Abstract
Objective
Supply-demand mismatch of medial femoral condyle (MFC) osteochondral allografts (OCAs) remains a rate-limiting factor in the treatment of osteochondral defects of the femoral condyle. Surface contour mapping was used to determine whether a contralateral lateral femoral condyle (LFC) versus ipsilateral MFC OCA differs in the alignment of donor:native subchondral bone for large osteochondral defects of the MFC.
Design
Thirty fresh-frozen human femoral condyles were matched by tibial width into 10 groups of 3 condyles (MFC recipient, MFC donor, and LFC donor) each for 3 cartilage surgeons (90 condyles). The recipient MFC was imaged using nano-computed tomography scan. Donor oval grafts were harvested from each matched condyle and transplanted into a 17 mm × 36 mm defect created in the recipient condyle. Following the first transplant, the recipient condyle was imaged and superimposed on the native condyle nano-CT scan. The donor plug was removed and the process repeated for the other donor. Surface height deviation and circumferential step-off height deviation were compared between native and donor subchondral bone surfaces for each transplant.
Results
There was no statistically significant difference in mean subchondral bone surface deviation (LFC = 0.87 mm, MFC = 0.76 mm, P = 0.07) nor circumferential step-off height (LFC = 0.93 mm, MFC = 0.85 mm, P = 0.09) between the LFC and MFC plugs. There were no significant differences in outcomes between surgeons.
Conclusions
There were no significant differences in subchondral bone circumferential step-off or surface deviation between ipsilateral MFC and contralateral LFC oval-shaped OCAs for 17 mm × 36 mm defects of the MFC.
Introduction
Osteochondral allograft (OCA) transplantation has been established as an effective treatment for full-thickness chondral defects of the femoral condyles.1 -6 In comparison to other cartilage restoration techniques, OCA has the benefit of being a single-stage procedure capable of treating the entire osteochondral unit without the risk of donor-site morbidity.5,7 -11 The goals of a successful OCA transplantation in the knee are to anatomically restore the articular surface of the recipient condyle with enough underlying bone to achieve stability.11 -16 Matching the differences in surface topography between donor and recipient condyles has proven to be a difficult but necessary task, as studies have shown that grafts that are 0.5 to 1 mm proud lead to as much as a 50% increase in local contact pressures.17 -21
In the knee, lesions of the medial femoral condyle (MFC) outnumber those on the lateral femoral condyle (LFC) by nearly 6-fold.22 -27 Due to the desire to match the individual variances in surface topography, most surgeons look to use an orthotopic graft (i.e., left MFC allograft for a left MFC lesion), putting MFC allografts in high demand. Statistics from JRF Ortho (data on file, Joint Restoration Foundation, Centennial, CO) indicate that 60% of OCAs requested are for MFC grafts. In contrast, 75% of grafts that are suitable and made available to surgeons are LFCs. The supply-demand mismatch in the available OCAs has led to exploration of the viability of non-orthotopic OCAs for transplant into MFC lesions.
The geometries of LFCs and MFCs have been shown to differ in shape, curvature, and size.28 -32 In addition, the articular cartilage thickness differs between LFCs and MFCs, with additional variations related to sex, race, weight, height, and applied stress during weightbearing.33 -42 Most surgeons opt for use of a single cylindrical OCA plug for lesions less than 25 mm with predictable restoration of the surface contour and articular step-off.37,43,44 Larger lesions are commonly treated with stacked or overlapping circular plug constructs. 4 In recent years, instrumentation has been developed to obtain and transplant a single oval-shaped OCA for larger defects36,45 -47; however, there are minimal clinical outcome reports available. Computer simulated modeling of large oval OCAs suggest that topographic restoration may be difficult to achieve, especially with a non-orthotopic graft.48,49
We have previously demonstrated that oval contralateral LFC OCA can attain an acceptable cartilage surface contour match compared with an oval ipsilateral MFC OCA for large oval defects of the MFC. 50 Similar results have been found in computer-simulated models of circular and oval defects in multiple locations along the MFC for both articular cartilage and subchondral bone contour, although the subchondral bone of non-orthotopic grafts has been more challenging.44,51 Additional prior work in the patella demonstrated that differences in the underlying subchondral bone contour between homologous and non-homologous OCA grafts may be larger than those seen on the cartilage surface. 52 These differences have been shown to result in abnormal force attenuation and distribution, especially in the region near the graft:host interface. 53
The purpose of this study was to use surface contour mapping to determine whether using a contralateral LFC versus an ipsilateral MFC OCA graft plays a role in the ability of donor subchondral bone to align with the native condyle subchondral bone when treating large oval osteochondral defects of the MFC.
Methods
Ninety fresh-frozen human femoral condyles were provided by JRF Ortho (Centennial, CO). Mean donor age was 28 ± 9.9 years (range, 21-35 years), and 94 of 120 (78.3%) of the grafts were male. Thirty MFCs were chosen as recipients. Each recipient condyle was then size-matched using tibial width to an ipsilateral MFC and contralateral LFC donor to produce 30 size-matched groups of 3 condyles. While there is no consistently agreed-upon threshold for size matching within the orthopedic community, reported acceptable ranges are from ±2 mm to ±5 mm.54,55 Eighty percent of the condyles were matched within 5 mm of tibial width. The mean matching deviation for the other 20% was 9.2 mm. Transplants were performed by 3 sports medicine or joint preservation fellowship-trained surgeons (W.D.B., T.S.M., J.A.G.) with a clinical practice focus in cartilage transplant. Each surgeon performed transplants on 10 groups of 3 condyles. Each condyle was refrigerated in a proprietary medium when not in use.
Computed Tomography Scanning and Allograft Transplantation
All recipient condyles underwent an initial nano-computed tomography (nano-CT) scan to determine the native subchondral bone surface morphology. Individual condyles were scanned using a Phoenix Nantom M with Phoenix Datos|x 2 Acquisition, version 2.6.1 (GE Inspection Technologies, LP, Skaneateles, NY). Scan settings of 80 kV, 400 µA, 3 frames averaged, 1 skip, 500 ms exposure time, and 360 images per scan, with a 0.381 mm aluminum filter were used. Scans were reconstructed at 60 µm resolution using Phoenix Datos|x 2 reconstruction v 2.6.1-RTM (GE Inspection Technologies, LP, Skaneateles, NY). 32
After the initial nano-CT scan, a 17 mm × 36 mm oval “defect,” with the anterior edge located just posterior to the roof of the intercondylar notch, was created in the recipient MFC with a commercially available OCA oval dowel instrument set. There were 2 donor transplant groups: ipsilateral MFC oval dowel and contralateral LFC oval dowel.
Using a random number generator, either the medial or lateral condyle donor was chosen to be transplanted first. This randomization process was performed to prevent any confounding error in the measurements due to cartilage deformation during the first transplant or while removing the first transplant. The commercial instruments and technique were used to harvest a 17 mm × 36 mm oval plug from a homologous location on the first donor condyle. The plug template was used to ensure the thickness was flush and adjustments to the plug thickness were made as necessary. The plug orientation was marked at the 12 and 3 o’ clock positions. The edges of the donor plug were bulleted to allow ease of placement. The donor plug was then manually placed into the recipient defect and advanced until flush (
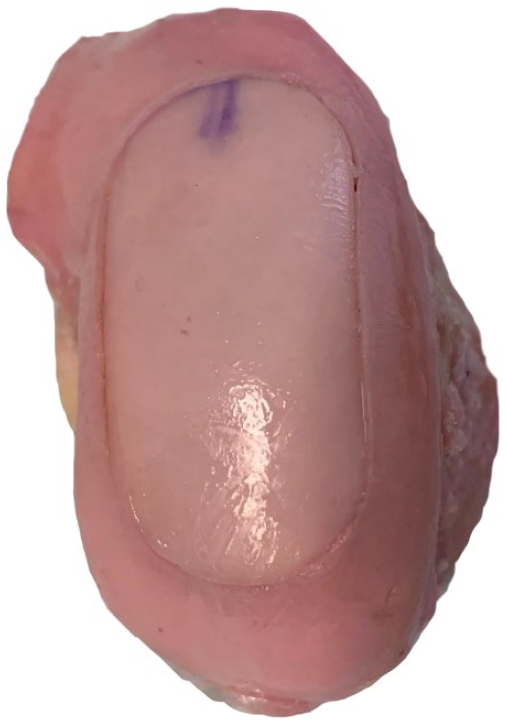
Implanted oval cartilage graft.
Surface Mapping
Reconstructed nano-CT scans of the recipient condyle were imported into Dragonfly 2021.3 (Object Research Systems, Montreal, Quebec, Canada), and the 3 volumes (native plus 2 transplants) from each group were aligned with each other using mutual information registration with smallest iterative steps of 0.5 degrees and 18 µm. A structured grid was extracted from a volume of interest (VOI) box (including the entire graft region) within each registered volume to align voxels for accurate measurement between surfaces. Condylar subchondral bone was then segmented from background and cartilage using a global threshold, and internal voids were filled to achieve a solid segmentation, including empty trabecular space within each condyle. These segmentations were converted to surface meshes that smoothly transect bounding voxels and fit the subchondral bone surface contour within the VOIs. Finally, surface deviation was measured directly as the normal distance between each mesh vertex on a graft region—corresponding with the center of each voxel—and the native subchondral bone surface. There were over 200,000 measurement points per graft. These measurements identified the amount of surface deviation (proud or sunken) between the native subchondral bone surface and the subchondral surface of the transplanted donor graft (
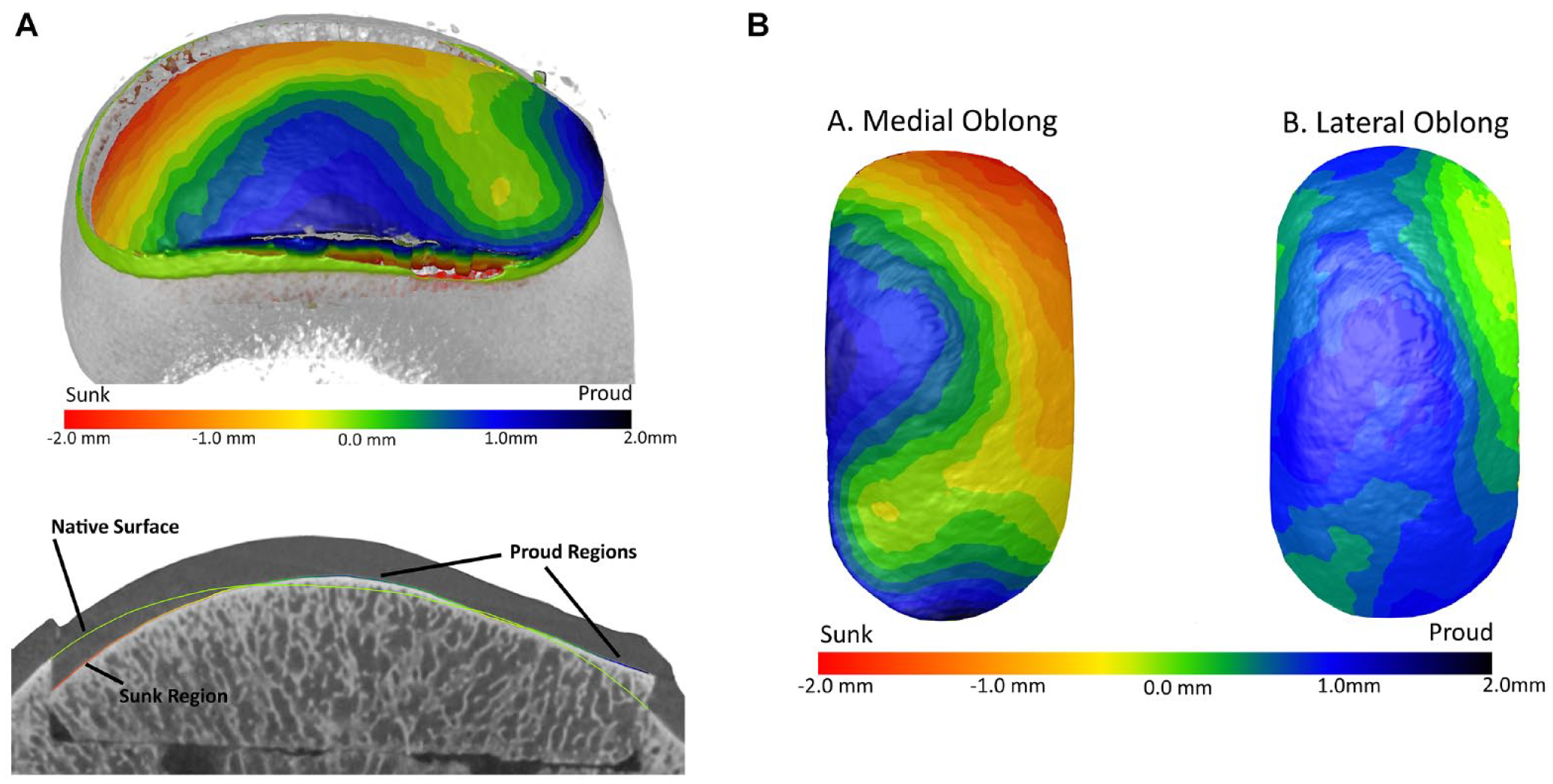
Surface deviation. (
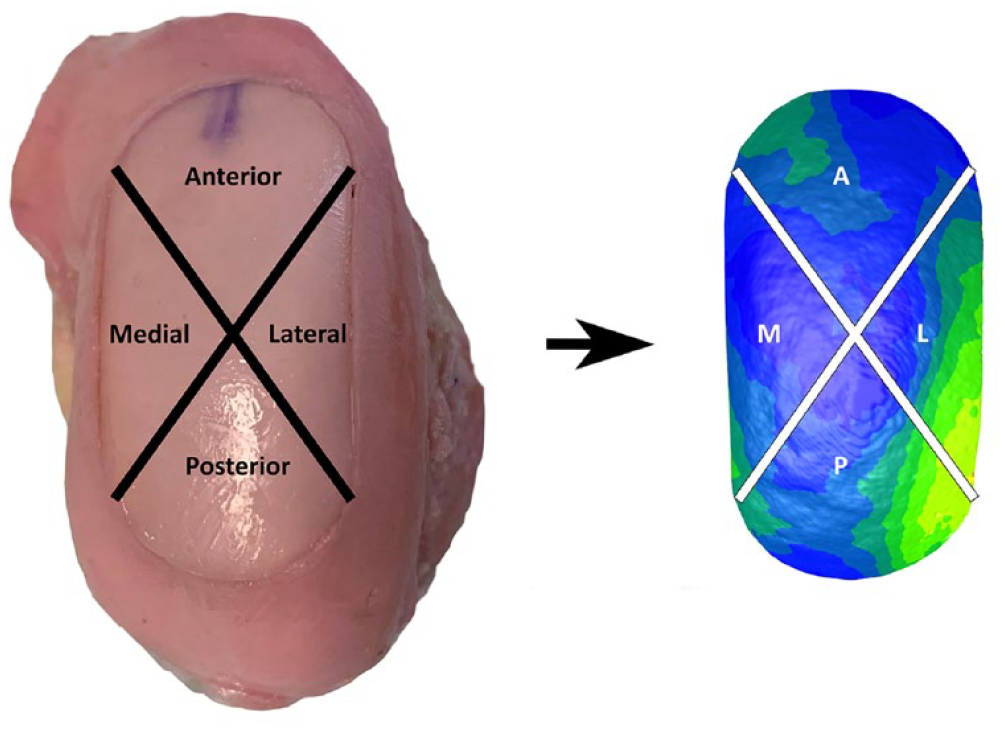
Division of the cartilage grafts into anterior, posterior, medial, and lateral segments. Due to the oval shape of the transplant, the segments were not all equal size.
Step-Off Height Measurements
Step-off height was measured directly as the surface deviation in a narrow margin of 5 voxels (0.3 mm) along the entire edge of the graft. This measure represented the difference in subchondral bone height between the graft and the surrounding native bone at the native:graft interface (
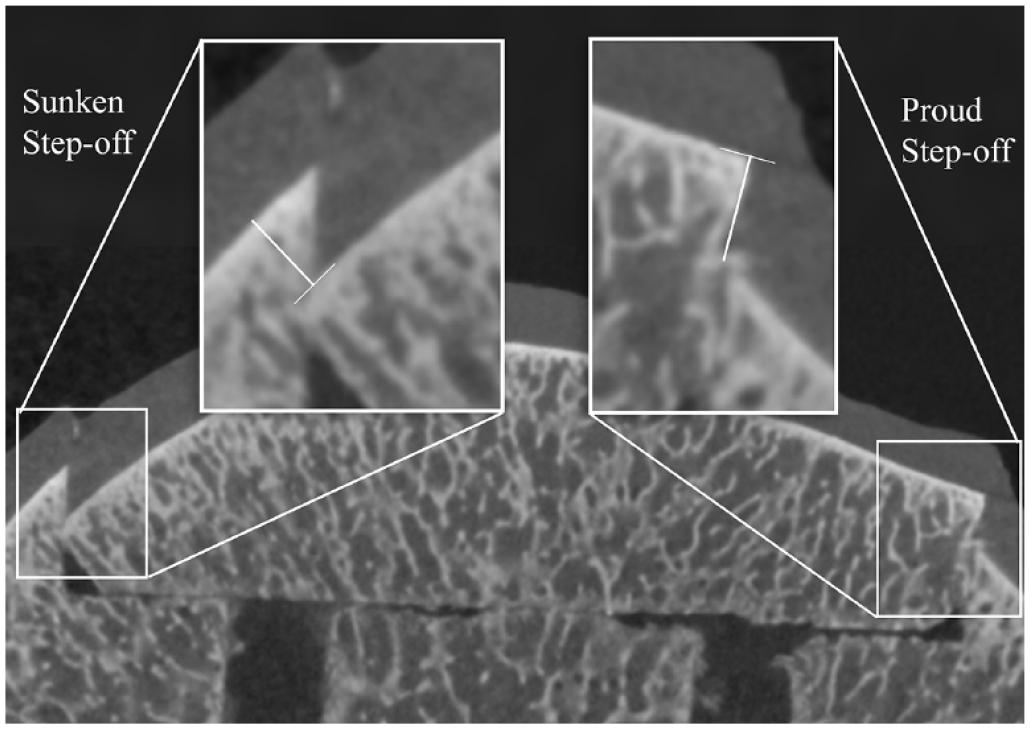
Measurement of subchondral bone step-off height at the interface between the graft and the surrounding native bone. Step-off height (
In order to determine the total amount of surface and step-off height deviation, both proud and sunken, without having the proud and sunken regions canceling each other out, the root mean square (RMS) of the difference in height between the native subchondral bone surface and the transplanted plug surface was then calculated for both the whole condyle, as well as for each segment.
Statistics
Student t tests and 1- and 2-way analyses of variance (ANOVAs) were used to determine the effect of transplant type (contralateral LFC or ipsilateral MFC) and graft segment (anterior, posterior, medial, lateral) on step-off height and surface deviation. Sidak’s multiple comparisons were used to compare step-off heights and surface deviation differences between each pair of segments. Statistical analyses were performed with Stata Version 12.1 (StataCorp, College Station, TX). Statistical significance was set at P < 0.05.
Previous work with single circular medial and lateral femoral condyle transplants demonstrated standard deviations of RMS surface height deviation to be between 0.21 and 0.38 mm.37,43 Given an increase in the expected variability of the surface shape for the larger oval grafts, a more conservative standard deviation of 0.5 mm was used. A clinically relevant difference of 0.75 mm (slightly less than the acceptable 1 mm prominence) between groups was used with a power of 0.8 in a 2-tailed test (α < 0.05) to determine a sample size of 7 recipient condyles per group. To provide a safety factor, the study sample size was increased to 10 recipient condyles per group. In order to evaluate whether there was an effect due to the surgeon, 3 surgeons each performed transplants on 10 condyle groups.
Results
A total of 60 transplants were performed. During recipient socket preparation, one socket wall of one recipient condyle was cracked preventing a press-fit. This recipient was therefore excluded resulting in n = 29 for both the MFC and LFC donor groups. There was no statistically significant difference in mean subchondral bone surface deviation between contralateral LFC and ipsilateral MFC plugs (LFC = 0.87 ± 0.22 mm, MFC = 0.76 ± 0.24 mm, P = 0.07). There were no significant differences in the RMS surface deviation between the 4 segments of the graft (anterior, posterior, medial, lateral) within each graft type or across graft types (MFC vs. LFC;
Table 1
,
Root Mean Square Surface Deviation (mm) for the Contralateral Lateral Femoral Condyle Oval Graft (n = 29) and Ipsilateral Medial Femoral Condyle Oval Graft (n = 29) in Each Quadrant of the Transplanted Plug.
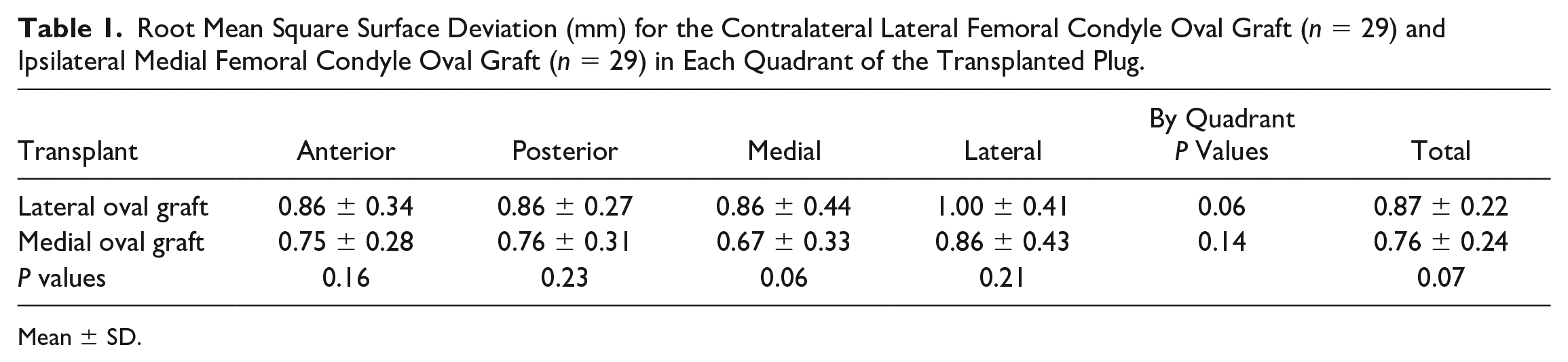
Mean ± SD.
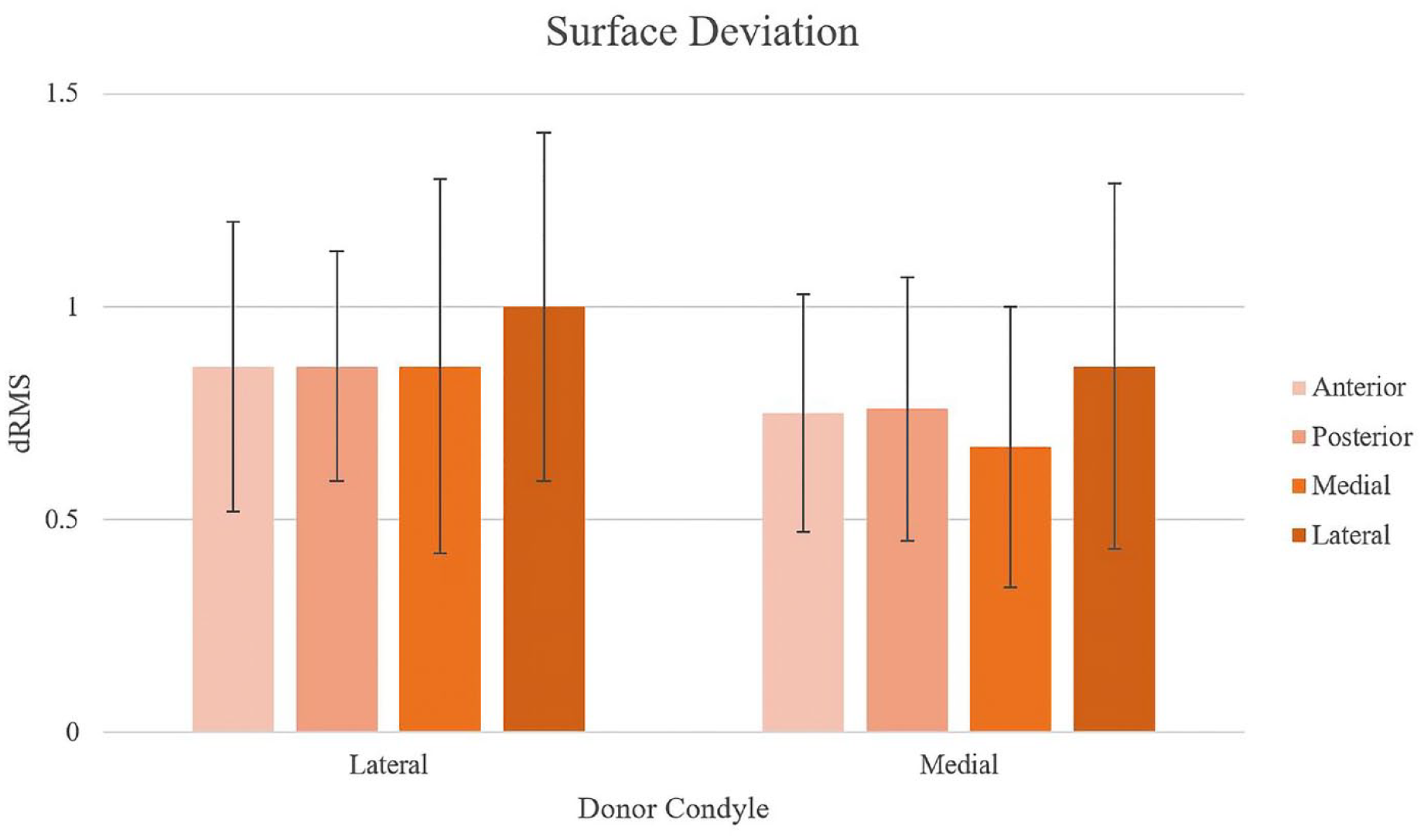
Subchondral bone surface deviation (dRMS) was not significantly affected by donor condyle or graft quadrant.
At the interface between the donor plug and the surrounding native subchondral bone, there was no significant difference in the overall circumferential step-off height between the contralateral LFC and ipsilateral MFC plugs (LFC = 0.93 ± 0.18 mm, MFC = 0.85 ± 0.21 mm, P = 0.09;
Table 2
,
Root Mean Square Step-Off Height (mm) for the Contralateral Lateral Femoral Condyle Oval Graft (n = 29) and Ipsilateral Medial Femoral Condyle Oval Graft (n = 30) in Each Quadrant of the Transplanted Plug.
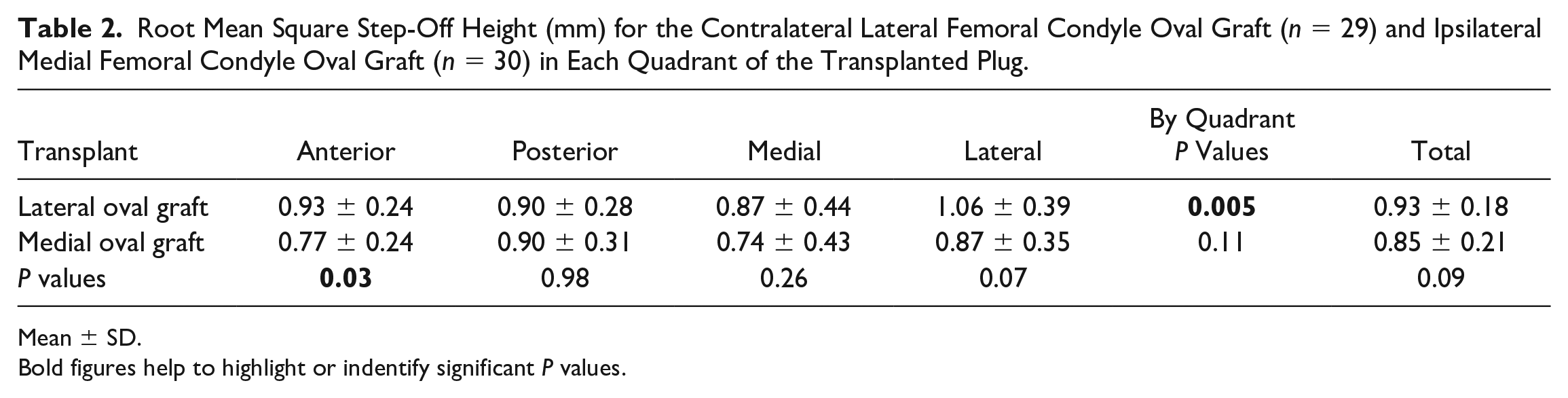
Mean ± SD.Bold figures help to highlight or indentify significant P values.
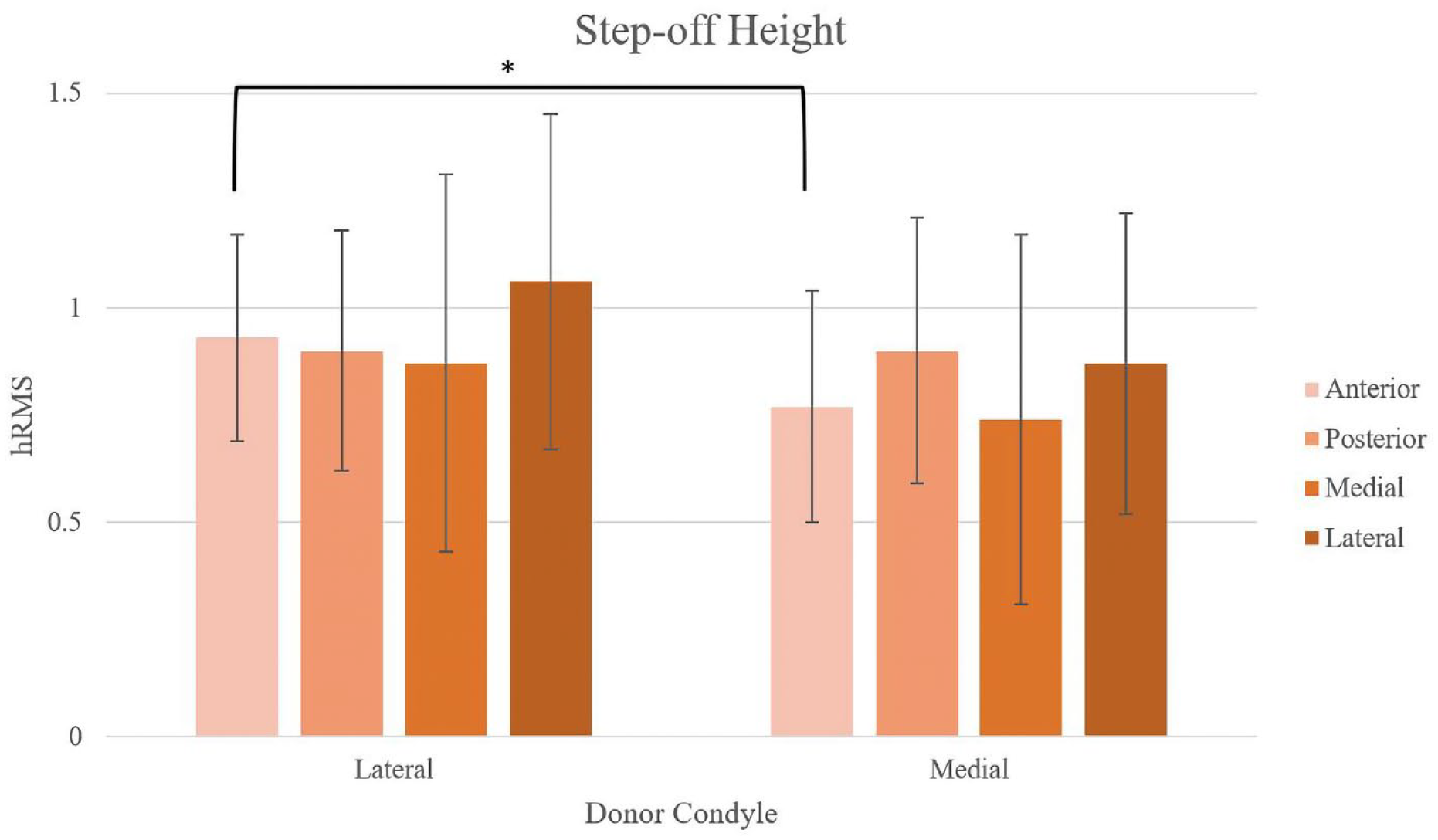
Subchondral bone step-off height (hRMS) was not significantly affected by donor condyle or graft quadrant except in the anterior quadrant between the lateral femoral condyle and medial femoral condyle donors (*P = 0.03).
Post hoc power testing based on the standard deviations found in this study (
Tables 1
and
Discussion
Our results demonstrate that there were no significant differences in subchondral bone surface deviation or overall step-off height between ipsilateral (orthotopic) MFC and contralateral (non-orthotopic) LFC oval-shaped allografts. There were some small differences (<0.2 mm) in step-off height between some segments of the grafts which are not likely to be clinically relevant. This suggests that contralateral LFC allografts can be as effective as ipsilateral MFC allografts in replicating subchondral bone geometry of the native MFC.
In order for OCA transplantation to be successful, anatomic restoration of the articular surface in the area of pathology must be achieved. Many studies have been conducted with the goal of determining the acceptable range of surface geometry mismatch when performing osteochondral transplants.19,37,43 While previous studies have suggested that the articular cartilage surface should be matched within 1 mm recession and 0.5 to 1 mm elevation to reliably produce acceptable outcomes,19,56 there is minimal data related to the acceptable range of subchondral bone mismatch for osteochondral transplants. Ackermann et al. investigated the predictive associations between OCA morphology and postoperative OCA appearance at the graft-host junction. Their results indicated that grafts <5 mm thick were more likely to demonstrate cystic changes (odds ratio [OR] = 4.9, 95% confidence interval [CI], 1.5-16.1; P = 0.009), while grafts >9 mm thick were more likely to present with residual osseous cleft (P = 0.1). 13 While these data support a desired range for overall graft thickness, to our knowledge there are no studies that have investigated acceptable ranges for subchondral bone step-off height or surface deviation. Previous work investigating subchondral bone alignment in OCA of patellar defects identified that step-off height and surface deviation at the subchondral bone were approximately 0.5 mm greater than the differences in the cartilage surface, suggesting greater variability in subchondral bone alignment than there is at the cartilage surface. 52 Further study of subchondral bone step-off and height deviation is necessary to determine acceptable values for subchondral bone alignment compared to the surrounding native bone.
The role of the subchondral bone beneath and surrounding OCA grafts has become an area of interest in determining how to optimize graft survival. Trabecular incorporation of graft subchondral bone has been shown to be a prognostic indicator of allograft health, viability, and functional outcomes.16,57 The presence of healthy subchondral bone is crucial for the absorption of forces generated during impulse loading, which protects the overlying layer of cartilage.14,58 Bernstein et al. 54 posited that subchondral bone mismatch between donor and host may translate to a shear stress gradient at the graft-host interface. D’Lima et al. 56 found that peak compressive strains were found at the margins of the osteochondral defect at the junction of the cartilage and subchondral bone, which they hypothesized to be a potential site for initiation of progressive cartilage damage. These variations in local stresses have been thought to be great enough to kill chondrocytes or inhibit graft integration, potentially leading to graft failure. 53
Several animal studies have provided a foundation for the idea that the subchondral bone is crucial for graft survival. In their in vivo sheep and goat models of femoral condyle OCA, Von Rechenberg et al. 57 and Pallante-Kirchura et al. 59 found that step-off at the subchondral bone level was a risk factor for subchondral cyst formation. The authors suggested the interaction of the subchondral bone with synovial fluid at the cartilage:bone interface as a potential mechanism for subchondral cyst formation, such as in the setting of subchondral bone step-off. 59 Also using a goat model of femoral condyle OCA, Chan et al. 60 found that deviations at the bone-cartilage interface were associated with deviations at the articular surface and lower cartilage stiffness over time. When the bone-cartilage interface deviated by <0.15 mm, deviations at the articular cartilage surface were significantly fewer than those with bone-cartilage interface deviations >0.15 mm. An inverse effect of cartilage surface variation affecting deviations at the bone-cartilage interface was not found to be true, indicating that the intraoperative emphasis placed on cartilage surface congruity may not be sufficient to protect from the effects of subchondral bone mismatch.52,60 The relative difference in donor and recipient cartilage thickness, variation in the donor cartilage thickness over the whole surface of the donor plug, and the fact that there is a greater opportunity for regional differences in cartilage thickness in larger plugs are all potential factors that could contribute to differences in subchondral bone step-off at the donor:recipient interface. Future studies would therefore be beneficial to further investigate these factors.
The ultimate success of OCA transplantation is dependent on surgical technique in addition to graft integration and biomechanics. As defect diameter increases, restoration of the anatomic surface contour and articular step-off is made more challenging by the differences in radius of curvature and surface topography of the femoral condyle. 40 Techniques historically used for these larger lesions include shell allografts 14 and overlapping OCA plugs.4,6 In 2015, instrumentation allowing the ability to harvest and implant a single oval-shaped graft became available. For the current study, a 17 mm × 36 mm defect was chosen to represent a moderately large defect and allowed for 4 mm of bone along the medial and lateral aspects of an MFC that is 25 mm wide. While there are no clinical outcome studies currently reporting solely on this single oval graft technique, attributable to the recent availability of the instrumentation, computer modeling studies have demonstrated the efficacy of oval grafts of the MFC to provide acceptable biomechanical matches for MFC longitudinal defects. 51
While there remains a gap in the literature regarding subchondral bone alignment in OCA transplants, comparisons can be drawn between existing computer simulation studies and prior work regarding oval OCAs for larger MFC defects.44,50,51 In their study of circular OCAs from the LFC and MFC for treatment of MFC defects, Urita et al. 44 analyzed the resulting articular cartilage subchondral bone contours from each graft source and found that there was no significant difference among donor groups or defect location. In a subsequent 2020 study, Urita et al. 51 analyzed the topographic matching of oval OCAs from MFCs, ipsilateral LFCs and contralateral MFCs for oval defects of the MFC. They found that for defect sizes of 17 mm × 30 mm and 20 mm × 30 mm, the subchondral bone surface deviation was significantly greater than articular cartilage surface mismatch and was greater in ipsilateral LFC grafts than for MFC grafts. Similar to the present study, Urita et al. 51 did not find a significant difference in subchondral bone surface deviation between contralateral LFC and ipsilateral MFC grafts at 20 mm × 30 mm; however, the contralateral LFC grafts for the 17 mm × 30 mm defects exhibited greater subchondral bone surface deviation than the MFC grafts. Overall, the current study demonstrated mean surface deviations for both ipsilateral MFC and contralateral LFC oval grafts that were lower than Urita’s MFC and contralateral LFC values. This may be attributable to measurement technique, the use of computer simulation versus clinical transplantation, or our larger sample size. 51
The results of the current study suggest that contralateral LFC allografts can be as effective as ipsilateral MFC allografts in replicating the subchondral bone geometry of the native MFC. In the setting of a supply-demand mismatch between highly available LFC grafts and scarcer MFC grafts, our findings provide support for the economic use of available donor osteochondral tissue without compromising the efficacy of restoring the articular surface of the femoral condyle within acceptable parameters. This will decrease patient wait times for size-matched grafts and help decrease the wastage of donated LFC tissue. An emerging body of literature suggests that matching the underlying subchondral bone contour may have similar or even greater implications than cartilage surface contour on contact stresses at the joint. 8 Finite element analysis is now required to determine whether differences in subchondral bone surface deviation or step-off at the graft:host interface affect local cartilage force distribution, with a goal of determining clinically relevant values for subchondral bone step-off and surface deviation in OCA transplant.
The present study had several limitations. First, the study was performed ex vivo using fresh-frozen cadaveric tissue which limits the clinical or biomechanical conclusions. In addition, we made use of a single proprietary OCA transplantation instrument system, which limits the generalizability of our results across graft procurement techniques. The objective of direct comparison between 2 graft constructs within the same recipient condyle introduced possible variation in defect contour with extraction of the first graft and implantation of the second. We also made use of size-matched condyles for this study (80% matched within 5 mm tibial width), while in practice surgeons may have differing preferences for donor size relative to the patient’s tibial or condylar width. Size-matching the condyles for the present study occasionally resulted in difficulty obtaining a suitable location for donor graft harvest due to small LFC size, at which times the oval donor plug was taken from a non-homologous location compared to the defect. Finally, measurement of the subchondral bone surface using Dragonfly often involved hand-correction of the desired region of interest, which could have introduced minor variability into the resulting measurements.
Conclusion
The results of the present study demonstrated no significant differences in overall subchondral bone circumferential step-off or surface deviation at the graft-host interface between ipsilateral MFC and contralateral LFC oval-shaped allografts for 17 mm × 36 mm defects of the MFC.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Study was funded by JRF Ortho and Arthrex Inc. Research reported in this publication was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under award number P30AR069620.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kelly M.R. Taylor: None declared; Conor S. Locke: None declared; Timothy S. Mologne: JRF Ortho, Arthrex; William D. Bugbee: JRF Ortho, Arthrex; John A. Grant: JRF Ortho, Arthrex, Vericel, Aesculap Biologics.
Ethical Approval
The study was considered exempt by the University’s Institutional Review Board (HUM00162355).
