Abstract
Objectives
α2-Macroglobulin (A2M) can prevent cartilage degeneration by blocking many types of cartilage-degrading enzymes, but the mechanism remains to be clarified. This study aimed to test that A2M protects against cartilage degeneration by promoting chondrocyte proliferation and cartilage matrix synthesis via inducing proliferating cell nuclear antigen (PCNA).
Design
The cartilage degeneration of the anterior cruciate ligament transection (ACLT) model was evaluated by Safranin O-fast green staining, and articular cartilage degeneration was graded using the Osteoarthritis Research Society International (OARSI)-modified Mankin criteria. The chondrocyte proliferation was detected by 5-Bromodeoxyuridinc (BrdU), MTT, and Cell Counting Kit-8 (CCK8) methods. The chondrocyte apoptosis was detected by lactate dehydrogenase (LDH) assay and Annexin PI staining with the flow cytometer. The glycosaminoglycan (sGAG) and aggrecan in culture supernatant were measured by enzyme-linked immunosorbent assay (ELISA). Reverse transcription-quantitative polymerase chain reaction (RT-qPCR) was used to analyze the type II collagen and aggrecan mRNA expression. The PCNA protein expression was analyzed by western blot and immunofluorescent staining.
Results
A2M can attenuate cartilage degeneration in ACLT rats. The OARSI scores for cartilage degeneration in the A2M group were lower than those in the phosphate-buffered saline (PBS) group. A2M can promote chondrocyte proliferation and inhibit chondrocyte apoptosis, promote the cartilage matrix synthesis in chondrocytes (type II collagen and aggrecan), and culture supernatant (sGAG and aggrecan). At the same time, it also up-regulated the PCNA protein expression in chondrocytes.
Conclusions
A2M can promote chondrocyte proliferation and cartilage matrix synthesis via inducing PCNA expression.
Introduction
Osteoarthritis (OA) is the most common form of arthritis worldwide; the number of OA sufferers will increase as the life expectancy of the global population rises. 1 OA is a degenerative joint disease characterized not only by progressive cartilage matrix degradation but also by synovial membrane inflammation, osteophyte formation, and subchondral bone sclerosis. 2
Cartilage is a tissue composed of only one cell type, chondrocytes, wrapped in a collagen-rich extracellular matrix they synthesize. During OA onset, chondrocytes undergo a panel of modifications induced by the cytokines and other injuries: proliferation, autophagy, cell death, induction of catabolic molecules, and repression of anabolic events. It is generally believed that degeneration of cartilage in OA is characterized by 2 phases: a biosynthetic phase, during which the cells resident in cartilage, the chondrocytes, attempt to repair the damaged extracellular matrix; and a degradative phase, in which the activity of enzymes produced by the chondrocytes digests the matrix, matrix synthesis is inhibited, and the consequent erosion of the cartilage is accelerated. 3 Up-regulation of cartilage catabolic cytokines and enzymes is thought to be a key mechanism of cartilage damage. Thus, inhibition of these molecules will likely slow or prevent the progression of osteoarthritis.
α2-Macroglobulin (A2M) is a member of the α-macroglobulin family of proteins found in the circulation of a broad range of species. 4 A2M is a major serum protease inhibitor, which can inhibit all classes of endoproteases.5,6 Our group indicated that A2M can prevent cartilage degeneration by blocking many types of cartilage-degrading enzymes. 7 Our hypothesis was that the cartilage-protective effect of A2M may promote the chondrocyte proliferation and cartilage matrix synthesis via inducing the proliferating cell nuclear antigen (PCNA) expression. In order to verify the hypothesis, we observed the chondrocyte proliferation, apoptosis, the cartilage matrix components synthesis, and the PCNA expression in cells and its supernatant.
Methods
Rat ACLT OA Model and Treatment With Supplemental Intra-Articular A2M
The study was approved by the Institutional Animal Care and Use Committee of Rhode Island Hospital. The animals were a subset of those used for a previous study. 8
Two-month-old male Wistar rats were purchased from Charles River and randomly divided into 7 groups and treated with different doses of A2M and its variants. This study only observed 3 groups: (1) sham; (2) anterior cruciate ligament transaction (ACLT) + PBS; or (3) ACLT + A2M (0.153 mg). ACLT or sham surgery was performed on the left knee. A2M dissolved in 20 μL of PBS was injected intra-articularly 24 hours and 14 days after ACLT. Animals in sham group and ACLT + PBS group received an equivalent volume of PBS at identical time points to the A2M group in their left knees to control for any procedural effects. All animals were euthanized at week 8 after the operation.
Safranin O-Fast Green Staining
The proximal tibiae were removed from the harvested joints and immersed in 10% formalin for 72 hours. The specimens were decalcified in 20% ethylenediaminetetraacetic acid solution (pH 7.2). The specimens were embedded in the paraffin block. The blocks were trimmed to expose the cartilage. Two serial 6-μm-thick sections were stained with Safranin O-fast green. 9 The Osteoarthritis Research Society International (OARSI)-modified Mankin was used to measure tibial cartilage degeneration in each joint compartment.
Chondrocyte Culture
ATDC5 (sigma, 99072806) were cultured in Dulbecco’s Modified Eagle’s Medium/Nutrient F-12 Ham (DMEM/F12) (Gibco), including 10% fetal bovine serum (FBS, Gibco), 100 μg/mL streptomycin, and 100 U/mL penicillin (Gibco) at 37°C in 5% CO2. When ATDC5 cells reached 80% confluence, the medium was changed to the differentiation medium which contains 1% Insulin-Transferrin-Selenium Supplement (ITS, Gibco). After that, ATDC5 cells were plated in 8-well chambers at 1 × 105 cells/well or in 6-well culture plates or in a 10 cm dish. At 80% confluence, the cells were treated with different concentrations of A2M (Sigma, M6159).
BrdU Labeling
The ATDC5 were treated with A2M (200 nM) for 24 hours. BrdU was added to the culture medium before 24 hours. BrdU staining was performed using the protocol provided by the ZYMED BrdU staining kit (Streptavidin-biotin system for BrdU Staining, CAT.NO. 93-3943). Remove the labeling medium from cells and wash in several changes of 1× PBS. Fix cells in 70% alcohol for 30 minutes at 4°C. Wash in 3 changes of distilled water for 2 minutes each. Incubate with denaturing solution for 30 minutes. After incubation, rinse in 1× PBS (2 min, 3 times). Apply a sufficient quantity of blocking solution to cover the specimen. Incubate at room temperature and drain or blot off the solution. Add sufficient biotinylated mouse anti-BrdU antibody to cover the specimen. Incubate at room temperature. Rinse with 1× PBS (2 min, 3 times). Add sufficient streptavidin-peroxidase antibody to cover the specimen. Incubate at room temperature. Rinse with 1× PBS (2 min, 3 times). Add a sufficient quantity of the 3,3’-Diaminobenzidine (DAB) mixture to cover the specimen. Incubate for 3 minutes. Rinse well with distilled water. Counterstain with a sufficient quantity of hematoxylin to cover the specimen. Wash slides in tap water. Put slides into 1× PBS until sections turn blue. Rinse in distilled water. Dehydrate slides in a graded series of alcohol and clear with xylene. Add 2 drops of histomount and coverslip.
Cell Counting Kit-8
The ATDC5 were treated with A2M (200 nM) for 24 and 48 hours. The cell counting was performed using the protocol provided by the Cell Counting Kit-8 (CCK8, Sigma-Aldrich, USA). ATDC5 cells were incubated overnight in a 96-well plate (2,000 cells/well; 100 μL). Add 10 μL of the CCK-8 solution to each well of the plate. Incubate the plate for 2 hours in the incubator. Measure the absorbance at 450 nm using a microplate reader (Spectra Max 190 absorbance microplate [A450] drawer). Prepare a calibration curve using the data obtained from the wells that contain known number of viable cells.
MTT
The ATDC5 were treated with A2M (200 nM) for 24 and 48 hours. The cell proliferation was detected by MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) method. MTT (Sigma-Aldrich, USA) solution (5 mg/mL) was prepared in 1× PBS and mixed by the vortex. The solution was filtered after adding MTT. ATDC5 cells were incubated overnight (2,000 cells/well; 100 μL). Discard media from cell cultures. Add 100 μL of serum-free media and 10 μL of MTT solution into each well. Incubate the plate at 37°C for 2 hours. After incubation, add 150 μL of dimethyl sulfoxide (DMSO) into each well. Wrap plate in foil and shake on an orbital shaker for 15 minutes. Read absorbance at OD 490 nm.
Reverse Transcription-Quantitative Polymerase Chain Reaction of Matrix Synthesis
The ATDC5 were treated with A2M (200 nM) for 24 hours. The mRNA levels of COLII and aggrecan were quantified by RT-qPCR. Total RNA was isolated from 24-hour culture cells with the RNeasy isolated kit (Qiagen). One microgram of total RNA was transcribed into cDNA by using the iScript cDNA synthesis kit (Bio-Rad Laboratories), and 40 ng/μL of the resulting cDNA was used as the template to quantify the relative content of mRNA by using the QuantiTect SYBR Green PCR kit (Qiagen) with the CFX Connect Real-Time System (BIO-RAD). Primer pairs were as follows: for mouse COL II, AGG-GCA-ACA-GCA-GGT-TCA-CAT-AC (forward) and TGT-CCA-CAC-CAA-ATT-CCT-GTT-CA (reverse); for mouse Aggrecan, CAG-TGC-GAT-GCA-GGC-TGG-CT (forward) and CCT-CCG-GCA-CTC-GTT-GGC-TG (reverse). 8 The 18S RNA was amplified as the internal control. The cycle threshold values for target genes were measured and calculated with computer software (BIO-RAD). Relative transcript levels were calculated according to the equation x = 2−∆∆Ct, where ∆∆Ct = CtE − ∆CtC (∆CtE = Ct exp-Ct18S, ∆CtC_CtC_Ct18S). 10
Measurements of Glycosaminoglycan Release
The ATDC5 were treated with A2M (200 nM) for 48 hours. Cumulative glycosaminoglycan (sGAG) concentration in the culture supernatant was determined photometrically using the dimethyl methylene blue dye assay with bovine chondroitin sulfate as a standard control at a wavelength of 525 nm, as previously reported. 11
Measurement of Aggrecan in the ATDC5 Culture Supernatant
The ATDC5 were treated with A2M (200 nM) for 48 hours. Aggrecan concentration in the ATDC5 culture supernatant was determined by the enzyme-linked immunosorbent assay (ELISA; GWB-DE7FB7, GENWAY).
Measurement of Lactate Dehydrogenase Activity
The ATDC5 were treated with A2M (50, 100, 200 nM) for 24 and 48 hours. The cell toxicity was assessed by quantitative measurement of lactate dehydrogenase (LDH) release into the culture medium, using the LDH assay kit (CytoTox 96 Non-Radioactive Cytotoxicity Assay, Promega). ATDC5 was lysed by adding 15 μL of lysis 10x Solution per 100 μL of culture medium, followed by incubation at 37°C for 45 minutes. Sample supernatant (50 μL) was added to each supernatant sample, and the enzymatic reaction was allowed to proceed for 30 minutes at room temperature, protected from light. The enzymatic assay was then stopped by adding 50 μL/well of the Stop Solution. The plate can be read at 490 nm using an ELISA plate reader. The number of cells present would be directly proportional to the absorbance values, which represent LDH activity.
Annexin PI Staining With Flow Cytometry
The ATDC5 were treated with A2M (200 nM) for 48 hours. The cell number was determined by Annexin PI staining with flow cytometry. Wash 5 × 104 cells in 200 μL of 1× Binding Buffer and centrifuge at 300×g for 10 minutes. Aspirate supernatant completely. Resuspend pellet in 20 μL of 1× Binding Buffer per well. Add 2 μL of Annexin V-FITC per well. Mix well and incubate for 15 minutes in the dark at room temperature. Wash cells by adding 200 μL of 1× Binding Buffer per well and centrifuge at 300×g for 10 minutes. Aspirate supernatant completely. Resuspend cell pellet in 50 μL of 1× Binding Buffer per well. Add 2 μL of the PI solution immediately prior to analysis by flow cytometry.
Immunofluorescent Staining
The ATDC5 were treated with A2M (200 nM) for 48 hours and in fixative for 30 minutes at room temperature. Wash 2 minutes ×3 times with 1× PBS. 100 μL of PCNA primary antibody (sc-56, Santa Cruz Biotechnology) was incubated in a moist chamber for 1 hour at 37°C (PCNA, 1:50). Wash 2 minutes ×3 times with 1× PBS. 10 μL mouse rhodamine fluorescein (1:500 in 1% bovine serum albumin (BSA) in 1× PBS containing 1:1,000 Hoechst) was incubated for 30 minutes at 37°C. Wash 2 minutes ×3 times with 1× PBS. After mounting, laser confocal microscopy (LSM800, Leica) was used for observation.
Western Blot Analysis of PCNA
The ATDC5 were treated with A2M (200 nM) for 48 hours. The protein level of PCNA was determined by Western blot. Total protein was extracted from 48-hour culture cells using the RIPA lysis (9806, Cell signaling) and quantified by BCA Protein Assay Kit (Pierce). Fifteen micrograms of protein was separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to nitrocellulose membranes for immunoblot analysis and stained with specific primary antibodies. The following primary antibodies were used: PCNA antibody (1:1000, sc-56, Santa Cruz Biotechnology) and β-actin antibody (1:1000, 13E5, Cell Signaling Technology). Fluorescence-labeled secondary antibodies were detected with a fluorescence scanner (Odyssey; LI-COR Biosciences). Band densities were quantified using Image software.
Statistical Analysis
Data were reported as mean ± SD. Two-tailed paired t tests were used to compare the difference between the control group and the experiment group. Statistics and creation of the graphs were carried out in GraphPad Prism 7.0.0. Statistical analyses were performed using SPSS software (v18.0; IBM Corp). P-values ≤ 0.05 were considered statistically significant.
Results
A2M Can Attenuate Cartilage Degeneration
Safranin O-fast green staining showed that, compared with ACLT + PBS rats, tibial cartilage from ACLT + A2M rats showed more intense staining that spanned the superficial, middle, and deep cartilage zones, indicating higher sGAG content, smoother, less fibrillated cartilage surfaces, and preservation of superficial zone features, including larger number of superficial zone chondrocytes (
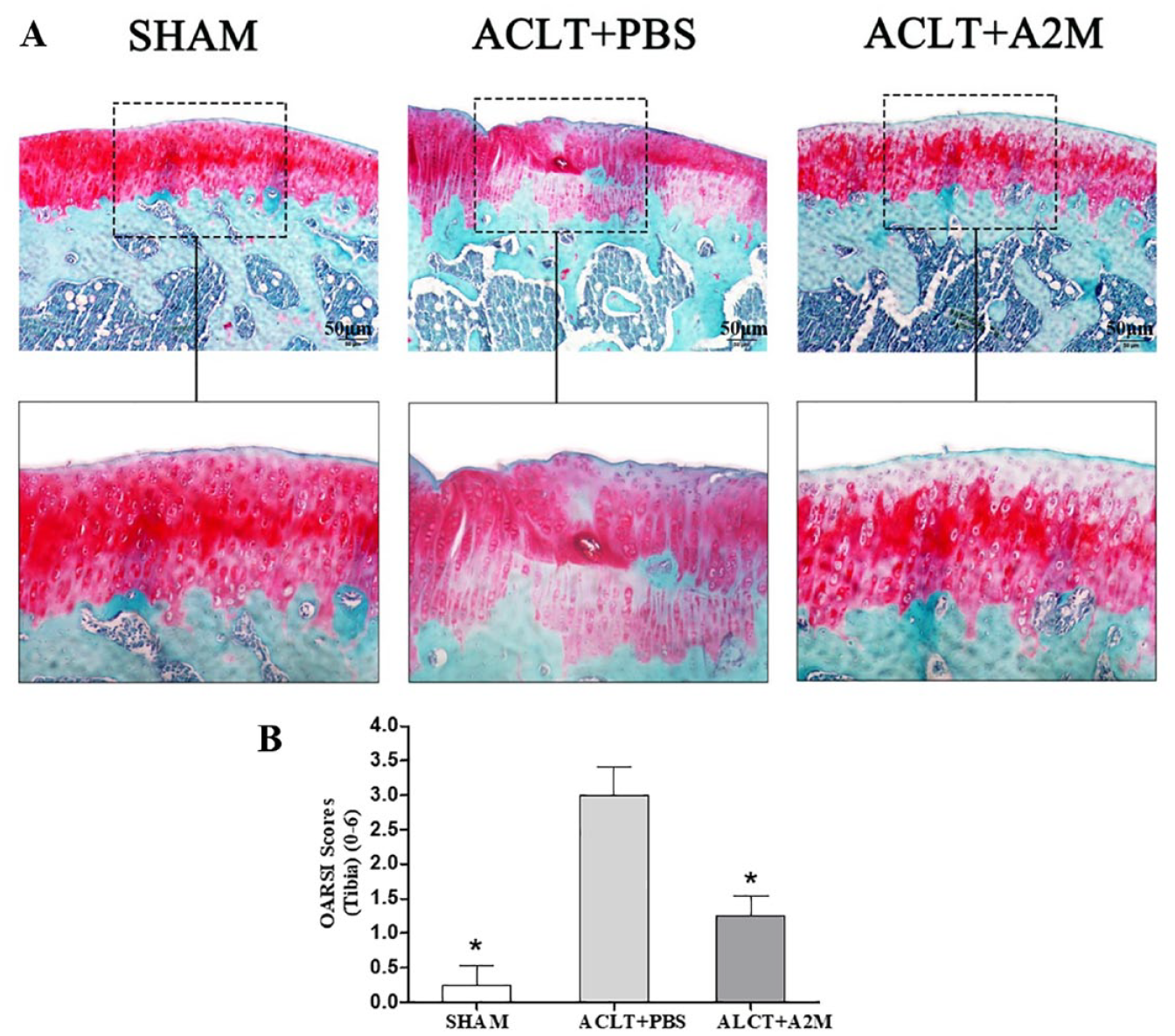
A2M attenuates cartilage degeneration in ACLT rats. (
A2M Can Promote the Chondrocyte Proliferation
Compared with the control group, the BrdU labeling results revealed that A2M can promote ATDC5 proliferation significantly (
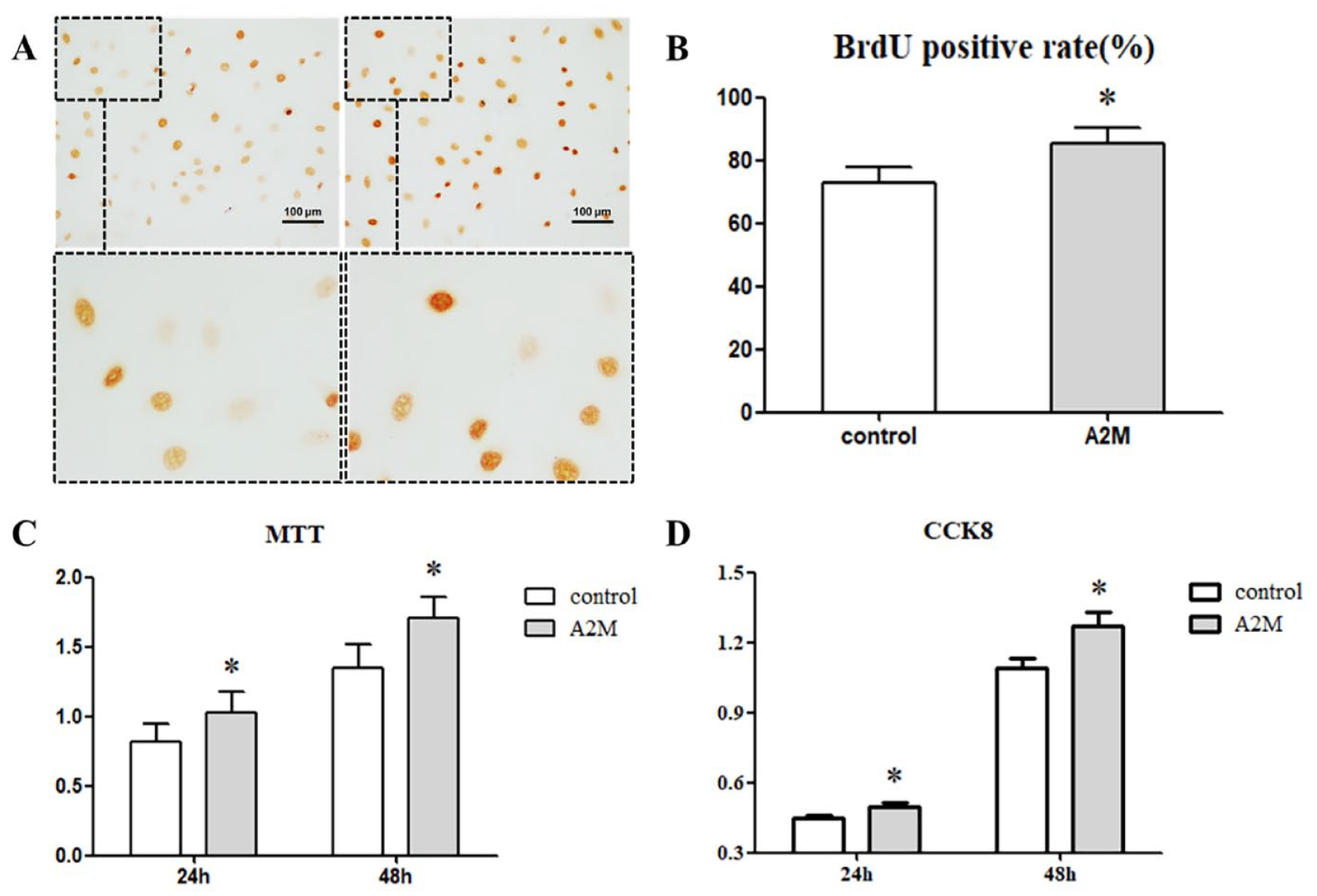
A2M promotes chondrocyte proliferation. (
A2M Can Reduce the Chondrocyte Apoptosis
We use the LDH assay to observe the ATDC5 apoptosis, and the results showed that A2M can inhibit the secretion of LDH in ATDC5. First, we observed the LDH level in the different concentrations of A2M groups. The results showed that the 3 different concentrations of A2M can inhibit the LDH secretion in the ATDC5 within 24 hours (
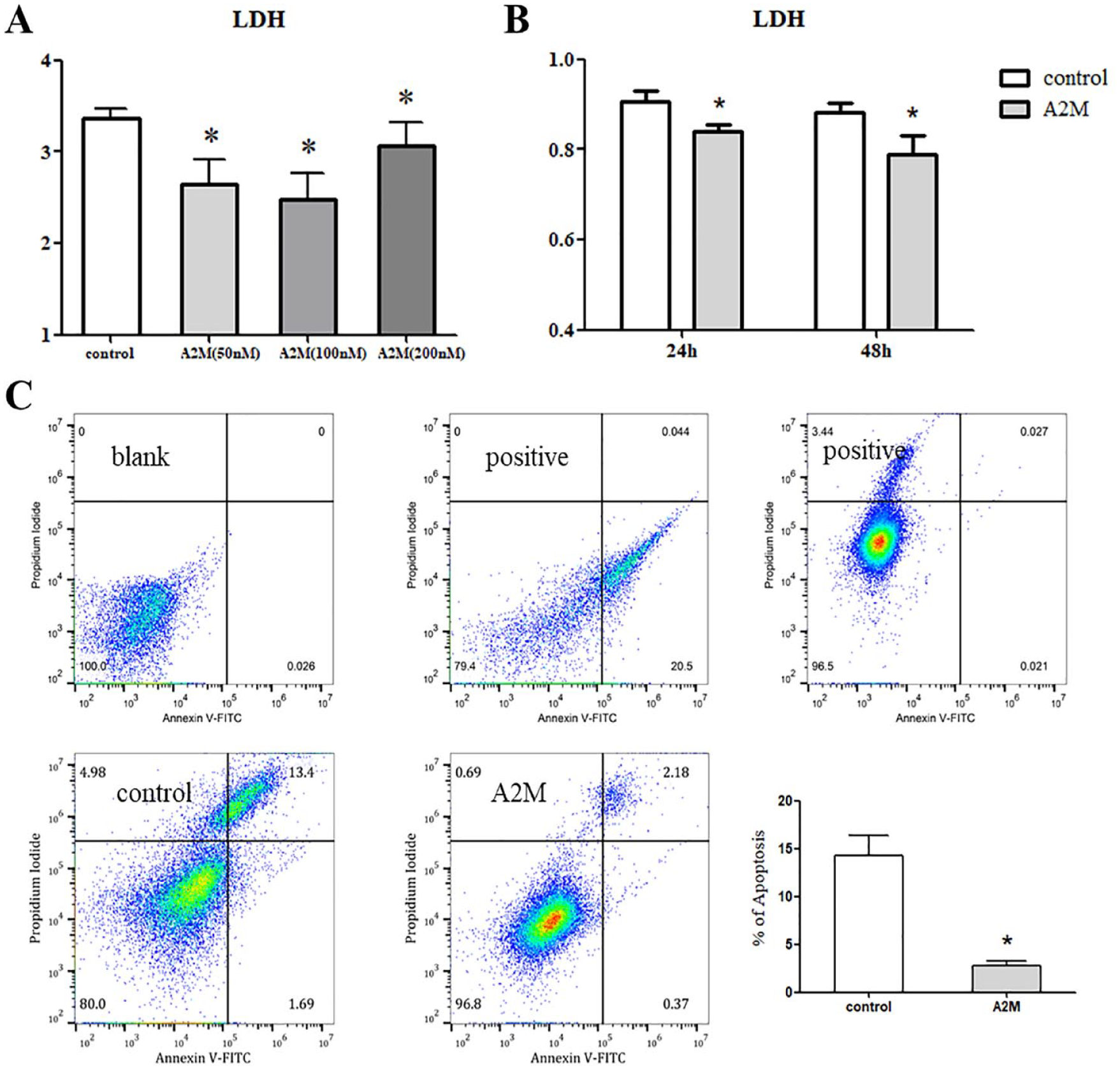
A2M reduced the chondrocyte apoptosis. (
Meanwhile, the flow cytometry method was used to estimate the ATDC5 apoptosis after treatment with A2M for 48 hours. Compared with the control group, A2M can reduce the ATDC5 apoptosis rate significantly (
A2M Promote the Secretion of sGAG and Aggrecan in the Culture Supernatant
Sulfated glycosaminoglycan (sGAG), mainly situated on aggrecan, has been considered to play a central role in cartilage homeostasis, since its negative charge attracts water into cartilage, creating a high osmotic pressure.
12
The sGAG released in the culture supernatant was detected in the A2M group of 48-hour samples, and the sGAG release in the A2M group was significantly higher than that in the control group (P < 0.05) (
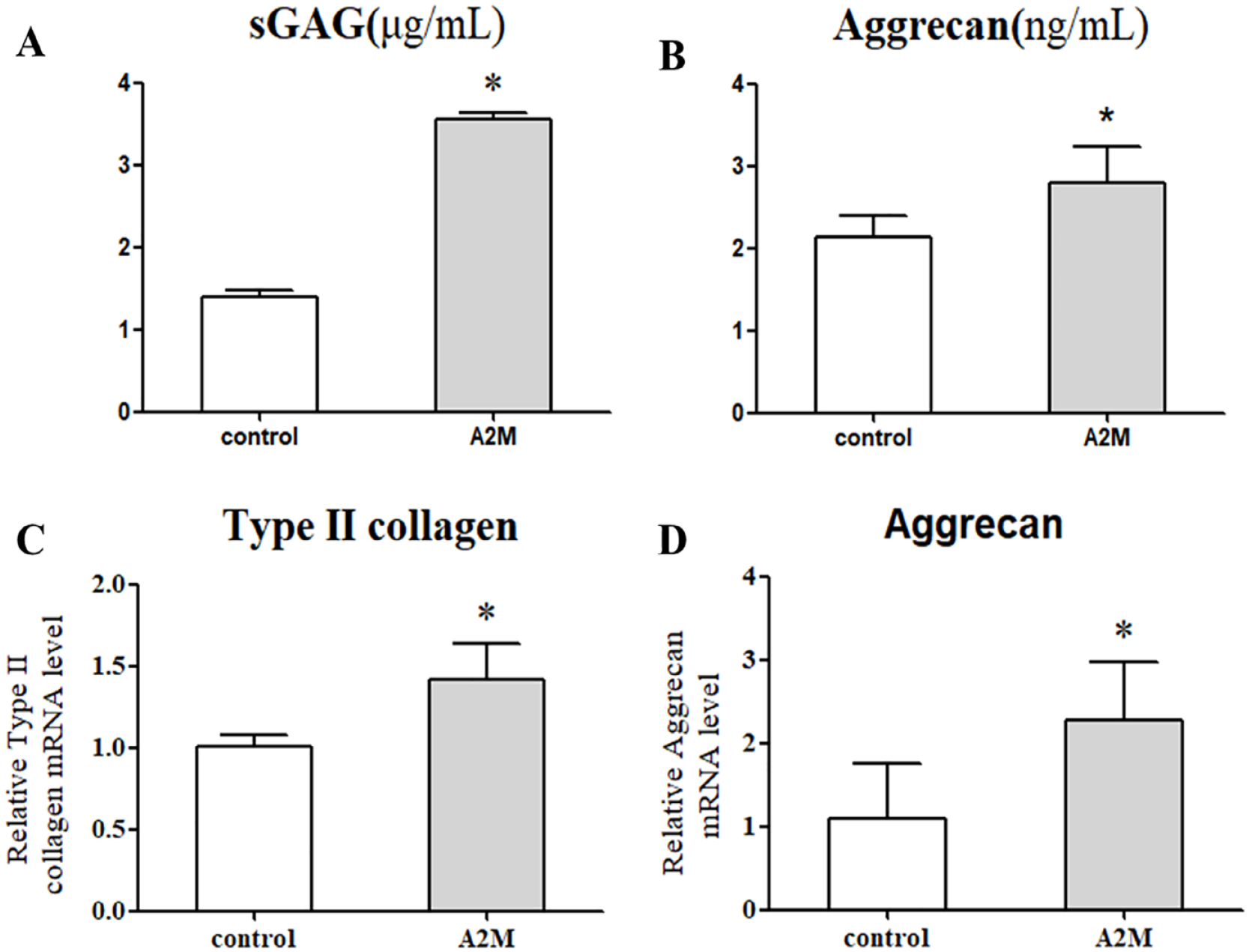
A2M promotes the expression of cartilage matrix in chondrocytes. (
A2M Increases the Expression of Cartilage Matrix Synthesis in the Chondrocyte
The expression of cartilage matrix gene in the chondrocyte was analyzed by RT-qPCR. The results showed that type II collagen and aggrecan mRNA level in the A2M group was significantly higher than that in the control group (
A2M Promotes the Expression of PCNA Protein in the Chondrocyte
The PCNA immunofluorescent staining results showed that the PCNA expression in the A2M group was significantly higher compared with the control group (
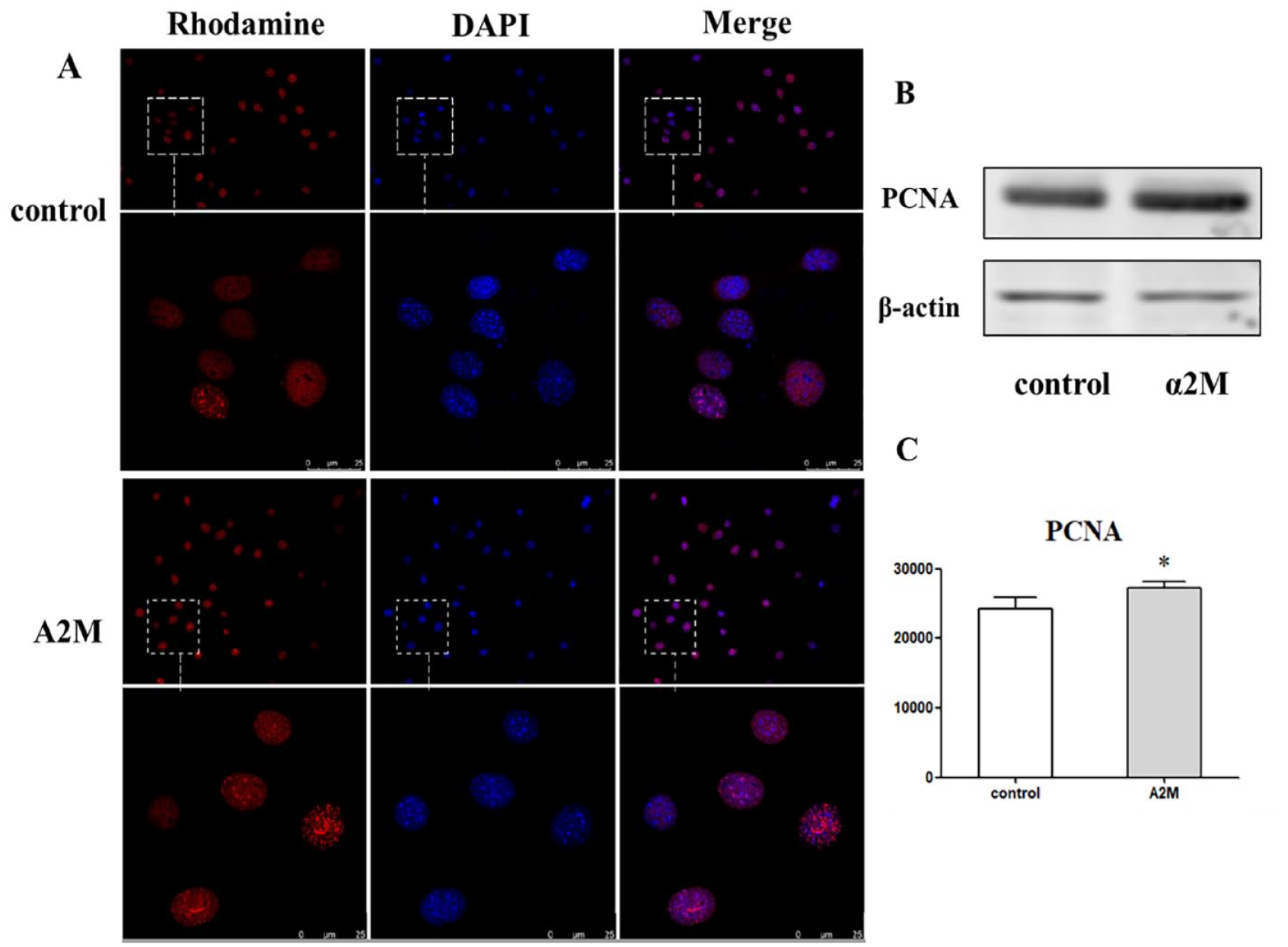
A2M promotes the expression of PCNA protein in the chondrocytes. (
These findings indicate that A2M can promote the PCNA protein expression in the chondrocyte.
Discussion
OA is already the leading cause of disability among the elderly, and its prevalence is rising. 13 OA also involves the progressive erosion of the structures of the joint, which occur as a largely independent pathophysiological process. The decrease of chondrocytes and matrix components (aggrecan and type II collagen) plays an important role in the pathogenesis of osteoarthritis. Chondrocytes are responsible for maintaining a balanced cartilage turnover and responding to changes induced by joint loading, cytokines, growth factor, and the presence of fragmented matrix molecules in the extracellular matrix (ECM) of cartilage. 14 So, it is very important to promote chondrocyte proliferation and cartilage matrix synthesis and inhibit chondrocyte apoptosis for the prevention and treatment of osteoarthritis.
A2M, a general endoproteinase inhibitor, is a noncovalent tetramer of two 370-kDa disulfide-linked homodimers that circulates in the blood at concentrations of 2 to 4 mg/mL and is also found in the joint fluid at similar concentrations. 15 A2M clinical treatment is now widely offered to patients suffering from arthritis and/or degenerative disk disease; however, little clinical evidence exists supporting the efficacy of this treatment at this time. Studies have confirmed that the loss of A2M inhibits aggrecan secretion in OA synovial mesenchymal progenitor cells (MPCs), whereas overexpression of A2M rescues the normal secretion of aggrecan. 16 Previous studies demonstrated that A2M did prevent OA cartilage degeneration, 8 but the mechanism is unclear.
The results of this study showed that A2M can promote the chondrocytes’ proliferation and inhibit the chondrocytes’ apoptosis. The BrdU labeling results showed that A2M can promote chondrocyte proliferation significantly. At the same time, the MTT and the CCK8 results also demonstrated that the A2M can promote the chondrocyte proliferation. We use the LDH assay to assess the cell toxicity of A2M, which was released into the culture medium. The different concentrations of A2M can inhibit the level of LDH in the chondrocytes, which suggest that A2M can reduce the cytotoxicity of chondrocytes. The flow cytometry results also showed that the A2M can reduce the chondrocyte apoptosis rate significantly.
The aggrecan is necessary for structure and function of cartilage. 17 Intra-articular aggrecan injection was an effective treatment for limiting joint pathology in ACL-X joints through inducing cartilage repair. Loss of aggrecan from the cartilage extracellular matrix is an early and sustained feature of osteoarthritis. 18 Type II collagen is rigid in nature and has a very slow rate of turnover in healthy tissue, while aggrecan, which undergoes degradation and regeneration at a more rapid pace, provided cartilage with its shock-absorptive property. 19 Excessive degradation of type II collagen and aggrecan by collagenases and aggrecanases was recognized as a pivotal event in the development of OA. 20 In order to further analyze the mechanism of A2M in preventing and treating OA, we observed the effects of A2M on the matrix synthesis in ATDC5. The qPCR results showed that A2M can promote the expression of aggrecan and type II collagen mRNA in the ATDC5. At the same time, the ELISA results showed that A2M promotes the aggrecan expression in the cell culture supernatant. A previous study showed that A2M can enhance cartilage matrix synthesis in the collagen II-induced arthritis model, including type II collagen and aggrecan, which suggested that A2M may have a cartilage repair function and/or facilitate for cartilage matrix synthesis to proceed. 21 A high dose of A2M did not induce chondrocyte death but rather increase collagen type II and aggrecan production. 8 A2M protected cartilage following joint injuries that can progress to OA and recombinant A2M variants were more protective than the wild type. 8 At the same time, our previous study also showed that A2M can inhibit interleukin (IL)-1β within chondrocytes and thus attenuate multiple downstream catabolic pathways including that of nuclear factor kappa B (NF-κB) and the matrix metalloproteinases (MMPs). 22
PCNA had an essential role in the processivity of DNA replication, providing a central scaffold that controls the dynamic engagement of a plethora of factors with the replication machinery. 23 PCNA interacted directly with a host of proteins involved in many different cellular processes. PCNA provided a molecular platform that facilitated the myriad protein-protein and protein-DNA interactions that occur at the replication fork. Numerous PCNA-associated proteins competed for binding to a common surface on PCNA; hence, it is very important to study the effect of PCNA on A2M, for exploring the mechanism of A2M regulating the cartilage metabolism. On the basis that A2M can promote the chondrocyte proliferation and the cartilage matrix synthesis, we further observed the PCNA expression by immunohistochemical method and western blot. The results showed that A2M can promote the PCNA protein expression in the chondrocytes. These results suggested that A2M can regulate cell proliferation by promoting PCNA protein and improving cartilage metabolism.
Conclusion
This study provided evidence that A2M prevents cartilage degeneration by promoting chondrocyte proliferation and cartilage matrix synthesis via inducing the PCNA expression.
Footnotes
Author Contributions
Study concept and design: Hailing Guo, Shaowei Wang, and Yang Zhang. Acquisition of material and data: Jian Sun, Li Guo, and Jian Pang. Preparation of the manuscript: Hailing Guo and Hongsheng Zhan. Critical reviewing and approval of the manuscript: all authors.
Acknowledgments and Funding
We thank all study participants of the study. We thank all the members of our groups. We also thank Lei Wei, Xiaojuan Sun, and Li Yue for their help in the experiment. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Shanghai Clinical Research Center for Musculoskeletal Health (20MC1920600), the program for Shanghai High-Level Local University Innovation Team (SZY20220315), the Key Clinical Discipline Construction Project “Orthopedics and Traumatology of Traditional Chinese Medicine” (2017Z02024), Shanghai Municipal Key Clinical Specialty Project (shslczdzk03901), Research Project Supported by Shanxi Scholarship Council of China (HGKY2019099), and Project of Shanghai Science and Technology Commission (22Y21920200).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Institutional Animal Care and Use Committee of Rhode Island Hospital.
