Abstract
Objective
The superficial zone (SZ) of articular cartilage is responsible for distributing shear forces for optimal cartilage loading and contributes to joint lubrication through the production of proteoglycan 4 (PRG4). PRG4 plays a critical role in joint homeostasis and is chondroprotective. Normal PRG4 production is affected by inflammation and irregular mechanical loading in post-traumatic osteoarthritis (PTOA). THe SZ chondrocyte (SZC) phenotype, including PRG4 expression, is regulated by the actin cytoskeleton in vitro. There remains a limited understanding of the regulation of PRG4 by the actin cytoskeleton in native articular chondrocytes. The filamentous (F)-actin cytoskeleton is a potential node in crosstalk between mechanical stimulation and cytokine activation and the regulation of PRG4 in SZCs, therefore developing insights in the regulation of PRG4 by actin may identify molecular targets for novel PTOA therapies.
Materials and methods
A comprehensive literature search on PRG4 and the regulation of the SZC phenotype by actin organization was performed.
Results
PRG4 is strongly regulated by the actin cytoskeleton in isolated SZCs in vitro. Biochemical and mechanical stimuli have been characterized to regulate PRG4 and may converge upon actin cytoskeleton signaling.
Conclusion
Actin-based regulation of PRG4 in native SZCs is not fully understood and requires further elucidation. Understanding the regulation of PRG4 by actin in SZCs requires an in vivo context to further potential of leveraging actin arrangement to arthritic therapeutics.
Keywords
Superficial Zone Chondrocytes: A Target for Preventing Osteoarthritis
The superficial zone (SZ), also called the tangential zone, is the thinnest layer of articular cartilage that makes up the first 10%-20% of cartilage thickness
1
(
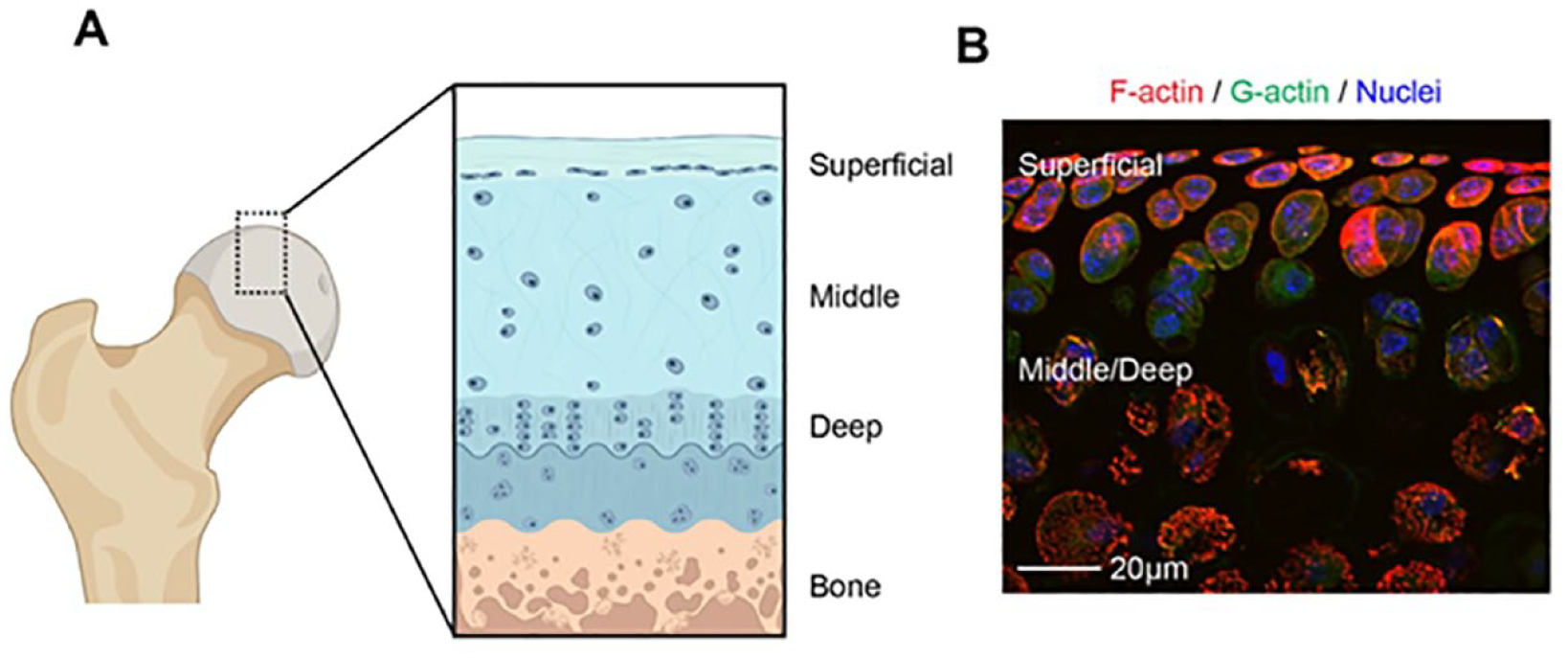
Cellular zonal organization of articular cartilage. (
Lubricin or proteoglycan 4 (PRG4), synonymous with megakaryocyte-stimulating factor, is a mucinous glycoprotein present at the articular surface that functions as a boundary lubricant and is essential for the maintenance of cartilage homeostasis. 9 While also expressed by synoviocytes to make up a component of synovial fluid, PRG4 from articular cartilage is exclusively expressed, produced, and secreted by SZCs. 10 PRG4 is regarded as an essential friction-reducing component between joint surfaces 11 and participates in lowering the coefficient of friction to the order of ~0.01 or less. 12 It works synergistically with hyaluronic acid to produce a lubricating effect.11,13
PRG4 is essential for joint health. In humans, loss-of-function mutations within the PRG4 gene cause camptodactyly-arthropathy-coxa vara-pericarditis syndrome, characterized by pre-adolescent joint abnormalities resulting in precocious joint failure requiring arthroplasty in the third to fourth decade.14,15 PRG4 loss may contribute to OA progression. In rodent16,17 and sheep joint instability models, 18 PRG4 can be found downregulated in OA16-18 and in another sheep study was associated with elevated friction in cartilage contacts. 19 In antigen-induced OA in murine joints, PRG4 expression is reduced at early timepoints. 16 In murine, the lubrication that PRG4 provides is cytoprotective and prevents matrix degradation. 9 In murine knockout models, an increase in friction between joint surfaces causes mitochondrial dysfunction in chondrocytes. 20 PRG4 also plays an anti-inflammatory role by binding to Toll-like receptors 2, 4, and 5 21 in the synovium, 22 resulting in the inhibition of matrix metalloproteinase activity.23-25 This results in joint failure characterized by synovium thickening, cartilage degeneration, and progressive loss of proteoglycans.26,27
While these studies may support that PRG4 plays a role in PTOA progression, other in vivo studies have shown an increase in PRG4 in synovial fluid during OA.28-31 These increases in PRG4 can coincide with an increased coefficient of friction. 32 Therefore, despite elevated synovial PRG4 levels, OA progression still ensues, 33 leading to the contention that the loss of PRG4 is not a driving force of PTOA; although fragmentation of PRG4 remains a possibility which may artifactually raise PRG4 levels. Of note, in OA, the PRG4 secreted is mainly by synoviocytes and under inflammatory conditions may display truncated O-linked glycosylations, 34 impairing its lubricative properties. 35 While there are mixed findings on the regulation of PRG4 expression in PTOA, it has been shown that intra-articular (IA) supplementation of PRG4 to murine joints12,36-38and porcine joints 39 in surgically induced PTOA models prevents chondropathy. In a similar fashion, PRG4 overexpression in mice protects against surgically induced PTOA by inhibiting cartilage catabolism and hypertrophy. 40
Due to the critical role PRG4 plays in healthy, functional joints and its chondroprotective effect, understanding the molecular regulation of PRG4 may lead to the development of novel OA therapies. Studies have indicated several pathways that regulate PRG4 including transforming growth factor beta (TGF-β), Wnt signaling, the epidermal growth factor receptor (EGFR), transient receptor potential vanilloid channel, prostaglandin E2, and parathyroid hormone–related peptide which have been extensively reviewed.17,41-43 In addition, a connection between SZC form and function is demonstrated such that the actin cytoskeleton regulates SZC phenotype, specifically PRG4 expression in vitro44-46 ( Table 1 ). As a critical node in PRG4 signal transduction, the actin cytoskeleton could provide a target to stimulate PRG4 expression.47,48 In this review, we aim to highlight the mechanisms by which actin regulates PRG4 and the potential connections between actin signaling and other known regulators of PRG4 including chemical and mechanical stimuli.
Actin/Actin Signaling Modulation Regulates PRG4.
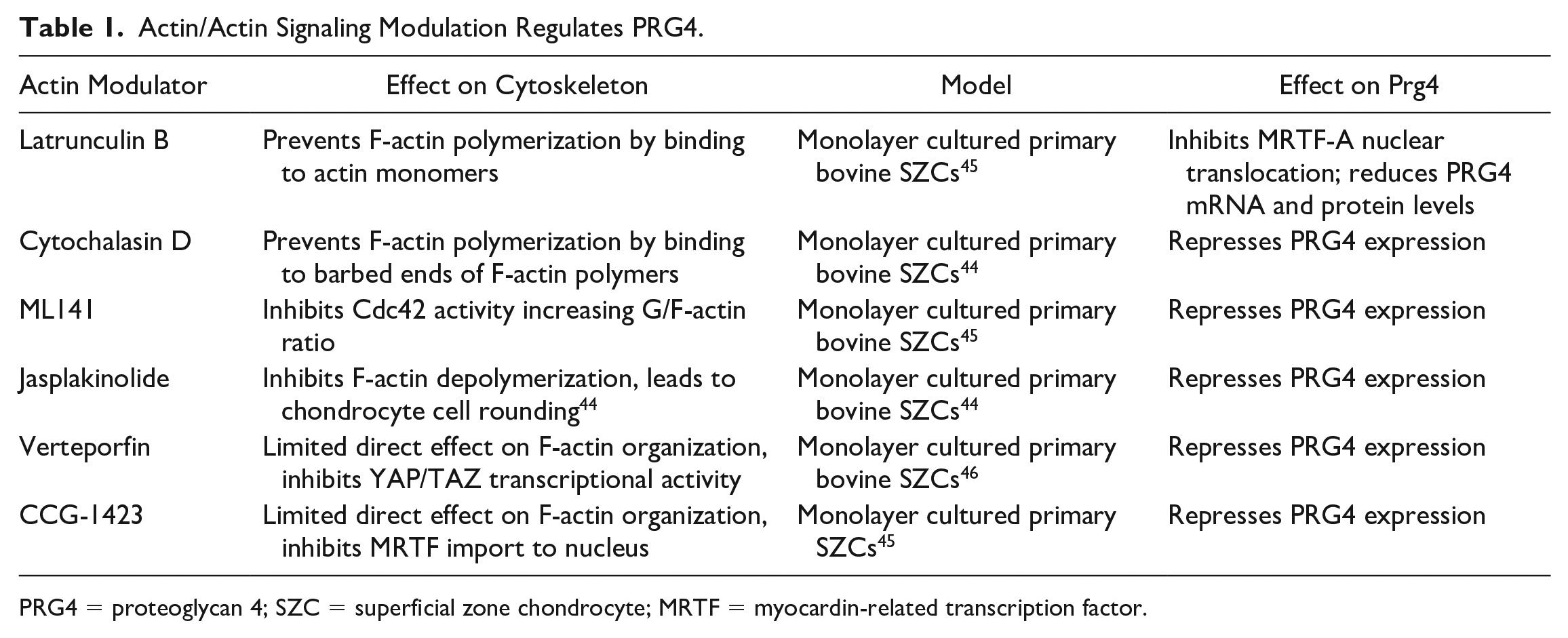
PRG4 = proteoglycan 4; SZC = superficial zone chondrocyte; MRTF = myocardin-related transcription factor.
SZCs Have Unique Cell Morphology and Cytoskeletal Structure
SZCs are thin and relatively densely packed and horizontally arranged parallel to the cartilage surface. SZC are small and densely packed as compared to deep zone chondrocytes (DZCs) which are more large, round and sparsely distributed1,49 (
In comparison with DZCs, SZCs have a unique F-actin organization. While immunofluorescence for F-actin is relatively uniform throughout different cartilage zones in native tissue, western blot analysis of total actin from isolated proteins of native cartilage shows a larger amount of actin in SZCs, suggesting a larger pool of soluble globular (G-)actin in SZCs. 50 A greater pool of G-actin in SZCs may allow for dynamic actin remodeling. Supporting this, when SZCs are isolated in tissue culture, they demonstrate a greater ratio of F/G-actin and a greater propensity to form mature focal adhesions than DZC counterparts. 55 Focal adhesions and their physical connection to mechanosensitive proteins function to drive actin reorganization in response to mechanical stimuli. RNA sequencing data comparing human SZCs to DZCs identifies differential expression of cytoskeletal associated molecules, linked to Rho GTPase cell division control protein 42 homologue (Cdc42) signaling. 56 Taken together, it is likely that SZC F-actin networks are maintained by unique signaling pathways and that remodeling of F-actin networks regulates SZC phenotype.
The Actin Cytoskeleton is a Regulator of Chondrocyte Phenotype Including PRG4
The regulation of the chondrocyte phenotype, specifically that of the SZC phenotype, by cytoskeletal elements is best exemplified by the regulation via F-actin ( Table 1 ). While F-actin is known to regulate intercellular force generation, F-actin dynamics is emerging as a key node in intracellular signaling molecule. 57 The F-actin network–associated proteins is thought to be substantially altered in human and murine OA pathogenesis as transcriptional analysis of differentially expressed genes show the greatest changes in actin cytoskeleton regulatory pathways.58,59 In addition, F-actin reorganization is a potent regulator of chondrocyte phenotype with ability to regulate cartilage matrix, fibroblast matrix, contractile, proliferative, apoptotic, and transcriptional molecule expression, mostly exemplified in vitro.60-62 Notably, changes in F-actin organization are thought to trigger a phenotypic switch in chondrocytes. F-actin reorganization from a cortical arrangement in primary cells to F-actin stress fibers in passaged cells leads to decreased type II collagen and with increased type I collagen expression. In addition to the regulation of cartilage matrix expression, actin reorganization is a regulator of the SZC phenotype including PRG4 expression.44-46,55 Treatment of primary SZCs with latrunculin B, which binds and sequesters actin monomers to prevent F-actin polymerization, 63 increases the ratio of G- to F-actin in cells, leading to a decrease in PRG4 messenger RNA (mRNA) levels 45 ( Table 1 ). Similar effects in SZCs are also observed with cytochalasin D. Cytochalasin D binds to barbed ends of F-actin which also prevents F-actin polymerization in SZC and reduces PRG4 mRNA and protein levels 64 ( Table 1 ).
The downstream regulation of PRG4 via actin has been shown to be through regulating the subcellular localization of 2 signaling transcription factors, Yes-associated protein and its paralog transcriptional coactivator with PDZ-binding motif (YAP/TAZ), and myocardin-related transcription factor (MRTF). 61 YAP/TAZ and MRTF are found in primary SZCs. 55
The F-actin cytoskeleton regulates the subcellular localization of YAP/TAZ, through angiomotin (AMOT). When YAP/TAZ is bound to angiomotin, the YAP/TAZ/AMOT complex localizes in the cytoplasm. Angiomotin contains a conserved actin-binding domain in its N-terminus and competitively binds to F-actin.
65
During F-actin polymerization, YAP/TAZ is liberated from angiomotin and is free to be shuttled into the nucleus (
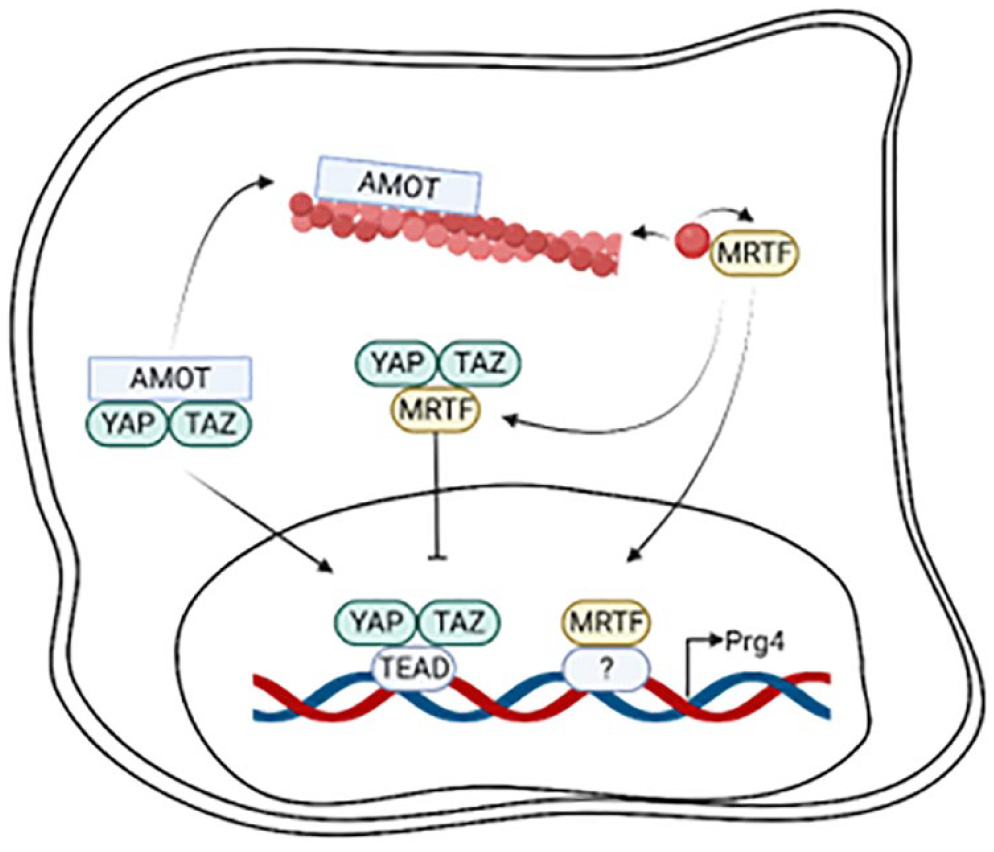
F-actin polymerization drives PRG4 expression through Yes-associated protein and its paralog transcriptional coactivator with PDZ-binding motif (YAP/TAZ). Angiomotin (AMOT) competitively binds to polymerized F-actin, freeing YAP/TAZ to enter the nucleus to bind to cofactor TEAD to induce PRG4 transcription. MRTF has a high affinity for G-actin, and upon F-actin polymerization, MRTF is freed to enter the nucleus to bind to transcriptional cofactors to drive PRG4 expression. Conversely, MRTF can colocalize to YAP/TAZ to sequester the complex in the cytoplasm. PRG4 = proteoglycan 4; AMOT = angiomotin; TEAD = TEA Domain; MRTF = myocardin-related transcription factor. Created with BioRender.com.
F-actin polymerization also regulates MRTF subcellular localization (
F-actin polymerization in SZCs drives PRG4 expression by increasing nuclear localization of YAP/TAZ and MRTF.45,46 YAP/TAZ and MRTF-A signaling may also intersect; MRTF is essential for TAZ expression and the 2 proteins can colocalize to inhibit each other’s nuclear accumulation, 69 suggesting coordinated regulation of PRG4 downstream of actin polymerization.
F-actin as a Critical Node in the Regulation of PRG4 by Growth Factors and Cytokines
The mechanistic regulation of PRG4 by growth factors/cytokines has led to the identification of various signaling pathways that contribute to PRG4 expression which intersect upon the F-actin cytoskeleton (
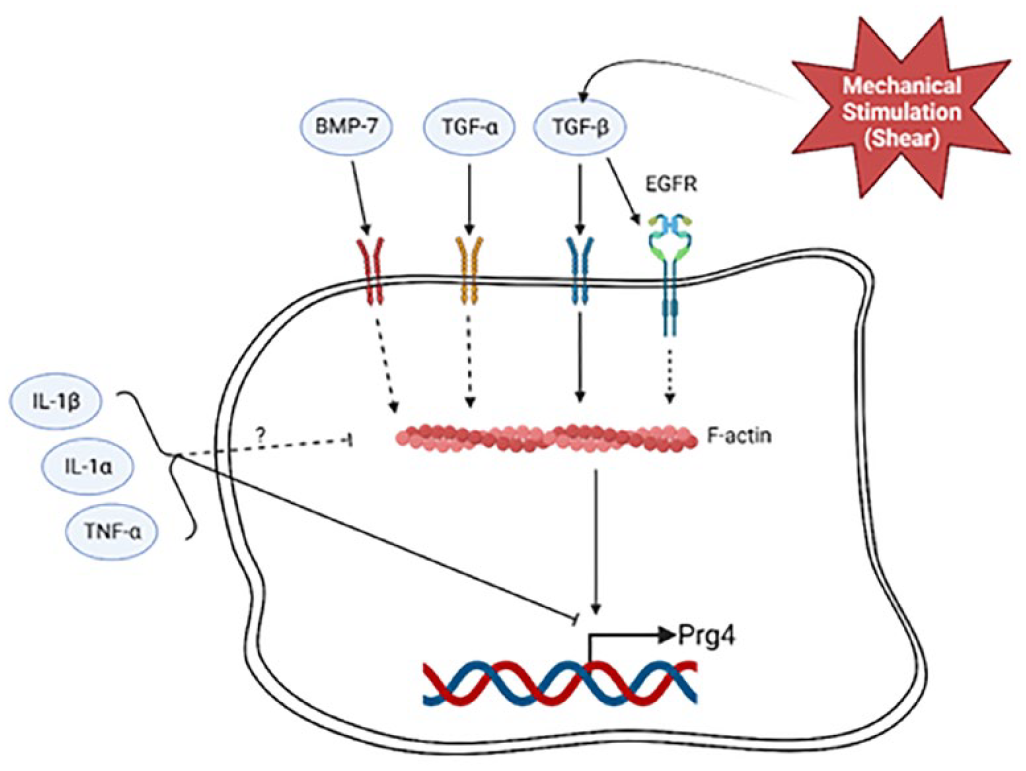
Schematic of converging signaling pathways that mediate PRG4 expression upon the actin cytoskeleton. Dashed lines depict regulatory mechanisms elucidated in cell types other than SZCs. Growth factor stimulation via BMP-7, TGFβ, and TGFα stimulates PRG4 and promotes F-actin polymerization. Mechanical stimulation, in part, regulates PRG4 through TGFβ. Prg4 is inhibited by TNFα, IL1α, and IL1β which may decrease actin polymerization, though there remains confounding evidence. PRG4 = proteoglycan 4; SZC = superficial zone chondrocyte; BMP7 = bone morphogenic protein 7; TGFβ = transforming growth factor beta; TGFα = transforming growth factor alpha; IL1α = interleukin-1 alpha; IL1β = interleukin-1 beta. Created with BioRender.com.
TGFβ also activates EGFR-mediated pathways. TGFβ2 through EGFR, increases PRG4. This requires the binding of transcription factor Creb5 to 2 proximal cis-elements, E1 and E2. 77 E3, a distal element, drives the transcriptional activity induced by TGF-β2. In epithelial cells and fibroblasts, activation of EGFR promotes F-actin polymerization and nuclear MRTF localization.78,79 Furthermore, the EGFR itself has an actin-binding domain (residues 984-996) and associates with F-actin, 80 although the nature of this interaction in regulating downstream gene expression is unknown.
Bone morphogenic protein 7 (BMP7) is another member of the TGFβ superfamily, which works synergistically with TGFβ to increase PRG4 accumulation.70-72,81-85 While it is unclear whether BMP7 induced changes to the cytoskeleton in SZCs, in Swiss3T3 cells, BMP7 induces rapid F-actin polymerization, even more so than TGFβ. 86 The regulation of F-actin by BMP7 in SZC to regulate PRG4 requires further investigation.
In contrast to growth factors, exposure of SZCs to inflammatory cytokines, including interleukin-1 alpha (IL1α), interleukin-1 beta (IL1β), and tumor necrosis factor alpha (TNFα), significantly reduces PRG4 accumulation.72,85,87 Treating both isolated rat chondrocytes 88 and bovine cartilage explants 89 with IL1α reduces levels of PRG4. Notably, this change in expression is rescued in monolayer cultures via growth factor treatment by either TGFβ173,88 or BMP7. 72 Inflammatory cytokines influence chondrocyte cell morphology and cytoskeleton arrangement. IL1α depolymerizes F-actin networks in SZCs via increased Piezo1 activity. 90 IL1β changes chondrocyte cell morphology as treatment condenses chondrocyte shape yet maintains F-actin protrusions. 91 Confounding data shows IL1 increased F-actin in both isolated chondrocytes and cartilage explants in vitro. 92 This may be via Rho GTPase activity as both TNFα and IL1 activate Cdc42 in fibroblasts. 93 Taken together, a connection between F-actin polymerization and inflammatory cytokines in vitro exists.
Mechanical Stimulation as a Regulator of PRG4
Mechanical stimulation is critical for PRG4 expression. Movement of joints with natural loading stimulates PRG4; mice housed with running wheels express more PRG4 expression in their knee joints than those without a running wheel. 94 In larger scale joint models, continuous passive motion applied to bovine joints ex vivo increases PRG4 synthesis and secretion. 95 Bovine cartilage explants under dynamic shear stimulation secrete more PRG4 than compression alone. 96 Mechanical stimulation required for PRG4 synthesis is dependent on the TGFβ pathway as inhibiting TGFβ signaling decreases mechanically induced PRG4. 97 Interestingly, injurious mechanically overloading cartilage tissue results in initial elevation of PRG4 synthesis from SZCs at 2 days post-load but decreases by 6 days in culture. 98 This is concurrent with an increased coefficient of friction of tissue surface.32,98 The early increase of PRG4 may indicate a compensatory mechanism as SZC attempt to repair the injury, which eventually may be overcome by cellular death. In human ankle cartilage, loading applied in a bioreactor not only stimulates PRG4 production but also reduces SZC viability. 99
In 2-dimensional (2D) and 3D chondrocyte culture, PRG4 is upregulated under application of cyclic tensile strain, 100 shear stress, 94 and a combination of multi-axial loading. 101 Cellular responses due to mechanical stimulation may depend heavily on F-actin cytoskeleton reorganization as mechanical stimulation directly affects F-actin polymerization. Mechanical forces can be used to induce both polymerization and depolymerization of F-actin. Polymerization is force-dependent; at low levels of force (50 pN), G-actin polymerizes into F-actin, while strong forces (150 pN) cause the F-actin to depolymerize. 102 The type of mechanical loading also affects polymerization; F-actin dissociates under dynamic loading, and to a lesser extent under static loading. 103 F-actin arranges to form semiflexible polymers to mechanically stabilize the intracellular system. 104 F-actin architecture is regulated by actin binding proteins such as crosslinkers that confine filament movement and provide higher order structure. Furthermore, actin-binding proteins themselves can attune their response to mechanical stimulation of F-actin. For example, the binding rate of cofilin, an F-actin-severing protein, is decreased when F-actin is tensed at low levels. F-actin-binding proteins can regulate the behavior of F-actin networks in response to mechanical stimuli which drives responsive cellular functions.
Actin-Binding Proteins as Regulators of F-Actin Organization and the SZC Phenotype
F-actin-binding proteins regulate polymerization dynamics via different functions, maintaining a pool of G-actin readily available for polymerization, nucleation of new filaments, filament capping and severing, sustaining filament elongation, and capping filaments. 105 In passaged chondrocytes, knockdown of cofilin-1, an actin-depolymerizing factor, increases actin polymerization and collagen type I expression. 61 Adseverin, an actin capping and severing protein, also regulates chondrocyte phenotype by maintaining the proportion of G- and F-actin and expression of aggrecan and the chondrogenic transcription factor Sox9. 106 Proteomic analysis of chondrocytes derived from human patients shows actin-binding proteins destrin and cofilin-1 are downregulated in OA, and cofilin-2 and gelsolin are upregulated. 107 This suggests that actin-binding protein composition is critical in chondrocyte homeostasis and distinguishes diseased cartilage from healthy. In OA, further understanding of the mechanisms that regulate F-actin may provide new insights into the causes of F-actin-dependent reduction of PRG4 expression. Of particular interest is understanding the actin-binding proteins that confer F-actin stability, such as tropomyosins (Tpms). There are over 40 non-functionally redundant Tpm isoforms found in both muscle and non-muscle cells, each providing a unique function in organizing actin structures critical for metabolic and locomotive activities. 108 Tpms are elongated proteins that directly bind along 7 consecutive actin subunits on the long-pitch helix of actin filaments. After binding, Tpms polymerize end-to-end and regulate the affinity of F-actin to various actin-binding proteins with different functions such as severing, capping, contractility, cross-linking, and depolymerizing.108,109 Thus, Tpms are considered master regulators of F-actin as specific Tpm isoforms define F-actin organization. In our previous studies, we have determined that F-actin organization in lens epithelial cells and tenocytes is dependent on the specific isoform Tpm3.1.110,111 The regulation of SZC and the overall chondrocyte phenotype by specific Tpm isoforms or other actin-binding proteins requires future exploration.
Toward Understanding the Mechanisms Regulating SZC PRG4 by F-actin
Mechanistic studies on the regulation of PRG4 by F-actin have been performed using isolated SZCs in vitro. It is critical to study actin regulation of SZCs in the context of the native tissue environment because of culture dependent F-actin reorganization. To counterbalance the mechanical stiffness of the tissue culture environment, chondrocytes grown in monolayer form F-actin stress fibers which drives phenotypic changes.61,112 It is still undetermined whether the stress fibers that form in cultured chondrocytes are biologically relevant to chondrocytes in native cartilage. Since F-actin structure and function are culture context-specific, 113 it is necessary to study F-actin regulation of SZCs in physiologically and mechanically representative environments, ideally in vivo, although this poses technical challenges.
Existing tools may be used to study actin regulation of SZCs in more fitting native environments than monolayer cultures on 2D tissue culture plastics. F-actin in native SZCs has been visualized in 3D by reconstructing confocal images of sections of cartilage tissue, 38 as well as en face in situ hybridization which shows punctate actin around the cortex of cells. 41 Since these native tissue studies require fixed tissues, it remains undetermined whether F-actin is dynamic in native tissue as in vitro. Future studies should aim to delineate if F-actin reorganizes in native SZCs, and if SZC actin dynamics play the same role in vivo as in vitro.
Live imaging of 3D culture of SZCs may provide a promising ex vivo approach with continuum of in vitro to in vivo experimentation. Like native tissue, 3D-cultured chondrocytes have cortical F-actin arrangement. 74 To study actin dynamics in 3D culture, transduction of plasmids containing green-fluorescent-protein (GFP)-actin has been used to examine the real-time effects IL1α and TGFβ have on cytoskeletal organization in chondrocytes. 74 LifeAct-GFP, a small peptide labeled with GFP with affinity for microfilaments, has been shown to be a better alternative with less interference upon actin dynamics and provides clearer actin visualization,114,115 however not without LifeAct artifacts. 116 Transgenic LifeAct-GFP mice are promising for live imaging tool as they have normal growth and development and provide observation of live actin dynamics. 117 Future studies may use these tools to study SZC actin regulation of PRG4 in real time in the context of 3D or native tissue environments.
Targeting PRG4 via the F-actin Cytoskeleton as a Therapy for PTOA
Currently, non-surgical interventions to prevent PTOA after injury are limited, thus there is a need to identify preventative therapies. Targeting the F-actin cytoskeleton could prevent the loss or restore PRG4 levels in joint disease. For several diseases, drugs that modify the actin cytoskeleton are currently being assessed in phase 4 clinical trials including fluvoxamine, which inhibits actin polymerization, and pimozide, which delays actin nucleation via ARP2/3. 43 To promote PRG4 production in SZCs, F-actin structures that are critical for mediating downstream entry of transcription factors to the nucleus would be preserved. To our knowledge, there are limited drugs that stabilize F-actin in vivo. Jasplakinolide, a cytotoxic peptide from the marine sponge, can promote F-actin in vitro, but has confounding effects of F-actin destabilization in vivo. 118 To our knowledge, jasplakinolide is not clinically used.
A challenge in the development of actin-modifying therapeutics for PTOA treatment is delivering the drug in a joint-specific manner. Because actin is an abundant protein, it may be unlikely that an actin-modifying drug could be admin that an actin-modifying drug could be administered systemically as the therapeutic may have off-target effects. General actin depolymerization would be detrimental as demonstrated by latrunculin’s potential to kill cancer cells. 119 A reasonable drug delivery would be IA injections to deliver therapeutics directly to the joint. In addition, a drug targeting–specific F-actin networks could modulate actin-binding proteins such as Tpms. Tpms are emerging as a promising anti-cancer target as specific isoforms are overexpressed in cancers such as urothelial bladder cancer, epithelial ovarian cancer, and renal cell carcinoma.120-124 Tpms that are aberrantly expressed provide druggable targets and have potential to aid in cancer treatment. A further understanding of Tpms in PTOA may provide new therapeutic opportunities.
Since a fine-tuned balance between F-actin polymerization and depolymerization is necessary for proper chondrogenic expression, targeting actin may be suitable for preventing PTOA. After a joint injury leading to PTOA, aberrant mechanical loading and/or an influx of inflammatory cytokines may cause alterations in F-actin structure, leading to dysregulation of chondroprotective molecules like PRG4. If an actin-binding protein is found to promote a favorable actin arrangement for downstream transcription of PRG4, this may be sufficient target for an OA therapy. By studying how actin networks become altered early in arthrosis before tissue remodeling occurs may elucidate molecular targets for OA. Further understanding of in vivo actin dynamics in healthy versus diseased cartilage and key actin-binding proteins that mediate actin reorganization is necessary for the development of novel actin-based therapeutics.
Supplemental Material
sj-jpg-1-car-10.1177_19476035231223455 – Supplemental material for The Actin Cytoskeleton as a Regulator of Proteoglycan 4
Supplemental material, sj-jpg-1-car-10.1177_19476035231223455 for The Actin Cytoskeleton as a Regulator of Proteoglycan 4 by Sofia Gonzalez-Nolde, Cameron J. Schweiger, Elizabeth E.R. Davis, Thomas J. Manzoni, Samer M.I. Hussein, Tannin A. Schmidt, Stephanie G. Cone, Gregory D. Jay and Justin Parreno in CARTILAGE
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Delaware Center for Musculoskeletal Research from the National Institutes of Health’s National Institute of General Medical Sciences under grant P20GM139760; and, by the Delaware INBRE program from the National Institute of General Medical Sciences – NIGMS (P20 GM103446) from the National Institutes of Health and the State of Delaware.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Schmidt and Jay have interest stakes in Lubris, LLC.
Ethical Approval
Since this is a narrative review of previously published data, ethical approval was not needed for this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
