Abstract
Objective
Collagen disruption is one of the underlying causes of knee pain in patients with osteoarthritis and/or diverse cartilage defects. Atelocollagen is a type of collagen that lacks telopeptides and thus has reduced antigenicity. The intra-articular injection of type I atelocollagen supplements collagen levels in the disrupted articular cartilage. This randomized controlled trial evaluated the effects of the intra-articular injection of atelocollagen for the management of knee pain.
Design
Two hundred patients with osteoarthritis, chondromalacia, or other cartilage defects were randomly assigned to receive a 3-mL intra-articular injection of atelocollagen (BioCollagen group) or saline (Placebo group). Clinical improvement was evaluated over a 24-week period using the 100-mm visual analogue scale (VAS), the Western Ontario and McMaster University Osteoarthritis Index (WOMAC), and the 36-item Short-Form Health Survey (SF-36).
Results
VAS scores were significantly better in the BioCollagen group as compared with the Placebo group at 24 weeks. More patients in the BioCollagen group reported exceeding 20% and 40% VAS improvements. The WOMAC and SF-36 scores were also significantly improved from baseline after the intra-articular injection of atelocollagen; although, the differences between the BioCollagen and Placebo groups were not significant. There were no unexpected or severe adverse events reported for either group.
Conclusions
The results show that an intra-articular injection of atelocollagen effectively alleviates knee pain, as intended. Therefore, the intra-articular injection of atelocollagen can be considered an alternative solution to controlling knee pain due to osteoarthritis and diverse cartilage defects.
Introduction
Articular cartilage, composed mostly of collagen and extracellular matrix proteins, protects joints and enables smooth locomotion. When the articular cartilage is damaged, the collagen network is disrupted, and cartilage cells are no longer protected. The subsequent degradation of the collagen fibrils and other internal matrix molecules can lead to a loss of water from the cartilage, which affects the stability of the osmotic pressure and results in a gradual loss of proteoglycans. These degenerative processes are exacerbated by repetitive strain on the cartilage during joint movement and eventually leads to the development of osteoarthritis.1-3 Osteoarthritis, the most prevalent joint disease in humans, results from the breakdown of articular cartilage.1,4 Osteoarthritic symptoms include joint pain, joint stiffness, and muscle contracture. 5 It mostly affects the quality of life of older people, with the World Health Organization estimating that 10% to 15% of adults aged older than 60 years were affected by osteoarthritis in 2013.1,5
Various options are available for the treatment of osteoarthritis, including exercise, physiotherapy, pharmacotherapy, and intra-articular injections.6,7 Intra-articular injections are usually administered to patients who fail to respond to physiotherapy and pharmacotherapy. Intra-articular corticosteroids and hyaluronic acid (HA) are commonly injected for pain relief and to facilitate movement. However, these treatments have clinical limitations and may further damage the articular cartilage.8-10 Corticosteroid joint injections lead to pain relief that lasts for only a few months, 11 and repeated injections can cause adverse events, such as local tissue atrophy, long-term joint damage, infection, and systemic complications. 12 Although injections of HA lubricate the joint, allowing it to move smoothly, there is no concrete evidence to support the efficacy of HA because of its dynamic turnover rate and short half-life.13-16 Also, platelet-rich plasma (PRP) is an alternative option for intra-articular injection as it can release the growth factor for potential regeneration, and PRP showed comparable clinical improvement to HA for the functional status of knee.6,17-19
The current suite of agents available for intra-articular injections cannot resolve the underlying problem of the cartilage defect, which is linked to a disrupted collagen organization. A better clinical outcome could be expected if a collagen substitute were used to restore the disrupted collagen in the cartilage tissue. Such restoration is possible because of the stable, triple-helical structure and long half-life of collagen: these attributes protect collagen from the enzymatic breakdown that typically affects other injectable substances, such as HA and corticosteroids. 20 Type 1 porcine atelocollagen has been used successfully to restore cartilage when implanted into cartilage defects in combination with bone marrow stimulation techniques, such as microfracture.21-25 We therefore hypothesized that an intra-articular injection of atelocollagen in patients with knee pain due to osteoarthritis or other chondral defects would alleviate joint pain by supplementing collagen amounts in the cartilage defect. This double-blind, randomized controlled trial investigated the clinical efficacy and safety of an intra-articular injection of atelocollagen for pain relief.
Methods
We conducted a multicenter, randomized, double-blind study to provide level I evidence for or against our hypothesis. Our placebo-controlled trial was conducted in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. It was approved by the institutional review board (IRB) of each trial center: Yeouido St. Mary’s Hospital (XC14DSMI0001S); Konkuk University Medical Center (KUH1060071); Samsung Medical Center (SMC2014-01-109); Seoul St. Mary’s Hospital (XC14DSMI0001K); Chung-Ang University Hospital [C2014013(1209)].
Patients
Informed consent was obtained from all participants. Patients who fulfilled the selection criteria and gave written informed consent were included in the clinical trial. The inclusion criteria were as follows: (1) age ≥19 years; (2) presence of knee joint pain due to osteoarthritis, chondromalacia, or other cartilage defects; (3) a Kellgren-Lawrence grade of ≤3; and (4) a visual analogue scale (VAS) score of ≥40 mm. The exclusion criteria were as follows: (1) patients (or family members) with a history of (or ongoing) autoimmune disease; (2) hypersensitivity to porcine protein; (3) the presence of severe effusion; or (4) the presence of other critical diseases (e.g., liver, heart, or kidney disease; cancer; diabetes; mental illness) that may affect the study results. Based on these criteria, a total of 200 patients from the 5 medical centers were enrolled by the participating physicians.
A computer-generated list of random numbers was used to allocate patients to either the BioCollagen group for an intra-articular injection of atelocollagen (CartiZol; Sewon Cellontech, Seoul, Korea) or the Placebo group for an intra-articular injection of saline (Daihan, Gyeonggi, Korea). Atelocollagen used in this trial was type I atelocollagen, which was extracted from porcine skin and designed to maintain collagen’s natural triple-helix structure without using additives for cross-linking.
An independent statistician generated the randomization sequence, which was center-stratified with a 1:1 allocation and block size of 4 using SAS version 9.3 statistical software (SAS Institute, Inc., Cary, NC, USA). Blinding was accomplished by preparing treatment material in a prefilled syringe of the same design as that used to inject saline, with opaque stickers on the syringes that kept the contents hidden from the patients and the physicians throughout the trial. Each patient received a 3-mL dose as a single, intra-articular injection to one affected knee.
Clinical Follow-up
Patients were asked to visit the hospital at 4, 12, and 24 weeks after injection, within ±7 days of the scheduled date. The primary endpoint was the 100-mm VAS at 24 weeks. Secondary endpoints were (1) the Western Ontario and McMaster University Osteoarthritis Index (WOMAC) and 36-item Short-Form Health Survey (SF-36) at 4, 12, and 24 weeks; (2) 100-mm VAS at 4 and 12 weeks; (3) patient satisfaction; (4) physician satisfaction; and (5) physical examination. Descriptive statistics were used to analyze the frequency of adverse events and their relation to the intra-articular injection. Additionally, 20% and 40% improvement in 100-mm VAS was evaluated as an explorative analysis to identify clinically relevant improvement.
Statistical Analysis
A power analysis was conducted to calculate the number of patients required to achieve 80% power to detect significant differences of 28 mm in terms of 100-mm VAS, with an effect size of 13 between the groups and reliability of 95%. Considering the projected dropout of 50%, we needed 100 patients in each group. For the primary analysis, the mean VAS scores were compared between the 2 groups using a full analysis set using a last-observation-carried-forward analysis. Statistical analyses were performed with SAS version 9.3. VAS, WOMAC, and SF-36 were statistically analyzed using an unpaired t test. Safety data were statistically analyzed using an unpaired t test, Wilcoxon rank-sum test, χ2 test, and Fisher’s exact test. Data are expressed as the mean ± SD. Values of P ≤ 0.05 were considered to indicate statistical significance.
Results
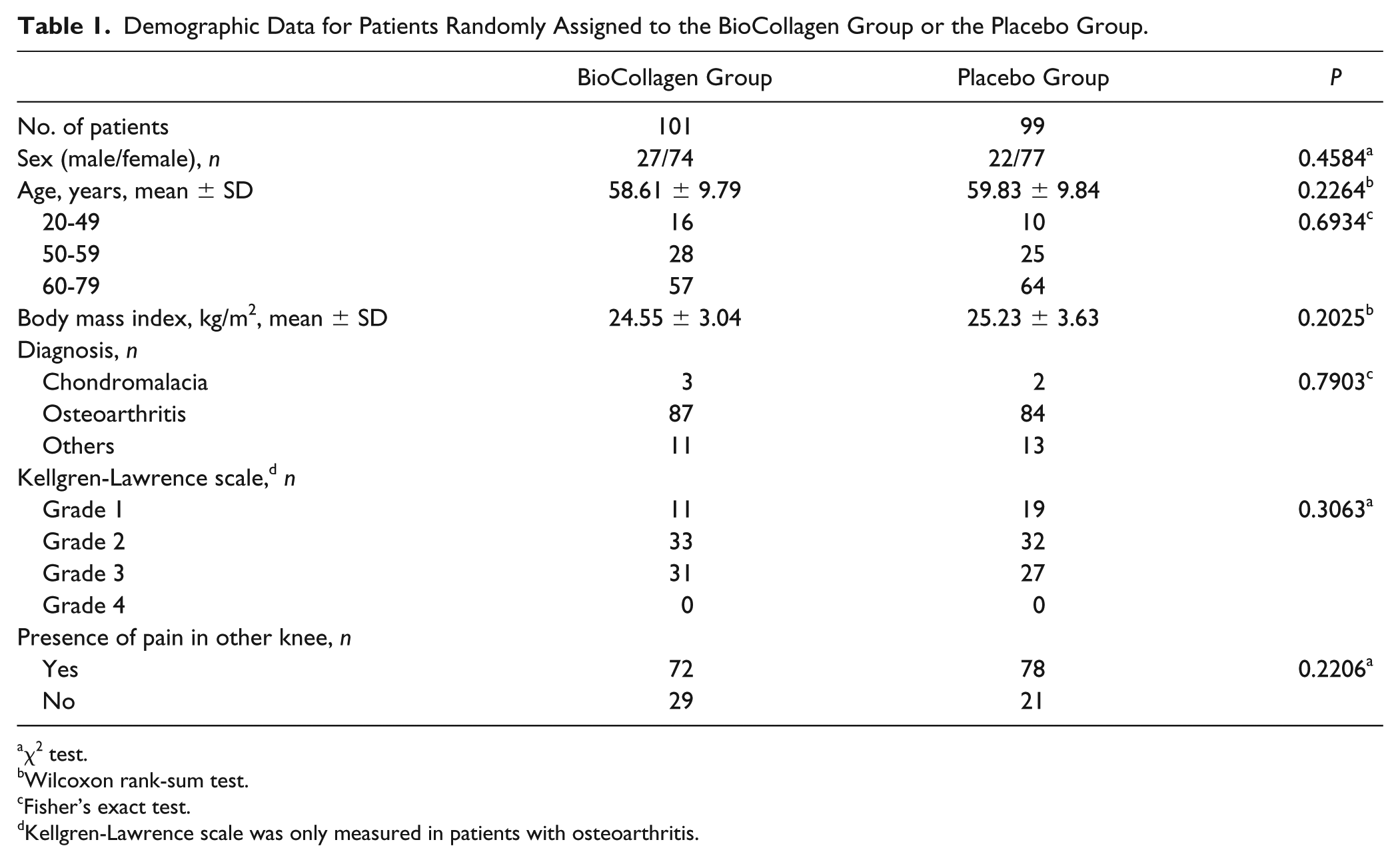
Two hundred patients with osteoarthritis, chondromalacia, or other cartilage defects caused by internal change in the knee joint, meniscal tear, patella fracture, or patellofemoral disorder were recruited between July 8, 2014 and January 29, 2015. Among them, 101 patients were randomly assigned to the BioCollagen group and 99 patients to the Placebo group in consideration of their demographic data ( Table 1 , Fig. 1 ). The clinical evaluation of patients at each visit was recorded and analyzed using a full analysis set.
Demographic Data for Patients Randomly Assigned to the BioCollagen Group or the Placebo Group.
χ2 test.
Wilcoxon rank-sum test.
Fisher’s exact test.
Kellgren-Lawrence scale was only measured in patients with osteoarthritis.
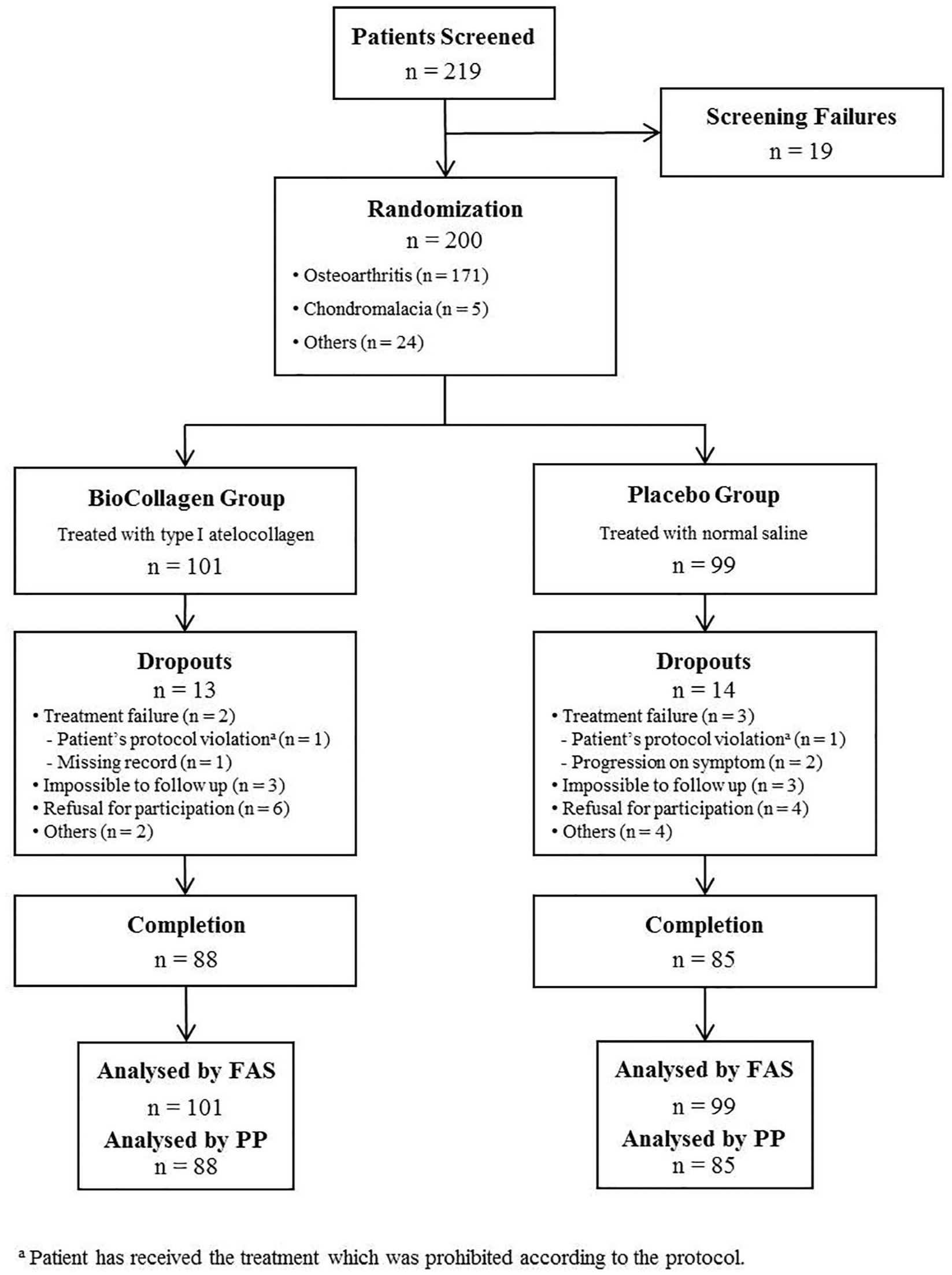
Enrolment and outcomes. Patient assignments for the randomized controlled trial. FAS, full analysis set; PP, per protocol.
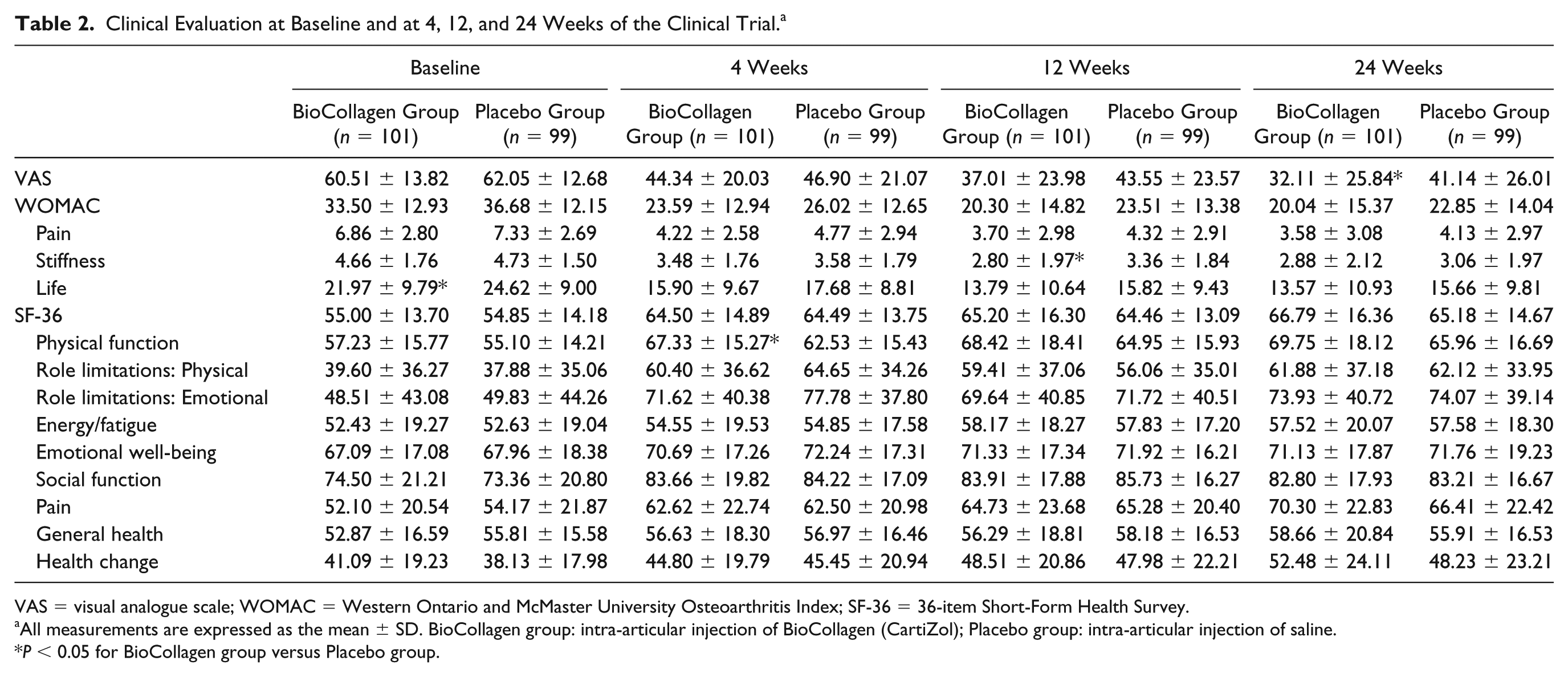
The VAS scores of the BioCollagen group at 24 weeks after injection were significantly better than those of the Placebo group (P = 0.0147) ( Table 2 ). Yet, the VAS scores of the BioCollagen group were not significantly different to those of the Placebo group at 4 and 12 weeks (P = 0.3791 and P = 0.0534, respectively). We further determined the number of patients whose 100-mm VAS improved by 20% and 40% as an exploratory analysis for clinical relevance. More patients in the BioCollagen group than in the Placebo group reported a greater than 20% or 40% improvement in VAS (P = 0.0150 and P = 0.0239, respectively; Fig. 2 ). No significant relations were found between the improvements of VAS and individual patient factors, such as age, sex, height, weight, body mass index, presence of pain on the opposite knee joint, or personal medical history.
Clinical Evaluation at Baseline and at 4, 12, and 24 Weeks of the Clinical Trial. a
VAS = visual analogue scale; WOMAC = Western Ontario and McMaster University Osteoarthritis Index; SF-36 = 36-item Short-Form Health Survey.
All measurements are expressed as the mean ± SD. BioCollagen group: intra-articular injection of BioCollagen (CartiZol); Placebo group: intra-articular injection of saline.
P < 0.05 for BioCollagen group versus Placebo group.
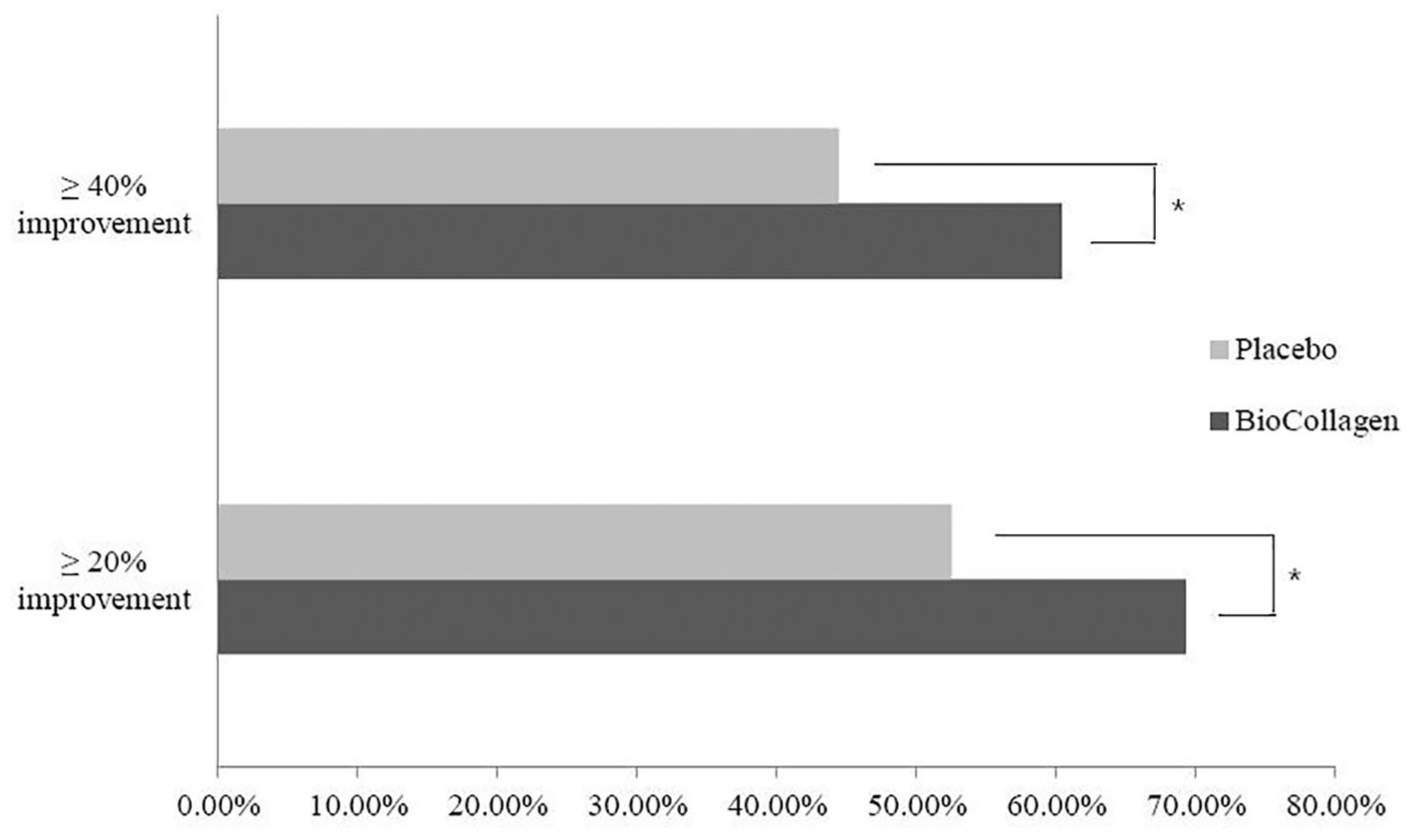
Visual analogue scale (VAS) improvement rates for patients in the BioCollagen and Placebo groups. In the BioCollagen group, 69.31% patients reported a 20% improvement, and 60.40% patients reported a 40% improvement. In the Placebo group, 52.53% patients reported a 20% improvement, and 44.44% patients reported a 40% improvement (χ2 test : *P < 0.05).
The WOMAC of the BioCollagen group was not significantly different from that of the Placebo group at 4, 12, or 24 weeks (P = 0.1816, P = 0.1099, and P = 0.1790, respectively). The SF-36 of the BioCollagen group was also not significantly different to that of the Placebo group at all 3 time points (P = 0.9956, P = 0.7244, and P = 0.4637, respectively). Rates of satisfaction were assessed at 24 weeks for both patients and physicians. Seventy-two patients in the BioCollagen group were satisfied with the result, whereas 57 patients from the Placebo group were satisfied (P = 0.0253). Similarly, physicians were satisfied with 73 clinical results from the BioCollagen group and 61 from the Placebo group (P = 0.0682). No significant changes were observed during the physical examination over the follow-up period.
No significant differences in the rate of total adverse events or rate of occurrence of each adverse event were found between the BioCollagen and Placebo groups ( Table 3 ). All adverse events were resolved by conventional treatment without complications, with none in direct relation with the use of atelocollagen.
Adverse Events Reported During the Clinical Trial.
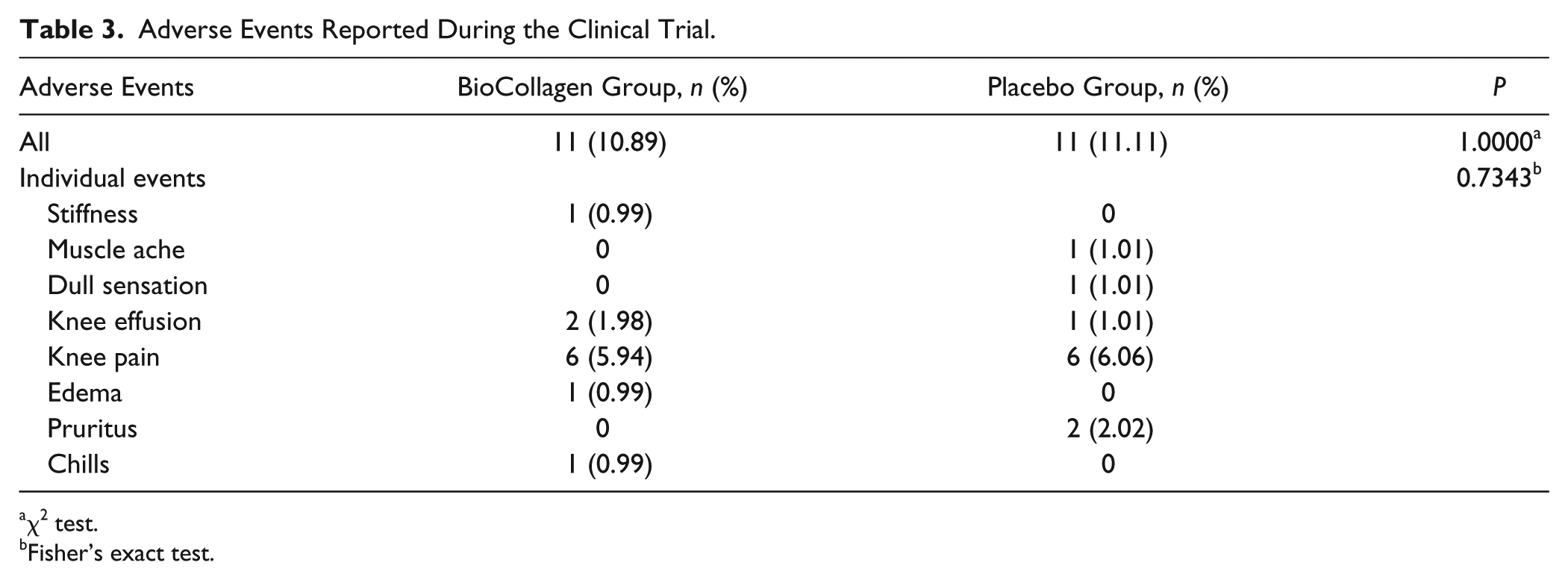
χ2 test.
Fisher’s exact test.
Discussion
The advantages of corticosteroid or HA intra-articular injections for osteoarthritis are unclear. Corticosteroids must be injected repeatedly over time for effective treatment, which can lead to serious adverse events. 12 HA, which alleviates the pain, is unable to correct the damage to the articular cartilage and has a short half-life.13-16 Because both treatments are intended only to relieve pain without correcting the damaged articular cartilage, they may lead to even more serious articular cartilage defects. 8 Hence, there is a need for an intra-articular injection that aims to correct the underlying pathophysiological process as well as provide pain relief.17,26,27 Collagen, the main component of articular cartilage, has a strong triple-helix structure with a longer half-life than corticosteroids or HA. As such, collagen could serve as a good alternative candidate for intra-articular injection with a low risk of serious adverse events.28-31
Collagen has been used in diverse areas of tissue reconstruction and medical therapeutics. Atelocollagen has been applied to the reconstruction of various tissues, including cartilage,21-25 ligaments,32,33 tendons,33,34 dermis,35,36 bone,37,38 and for wound healing. 39 The triple helical structure and lack of antigenicity of atelocollagen ensure its biocompatibility and safety for medical applications.
Previous studies have examined the efficacy of intra-articular injections of collagen for the treatment of knee osteoarthritis. Furuzawa-Carballeda et al. 40 compared the clinical efficacy of multiple injections of polymerized collagen and HA. After 6 months, VAS improvement in the collagen group was significantly better than that in the HA group. 40 Another group studied the clinical efficacy of an intra-articular injection of collagen of porcine origin as compared with HA. 41 They found that, after 6 months, the VAS of the collagen group had significantly improved from baseline, but there was no significant difference between the collagen- and HA-treatment groups. These studies showed that an intra-articular injection of collagen provided a likelihood of clinical improvement in terms of alleviating knee pain.
In this clinical trial, when atelocollagen was injected into the articular capsule, significant pain relief was observed within 6 months among patients with osteoarthritis progression and diverse cartilage defects for both sexes and all ages. Also, statistically significant improvements in the 20% and 40% VAS improvement rate were found, indicative of clinical relevance. Considering these results, the intra-articular injection of atelocollagen can be regarded as an effective treatment for knee pain.
In this investigation, a significant difference was found in the VAS between the BioCollagen group and the Placebo group. Although there was no statistically significant difference between the BioCollagen and Placebo groups for either the WOMAC or SF-36 evaluations, the BioCollagen group showed statistically significant functional improvement after the joint injection as compared with baseline. One possible explanation for this discrepancy is that the standard questions surveyed in the VAS, WOMAC, and SF-36 tests focus on different views and time points. The VAS survey questions are limited to the degree of pain in the treated knee at the time of survey, whereas the questions for the WOMAC focus on the condition of the knee during the past week prior to the survey. In contrast, the SF-36 survey measures patient quality of life over the past 4 weeks as a result of treatment. In addition, considering that 150 patients (75%) previously had pain in their other knee, patients may have confused the condition and pain when scoring the WOMAC and SF-36 tests. Therefore, it is not surprising to find a lack of significance for these 2 tests.
There are some study limitations. The follow-up period was 6 months, which is a generally accepted time to evaluate pain alleviation due to intra-articular injection.42,43 However, it was not intended to observe the long-term effect of pain relief nor any regeneration of articular cartilage. Therefore, additional studies with a longer follow-up period of at least 24 months may be necessary to determine any additional effect of intra-articular injection of type I atelocollagen.
Conclusion
This clinical trial demonstrated that knee pain could be alleviated with a single, intra-articular injection of type I atelocollagen. Thus, this study demonstrates that the intra-articular injection of atelocollagen is a simple, safe, and effective treatment for alleviation of joint pain at 6 months in patients with osteoarthritis, chondromalacia, or other cartilage defects of the knee.
Footnotes
Acknowledgments and Funding
We thank Professor Dong Jae Kim and Research Assistant Su Jung Kim (Department of Medical Life Science School of Medicine, The Catholic University of Korea) for analyzing and reviewing our statistical results. We thank Peter Mittwede, MD, Ph.D. for providing scientific review and Rebecca Jackson, PhD, for editing a draft of this manuscript on behalf of Edanz (![]() ).
).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Our placebo-controlled trial was conducted in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. It was approved by the institutional review board (IRB) of each trial center: Yeouido St. Mary’s Hospital (XC14DSMI0001S); Konkuk University Medical Center (KUH1060071); Samsung Medical Center (SMC2014-01-109); Seoul St. Mary’s Hospital (XC14DSMI0001K); Chung-Ang University Hospital [C2014013(1209)].
Informed Consent
Written informed consent was obtained from all participants before the study.
Trial Registration
Not applicable.
