Abstract
Objective
Activation of sympathetic tone is important for cartilage degradation in osteoarthritis (OA). Recent studies reported that sympathetic signals can affect the mitochondrial function of target cells. It is unknown whether this effect exits in chondrocytes and affects chondrocyte catabolism. The contribution of mitochondrial dynamics in the activation of α2-adrenergic signal-mediated chondrocyte catabolism was investigated in this study.
Design
Primary chondrocytes were stimulated with norepinephrine (NE) alone, or pretreated with an α2-adrenergic receptor (Adra2) antagonist (yohimbine) and followed by stimulation with NE. Changes in chondrocyte metabolism and their mitochondrial dynamics were investigated.
Results
We demonstrated that NE stimulation induced increased gene and protein expressions of matrix metalloproteinase-3 and decreased level of aggrecan by chondrocytes. This was accompanied by upregulated mitochondriogenesis and the number of mitochondria, when compared with the vehicle-treated controls. Mitochondrial fusion and fission, and mitophagy also increased significantly in response to NE stimulation. Inhibition of Adra2 attenuated chondrocyte catabolism and mitochondrial dynamics induced by NE.
Conclusions
The present findings indicate that upregulation of mitochondrial dynamics through mitochondriogenesis, fusion, fission, and mitophagy is responsible for activation of α2-adrenergic signal-mediated chondrocyte catabolism. The hypothesis that “α2-adrenergic signal activation promotes cartilage degeneration in temporomandibular joint osteoarthritis (TMJ-OA) by upregulating mitochondrial dynamics in chondrocytes” is validated. This represents a new regulatory mechanism in the chondrocytes of TMJ-OA that inhibits abnormal activation of mitochondrial fusion and fission is a potential regulator for improving mitochondrial function and inhibiting chondrocyte injury and contrives a potentially innovative therapeutic direction for the prevention of TMJ-OA.
Introduction
Temporomandibular joint osteoarthritis (TMJ-OA) is a subtype of temporomandibular disorders (TMDs). This condition is characterized by progressive cartilage degradation. 1 Because the mechanism of cartilage degradation is still unknown, current treatment strategies for TMJ-OA are predominantly palliative in nature, with the aim of relieving pain and inflammation. There is presently no effective disease-modifying strategy to block or delay OA progression.2,3
Sympathetic tone plays an important regulatory role in many physiological and pathological processes. Regulation is achieved by the secretion of norepinephrine (NE) to activate the adrenergic receptors of the target organs.4,5 Upregulation of NE, the major sympathetic neurotransmitter, is detected in the synovial fluid of patients with trauma-induced OA.5,6 Sprouting of sympathetic nerve fibers is identified in subchondral bone during the early stage of OA. Those nerve fibers extend into the overlying cartilage via vascular channels.7,8 Chondrocytes express different kinds of adrenergic receptors to regulate different physiological activities such as chondrocyte proliferation, differentiation, and apoptosis after they are activated by NE. 9 A low concentration of NE (10−7 mol/L), delivered via α-adrenergic signaling, promotes cartilage degradation through induction of chondrocyte apoptosis. 10 These results suggest that sympathetic signal promotes cartilage degeneration in OA by activating the α2-adrenergic receptors (Adra2) in chondrocytes. 10 Nevertheless, the biological triggers involved in these processes and the associated mechanisms are unknown.
Mitochondrial dynamics is now perceived as a core player in tissue homeostasis. This process refers to the coordinated cycles of biogenesis, fusion, fission, and degradation, which mitochondria constantly undergo to maintain their integrity, distribution, and size. 11 These mechanisms represent an early response to mitochondrial stress, recycling organelle portions that are irreversibly damaged and preserving mitochondrial function.12,13 The upregulation of mitochondrial dynamics has been shown to be associated with chondrocyte damage and TMJ-OA progression in different disease models. 14 In most cases, mitochondrial dynamics is considered a protective or adaptive mechanism, given its ability to clear defective mitochondrial built-up. Nonetheless, uncontrolled or excessive upregulated mitochondrial dynamics may result in the shortage of functional or healthy mitochondria for ATP generation, leading to compromised cell survival. 15 These events can promote the production of cytokines and oxidative stress, triggering inflammation, which can damage the integrity of mitochondrial DNA and their repair capacity, ultimately resulting in chondrocyte death.16,17 Recent studies have shown that sympathetic signals can trigger mitochondrial dysfunction in adipocytes and cardiomyocytes to promote the onset of metabolic diseases such as obesity and hypertension. 18 However, it is unknown whether mitochondrial dynamics is involved in sympathetic tone-induced chondrocyte degeneration.
Accordingly, the objective of this study was to test the hypothesis that Adra2 activation promotes cartilage degeneration in TMJ-OA by upregulating mitochondrial dynamics in chondrocytes. To test this hypothesis, the effect of sympathetic signals on mitochondrial genesis by chondrocytes was examined in vitro after treatment with NE. Changes in mitochondrial morphology and function after Adra2 activation were also examined.
Materials and Methods
Primary Chondrocyte Isolation and NE Stimulation
Chondrocytes were isolated from the condylar cartilage of 3-week-old female Sprague-Dawley rat TMJs. In each batch, we isolated 20 mandibular condyles from 10 rats, and carefully separated the condylar cartilage from the condyles under stereomicroscope. The primary chondrocytes from every 20 mandibular condylar cartilages were pooled together as one independent sample. The TMJs were digested with 0.25% trypsin (MilliporeSigma, St. Louis, MO) for 20 minutes, followed by 0.2% type II collagenase (PC-50995; PlantChemMed Biology Co., Ltd., Shanghai, China) for 2-3 hours. The chondrocytes were plated in 60-mm-diameter plates at a density of 1.5 × 106 cells/plate and cultured with control medium. The control medium consisted of Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12 medium (DMEM/F12; Gibco, Thermo Fisher Scientific, Waltham, MA) supplemented with 10% fetal bovine serum (FBS; PC-00001; PlantChemMed Biology Co., Ltd.) and 1% penicillin/streptomycin (PC-86115; PlantChemMed Biology Co., Ltd.). The chondrocytes were harvested after culturing for 5 days. Secondary cultures were performed in 6-well plates at a density of 5 × 105 cells per well for subsequent experiments. As previously reported, 10 the effects of NE on chondrocyte expression of aggrecan and matrix metalloproteinase (MMPs) were most apparent when its concentration was 10−7 M. These changes were progressively attenuated as the concentration of NE increased, with no effects observed when the NE concentration was increased to 10−6 M. At low concentrations (≤10−7 M), NE is mediated mainly via α-adrenoreceptors, whereas at high concentrations (>10−7 M), NE acts preferentially via the β-adrenoreceptors. Hence, the procatabolic changes induced by NE on adult chondrocytes are likely to be mediated by α-adrenergic signaling. Only the α2-adrenoreceptor antagonist could reverse NE-induced expression of procatabolic mediators in chondrocytes. As previously reported, 10 in vivo experiments showed that the effective intra-articular injection dose of yohimbine was 100 μg, 50 μl once a week for 4 weeks, and that in vitro experiments declared the effective dose of yohimbine (a selective Adra2 antagonist; Y3125; MilliporeSigma) was 10−5 M for 1 hour pretreatment. Therefore, the chondrocytes were treated with 10−7 M NE for 1 hour, or first pretreated with 10−5 M yohimbine for 1 hour before stimulation with 10−7 M NE for 1 hour.
Transmission Electron Microscopy
Chondrocytes in the control and experimental groups were fixed with 2.5% glutaraldehyde, post-fixed with 1% osmium tetroxide, dehydrated with an ascending ethanol series, immersed in propylene oxide as transition medium and embedded in epoxy resin. Thin sections (90 nm thick) were collected on Formvar- and carbon-coated Ni grids and examined with a JEM-123 transmission electron microscope (TEM; JEOL, Tokyo, Japan) at 110 kV.
Immunofluorescence
Chondrocytes in the control (culture medium only) and experimental groups were fixed with 4% paraformaldehyde, incubated with protein-blocked serum-free solution (Invitrogen, Thermo Fisher Scientific) and exposed to primary antibodies (n = 6) at 4 °C overnight. The antibodies used were anti-aggrecan antibody (1:200, ab36861; Abcam, Cambridge, UK), anti-collagen II antibody (1:250, ab34712; Abcam), anti-mitochondria antibody (1:200, MA5-12017; Thermo Fisher Scientific), anti-matrix metalloproteinases-3 antibody (1:250, ab52915; Abcam), anti-dynamin-related protein-1 antibody (1:150, ab3456; Abcam), anti-mitofusin-2 antibody (1:300, ab124773; Abcam), and anti-LC3B antibody (1:200, ab63817; Abcam). After washed by phosphate-buffered saline (PBS), cells were incubated with second antibodies (1:800, ab150113, ab150080, ab150077; Abcam) for 1 hour in the dark. The cells were mounted in antifade mountant and stained with 4′,6-diamidino-2-phenylindole (DAPI; Invitrogen, Thermo Fisher Scientific) for confocal laser scanning microscopy (CLSM; FV1000; Olympus, Tokyo, Japan). The primary chondrocytes from every 20 mandibular condylar cartilage were pooled together as one independent sample, and “n = 6” means that in each group, six pooled samples were used for experiments and statistical analysis.
Quantitative Real-Time Polymerase Chain Reaction
Factors related to cartilage metabolism, mitochondriogenesis-related genes, mitochondria fusion-related and fission-related genes, and mitophagy-related genes were detected by quantitative real-time polymerase chain reaction (qRT-PCR). Briefly, total RNA was extracted using TRIzol (Thermo Fisher Scientific). Primers for the target genes are listed in Suppl. Table 1. Gene expression was analyzed with the 7500 real-time PCR (Thermo Fisher Scientific), using glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as the internal control. The amount of target cDNA relative to GAPDH was calculated using the 2−ΔΔCT method. Results were calculated as the relative quantification compared with the control group, which was set at 1-fold. Every 20 TMJs were collected and their cartilages were mixed into one independent sample. Data were collected from six independent pooled samples.
Enzyme-Linked Immunosorbent Assay
After removal of cells and debris via centrifugation (1,000 rpm, 3 minutes), the supernatant was collected and used immediately for subsequent experiments or stored at −80 °C. The rat tumor necrosis factor (TNF)-α, interleukin (IL)-1β, IL-10, and IL-13 enzyme-linked immunosorbent assay (ELISA) Kit (Thermo Fisher Scientific) were used to measure the quantity of corresponding cytokines secreted into the cell culture supernatant, according to the manufacturer’s instructions. Sample absorbance was measured at 450 nm within 30 minutes.
Western Blot
The protein expression of factors related to mitophagy-related genes was detected by Western blot (WB). Briefly, total protein was extracted from each group, fractionated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), transferred onto a polyvinylidene fluoride membrane. After blocked with 5% nonfat milk, the membrane was incubated with primary antibodies against LC3B (1:1000, AL221; Beyotime Biotechnology, Jiangsu, China) and GAPDH (1:2000, ab245355; Abcam) was used as internal control. After membrane incubation with a horseradish peroxidase (HRP)-conjugated secondary antibody (1:5000; Zhongshan Golden Bridge Biotechnology, China) and Immobilon Western Chemiluminescent HRP Substrate (WBKLS0100; Millipore, Billerica, MA), signals were detected by Ultimate Western System (Azure Biosystems C600). The stained bands were quantified using image J software. Every 20 TMJs were collected and their cartilages were mixed into one independent sample. Data were collected from six independent pooled samples.
Active Mitochondria Staining
The MitoTracker Green FM stain (Thermo Fisher Scientific) was used to evaluate active mitochondria in the chondrocytes. Chondrocytes were stimulated by NE or NE with yohimbine (NE + Yoh) for 1 hour. The stimulated chondrocytes were washed twice with serum-free medium and incubated in the dark with 1 ml of 200-nM MitoTracker Green FM in a humidified 5% CO2 atmosphere for 30 minutes (n = 6). The staining agent was removed and α-minimal essential medium (α-MEM) was added to the specimens. The processed cells were examined with CLSM. Images were analyzed using the Mitochondrial Network Analysis (MiNA 3.0.1; http://github.com/ScienceToolkit/MiNA), an Image J (version 1.8.0)-based utility for quantifying the mitochondrial network parameters in each group. The data of mitochondrial networks were quantified by randomly selecting three cells in one image of mitochondria immunostaining and the average of those data was used as one sample data, and six replicates were set up in each group.
Mitochondrial Membrane Potential
5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimidazolylcarbocyanine iodide (JC-1), a cell membrane-permeable fluorescent dye, resides exclusively in the mitochondria, especially in those with healthy physiological status that are distinguished by high mitochondrial membrane potential (ΔΨm). The ΔΨm of chondrocytes stimulated by NE or NE + Yoh was measured with the Mitochondrial Membrane Potential Assay Kit (Beyotime Biotechnology) using the cyanine dye JC-1 (MilliporeSigma) (n = 6). The stained cells were examined using CLSM and analyzed with ImageJ software. The primary chondrocytes from every 20 mandibular condylar cartilage were pooled together as one independent sample, and “n = 6” means that in each group, six pooled samples were used for experiments and statistical analysis.
Flow Cytometry
Chondrocytes were isolated and resuspended in 200 μL of PBS. Add 10 μl of 200-μM JC-1 dye (2 μM in the final concentration) and incubate the cells at 37 °C, 5% CO2 for 15-30 minutes. The JC-1 dye was excited using an argon laser at a wavelength of 488 nm. Both JC-1 aggregates and JC-1 monomers exhibited green fluorescence (peak emission at 527 nm) which was measured in the FL1 channel (530 nm). However, the JC-1 aggregates showed a red fluorescence (peak emission at 590 nm) which was detected and measured in the FL2 channel (585 nm). Finally, we determined the ratio of red fluorescence divided that of green fluorescence. Data analysis was performed using the FlowJo 10.0 software (FlowJo LLC, 583 Ashland, OR). Data were collected from six independent pooled samples.
Statistical Analysis
Data derived from each experiment was tested for their normality (Shapiro-Wilk test) and equal variance assumptions (modified Levene test) prior to the use of parametric statistical methods. If either of those assumptions was violated, the corresponding data were nonlinearly transformed to satisfy those assumptions. All quantitative data were expressed as means ± standard deviations. One-factor analysis of variance (ANOVA) and post hoc Holm-Šidák procedures were used for analysis of data obtained from immunofluorescence staining. For qRT-PCR, gene expression was analyzed by comparing with the Gapdh endogenous control using one-factor ANOVA and Holm-Šidák multiple comparison procedures. Statistical significance for all tests was preset at α = 0.05. Bars labeled with *** (P < 0.001), ** (P < 0.01), or * (P < 0.05) were significantly different. “n” represents the number of donors in rat experiments. All the statistical analyses were performed with GraphPad Prism software (version 9.2).
Results
Effect of Adra2 Activation on Chondrocyte Procatabolic Activities
Almost all chondrocytes isolated from TMJs were positive for aggrecan and type II collagen (Fig. 1A and
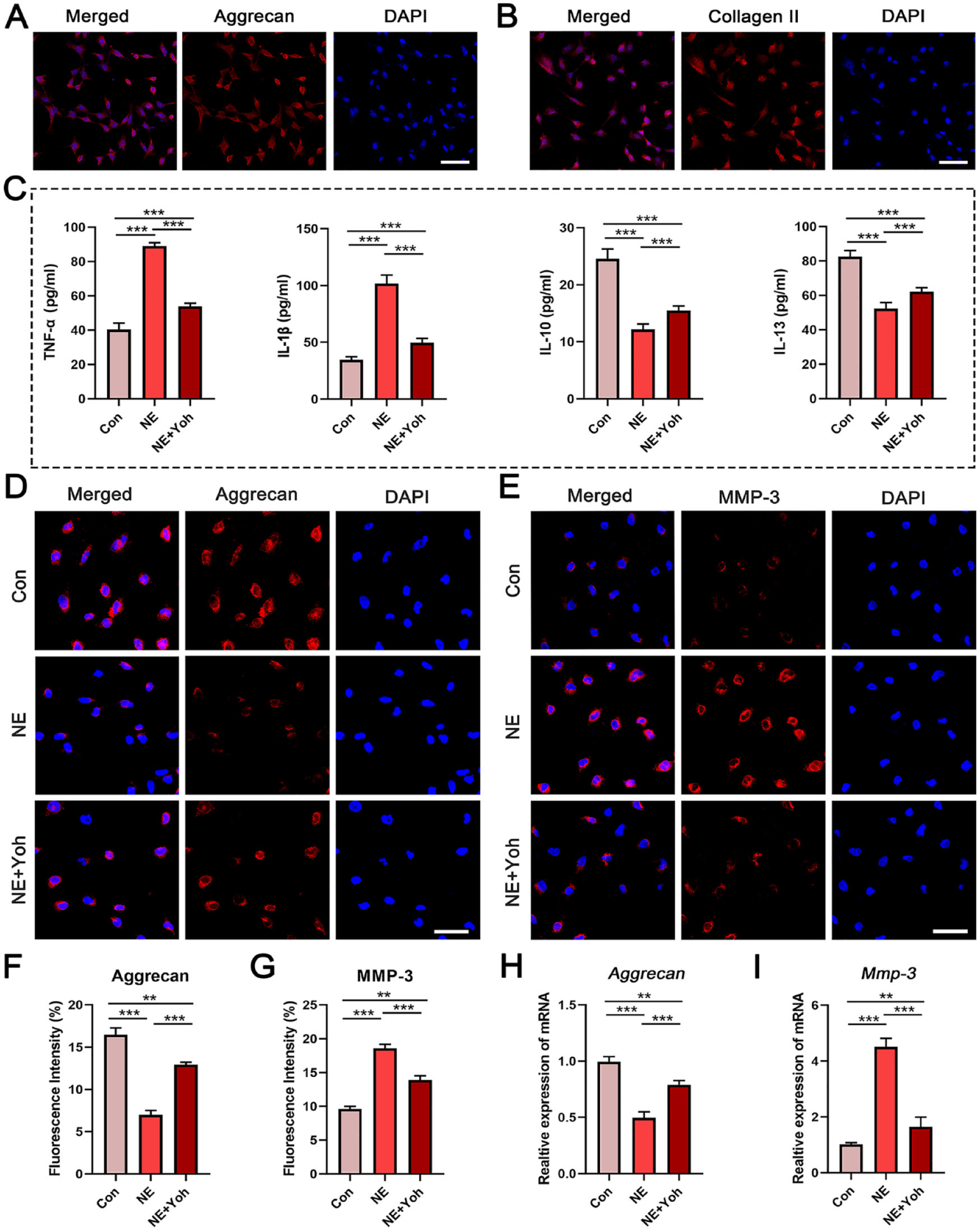
Effects of sympathetic signal on chondrocytes derived the different groups. (
Effects of Adra2 Activation on Mitochondriogenesis and Energy Production by Chondrocytes
MitoTracker Green FM staining was used to identify the mitochondrial distribution and network after the chondrocytes were stimulated with NE or NE + Yoh.
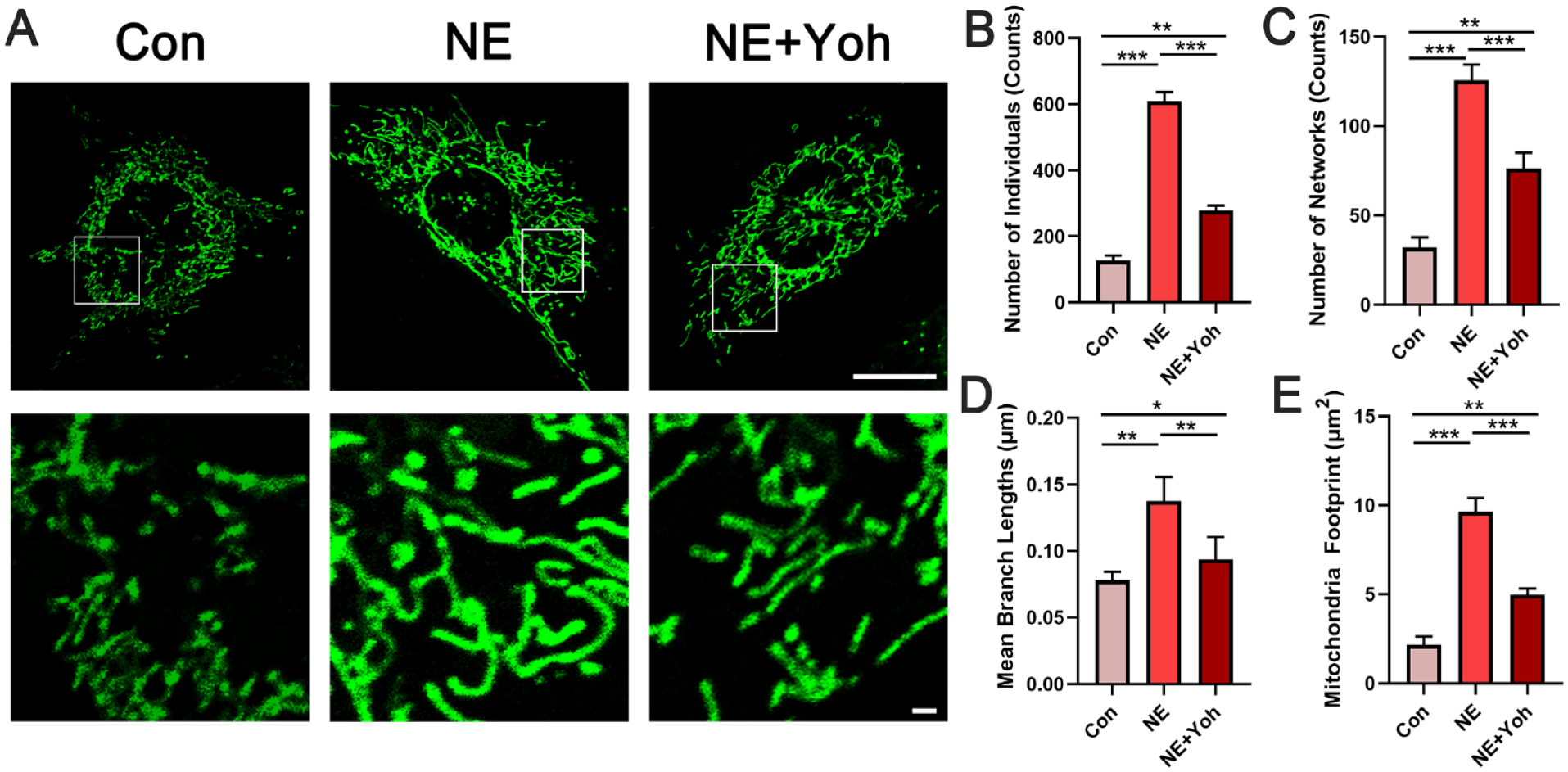
Changes in mitochondrial networks in chondrocytes derived from different groups.
Mitochondriogenesis in the chondrocytes stimulated by NE or NE + Yoh was detected via qRT-PCR. Gene expressions of the mitochondriogenesis-related markers peroxisome proliferator-activated receptor gamma coactivator 1-alpha (Pgc-1α), nuclear respiratory factor 1 (Nrf1), nuclear respiratory factor 2 (Nrf2), mitochondrial transcription factor A (mtTFA), and mitochondrial single-stranded DNA-binding protein (mtSSB) were significantly upregulated in the NE group (P < 0.001). These increases were attenuated in the NE + Yoh group (P < 0.001) (
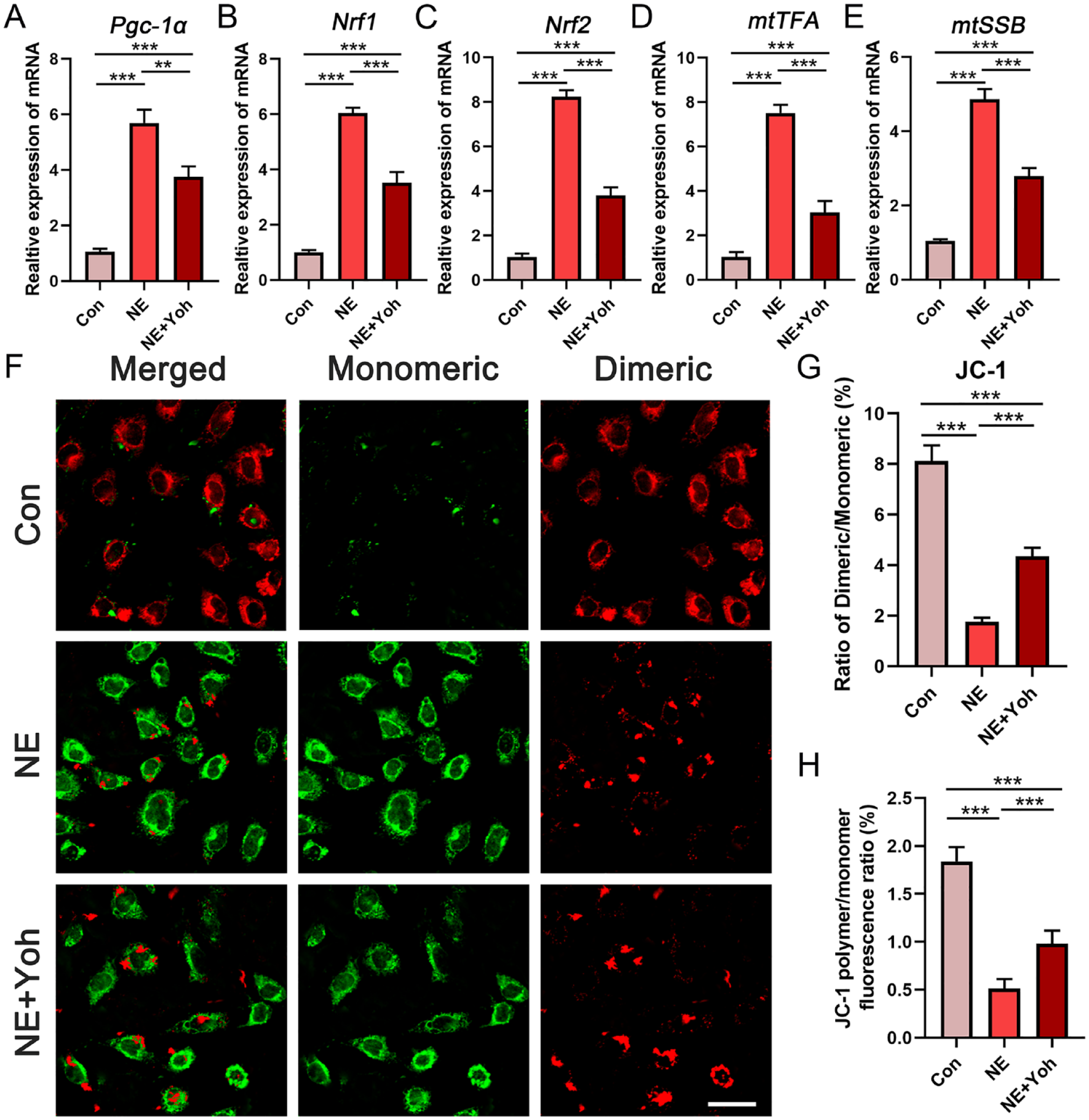
Effects of different treatments on mitochondrial biogenesis in chondrocytes in vitro. (
Contribution of Adra2 Activation to Mitochondrial Fusion and Fission
The status of fusion regulators and fission-related genes was evaluated with qRT-PCR. Expression of fission-related genes, such as dynamin-related protein-1 (Drp1), mitochondrial fission protein-1 (Fis1), and mitochondrial fission factor (Mff), was significantly upregulated in chondrocytes stimulated with NE for 1 hour. These increases were attenuated in chondrocytes treated with NE and yohimbine (P < 0.001) (
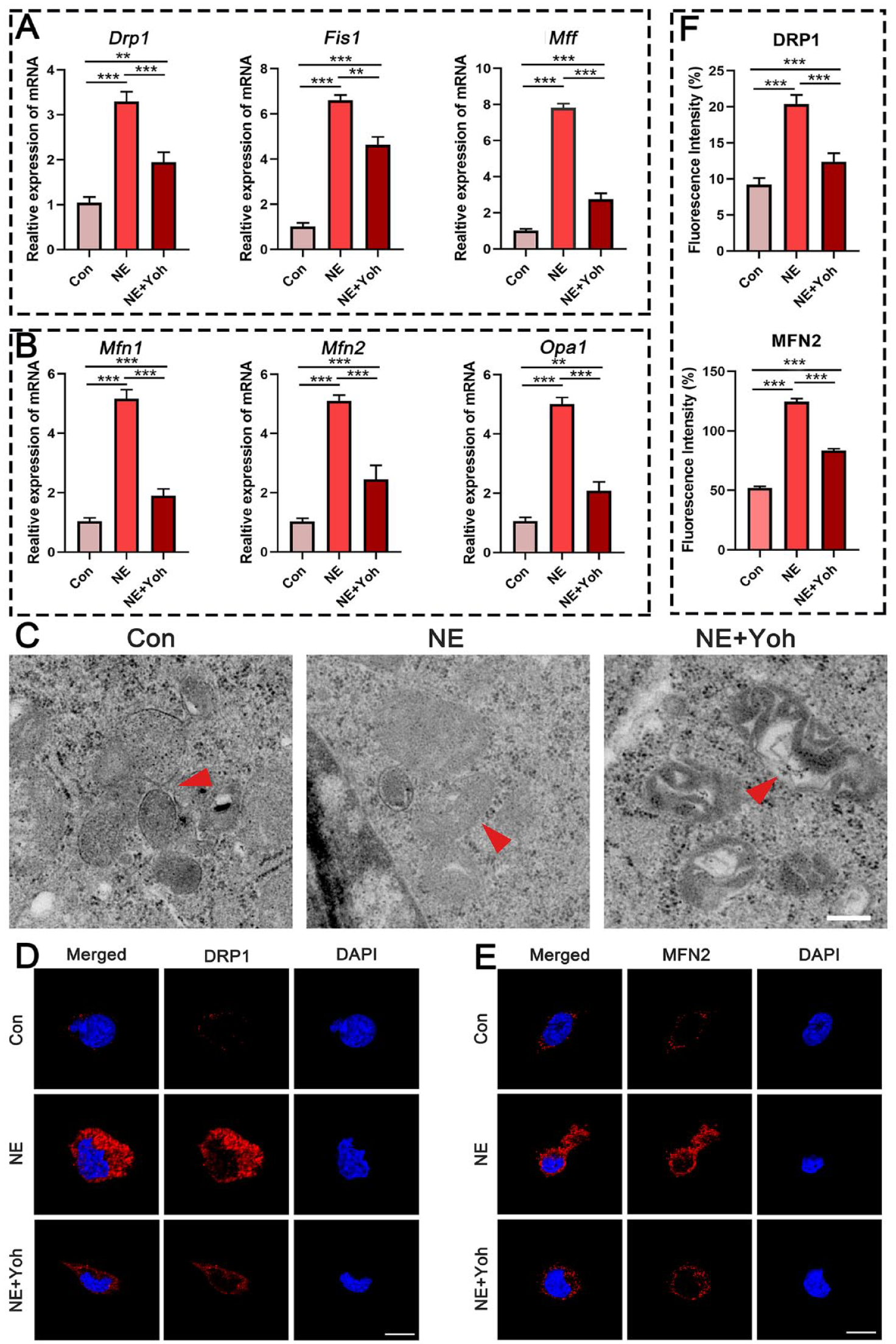
Changes in mitochondrial dynamics in chondrocytes derived from different groups. (
TEM was performed to confirm the activated states of fusion and fission in the mitochondria of chondrocytes. Yohimbine reversed the activation of mitochondrial fusion and fission after stimulation for 1 hour (red arrows indicated fused and fissured mitochondria) (
Effects of Adra2 Activation to Mitochondrial Autophagy
Experiments were conducted to clarify whether mitochondrial autophagy was related to cartilage degeneration when Adra2 was activated. TEM showed that the mitochondria in the NE group were found within autophagosomes (red arrows). Such a feature was characteristic of mitochondrial autophagy. In comparison, mitophagy was significantly inhibited after treatment with NE and yohimbine for 1 hour (
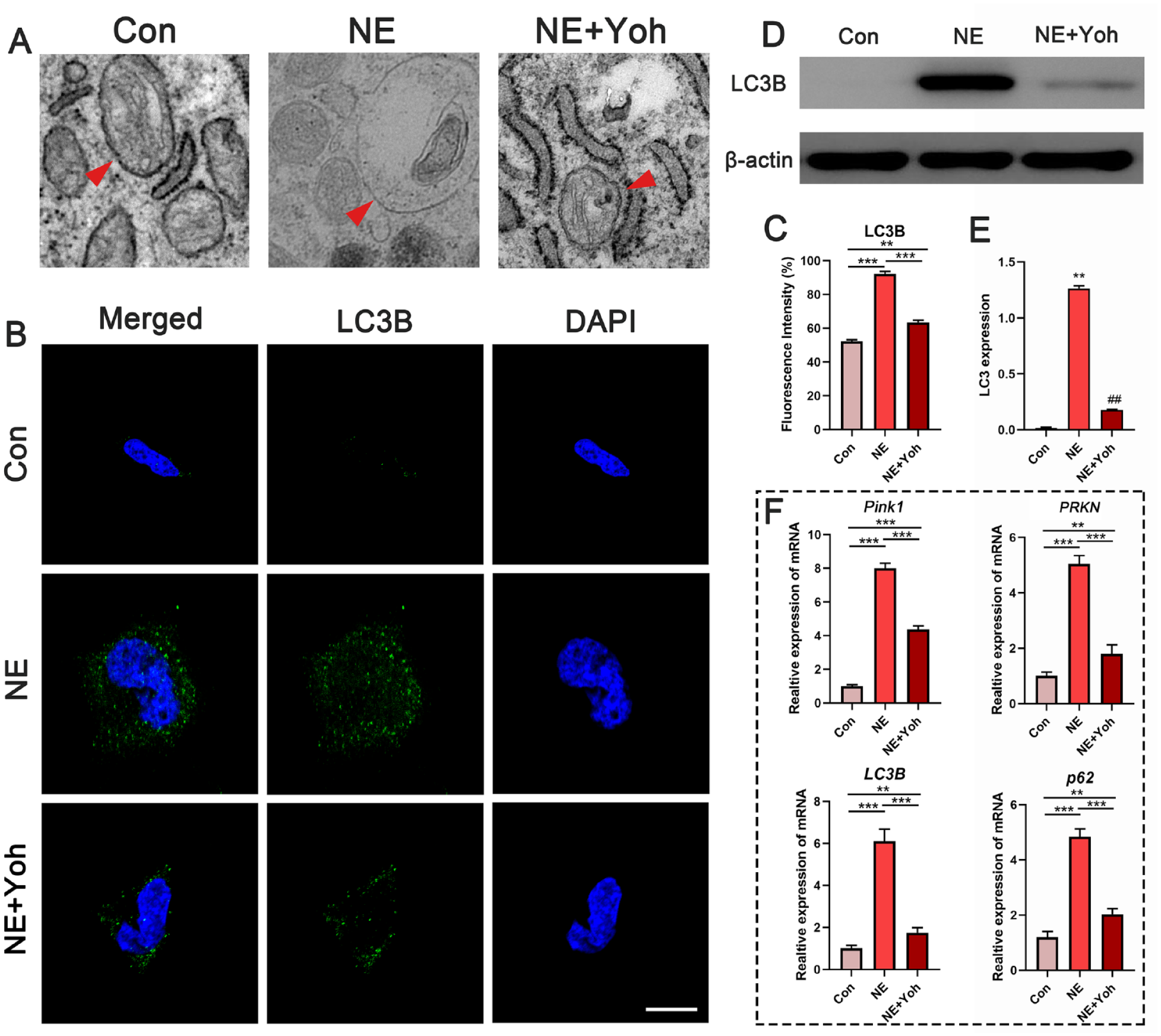
Autophagy in chondrocytes derived from different groups. (
To further detect the level of autophagy, LC3B, a mitophagy-related protein, was quantified by immunofluorescence staining. The immunofluorescent intensity of LC3B was significantly upregulated in chondrocytes stimulated by NE (P < 0.001) (
Discussion
This study demonstrated that activation of α2-adrenergic signals in chondrocytes promoted the secretion of MMP-3 and degradation of proteoglycans. Concomitantly, mitochondrial dynamics was upregulated, including mitochondrial biogenesis, fusion, fission, and mitophagy. Overactivated mitochondrial dynamics precipitated cell stress that, in turn, induced chondrocyte catabolism and subsequently apoptosis, resulting in osteoarthritic cartilage degradation.20,21 Inhibition of Adra2 attenuated chondrocyte catabolism and mitochondrial dynamics induced by NE. These data suggest that upregulation of mitochondrial dynamics is an important pathogenic mechanism of cartilage degeneration induced by the sympathetic tone.
The mitochondrial dysfunction of OA chondrocytes has been extensively studied, including increased oxidative stress of mitochondria, damaged mitochondrial DNA and matrix, and the resulting inflammatory response and matrix metalloproteinase production. 22 We have shown that activation of sympathetic signal played a detrimental role in the progression of TMJ-OA, and chondrocytes stimulated by 10−7 M NE for 1 hour exhibited decreased aggrecan expression but increased MMP-3 expression, similar to the pathological status of OA chondrocytes. 10 Meanwhile, the present results are further validated by using OA chondrocytes or chondrocytes stimulated by TNF-α, IL-1β, IL-10, and IL-13. The results showed that the expressions of pro-inflammatory cytokines (including TNF-α and IL-1β) secreted by NE-stimulated chondrocytes were significantly higher than those in the control group, and these increases were inhibited after NE + Yoh treatment. Conversely, the expressions of anti-inflammatory cytokines (including IL-10 and IL-13) showed the opposite trends, which were decreased in the NE group, whereas increased in the NE + Yoh group. Therefore, this study stimulated the primary chondrocytes with NE and studied the mitochondrial dynamics in the activation of α2-adrenergic signal-mediated chondrocyte catabolism. The results indicated that α2-adrenergic signal transduction in chondrocytes plays a detrimental role in the pathogenesis of TMJ degenerative remodeling.
Recent studies reported that sympathetic signals could induce mitochondrial dysfunction in pathological condition which promoted disease progression in turn.23,24 It is not clear whether this cause-and-effect relationship exists in chondrocytes and affects the catabolism of OA chondrocytes. Mitochondrial respiratory chain (MRC) damage has been reported in OA chondrocytes that causes decrease in Δψm and ATP synthesis and increase in oxygen free radicals. High level of oxygen free radicals initiates chondrocyte apoptosis through the destruction of mitochondrial membrane, collapse of Δψm, and damage of mitochondrial DNA.25,26 Hence, Δψm was measured with the fluorescence probe JC-1 in the present work. The results showed that activation of α2-adrenergic signals by NE intensified mitochondrial depolarization and significantly decreased Δψm, while blocking of Arda2 reversed the decrease in Δψm.
The biogenesis of chondrocyte mitochondria increases when existing mitochondria with abnormal structure and function cannot produce sufficient energy.
22
Results from the present work indicate that when chondrocytes were stimulated by sympathetic signal neurotransmitters, their mitochondria underwent a series of quantitative and morphological changes to meet the energy demands for repair.
27
When the chondrocytes were stimulated by NE, their mitochondriogenesis markers were significantly upregulated. The morphology of mitochondria also changed from spherical to tubular to enhance energy production (
Homeostasis of the mitochondrial network is determined by the balance between mitochondrial fusion and fission. 32 Mitochondrial fusion and fission respond actively to changes in cell metabolism. When the mitochondrial dynamics of chondrocytes is out of order, there is a significant impact on mitochondrial structure and function. 33 The expression of proteins related to mitochondrial fusion and fission in chondrocytes is significantly increased during OA. 34 This suggests that upregulation of mitochondrial fusion and fission in the chondrocytes during OA results in mitochondrial kinetic damage. When the fusion and fission of cartilage mitochondria were inhibited, the basal and maximum oxygen consumption rates were downregulated, whereas glucose uptake was upregulated. 35 These features infer that mitochondrial fusion and fission play an important role in regulating the metabolism of osteoarthritic chondrocytes. 36 A previous study reported that overexpression of mitochondrial fusion and fission in chondrocytes exerts a pro-inflammatory role by upregulating the expression of inflammation-related genes in OA. 37 Hence, upregulation of chondrocyte mitochondrial fusion and fission contributes to the inflammation that occurs during OA.
Apart from mitochondrial biogenesis, fusion, and fission, mitophagy is another phenomenon associated with mitochondrial dynamics. 34 Mitophagy is regarded as a survival mechanism. Its dysregulation has been linked to the pathogenesis of OA. 38 Mitophagy is involved in mitochondrial fragmentation and cell death in human primary chondrocytes. 39 In addition, defective mitophagy in OA chondrocytes results in abnormal accumulation of dysfunctioned mitochondria and disruption of metabolic function in those chondrocytes. 33 In this study, mitochondrial autophagy could be clearly identified by TEM. The markers associated with mitophagy also increased significantly after NE stimulation and that the application of yohimbine significantly reversed this effect. These observations are supportive of the involvement of mitophagy in chondrocyte degeneration.
In the present research, activation of fusion and fission was evident when chondrocytes were treated with NE. This observation suggests that fusion and fission is one of the key players involved in promoting cartilage degeneration in TMJ-OA. Previous work identified that fusion and fission were related to cardiomyocyte injury. Inhibition of fusion and fission could effectively improve mitochondrial function and prevent the decline of cardiac function. 40 None report is available on the relationship between fusion and fission and cartilage degeneration in TMJ-OA. The present work reveals the role of fusion and fission activation in cartilage degeneration after the chondrocytes were treated with NE. Although important progress has been made in identifying the role played by chondrocyte mitochondrial dynamics in OA, there are other problems that need to be addressed. For example, the signaling pathways responsible for the upregulation of mitochondrial dynamics by α2-adrenergic signals are not clear. Additional receptors that promote the upregulation of mitochondrial dynamics, apart from Adra2, remain to be found. Because the expression profiles of adrenoreceptors are altered during TMJ-OA progression, it is important to utilize NE to stimulate condylar chondrocytes derived from different osteoarthritic stages in future studies.
Although the time of yohimbine being effective to mitigate TMJ pain and aligned complications is unclear. Studies 41 have shown that reduction of inflammation in TMJ-OA is effective in relieving pain. Therefore, the time of inflammatory cytokine reduction after yohimbine treatment can be used to evaluate the time to effectively mitigate the TMJ pain and aligned complications. As previously reported, 10 after 4-week intra-articular yohimbine injection, the expression of inflammatory factors (such as MMP-3 and MMP-13) significantly decreased, which meant the alleviated pain. Furthermore, the pain-related behaviors need to be tested in further studies. Noteworthily, due to the anatomical and physiological differences between rats and humans, the application of yohimbine to mitigate the TMJ pain and aligned complications in humans remains to be further discussed. These results confirmed the potential application of yohimbine for the treatment of TMJ-OA. We will further verify it in follow-up clinical studies. The data on lethal dose, 50% (LD50); bioavailability; and half-life of yohimbine in vivo will be further studied for clinical treatment.
In conclusion, this study demonstrates that changes in mitochondrial dynamics play an important role in chondrocyte catabolism mediated by sympathetic signal activation. The hypothesis that “α2-adrenergic signal activation promotes cartilage degeneration in TMJ-OA by upregulating mitochondrial dynamics in chondrocytes” is validated. Actually, inhibiting abnormal activation of mitochondrial fusion and fission is a potential regulator for improving mitochondrial function and inhibiting chondrocyte injury and contrives a potentially innovative therapeutic direction for the prevention of TMJ-OA.
Supplemental Material
sj-docx-1-car-10.1177_19476035231189841 – Supplemental material for Upregulated Mitochondrial Dynamics Is Responsible for the Procatabolic Changes of Chondrocyte Induced by α2-Adrenergic Signal Activation
Supplemental material, sj-docx-1-car-10.1177_19476035231189841 for Upregulated Mitochondrial Dynamics Is Responsible for the Procatabolic Changes of Chondrocyte Induced by α2-Adrenergic Signal Activation by Jiaying He, Wenpin Qin, Yusong Zhang, Jianfei Yan, Xiaoxiao Han, Jialu Gao, Qihong Li and Kai Jiao in CARTILAGE
Footnotes
Jiaying He, Wenpin Qin, Yusong Zhang, Jianfei Yan, Xiaoxiao Han, Jialu Gao, and Kai Jiao are now affiliated to State Key Laboratory of Oral & Maxillofacial Reconstruction and Regeneration & National Clinical Research Center for Oral Diseases & Shaanxi Key Laboratory of Stomatology, School of Stomatology & Shaanxi Key Laboratory of Stomatology, School of Stomatology, The Fourth Military Medical University, Xi’an, China
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by grants from the National Natural Science Foundation of China (grant numbers: 81870787, 82170978), and Distinguished Young Scientists Funds of Shaanxi Province (grant number: 2021JC-34).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Three-week-old female Sprague-Dawley rat were provided by the Laboratory Animal Center of the Air Force Medical University (AFMU; Xi’an, Shaanxi Province, China). All experimental procedures, including animal experiments, were approved by the Institutional Ethics Committee of AFMU.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
