations based on the Kellgren–Lawrence (KL) grading system. These assessments were repeated at the 24th week of follow-up to monitor clinical progress. In addition, synovial fluid samples were obtained both before and at the 24th week for the laboratory analysis of cytokines and oxidative stress markers.
Medication Tracking
From the available records, it was noted that patients were allowed to use acetaminophen and NSAIDs as needed for pain management. Pharmacy data, clinical records, and patient self-reports were reviewed retrospectively to ensure that no other medications, such as additional painkillers, muscle relaxants, or corticosteroids, were used that could potentially influence the study results. This careful review ensured that only the allowed medications were taken, minimizing the risk of confounding effects from unapproved drug use.
Joint Lavage Procedure
The procedure was performed under sterile conditions by a single experienced orthopedic surgeon, following the method outlined by Ayral. 8 Patients were positioned supine on the treatment bed, knees were elevated 30 cm above the bed, and kept in a flexed position. After proper skin sterilization and draping, local anesthesia was administered at the injection site using 1% lidocaine. A 14G needle was inserted into the articular cavity through the suprapatellar bursa, adjacent to the patella, to aspirate synovial fluid (using a 10 mL syringe) for biochemical analysis. This needle was then connected to a manual pump infusion set containing sterile 0.9% saline solution. A second needle was inserted into the articular cavity, either medial to the patellar ligament, to serve as the outflow. To ensure effective lavage, the positions of the needles were adjusted as needed, the outflow needle was temporarily blocked, and the joint was passively mobilized along with manual compression of the joint cavity. A total of 2000 mL of saline was infused and drained from the knee joint using a Hemovac negative-pressure drainage system connected to the outflow system. Residual fluid was manually expelled from the joint at the end of the procedure. Finally, the entery sites were covered with sterile dressings, and the knee was wrapped with an elastic bandage for 2 hours postintervention.
Clinical Assessment
Patient outcomes were assessed using the WOMAC Numerical Rating Scale and VAS. These evaluations were performed before and 24th week after the joint lavage procedure. The WOMAC index and VAS were used to thoroughly measure changes in pain, functional ability, and overall patient satisfaction, offering a comprehensive assessment of treatment efficacy. To determine the clinical relevance of the changes in symptoms, we assessed the minimal clinically important difference (MCID) and patient acceptable symptom state (PASS). The MCID was calculated using the distribution-based approach, with a threshold of 0.5 times the standard deviation of the change in scores, and it was used to determine the achievement of clinically meaningful improvements. The PASS was assessed using an anchor question asking patients if they were “satisfied” or “not satisfied” with their current state.14-16 Patients achieving PASS were those who reported being satisfied, indicating a positive response to the treatment. This approach allowed us to evaluate both the achievement of clinical improvements through MCID and the level of patient satisfaction through PASS.
Biochemical Analysis
Fluid Collection and Processing
Synovial fluid was successfully aspirated from all participants, using standardized techniques involving passive joint mobilization and the application of negative pressure. Synovial fluid samples were collected at baseline, immediately before the joint lavage procedure, and 24 weeks postintervention. Approximately 1 to 3 mL of synovial fluid was aspirated during each collection. To manage the high viscosity of the synovial fluid due to hyaluronic acid, a hyaluronidase treatment was applied according to the manufacturer’s (Sigma-Aldrich Hyaluronidase from bovine testes) protocol 0.5 mg/mL enzyme solution of hyaluronidase from bovine testes was prepared in cold 20 mM sodium phosphate buffer (pH 7.0), with 77 mM NaCl and 0.1 mg/mL bovine serum albumin (BSA). One drop of the enzyme solution was added per milliliter of synovial fluid using a dropper. The samples were then centrifuged at 5000 rpm for 10 minutes, and the supernatants were transferred into Eppendorf tubes with a minimum volume of 0.3 mL per tube. All collected samples were stored at −80°C until enough samples were available to be analyzed in batches under identical conditions, minimizing variability. At the time of analysis, the samples were thawed and used for further evaluation of inflammatory mediators and cartilage degradation biomarkers.
Cytosine Analysis and Degradation Biomarkers with Enzyme-Linked Immunosorbent Assay
Cytokine concentrations in the synovial fluid were assessed using enzyme-linked immunosorbent assay (ELISA) kits, according to the manufacturer’s protocols (Sigma-Aldrich human ELISA kits, from Merck KGaA, Darmstadt, Germany). The cytokines measured included IL-1β, IL-6, and TNF-α. The detection ranges and sensitivity limits for the assays are as follows:
IL-1β: 0.48 to 1000 pg/mL, 0.23 pg/mL; IL-6: 1.37 to 1000 pg/mL, 3.0 pg/mL.; TNF-α: 1.65 to 400 pg/mL, 1.65 pg/mL. MMP-3, a degradation enzyme, was measured using a specific ELISA kit (Boster Biological Technology, product code EK0461). The detection range for MMP-3 was 15.6 pg/mL to 10,000 pg/mL with a sensitivity of <10 pg/mL.
Determining Oxidative Stress by Measurement of Total Oxidant Status, Total Antioxidant Status, and Oxidative Stress Index
The stored synovial fluid samples were analyzed for total oxidant status (TOS), total antioxidant status (TAS), and oxidative stress index (OSI) using the established Erel methodology. 17 A commercially available test kit (Rel Assay Diagnostics kit; Mega Tip, Gaziantep, Turkey) was used according to the manufacturer’s protocol. TAS levels were expressed in millimoles of Trolox equivalent per liter, whereas TOS levels were measured in micromoles of hydrogen peroxide equivalent per liter. The OSI was calculated using the following formula: OSI (arbitrary units) = (TOS [μmol H2O2 Eq/L]/TAS [μmol Trolox Eq/L]) × 100.
Statistical Analysis
The sample size calculation indicated that 59 patients were required in each treatment group to detect a standardized effect size of 0.4 with 85% power at a significance level of 5%. Considering a 20% drop-out/loss to follow-up rate, the total sample size was increased to approximately 70 patients. Power analysis was performed using the G*Power software which is a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. In this study, the conformity of continuous variables to a normal distribution was evaluated using the Shapiro–Wilk’s test. Continuous variables are expressed as mean ± standard deviation or median (25th percentile–75th percentile), and categorical variables are expressed as n (%). According to the normality test results, the preintervention and postintervention terms were compared using the Wilcoxon signed-rank test if the distribution was not normal. To assess correlations between changes in inflammatory cytokine levels and clinical outcome measures, Spearman’s rank correlation was performed due to the ordinal nature and potential nonnormal distribution of data. Baseline cytokine levels were divided into 3 groups based on the 25th and 75th percentile values to represent low, medium, and high cytokine levels. Differences in clinical outcome changes between these groups were analyzed using the Kruskal–Wallis test, a nonparametric method suitable for comparing independent groups without assuming normal distribution. When the Kruskal–Wallis test indicated significant differences, post-hoc pairwise comparisons were conducted using the Dunn–Bonferroni test to identify specific group differences while controlling for multiple comparisons. Logistic regression analyses were conducted to evaluate the effect of potential confounders on clinical outcomes, with both univariate and multivariable models applied to identify independent predictors. No formal adjustment for multiplicity was applied to the statistical analyses, as the primary objective was exploratory in nature, aiming to identify potential associations. All statistical analyses were performed using SPSS software (IBM Corp. Released 2012. IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY: IBM Corp.). The type I error was set at 5% for statistical comparisons.
Results
The distribution of age, gender, height, weight, BMI, and KL grade were presented in Table 1 . The mean volume of synovial fluid collected was 1.46 ± 0.60 mL preintervention and 1.14 ± 0.33 mL postintervention. Only the more painful knee of each patient was selected for intervention. No changes were observed in KL grade at the 24-week postintervention follow-up.
Distribution of Demographics.

The data are presented as median (25th percentile–75th percentile) interquartile range (IQR) formatting; mean ± standard deviation (minimum–maximum), and n (%).
IQR = interquartile range; BMI = body mass index.
Significant improvements in pain and function were observed postintervention. Both VAS and all components of the WOMAC scores, including pain, stiffness, function, and total, showed significant reductions compared to baseline (P < 0.001) ( Table 2 ).
Comparison of Preintervention and Postintervention VAS and WOMAC Scores.

IQR = interquartile range; VAS = visual analog scale; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index.
Wilcoxon Signed Rank Test; The data clearly with proper median and interquartile range (IQR) formatting for each measure.
Postintervention, synovial fluid analysis revealed substantial reductions in inflammatory cytokines, including IL-6, IL-1β, and TNF-α (P < 0.001 for each) (
Table 3
) (
Comparison of Preintervention and Postintervention Cytokine Levels in Synovial Fluid.

Data are presented as median (25th percentile–75th percentile) interquartile range (IQR).
IQR = interquartile range; IL = interleukin.
P values were calculated using the Wilcoxon Signed-Rank Test.
Comparison of Preintervention and Postintervention MMP-3 Levels in Synovial Fluid.

Data are presented as median (25th percentile–75th percentile) interquartile range (IQR).
IQR = interquartile range; MMP = matrix metalloproteinase.
P values were calculated using the Wilcoxon Signed-Rank Test.
Comparison of Preintervention and Postintervention Oxidative Stress Markers Levels in Synovial Fluid.
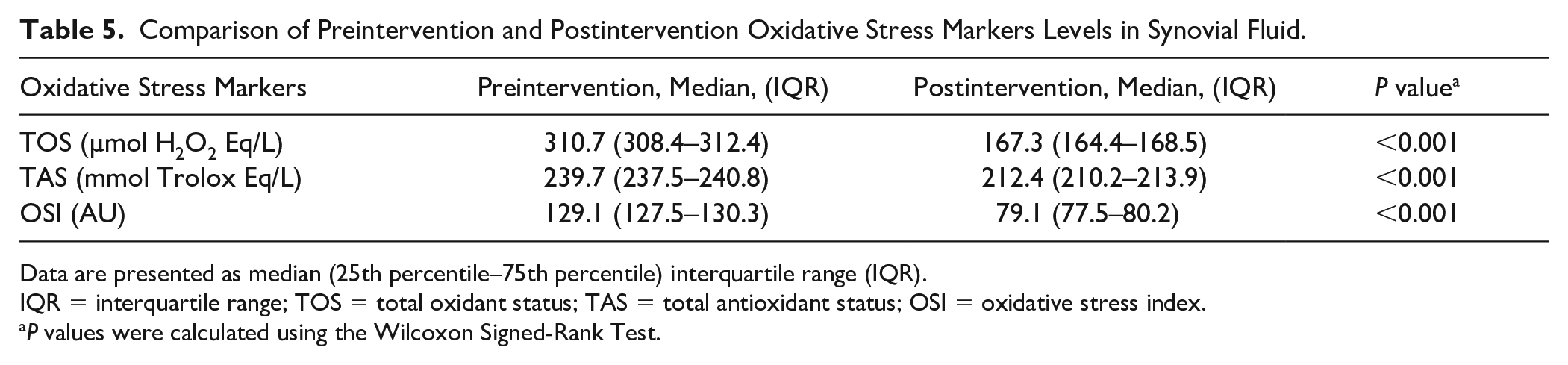
Data are presented as median (25th percentile–75th percentile) interquartile range (IQR).
IQR = interquartile range; TOS = total oxidant status; TAS = total antioxidant status; OSI = oxidative stress index.
P values were calculated using the Wilcoxon Signed-Rank Test.
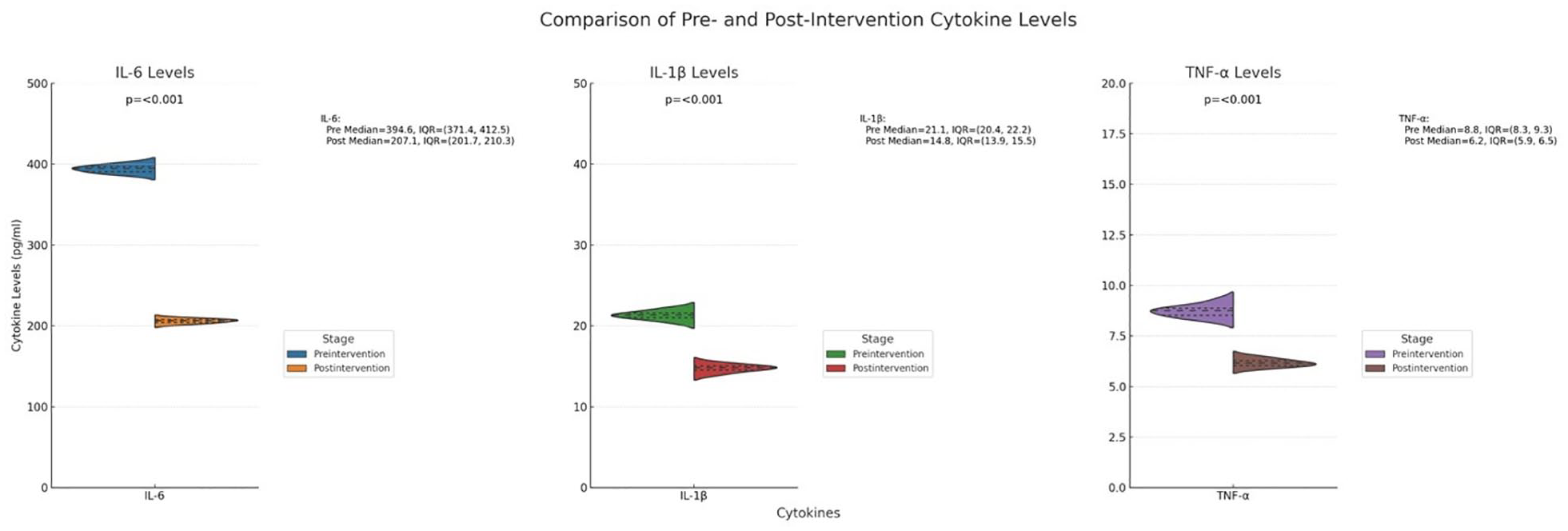
Changes in cytokine levels (IL-6, IL-1β, TNF-α) baseline 24-week follow-up. Data are shown as median with interquartile range (IQR). All cytokine levels significantly decreased from preintervention to postintervention (all P < 0.001).
All patients met the MCID thresholds across outcome measures, with 100% achieving clinically meaningful improvements in VAS and WOMAC scores. The PASS criteria showed that satisfaction rates varied across outcomes, with the highest satisfaction achieved in WOMAC Total (84.3%) and WOMAC Stiffness (81.4%) (
MCID and PASS Cut-Off Points and Achievement Rates for Outcome Measures.
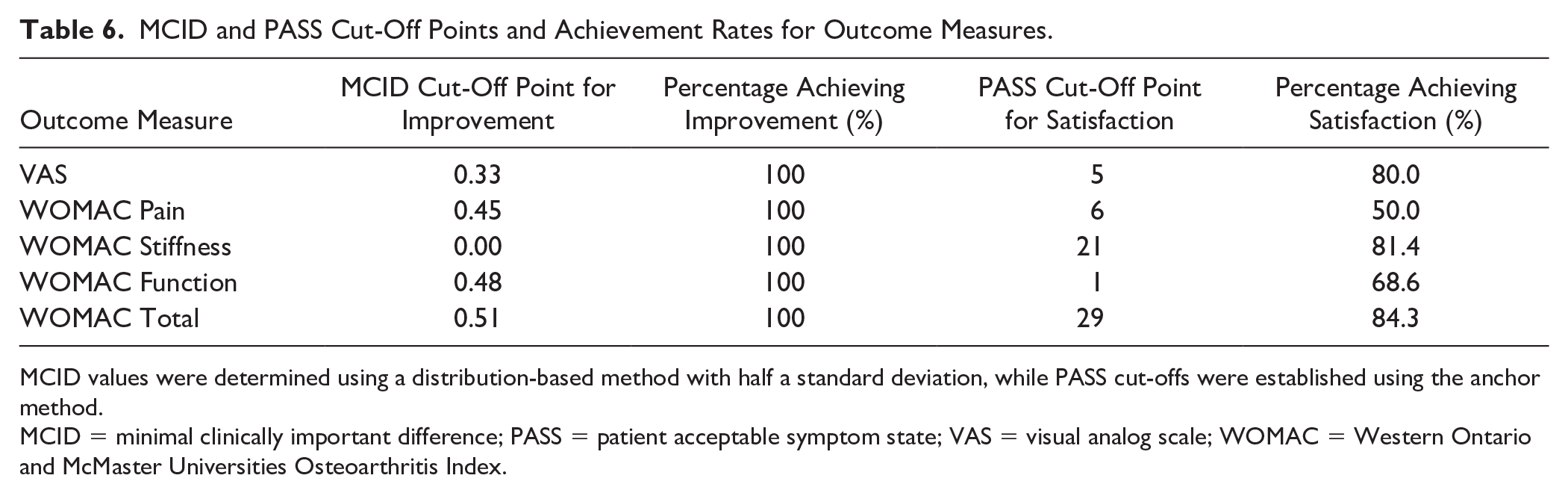
MCID values were determined using a distribution-based method with half a standard deviation, while PASS cut-offs were established using the anchor method.
MCID = minimal clinically important difference; PASS = patient acceptable symptom state; VAS = visual analog scale; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index.
In terms of correlation between cytokine changes and clinical outcomes, Spearman’s correlation analysis identified a moderate positive correlation between IL-6 and VAS scores (r = 0.35, P < 0.01), as well as between TNF-α changes and WOMAC Total scores (r = 0.41, P < 0.01) (
Correlation of Inflammatory Cytokine Changes with Clinical Outcomes in VAS and WOMAC Scores.

IL = interleukin; VAS = visual analog scale; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index. rs: Spearman correlation coefficient.
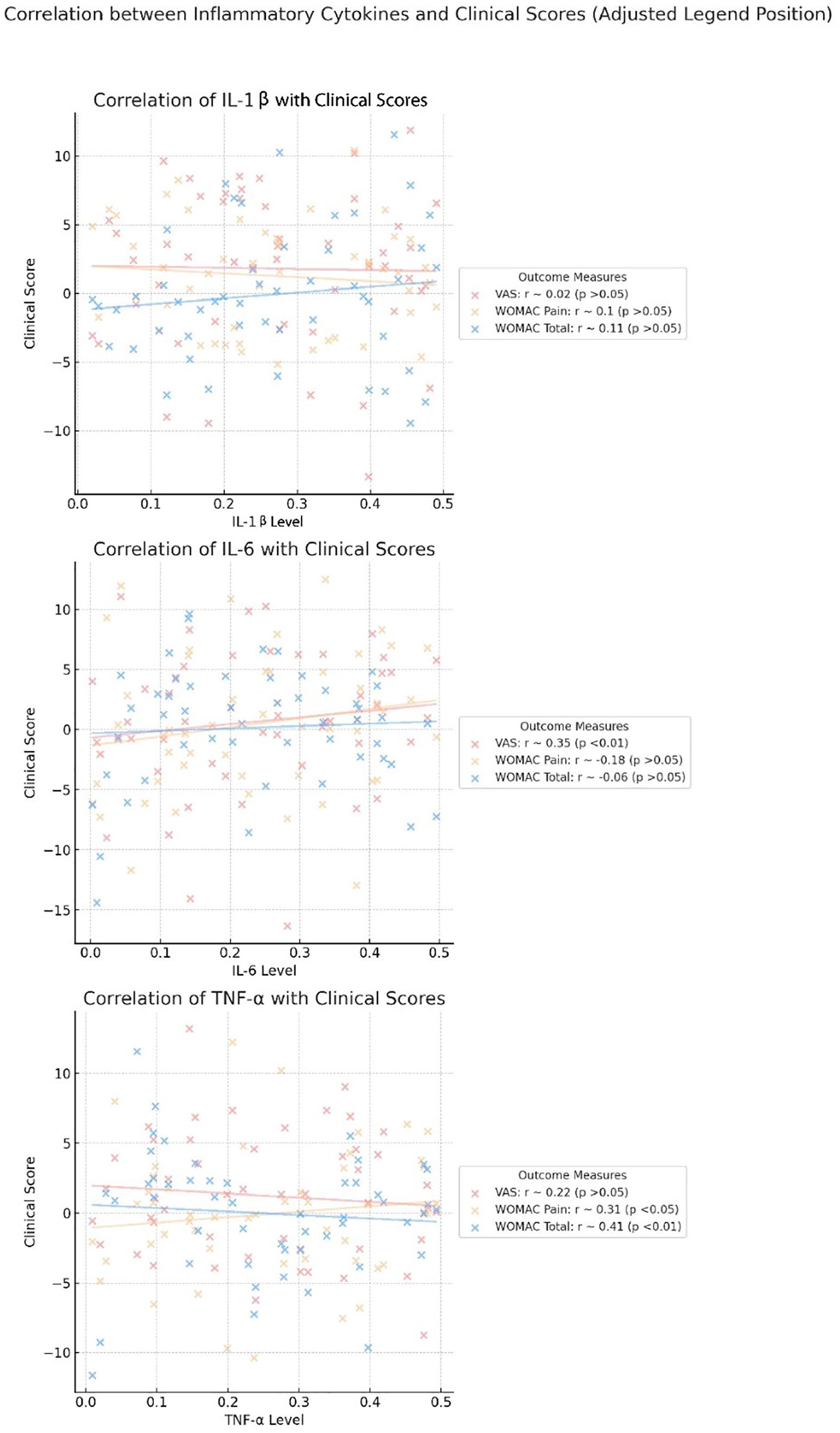
Spearman’s Correlation confounder’s adjusted cytokines changes and WOMAC and VAS.
Logistic regression analysis examined potential confounders, with univariate analysis indicating that higher pre-intervention WOMAC Function scores were associated with better outcomes (odds ratio [OR] = 1.85, p = 0.044). However, in multivariable analysis, none of the variables showed significant independent associations with clinical outcomes (P > 0.05) ( Table 8 ).
Effects of Potential Confounders on Clinical Outcomes.

Adj. OR = adjusted odds ratio; CI = confidence interval; BMI = body mass index; KL = Kellgren-Lawrence; VAS = visual analog scale; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index.
From the stratified analysis based on baseline cytokine levels (IL-6, TNF-α, IL-1β), Baseline TNF-α levels were significantly correlated with improvements in VAS, WOMAC pain, function, and total scores (P < 0.05) ( Table 9 ). Baseline IL-6 levels were correlated with improvements in VAS and WOMAC pain scores (P < 0.05) ( Table 10 ). In addition, baseline IL-1β levels were correlated with changes in VAS, WOMAC pain, and total scores (P < 0.05) ( Table 11 ).
Changes in Clinical Outcomes across TNF-α Baseline Levels.

VAS = visual analog scale; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index.
Changes in Clinical Outcomes across IL-6 Baseline Levels.

VAS = visual analog scale; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index.
Changes in Clinical Outcomes across IL-1β Baseline Levels.

VAS = visual analog scale; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index.
Discussion
Our results demonstrated that both the VAS and WOMAC scores significantly decreased following joint lavage, indicating notable reductions in pain and improvements in functional capacity. All patients achieved MCID for VAS and WOMAC scores, and a majority met the PASS criteria. These findings reflect significant clinical improvements and high levels of patient satisfaction with the intervention. In addition to these clinical improvements, our study showed a significant decrease in proinflammatory cytokines, including IL-1β, IL-6, and TNF-α, as well as a reduction in the cartilage-degrading enzyme MMP-3, which has not been previously documented in the literature for joint lavage interventions. From the stratified analysis based on baseline cytokine levels (IL-6, TNF-α, IL-1β), we found that higher baseline levels of these cytokines were significantly associated with greater improvements in pain and functional outcomes, as reflected in VAS and WOMAC scores. In addition, postintervention reductions in IL-6 and TNF-α showed moderate positive correlations with improvements in VAS and WOMAC Total scores, respectively, suggesting that both elevated baseline cytokine levels, and their subsequent reduction are linked to enhanced pain relief and functional recovery. Importantly, our analysis showed that confounders such as age, gender, BMI, and KL grade were not significantly correlated with clinical outcomes. Furthermore, oxidative stress markers measured by TOS, TAS, and the OSI were significantly reduced, suggesting that joint lavage may play a role in mitigating oxidative damage within the joint environment. This study is particularly notable as it is the first to demonstrate the effects of joint lavage on cytokines, degrading enzymes, and oxidative stress in knee OA.
Our study’s findings on pain and functional outcomes align with the majority of the existing literature on joint lavage for knee OA. For example, Li and colleagues 10 conducted a study of 82 patients, primarily using 500 mL of saline for joint lavage. They reported that more than half of the patients experienced over 50% improvement in pain at 6 months, accompanied by significant reductions in WOMAC pain scores, while in another study by Arden, 18 Seventy-one patients underwent tidal irrigation (TI) with 1 liter of saline and 79 were given intra-articular steroids. Both procedures showed short-term pain relief for at least 4 weeks; however, TI demonstrated a notably longer-lasting therapeutic effect for up to 26 weeks. Frias et al. 9 further supported these results, with 62 patients receiving joint lavage alone treated with 3000 mL of saline. Ravaud et al. 19 had 4 groups: IA corticosteroid (cortivazol), IA placebo, joint lavage, IA placebo or joint lavage, and IA corticosteroid (cortivazol). Joint lavage was performed using 1000 mL of saline. Significant pain relief was maintained for up to 24 weeks, particularly when lavage was combined with corticosteroids. Ike et al. 11 study involving 77 patients also showed that 1000 mL of saline used in TI provided superior pain relief and functional improvements over medical management, with benefits observed over a 14-week period. In a randomized controlled trial, Chang et al. 7 compared the outcomes of arthroscopic surgery with those of tidal knee irrigation in 32 patients. At the 12-month follow-up, the arthroscopy and closed-needle irrigation groups exhibited comparable outcomes. The authors concluded that most patients with non-end-stage knee OA could be effectively managed with nonsurgical treatments, suggesting that surgery may not always be necessary in such cases. Burki et al. 20 evaluated 89 patients who underwent 2-needle joint lavage with saline volumes ranging from 500 to 1000 mL. They found that nearly 60% of the patients were free from the need for surgery after 55 months, and both pain and function improved significantly over time. Parmigiani et al.’s 21 study involving 60 patients, showed no significant difference between the 2 groups; however, patients with more severe OA (KL 3) might benefit more from joint lavage with triamcinolone hexacetonide (TH). Bonakdar et al. 22 study also found that combining lavage with corticosteroids resulted in greater pain relief lasting up to 12 weeks than corticosteroid injections alone.
However, not all studies reported positive outcomes. Avouac et al. 23 conducted a meta-analysis of 6 randomized controlled trials involving 855 patients and found that joint lavage had a minimal impact on pain and function at 3 months, with no significant benefit over placebo. Reichenbach el al.’s 24 review of multiple trials similarly found only slight reductions in pain and minimal functional improvement, concluding that the clinical benefits of joint lavage might be limited, which may stem from variations in factors such as patient age, OA severity, saline volumes used, sample sizes, and study methodologies. Despite these inconsistencies, our study, like many others, demonstrated significant reductions in both pain and function, confirming the effectiveness of joint lavage as a valuable therapeutic intervention for managing knee OA.
In addition, patient-reported outcome measures (PROMs), such as VAS and WOMAC Subjective scores, often make it challenging to determine the true clinical relevance of findings. A statistically significant improvement on a PROM may not always translate into a clinically meaningful change for the patient, thereby limiting its value as robust evidence for treatment efficacy in clinical practice. To better evaluate clinical relevance beyond statistical significance, it is essential that the magnitude of improvement and patient satisfaction reach a level that patients perceive as significant.25,26 This is where psychometric measures such as the MCID and PASS are valuable. The MCID represents the smallest improvement in a PROM score that patients perceive as beneficial, signifying a clinically meaningful change, whereas PASS reflects the level of symptoms at which a patient feels well.15,27 Unlike previous studies, which have primarily focused on PROMs, our study is one of the first to assess the clinical relevance of joint lavage using MCID and PASS. Our results showed that all patients achieved MCID across outcome measures, and the majority met the PASS criteria, highlighting significant clinical benefits and high levels of patient satisfaction following joint lavage.
Furthermore, when examining confounders such as age, gender, BMI, and initial pain, our study did not observe significant independent associations between these variables and clinical improvements following joint lavage. However, it is important to note that our patient groups were relatively narrow, lacking diverse subgroups in terms of age, gender, BMI, and KL grades. Specifically, we did not have distinct cutoff groups for different age ranges, gender homogeneity, or BMI categories, nor did we have sufficient representation across all KL grades (1, 2, 3, and 4). This limitation in the distribution of subgroups may affect the generalizability of our findings, suggesting that the results might not fully reflect the effects of joint lavage across more varied populations. Future studies with broader and more well-defined subgroups will be necessary to confirm these outcomes.
In addition, we did not observe significant changes in the radiographic scores (KL grade) following the intervention, aside from the improvements in pain and function. The absence of changes in the KL score suggests that while joint lavage may effectively provide symptomatic relief, it may not significantly modify structural progression of the disease. Therefore, joint lavage may serve as a symptomatic treatment rather than a disease-modifying intervention. Long-term follow-up is crucial to determine whether joint lavage has any lasting impact on joint structure beyond short-term symptom relief. It would be valuable to outline plans for extended follow-up beyond 24 weeks to monitor potential structural improvements or stabilization in joint deterioration.
Knee OA is characterized by cartilage degeneration and chronic inflammation. As cartilage breaks down, debris, calcium-containing crystals, and matrix molecules accumulate in the joint, triggering synovitis and the release of proinflammatory cytokines such as IL-1β, TNF-α, and IL-6.28-31 These cytokines, in turn, activate MMPs, which are key enzymes involved in the breakdown of the cartilage matrix, with MMP-3 being particularly notable because of its broad substrate specificity. MMP-3 plays a pivotal role in the degradation of type II collagen, a crucial component of cartilage that accelerates cartilage degradation and further worsening OA. 32 The literature has indirectly suggested that joint lavage may reduce inflammation by clearing the joint of debris, soluble cytokines, and degrading enzymes, with studies such as those by Frías et al. 9 and Ravaud et al. 19 showing improvements in pain and function but not directly measuring the effect on inflammatory mediators. Fu et al. 33 conducted an experimental study using a rabbit model of induced OA and demonstrated a reduction in IL-1β and TNF-α levels following joint lavage, providing the first direct evidence of the anti-inflammatory effects of lavage. However, their study was limited to an animal model, which may not fully replicate the complexities of OA in humans. Our study is the first to investigate the direct effects of joint lavage in human patients with naturally occurring OA. We not only observed significant reductions in proinflammatory cytokines, including IL-1β, IL-6, and TNF-α, but also observed a decrease in MMP-3, a critical enzyme responsible for cartilage breakdown. This comprehensive reduction in both cytokines and degrading enzymes underscores the clinical effectiveness of joint lavage in mitigating inflammation and cartilage degradation, offering a more holistic approach to OA management.
Furthermore, previous studies have identified associations of many inflammatory cytokines with the destruction of cartilage, subchondral bone remodeling, synovitis, and OA severity and progression.34,35 In recent studies, the association of specific cytokines with levels of pain and clinical scores has been highlighted; therefore, determining the relationships between inflammatory biomarkers and knee symptoms has become increasingly important. Many studies have demonstrated the relationship between movement impairment, as measured by the WOMAC, and the elevation of proinflammatory cytokines.36,37 In particular, several researchers have reported associations between increased IL-6 and TNF-α levels and clinical scores of painful knee OA.38,39 However, some studies have revealed inconsistent relationships between knee joint symptoms and levels of IL-6, TNF-α, and C-reactive protein (CRP).40-43 Building on these previous findings, our study is the first to reveal notable correlations between changes in cytokine levels and clinical outcomes following joint lavage. Specifically, a moderate positive correlation was found between reductions in IL-6 levels and improvements in VAS scores, indicating that decreased IL-6 was associated with reduced pain perception. Similarly, a positive correlation was observed between changes in TNF-α levels and WOMAC Total scores, suggesting that decreases in TNF-α were linked to enhanced functional outcomes and overall symptom relief. Furthermore, we found that baseline levels of TNF-α, IL-6, and IL-1β were significantly associated with improvements in pain and function postintervention, with elevated baseline cytokine levels correlating with greater reductions in VAS and WOMAC scores. This finding underscores the dual importance of baseline cytokine levels and their reduction during treatment, as both appear to play critical roles in achieving better clinical outcomes. These correlations provide insight into the potential mechanisms through which joint lavage exerts its beneficial effects. By targeting and reducing key proinflammatory cytokines, joint lavage may mitigate the inflammatory processes driving pain and functional impairment in knee OA. Together, these results highlight the need to consider both baseline cytokine profiles and their modulation when tailoring interventions for knee OA.
However, it is important to acknowledge that OA, while a chronic degenerative disease, can exhibit variations in symptom severity over time, which may have contributed to the observed improvements in pain and function. In particular, the absence of a control group or sham procedure in our study means that we cannot rule out the influence of regression to the mean where patients experiencing heightened symptoms may naturally return to their typical state over time, independent of the treatment. Given that joint lavage was indicated for patients with persistent pain unresponsive to conservative measures, including previous intra-articular treatments, these patients may have been at a point of increased symptom intensity when treated. Therefore, while our findings suggest an association between joint lavage and reductions in pain and inflammation, we must interpret these results cautiously and recognize that they are exploratory in nature and should be viewed as an initial step toward understanding the potential impact of joint lavage in OA treatment. Future studies incorporating a control group are necessary to confirm the true effectiveness of joint lavage.
In addition to inflammatory mechanism, oxidative stress also plays a critical role in the progression of knee OA, driving both cartilage degradation and chronic joint inflammation.44,45 The imbalance between the excessive production of reactive oxygen species (ROS) and the body’s antioxidant defenses leads to oxidative damage to essential cellular components including lipids, proteins, and DNA.46,47 This damage accelerates the breakdown of cartilage and fuels synovial inflammation, creating a vicious cycle of joint destruction. ROS, such as hydrogen peroxide and superoxide anions, are generated in response to proinflammatory cytokines, further exacerbating oxidative damage and promoting the release of MMPs, which degrade cartilage matrix components.48,49 As the literature suggests, the oxidative stress burden overwhelms the joint’s natural antioxidant defenses, intensifying the destructive processes in OA. In this context, joint lavage offers a potential solution for reducing the accumulation of ROS and mitigating oxidative stress. In our study, we measured oxidative stress in synovial fluid using the TOS and OSI. Our findings demonstrate that joint lavage significantly reduced both TOS and OSI, indicating a substantial decrease in oxidative stress within the joint environment. By removing or diluting free radicals and oxidative molecules, joint lavage helps to lower the oxidative stress burden, which is a key driver of joint damage in OA. This study is the first to show that joint lavage not only reduces proinflammatory cytokines and cartilage-degrading enzymes, but also effectively addresses oxidative stress, offering a comprehensive therapeutic approach that targets both inflammatory and oxidative pathways in OA management.
This study has several strengths that distinguish it from previous research on knee OA treatments. This is the first study to demonstrate that joint lavage can effectively reduce not only proinflammatory cytokines and cartilage-degrading enzymes, such as MMP-3, but also oxidative stress markers (TOS and OSI). This comprehensive assessment provides new insights into the multifaceted mechanisms through which joint lavage may slow OA progression, while simultaneously targeting inflammation, oxidative stress, and cartilage degradation. The use of specific biomarkers to measure these processes adds scientific rigor and provides quantifiable outcomes.
Joint lavage compares favorably with other intra-articular modalities, such as corticosteroid injections, hyaluronic acid, and platelet-rich plasma (PRP).18,50 These injections provide symptomatic relief but often require repeated administration due to short-lived effects, which can carry risks such as cartilage damage and high costs. In contrast, joint lavage targets the direct removal of inflammatory mediators, cartilage debris, and free radicals, offering a multipronged therapeutic benefit. Compared to arthroscopy, which has shown limited long-term effectiveness and risks of postoperative complications, joint lavage offers a less-invasive, safer, and more cost-effective option.51,52 The ability to repeat lavage as needed or to combine it with other therapies makes it a versatile intervention for patients seeking to avoid or delay surgical procedures.
However, this study had certain limitations. This study was retrospective in nature, which brought several limitations. The use of existing clinical records limited our ability to standardize follow-up timing and ensure consistent data collection across all patients. The reliance on historical data also meant that certain variables might have been incompletely recorded or missing, leading to exclusions from the final analysis. In addition, our control over confounding factors was limited, and we could not implement real-time adjustments during patient follow-up and inability to consistently track changes from baseline to the endpoint. Moreover, the absence of imaging data, such as ultrasound or magnetic resonance imaging (MRI), to confirm and quantify the presence of synovial effusion is a limitation of our study. Effusion is often associated with active inflammation in OA, and the ability to aspirate synovial fluid from all participants may reflect a study population with a higher prevalence of effusion or subclinical inflammation. This introduces a potential selection bias, which may limit the generalizability of our findings to the broader OA population, particularly those without detectable effusion (dry knees). Future studies incorporating imaging modalities could provide a more comprehensive assessment of effusion and inflammation, enhancing the applicability of the results. A prospective study would allow for better control over the inclusion process, consistent and systematic data collection, and the implementation of standardized follow-up protocols. This would reduce biases and ensure a more rigorous assessment of the intervention’s impact. We acknowledge these limitations and suggest that future studies build on our findings using a prospective design to enhance the quality and validity of the results. The sample size, although sufficient for preliminary analysis, limits the generalizability of our findings to broader populations. Larger-scale studies with more diverse patient demographics are required to validate these findings. The lack of adjustment for multiplicity in this study may increase the risk of statistical error. Therefore, the results, especially for secondary outcomes, should be interpreted carefully. Future studies with larger sample sizes and appropriate adjustments are needed to confirm these findings. Furthermore, the follow-up period was limited to 24 weeks; therefore, long-term studies are required to assess the durability of the effects of joint lavage on both biochemical markers and clinical outcomes. In addition, while our study demonstrates short-term improvements, comparative studies are needed to assess the effectiveness of joint lavage versus other intra-articular applications, such as corticosteroids, hyaluronic acid, and PRP, as well as in comparison to arthroscopy. These studies will help clarify the long-term benefits and positioning of joint lavage within the treatment algorithms for OA. Finally, future research should aim to correlate biochemical findings with structural changes in cartilage using advanced imaging techniques to better understand the role of joint lavage in slowing OA progression.
In conclusion, our study demonstrated that joint lavage significantly reduced proinflammatory cytokines, cartilage-degrading enzymes, and oxidative stress markers in OA patients. These results suggest that joint lavage could be an effective treatment for OA by directly targeting the key pathological processes in the disease. Our findings highlight the need for further research to confirm these benefits across different OA populations and explore their long-term effects. Overall, this study provides valuable insights into the nonsurgical management of OA, offering the potential for improved therapeutic approaches and enhanced patient outcomes.
Footnotes
Author Contributions
MD and ÖCS contributed to the design and implementation of the research. MD contributed to the analysis of the results, to writing of manuscript, conceived the original manuscript, and supervised the Project.
Acknowledgments and Funding
The authors would like to acknowledge the outstanding statistical analysis of Dr Gökhan Ocakoğlu.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
This study was approved by the Clinical Research Ethics Committee of Bursa City Hospital, (date: May 29, 2024, decision number: 2024-9/5).
Data Access Statement
The datasets generated during and/or analyzed during this study are available from the corresponding author on reasonable request.
