Abstract
Objective
Cartilage degeneration is a key feature of osteoarthritis (OA) and rheumatoid arthritis and is thought to negatively impact patients’ quality of life. Diclofenac etalhyaluronate (DEH, SI-613/ONO-5704) is a hyaluronic acid (HA) derivative chemically bound to diclofenac (DF) that has been reported to improve OA symptoms; however, its effect on cartilage degeneration remains unknown. In the present study, we investigated the chondroprotective effect of DEH in rats with collagen-induced arthritis and interleukin-1β-stimulated human chondrocytes.
Design
Rats with collagen-induced arthritis were administered DEH and HA intra-articularly, and DF orally. Knee joint swelling, histological scores of articular cartilage, and inflammatory (Il1b) and catabolic (Mmp3 and Mmp13) gene expression in the synovial tissue and cartilage were evaluated. In vitro direct effects of DEH on matrix metalloproteinase (MMP)-3 and MMP-13 expression were examined in interleukin-1β-stimulated human chondrocytes.
Results
In a rat model of collagen-induced arthritis, a single intra-articular dose of DEH inhibited knee joint inflammation and cartilage degeneration. Daily oral administration of DF had similar effects. Conversely, HA administered as a single intra-articular dose had no effect. Only DEH inhibited Mmp3 gene expression in the cartilage, whereas DEH and DF inhibited Mmp3 and Mmp13 mRNA expression in the synovial tissue. In interleukin-1β-stimulated human chondrocytes, DEH and HA inhibited MMP-3 and MMP-13 production, whereas DF had no effect.
Conclusions
In this study, we demonstrated the chondroprotective effect of DEH in rats with collagen-induced arthritis and in interleukin-1β-stimulated human chondrocytes. Thus, DEH may suppress cartilage degeneration in patients with musculoskeletal diseases, such as OA.
Keywords
Introduction
The articular cartilage is a thin connective tissue that lines the epiphysis in synovial joints and is mainly composed of chondrocytes and extracellular matrices. The adult articular cartilage can be divided into four layers based on its histological characteristics: surface, intermediate, deep, and calcified layers, which are connected to the subchondral bone beneath it. 1 This tissue has unique viscoelastic properties, providing a low-friction surface for joint movement and cushioning the underlying bone during loading. 2 However, cartilage can degenerate as consequences of trauma, mechanical stress, and other molecular biological processes. 3 Degenerated cartilage is difficult to repair owing to its avascular nature, which results in limited self-renewal properties. 4 Osteoarthritis (OA) and rheumatoid arthritis are major diseases associated with cartilage degeneration that affect increasing numbers of patients worldwide. 5 Cartilage disorders impair physical function,6,7 contributing to the deterioration of patients’ quality of life. 8
In OA, which is mainly characterized by cartilage degeneration, the mainstay of therapy is pain management using drugs such as acetaminophen, non-steroidal anti-inflammatory drugs (NSAIDs), cyclooxygenase-2 inhibitors, hyaluronic acid (HA), and corticosteroids. HA has been suggested to have a chondroprotective effect on osteoarthritic cartilage9,10; however, current clinical evidence is limited. Despite recent advances regarding both drug (such as inhibitors of matrix-degrading enzymes, growth factors, Wnt signal inhibitors, senolytic agents, and bone resorption inhibitors) and cell therapies (allogeneic chondrocytes expressing transforming growth factor beta 1 and mesenchymal stem cells) that are expected to have disease-modifying effects on OA,11,12 there is no prospect for their practical application. Given the lack of available disease-modifying drugs for the treatment of cartilage degeneration, a re-evaluation of existing symptom-modifying drugs based on their chondroprotective effects may help optimize the treatment of diseases associated with cartilage degeneration.
Diclofenac etalhyaluronate (DEH; SI-613/ONO-5704) is a derivative of high-molecular-weight fermented HA (600-1,200 kDa), of which glucuronic acid moieties are chemically bound to the NSAID diclofenac (DF) via a 2-aminoethanol linker. DF is gradually released from DEH by the hydrolytic cleavage of the ester linkage, in a pH-dependent manner. Intra-articularly administered DEH remains in the joint for a long period 13 and locally releases DF in a sustained manner. 14 Furthermore, DEH has been shown to exert anti-inflammatory and analgesic effects in experimental animal models 14 ; in addition, DEH has been shown to improve knee and hip OA pain in clinical trials.15-18 In Japan, DEH has become clinically available as a drug to improve joint function in OA; however, the effect of DEH on cartilage degeneration remains unclear.
In the present study, we investigated the chondroprotective effect of DEH in vivo in rats with collagen-induced arthritis (CIA) and interleukin-1β (IL-1β)-stimulated human chondrocytes in vitro.
Materials and Methods
Materials
DEH (DF: approximately 11.8% [w/w]) and HA (600-1,200 kDa) were manufactured by Seikagaku Corporation (Tokyo, Japan). DF and 10% neutral-buffered formalin were purchased from Fujifilm Wako Pure Chemical Corporation (Osaka, Japan). Distilled water was purchased from Otsuka Pharmaceutical Factory Inc. (Tokushima, Japan). Incomplete Freund’s adjuvant was purchased from Becton Biosciences (Franklin Lakes, NJ). Bovine type II collagen was purchased from the Collagen Research Center (Tokyo, Japan). Phosphate-buffered saline (PBS), TRIzol reagent, PureLink RNA Mini Kit, High-Capacity cDNA Reverse Transcription Kit with RNase Inhibitor, and Dulbecco’s Modified Eagle’s Medium (DMEM)/F-12 were purchased from Thermo Fisher Scientific (Waltham, MA). Fetal bovine serum (FBS) was purchased from Sigma-Aldrich (St. Louis, MO). TB Green Premix Ex Taq II was purchased from Takara Bio, Inc. (Shiga, Japan). Recombinant human IL-1β and enzyme-linked immunosorbent assay (ELISA) kits for human matrix metalloproteinase (MMP)-3 and MMP-13 were purchased from R&D Systems (Minneapolis, MN).
Animals
Female Dark Agouti rats (9-10 weeks old) were purchased from Japan SLC, Inc. (Shizuoka, Japan). The animals were housed in specific pathogen-free conditions at a room temperature ranging from 20 to 26 °C, humidity of approximately 50%, and a 12-hour light/dark cycle. Animal studies were reviewed and approved by the Institutional Animal Care and Use Committee of Seikagaku Corporation and were performed under an animal husbandry/management system in an appropriate environment for animal welfare.
Induction and Treatment of CIA
The bovine collagen type II solution was emulsified by mixing it with an equal amount of incomplete Freund’s adjuvant. Rats were anesthetized with isoflurane inhalation and immunized by intradermal injection of the emulsion (0.05 mg/100 μL/site) at four sites on the back; this protocol results in an autoimmune reaction to collagen and the induction of arthritis. The normal control rats were non-treated or underwent the same procedures but were intradermally injected with physiological saline. Fourteen days after immunization, 50 μL of PBS, DEH (0.05-0.5 mg for dose-response analysis and 0.5 mg for other experiments), HA (0.5 mg), or a mixture of DF (59 μg, the same amount contained in 0.5 mg DEH) and HA (0.5 mg) was administered into the articular cavity of the knees of both hindlimbs under isoflurane anesthesia. DF was orally administered once daily from 14 to 21 or 28 days after immunization. The DF dose was set at 2 mg/kg, which is the same weight-adjusted dose of DF (maximum dose, 100 mg daily) prescribed for an OA or rheumatoid arthritis patient weighing 50 kg. DEH, HA, and the mixture of DF and HA were dissolved in PBS; DF was prepared in distilled water. Arthritis was induced in 81, 28, and 56 rats for joint swelling, histological, and gene expression analyses, respectively (see Supplementary Table S1-S6 for grouping). Forty-seven rats served as non-arthritic controls.
Histological Assessment
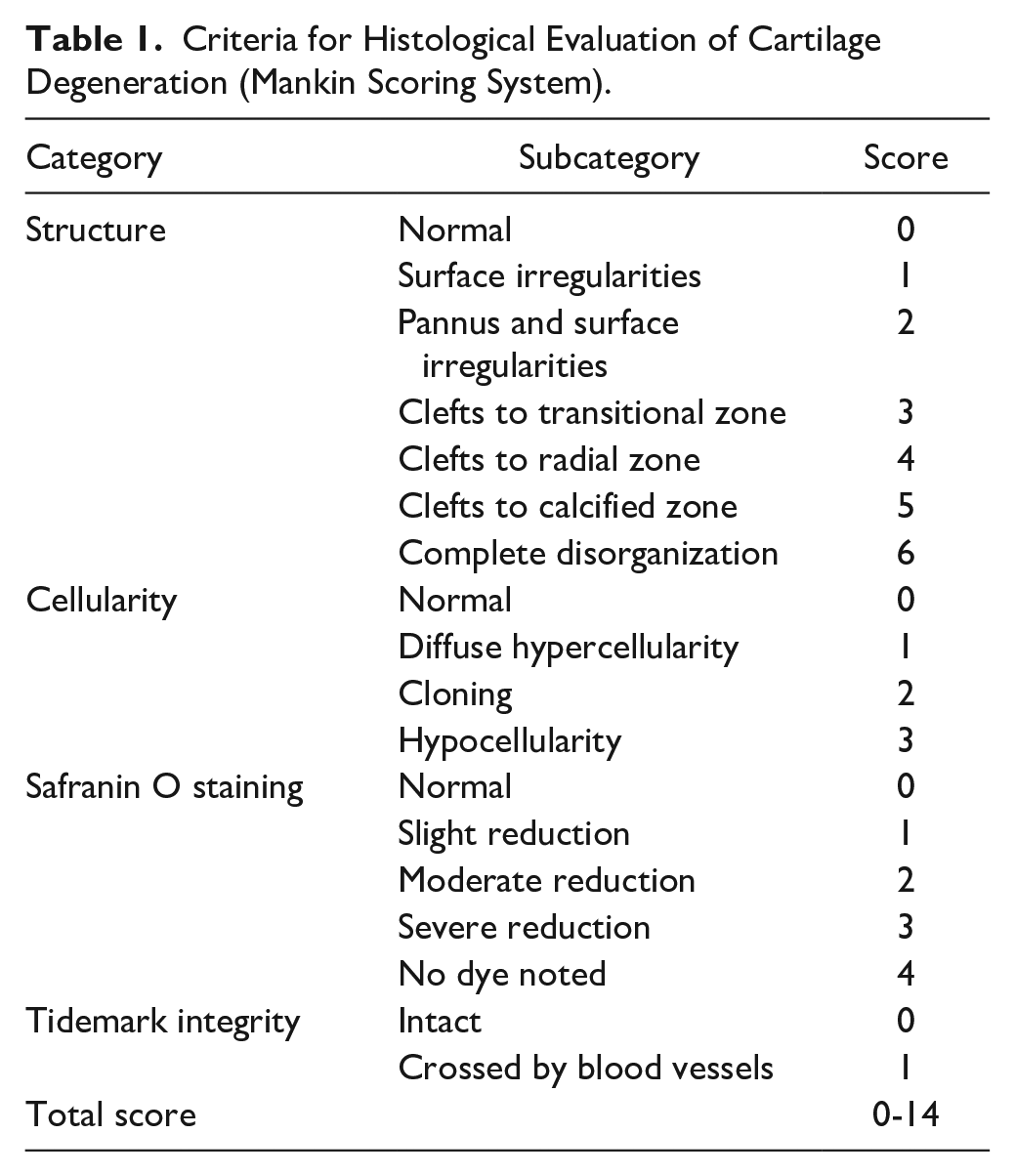
The rats were exsanguinated under isoflurane inhalation anesthesia, and the proximal end of the tibia at the hind limb knee joints was collected. Tissues were collected before immunization; at 7, 14, 21, and 28 days after immunization for a time-course study; and at 21 days after immunization for pharmacological evaluation. In the time-course study, the right knees of three rats were evaluated at each time point (Supplementary Table S1). In the pharmacological study, eight knees (the left and right knees of four CIA rats) per group were evaluated (Supplementary Table S2). In the non-arthritic control group, both knees of two rats were evaluated. Tissues were fixed in 10% neutral-buffered formalin and decalcified in formic acid formalin. The medial tibia was sectioned longitudinally and embedded in paraffin. The sections were stained with safranin O/fast green, and cartilage degeneration severity was scored according to the Mankin scoring system 18 ( Table 1 ), with a slight modification: the “Tidemark integrity” category was scored 1 when the tidemark was destroyed. The effects of the test materials were evaluated in the anterior one-third of the tibial cartilage, where degeneration due to model progression was evident. Scoring was performed in a blinded manner.
Criteria for Histological Evaluation of Cartilage Degeneration (Mankin Scoring System).
Assessment of Joint Swelling
Rats were anesthetized using isoflurane inhalation on the day before immunization and at 7, 13, 14, 15, 16, 17, 19, 21, 23, 24, 26, and 28 days after immunization to measure knee joint width. Nine CIA rats per group were evaluated in both dose-response and pharmacological studies (Supplementary Tables S3 and S4). In the non-arthritic control group, five rats were evaluated. The rats were fixed in a supine position, and the horizontal width (in mm) of the knee joints of the left and right hindlimbs was measured using a digital thickness gauge. Using the knee joint width on the day before collagen sensitization as the baseline, the change in the knee joint width (in mm) of both hindlimbs was calculated, and the mean was defined as knee swelling (in mm) for each animal after sensitization. Measurements after administration of the test materials were performed in a blinded manner.
RNA Extraction and Real-Time Polymerase Chain Reaction
The rats were exsanguinated under isoflurane inhalation anesthesia, and the synovial tissue and cartilage of the hind limb knee joints were collected and rapidly frozen in liquid nitrogen. Synovial tissue or cartilage from both knees of each rat were pooled in tubes to obtain sufficient RNA for real-time polymerase chain reaction (RT-PCR) analysis. Tissue collection was performed before immunization and at 7, 14, and 21 days after immunization for a time-course study, or 21 days after immunization for pharmacological evaluation. Eight rats per group were evaluated in both studies (Supplementary Tables S5 and S6). Tissues were homogenized in TRIzol reagent. Total RNA was extracted using the PureLink RNA Mini Kit according to the manufacturer’s instructions, and RNA concentrations were measured using a microvolume spectrophotometer. The cDNA samples were prepared using a high-capacity cDNA reverse transcription kit with an RNase Inhibitor. The expression levels of Il1b, Mmp3, and Mmp13 were quantified using the CFX384 Touch Real-Time PCR Detection System (Bio-Rad Laboratories, Inc., Hercules, CA) with TB Green Premix Ex Taq II. The thermal cycle program was one initial cycle at 95 °C for 30 seconds, followed by a 30-cycle sequence of denaturing at 95 °C for 5 seconds and annealing/extension at 60 °C for 30 seconds. The housekeeping gene Hprt1 was used as a reference gene for normalization. The primer sequences were: Mmp3, 19 5’-ATGATGAACGATGGACAGATGA-3’ (forward) and 5’-CATTGGCTGAGTGAAAGAGACC-3’ (reverse); Mmp13, 20 5’-TGGAACTAAAGAACATGGTGACTTCTA-3’ (forward) and 5’-CCCCGCCAAGGTTTGG-3’ (reverse). Commercially available primer pairs for Il1b (RA063423) and Hprt1 (RA015379) were purchased from Takara Bio, Inc. The expression level of each gene was quantified using a linearly regressed calibration curve and normalized to the expression level of Hprt1.
Chondrocytes Culture
Human chondrocytes (Lonza [Basel, Switzerland] and Cell Applications Inc. [San Diego, CA]) were cultured in DMEM/F-12 supplemented with 10% FBS at 37 °C in 5% CO2. Cells passed once or twice were seeded in 24-well plates at a starting density of 1.5 × 105 cells/mL per well, 22-24 hours before treatment. The culture medium was replaced with 1 mL of DMEM/F-12 containing 10% FBS and the corresponding materials: HA (2 mg/mL), DEH (0.5-2 mg/mL), or DF (0.03-3 µg/mL). The concentrations of DF were set below the cytotoxic range (>30 µg/mL) according to our preliminary experiments (data not shown). After a 30-minute incubation, IL-1β (0.1 ng/mL) was added and incubated for 16 hours, after which culture supernatants were collected. The concentrations of MMP-3 and MMP-13 in the culture supernatants were measured using an ELISA kit, according to the manufacturer’s instructions. Six wells were treated with each test material, and samples from each well were analyzed separately. Data are representative of several independent experiments.
Statistical Analysis
Statistical analyses were performed using the Statistical Analysis System (SAS; SAS Institute Inc., Cary, NC). In the in vivo study, the histological scores of each group, except for the normal group, were statistically analyzed using the Steel-Dwass test. The mean joint swelling values from days 14 to 28 after immunization were subjected to statistical analysis. The data for each group, except the normal group, were statistically analyzed using Williams’ test (for the dose-response study) or Tukey’s test (for the pharmacological study). Gene expression levels in each group were analyzed using Tukey’s test. In the in vitro study, MMP concentrations were statistically analyzed using Williams’ test or Student’s t test. Statistical significance was set at P < 0.05. Data are presented as mean and standard error of the mean.
Results
Cartilage Degeneration in a Rat CIA Model
Time-dependent cartilage degeneration in a rat model of CIA was histopathologically evaluated. Before sensitization, the cartilage surface was smooth without roughening (
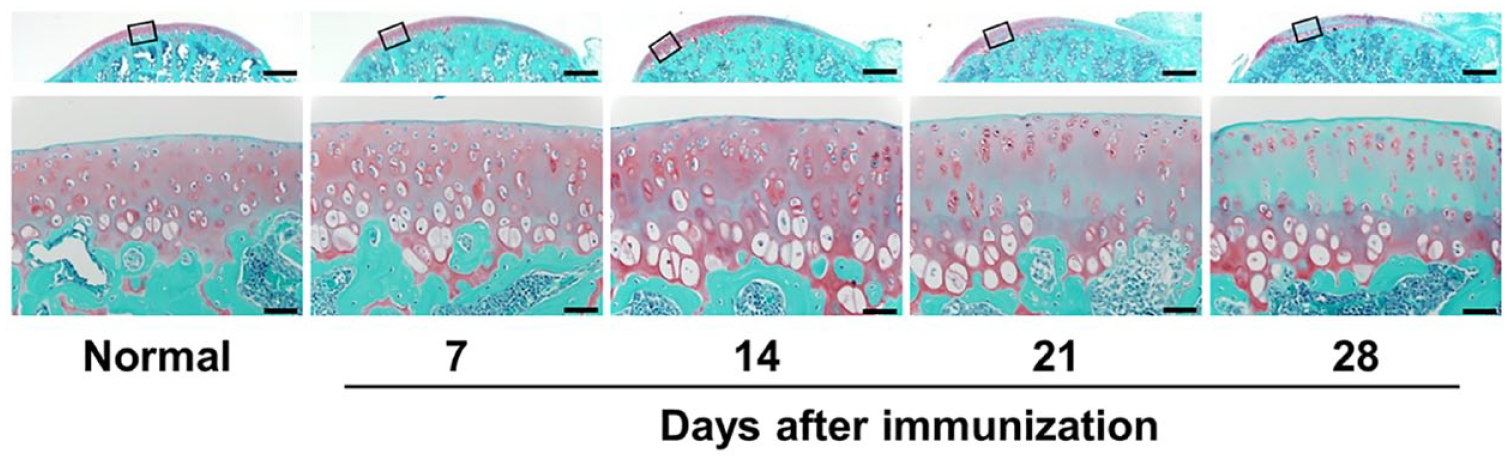
Sections of the tibial cartilage of a knee joint were stained with safranin O and fast green, where proteoglycan in the cartilage is in red and bone is in green. The upper images present an overview of the cartilage; the lower images correspond to the high magnification of the squared area in the upper image. Representative images are shown (n = 3). Scale bar = 500 µm (upper images) and 50 µm (lower images).
Effect of DEH on Joint Swelling, Il1b Transcription, and Cartilage Degeneration in a CIA Model
A dose-dependent inhibition of joint swelling by DEH was observed at 0.05-0.5 mg/joint doses (
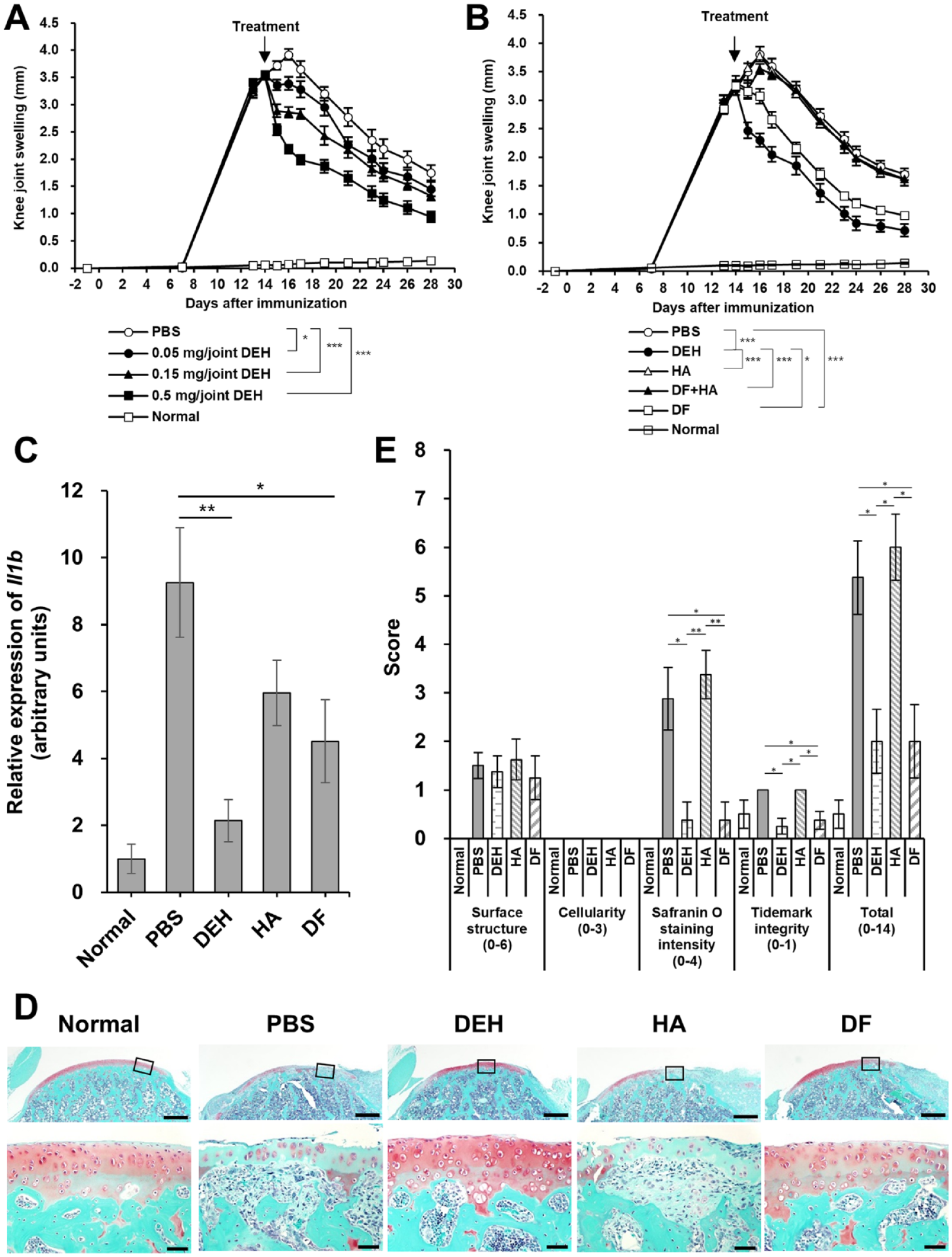
Anti-inflammatory and chondroprotective effects of DEH in CIA rats. (
Effect of DEH on Mmp3 and Mmp13 Expression in a CIA Model
To investigate the mechanisms underlying the chondroprotective effect of DEH, the Mmp3 and Mmp13 mRNA expression in the knee joints of CIA rats was measured using RT-PCR. We observed that, in this model, there was no change in Mmp3 or Mmp13 mRNA levels in the cartilage or synovial tissue 1 week after immunization. However, Mmp3 and Mmp13 mRNA levels increased 14 and 21 days after immunization compared to those in normal rats (
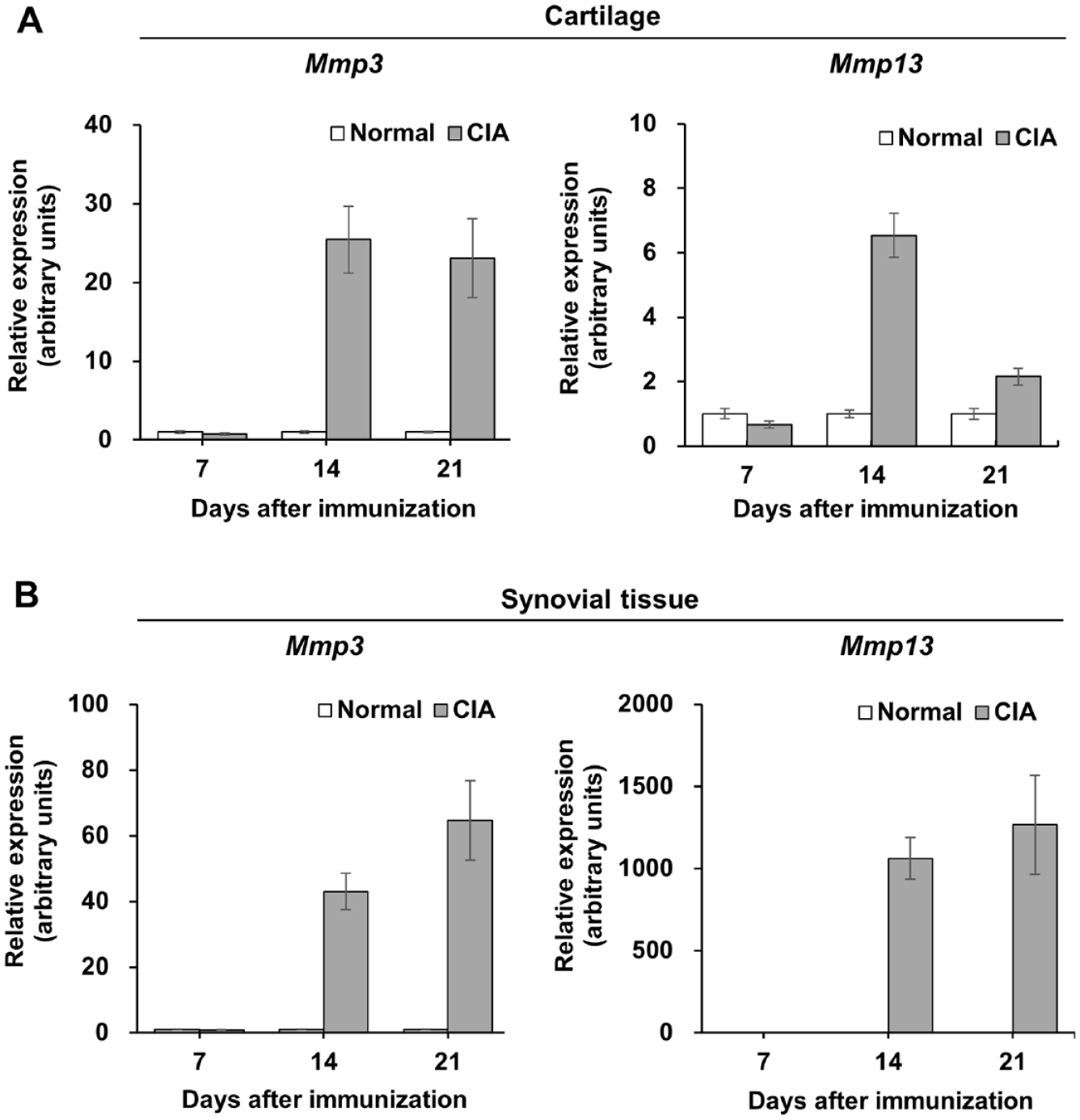
Upregulation of Mmp3 and Mmp13 mRNA in the knee tissues of rats with CIA. The expression levels of Mmp3 (left panel) and Mmp13 (right panel) in the cartilage (
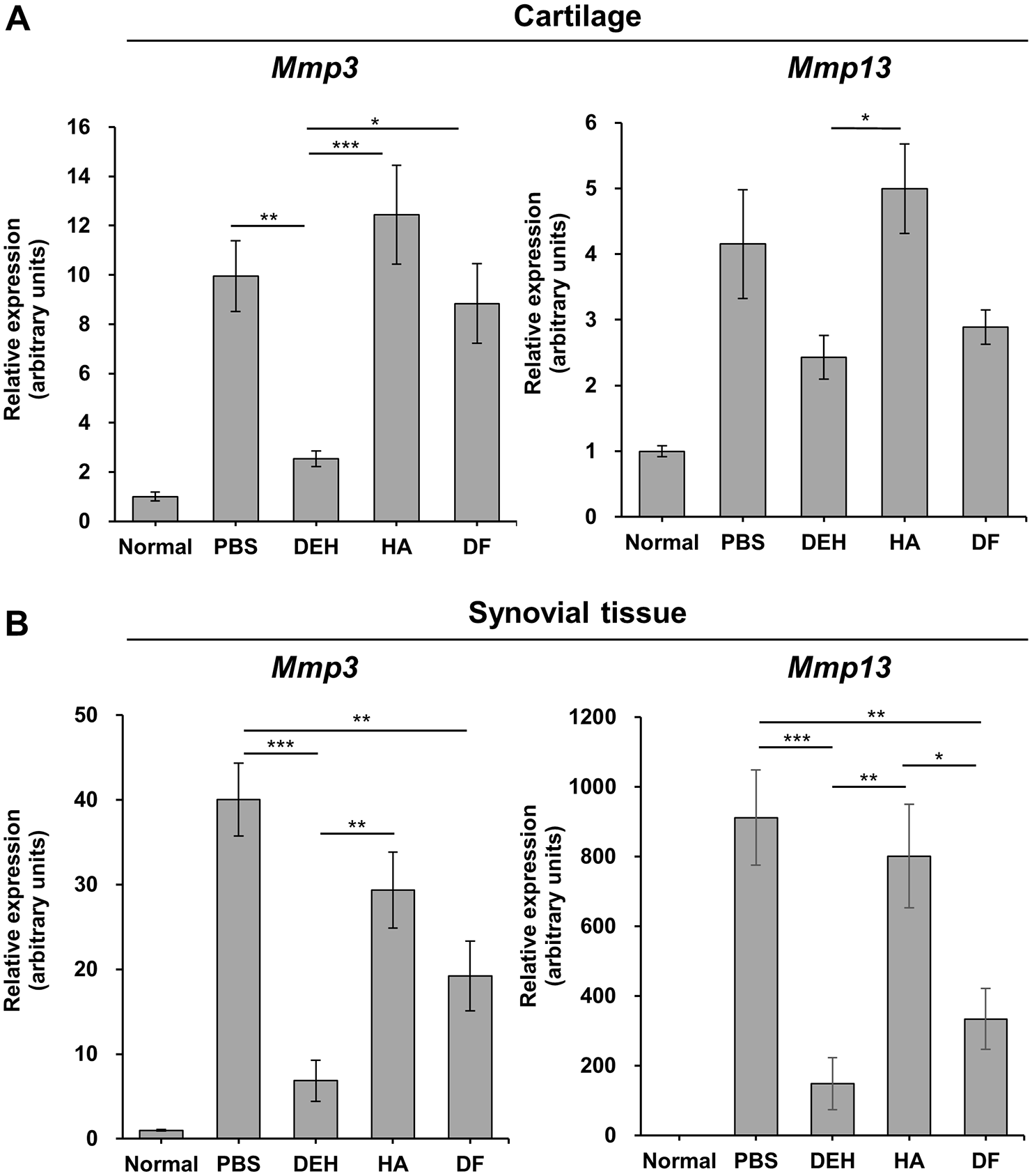
Reduction of Mmp3 and Mmp13 mRNA expression in CIA rats treated with DEH. The expression levels of Mmp3 (left panel) and Mmp13 (right panel) in the cartilage (
Effect of DEH on MMP-3 and MMP-13 Production on IL-1β-Stimulated Human Chondrocytes
To further investigate the inhibitory effect of DEH on MMP production in cartilage, we analyzed the protein expression of MMP-3 and MMP-13 in IL-1β-stimulated human chondrocytes. DEH decreased the levels of MMP-3 and MMP-13 in the culture supernatant in a concentration-dependent manner (
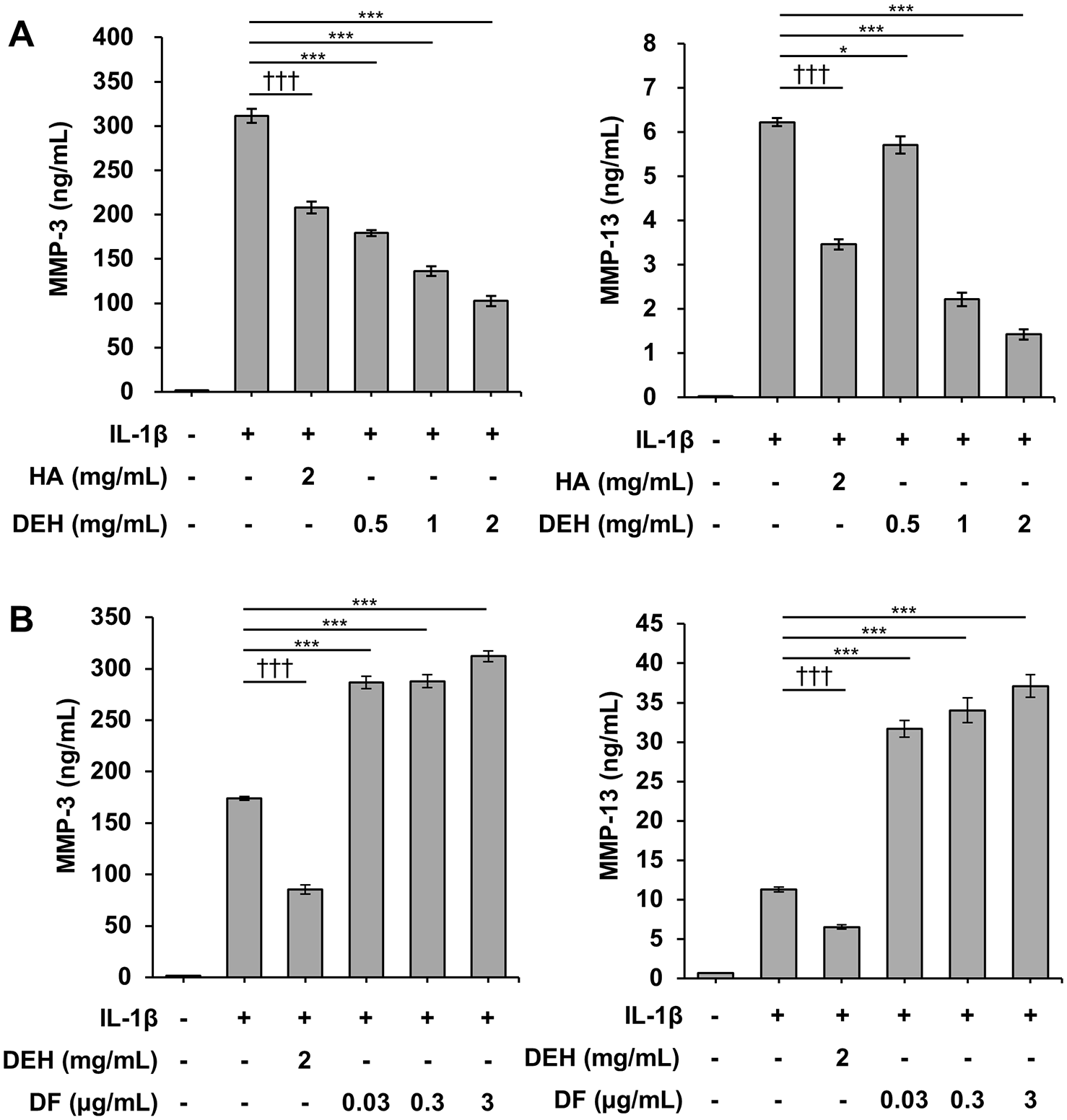
Inhibition of MMP-3 and MMP-13 expression by DEH in IL-1β-stimulated human chondrocytes. Concentrations of MMP-3 (left panel) and MMP-13 (right panel) in the culture supernatant were measured using an enzyme-linked immunosorbent assay (ELISA). (A) DEH reduced the production of MMP-3 and MMP-13 in a similar manner to HA. (B) By contrast, DF, a chemical component of DEH, increased the production of MMP-3 and MMP-13. *P < 0.05, **P < 0.01, ***P < 0.001, Williams’ test (vs. IL-1β-treated cells). †††P < 0.001, Student’s t test (vs. IL-1β-treated cells). The values represent the mean ± standard error. MMP = matrix metalloproteinase; DEH = diclofenac etalhyaluronate; IL-1β = interleukin-1β; HA = hyaluronic acid; DF = diclofenac; PBS = phosphate-buffered saline.
Discussion
In this study, we examined the pharmacological actions of DEH, an HA derivative chemically bound to DF, in a rat CIA model and in IL-1β-stimulated human chondrocytes. We demonstrated that DEH not only has analgesic and anti-inflammatory effects but also has a chondroprotective effect. Previously, we showed that DEH ameliorated symptoms of OA in clinical studies.15-17 We also found that DF locally released from DEH in joints was retained in the synovial tissue for approximately 4 weeks and showed persistent anti-inflammatory effects in non-clinical studies.13,14 Whether NSAIDs such as DF have beneficial effects on structural OA lesions is controversial. Both beneficial and neutral effects of NSAIDs on cartilage have been reported in in vitro and in vivo studies21-24; however, negative effects of NSAIDs on the structural pathology of OA have also been observed.25,26 The present study showed, for the first time, that DEH has a protective effect against cartilage degeneration caused by inflammation.
Our findings indicated that DEH suppressed Il1b transcription and joint swelling in the rat CIA model. These anti-inflammatory effects were not observed with HA (
Here, we found an inhibitory effect exerted by both DEH administered intra-articularly and DF administered orally on cartilage degeneration in the CIA model; no effect was observed for HA administration (
MMPs are metal-requiring proteases induced by inflammatory cytokines and mechanical stress and involved in cartilage degenerative disorders such as OA. The expression of MMP1, MMP3, MMP13, and MMP28 is increased in osteoarthritic cartilage
32
and the production of MMP-1, MMP-3, MMP-9, and MMP-13 is increased in synovial tissue.
33
The substrates of MMP-3 and MMP-13 include aggrecan, a core protein of proteoglycan, and collagen type II, the major component of cartilage
34
; therefore, MMP-3 and MMP-13 are thought to be involved in cartilage degeneration. To investigate the mechanism of the chondroprotective effect of DEH, we analyzed its effect on the expression of Mmp3 and Mmp13 in the cartilage and synovial tissue. DEH inhibited the expression of Mmp3 in the cartilage and Mmp3 and Mmp13 in the synovial tissue (
To further explore the chondroprotective effect of DEH, we assessed its effects on MMP production in IL-1β-stimulated human chondrocytes and found that DEH inhibited MMP-3 and MMP-13 production (
Inflammatory mechanisms (e.g., synovitis) are known to be involved in joint disease etiology, some of which also have a non-inflammatory component (e.g., mechanical stress), 40 such as OA. The experimental models used in this study, a rat CIA model and IL-1β-stimulated human chondrocytes, mimic the inflammatory conditions in joint diseases. Therefore, the chondroprotective effect of DEH observed in our study is limited to inflammatory conditions, and it remains unknown whether DEH affects joint diseases caused by non-inflammatory mechanisms. Age-related factors should also be considered. OA is a common disease in the elderly, and cartilage regenerative capacity is thought to be impaired in the patients. 41 In our in vivo study, CIA was induced in young rats; therefore, the chondroprotective effect of DEH may have been exaggerated compared to what can be achieved in patients with OA patients. To comprehensively understand the effect of DEH on cartilage degeneration, its inhibitory effect on cartilage degeneration should be evaluated in a clinically relevant OA model that includes non-inflammatory and advanced-age components. Furthermore, DEH is expected to have a structure-modifying effect on the degenerated cartilage in patients with OA.
Conclusion
In this study, DEH was found to have a chondroprotective effect in addition to its known analgesic and anti-inflammatory effects. The chondroprotective effects of DEH consist of a combination of indirect outcomes associated with the anti-inflammatory effect of DF, direct effects on chondrocytes similar to those of HA, and a novel effect via a specific DEH-mediated mechanism. Our findings indicate that DEH not only reduces pain in patients with OA, as demonstrated in clinical studies, but may also inhibit cartilage degeneration in these patients.
Supplemental Material
sj-xlsx-1-car-10.1177_19476035231224050 – Supplemental material for Effects of Diclofenac Etalhyaluronate (SI-613/ONO-5704) on Cartilage Degeneration in Arthritic Rats and Inflammatory Cytokine-Stimulated Human Chondrocytes
Supplemental material, sj-xlsx-1-car-10.1177_19476035231224050 for Effects of Diclofenac Etalhyaluronate (SI-613/ONO-5704) on Cartilage Degeneration in Arthritic Rats and Inflammatory Cytokine-Stimulated Human Chondrocytes by Shuhei Takada, Risa Nodera and Keiji Yoshioka in CARTILAGE
Footnotes
Acknowledgments and Funding
The authors thank Dr. Aisuke Nii for interpretation of the histological appearance of tissue sections and Mr. Ryoji Zuinen, Ms. Reiko Zuinen, Mr. Kazuhiro Kojima, Dr. Kei Toyama, Mr. Tomochika Kisukeda, Ms. Yaya Sugano, Ms. Yukie Saeki, and Ms. Haruka Mochizuki for their technical assistance in this study.
Author Contributions
All authors have contributed to study conception and design and acquisition and interpretation of data. S.T. and K.Y. contributed to drafting and revision of the article. All the authors have reviewed and approved the final version of the manuscript. All authors have also reviewed and approved the data presented in the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors of this paper are employees of Seikagaku Corporation.
Ethical Approval
Animal studies were reviewed and approved by the Animal Experiment Ethics Committee of the Seikagaku Corporation and were performed under the animal husbandry/management system in an appropriate environment for animal protection/welfare.
Availability of Data and Materials
The dataset supporting the conclusions of this study was stored at Seikagaku Corporation, Tokyo, Japan. Further inquiries regarding these data may be submitted to Shuhei Takada (
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
