Abstract
Objective
To directly compare clinical and MRI outcomes of multiple intra-articular injections of adipose-derived stromal cells (ASCs) or platelet-rich plasma (PRP) in patients with knee osteoarthritis (OA).
Design
We retrospectively compared 24-month outcomes in (1) 27 patients receiving 3-monthly intra-articular injections with a total of 43.8 million ASCs and (2) 23 patients receiving 3-monthly injections of 3-ml preparation of PRP. All patients had Kellgren-Lawrence grade 1, 2, or 3 knee OA with failed conservative medical therapy. The Numeric Pain Rating Scale (NPRS) scores; Knee injury and Osteoarthritis Outcome Score (KOOS) at baseline, 6, 12, and 24 months after the first injection; and the MRI Osteoarthritis Knee Score (MOAKS) at 12 and 24 months were considered as outcomes.
Results
No major complications occurred in any patient. Both groups significantly improved in pain NPRS score and KOOS at 6 months. At 12- and 24-month evaluations, the ASC group significantly decreased scores to a greater degree (P < 0.001) than the PRP group. MOAKS scores indicated a decrease in disease progression in the ASC group.
Conclusion
Both ASCs and PRP were safe and resulted in clinical improvement in patients with knee OA at 6 months; however, at 12 and 24 months, ASCs outperformed leukocyte-poor PRP in clinical and radiological outcomes.
Keywords
Introduction
Knee osteoarthritis (OA) is a worldwide chronic condition causing progressive pain and disability not only in the aging and/or obese but also within the younger and active populations. It is a multifactorial disorder leading to progressive cartilage damage, subchondral bone sclerosis, and synovial inflammation, resulting in impaired ability to exercise and perform physical activities of daily living (ADL). 1
Home-based exercise programs are standard treatments for knee OA, but these treatments are not always effective. 2 Nonsteroidal anti-inflammatory drugs (NSAIDs) have a limited benefit and are associated with potential risks. Moreover, knee arthroscopy is not always effective in improving OA symptoms with a lack of benefit beyond sham surgery and acceleration in progression to total knee replacement.3 -5 Total knee replacement remains the gold standard method for severe symptomatic OA. However, the latter solution is not without significant complications and residual pain after surgery; thus, it is recommended only as the last option after other treatments have failed.6,7 In that regard, orthobiologic injections such as platelet-rich plasma (PRP) have become increasingly popular for OA treatment due to their homeostatic, metabolic, and anti-inflammatory effects in the knee and articular cartilage.8,9 Despite conflicting results, they are reported to be safe and even more effective compared with other intra-articular injections such as hyaluronic acid, corticosteroids, or ozone.10,11 Using mesenchymal stromal cells (MSCs) is an attractive therapeutic option, thanks to their chondroprotective and anti-catabolic effects. First described in 2001, adipose-derived stromal cells (ASCs) are easy to harvest and include a higher proportion of MSCs, particularly after expansion.12 -16
Previous studies have reported that intra-articular injections of expanded ASCs may improve cartilage quality and delay OA progression.17 -19 However, there is a lack of comparative studies evaluating PRP with expanded ASCs. The aim of this study was therefore to compare the clinical and MRI outcomes of leukocyte-poor PRP (LP-PRP) and expanded ASC intra-articular injections in knee OA patients.
Methods
A retrospective analysis was conducted in our institution for knee OA patients who underwent orthobiologic injections from January 2015 to February 2019, with previous approval by the laboratory’s facility ethical committee (Clinical Trial MR0023). Two different methods were compared: LP-PRP and ASC injections. This retrospective cohort study was reported following the DOSES and TREND guidelines.20,21
Data Collection
After written informed consent was obtained from all patients, data were gathered from the patient’s medical records and processed anonymously, including clinical history, radiological Kellgren-Lawrence (KL) knee OA grading, Numeric Pain Rating Scale (NPRS) score, Knee Injury and Osteoarthritis Outcome Score (KOOS), 22 and MRI Osteoarthritis Knee Score (MOAKS) that were routinely collected from patients at 6, 12, and 24 months ( Table 1 ).
Outcome Measures Assessment Protocol.
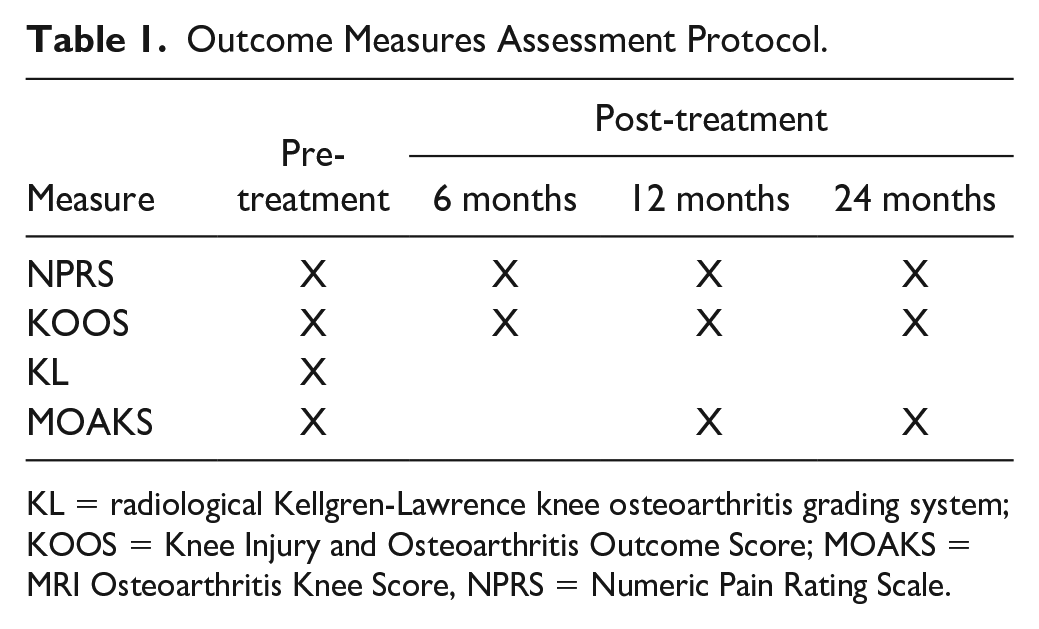
KL = radiological Kellgren-Lawrence knee osteoarthritis grading system; KOOS = Knee Injury and Osteoarthritis Outcome Score; MOAKS = MRI Osteoarthritis Knee Score, NPRS = Numeric Pain Rating Scale.
Participants
Patients were eligible for LP-PRP or ASCs if they fulfilled the following criteria: (1) unilateral knee OA; (2) Kellgren-Lawrence grade ranging I-III; (3) medial, lateral, and/or anterior compartment involvement; (4) no malalignment (less than 5° varus or valgus knee deformity as measured by the long mechanical axis of the knee on x-ray); (5) failed conservative treatment after an 8-week protocol including analgesia/anti-inflammatory medication, supplements, an exercise program, weight loss and/or nutritional management, and orthotics (when needed) under the professional supervision of a multidisciplinary team; (6) had a minimum Numerical Rating Scale (NRS) pain score of 5 (out of 11 points); and (7) were able to complete the patient-reported outcome questionnaires given by the investigators with minimal assistance.
Patients were excluded if they had (1) knee pain from causes different than OA (i.e., tumors or referred pain from the hip or lumbar spine); (2) previous surgery within the last 12 months; (3) previously received mesenchymal cell therapy due to any medical condition; (4) displaced meniscal tear on MRI; (5) grade IV chondral defect on MRI; (6) previous knee intra-articular injectable therapy within the last 6 months; (7) coagulation disorders (i.e., hemophilia); (8) a history of cancer; (9) a history of systemic illness or significant organ impairment/failure (i.e., renal failure); (10) a history of atypical chronic pain syndrome (i.e., chronic regional pain); (11) a history of allergy to any substances used within the treatments; and/or (12) being pregnant or breastfeeding.
Intervention
The treatment method was determined individually. Factors such as acceptance of the procedures and coverage by insurance were considered if patients fulfilled the criteria for conservative treatment.
Collection of adipose tissue and ASC processing
Before collection, the patient had to have normal prothrombin time, partial thromboplastin time, and platelet count. Adipose tissue (AT) was obtained from the periumbilical area. The procedure was done in an outpatient setting using 2% lidocaine with epinephrine as local anesthetics. A total of 2 to 5 ml of AT was obtained through multiple punctures with a reusable core biopsy instrument (BARDR® MAGNUM®, New Jersey, USA) and the corresponding biopsy needles. Sample processing and cell characterization are already described in previous studies. 23 AT was rinsed in Hanks’ Balanced Salt Solution (HBSS) (Gibco, Cat. No. 14,025,088) and incubated with collagenase (collagenase from Clostridium histolyticum, SIGMA, Cat. No. C9722). MSCs were counted and seeded in T25 cm2 tissue culture flask in complete media supplemented with 100 IU penicillin, 100 IU amphotericin, 100 IU streptomycin (Ritcher), and 10% autologous serum. When cultures reached 90% to 100% confluence, subculturing was performed using trypsin Tryple Select (Gibco, Cat. No. 12,563,029). At harvest, cells of all patients were in passages lower or equal to 3. Prior to injection, ASCs were tested for endotoxin, mycoplasma, and microbial contamination. For injection, ASCs were washed and suspended in 5 ml HBSS without antibiotics. Cell count and viability were confirmed using a manual hemocytometer. In the laboratory Regenerarr facility, ASCs were successfully grown in culture. Cells exhibit elongated nuclei and spindle-shaped cytoplasm, characteristic of a fibroblast-like morphology.
Characterization of ASCs
ASCs were stained with antibodies against CD73, CD90, CD105, CD45, CD34, CD11b, CD19 or CD79a, CD14, and HLA class II to determine surfer marker expression. In accordance with the International Society for Cellular Therapy (ISCT) recommendations, ≥95% of the ASC population expressed CD105, CD73, and CD90, as measured by flow cytometry. In addition, these cells lacked the expression (≤2% positive) of CD45, CD34, CD14 or CD11b, CD79a or CD19, and HLA-DR class II. Cells were able to differentiate to osteoblasts, adipocytes, and chondroblasts under standard in vitro differentiating conditions. FACSCanto II BD flow cytometer instrument was used for running the samples. The samples were immunostained with a determined panel of monoclonal antibodies (mentioned above) conjugated with different fluorochromes, and mouse isotype antibodies were used as control. The results were analyzed with the software Infinicyt version 1.7i.
PRP preparation
A 40-ml peripheral blood sample was extracted using a 20-gauge needle for a Vacutainer holder system. It was placed in 8 tubes with 8 ml of EDTA serum. Two of them were used to perform routine serologic testing, hematology control, and immunohematological donor/patient tests, and the remaining 6 tubes, for a total of 30 ml of blood (5 ml blood per tube) with 6 ml EDTA (1 ml per tube), was devoted to obtaining PRP. We utilized a 2-spin protocol. The blood was first centrifuged for 5 minutes at 1,600 rpm. The resulting plasma and buffy coat (an average of 5 ml per procedure) were separated from red cells under laminar flow. This product was referred to a 10-ml tube and was centrifuged one more time for 5 minutes at 1,800 rpm. A product of 4 ml LP-PRP was achieved.
Before PRP injection, quality control was performed on the baseline blood and the final PRP product through a hematology analyzer (BenchMark XT; Roche®, Basel, Switzerland). This assessment included platelet count per mm3, total leukocytes per mm3, and specific leukocyte formula. All patients were treated with LP-PRP (white blood cell concentration below baseline levels).
According to the PAW (platelets, activation, white blood cells) classification, 5 all patients were P4-Bß (P4: >1,250,000 platelets/μl, white blood cell concentration below baseline, neutrophil concentration below baseline). The mean concentration of platelets delivered per PRP injection was 1,280,320 ± 222.07/mm3, representing a 5.76-fold increase in the average baseline value. The mean final concentration of leukocytes was 1,213.8/mm3, representing a 5.59-fold decrease in the average baseline value. The mean final concentration of neutrophils was 700/mm3. No endogenous or exogenous activation was used.
Intra-articular injection of ASCs and LP-PRP in the knee
ASCs were injected at an average of 1 month after tissue collection by the senior author (M.A.K.). The cultured ASCs were washed twice and suspended in 3 ml of HBSS. Before injection, the skin was prepared with an aseptic technique with iodine solution. An average of 14.6 million ASCs were injected monthly into each affected knee 3 times using a superolateral portal under ultrasound guidance. LP-PRP volume was 5 ml in each injection. LP-PRP and cells were delivered intra-articularly without local anesthetics.
Post-procedure protocol
Immediately after the injection, the patients were kept supine for ~15 minutes. Patients were sent home with instructions to rest from daily activities for approximately 24 hours. If necessary, patients were allowed to use acetaminophen, but nonsteroidal anti-inflammatory medication was not allowed. After 48 hours, patients were instructed with a 2-week standardized protocol under the supervision of a physical therapist. LP-PRP patients were not allowed to participate in active or heavy sports for 72 hours, and ASC patients for a week.
MRI Assessment
The MOAKS is a validated semi-quantitative tool for evaluating knee OA. The MOAKS was used to describe OA progression over time of medial, lateral, and patellofemoral compartments. 15 Two experienced sports trauma radiologists performed the interpretation. We used a standardized and reproducible scoring system (Suppl. Material 1). The equipment used was a 1.5-T MRI scanner Flex-M Coil (Philips®, Eindhoven, The Netherlands). Short tau inversion recovery (STIR) coronal, axial, and sagittal views (field of view [FOV] = 130 mm, TR = 3,500, TE = 50, TI = 100) were evaluated, and thickness was 3 mm.
Minimal Important Clinical Difference
The minimal important clinical difference (MICD) is the smallest difference between two measurements deemed necessary for a patient to perceive beneficial clinical improvement. The MICD in knee OA is currently suggested to be 8-10 in KOOS. MICD is referred to the change, compared with the baseline, that a patient recognizes. MICD in patients with knee OA was considered for each domain of KOOS. Pain improvement: 13.4; symptoms: 15.5; ADL: 15.4; sport/recreation; 19.6; and for quality of life (QoL): 21.1. 22
Statistical Analysis
Data were processed with R software (R Foundation for Statistical Computing, Vienna, Austria [https://www.R-project.org/]). Results are shown as mean ± standard deviation (SD) for numerical variables and percent for categorical ones. The normality of the distribution of numerical variables was verified using the Shapiro-Wilk test.
Hierarchical linear modeling (HLM) with PRP as a reference group was used to assess change over time and the effect of treatment on each KOOS scale: symptoms, pain, ADL, sport, and QoL at 0, 6, 12, and 24 months. The intercept predicts the mean value in the reference group. A time variable was included in determining whether the evolution of the dependent variable fits a specific pattern. The spline model was used to evaluate time points where the slope changes (at 6 months). The following terms were included: baseline ASC group in relationship to PRP (baseline difference related to the control group) and monthly change from baseline to 6 and 24 months. Gender, age, body mass index (BMI), and OA were included in the model as independent variables only if results were statistically significant, as well as interaction terms between time and treatment. The significance level of the statistical tests was set at P < 0.05.
Results
A total of 23 patients in the LP-PRP group and 27 in the ASC group were analyzed; with no statistically significant intergroup demographic differences ( Table 2 ).
Baseline Characteristics of the ASC and PRP Groups.
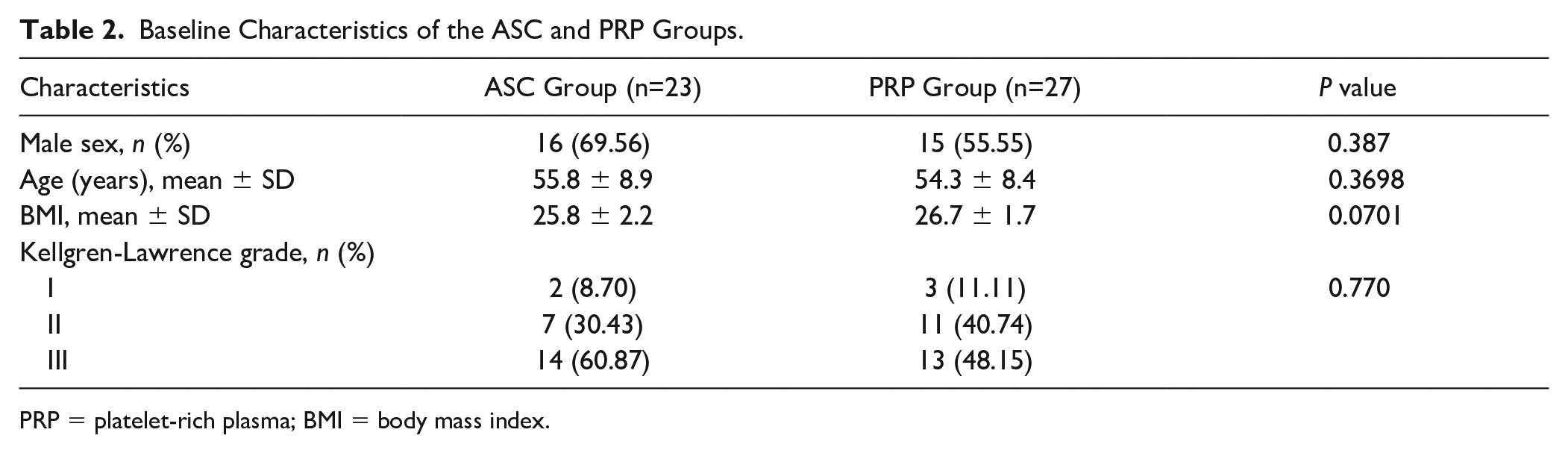
PRP = platelet-rich plasma; BMI = body mass index.
Clinical Outcomes
The mean clinical outcome scores at baseline, 6, 12, and 24 months for the ASC and PRP groups are summarized ( Table 3 ).
Mean Scale Values at Baseline, 6, 12, and 24 Months for the ASC and PRP Groups.
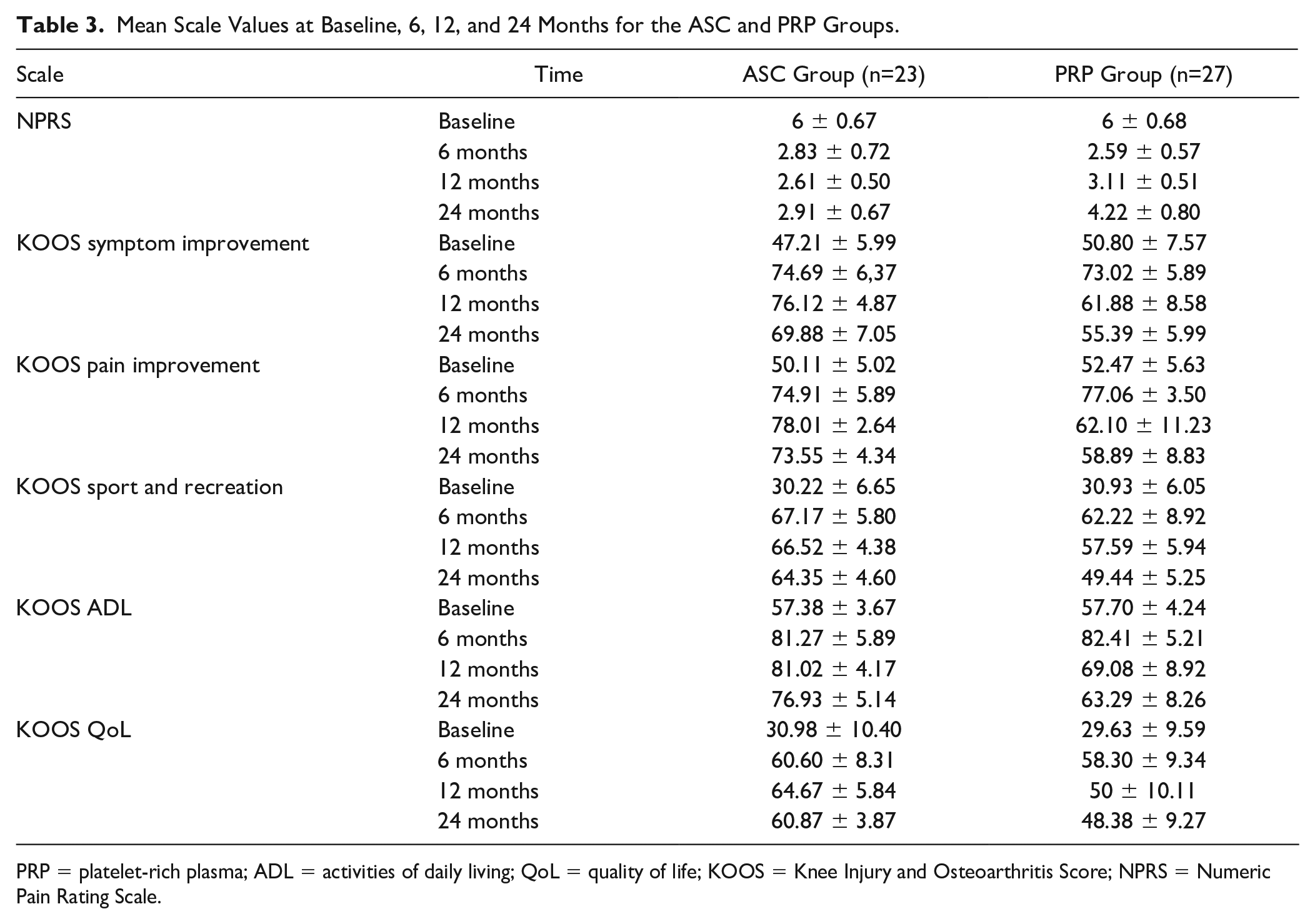
PRP = platelet-rich plasma; ADL = activities of daily living; QoL = quality of life; KOOS = Knee Injury and Osteoarthritis Score; NPRS = Numeric Pain Rating Scale.
NPRS and KOOS subscale analysis revealed improvement in all subscales at 24 months of follow-up ( Figure 1 ).
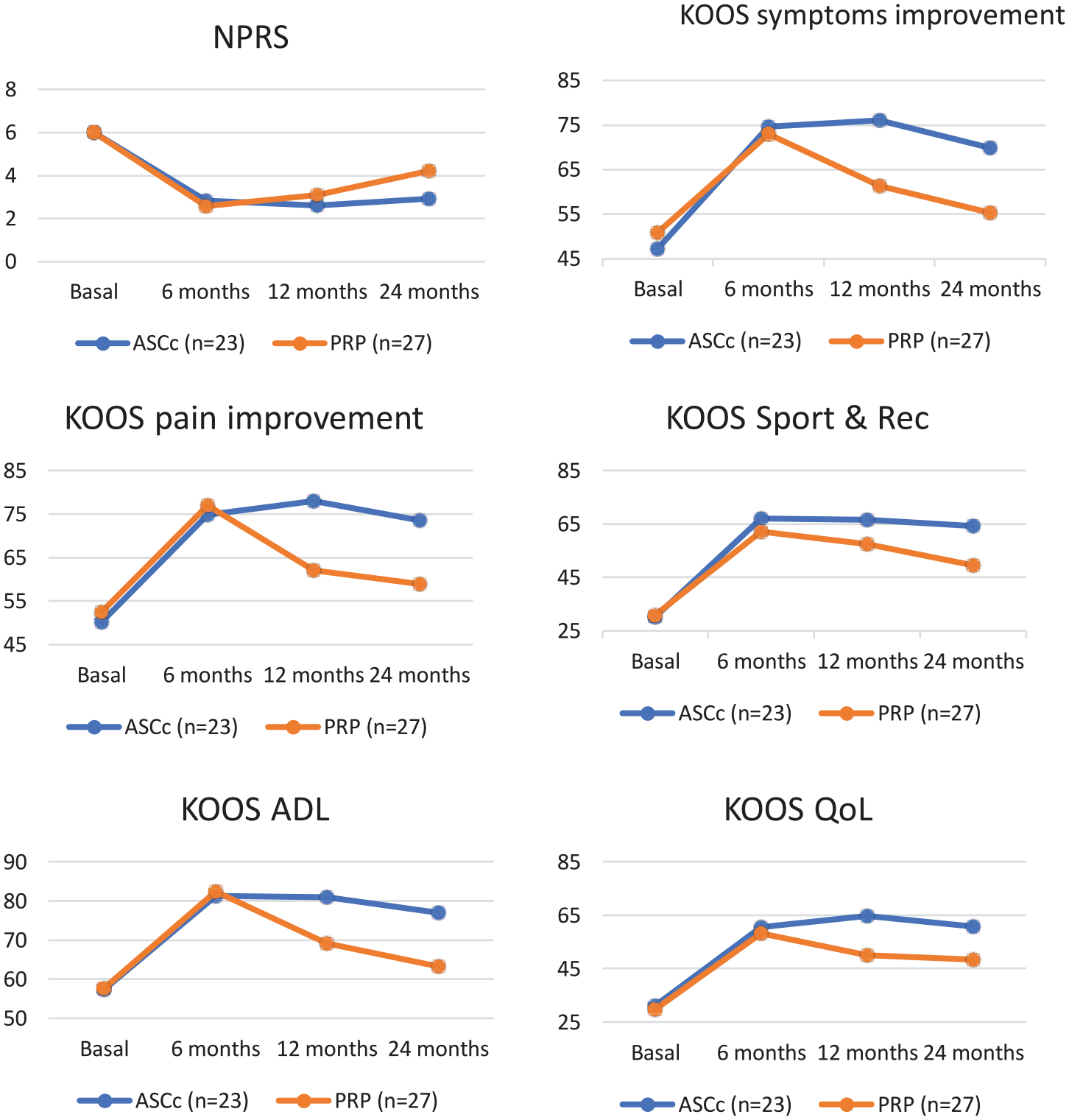
NPR and KOOS subscales (symptom improvement, pain improvement, sport and recreation). NPR = Numeric Pain Rating Scale; KOOS = Knee Injury and Osteoarthritis Outcome Score; ADL = activities of daily living; QoL = quality of life.
The slope progression of the KOOS subscales showed biphasic behavior. During the first 6 months, both groups sustained score improvement. Subsequently, the slope leveled off in both groups, but significantly more in the PRP group. The spline (trend change over time) was statistically significant in all subscales. HLM included terms for time trends and differences between groups. HLM with PRP as a control group is shown in Table 4 .
Hierarchical Linear Modeling (HLM) for KOOS Scales as the Outcome (Dependent Variable).
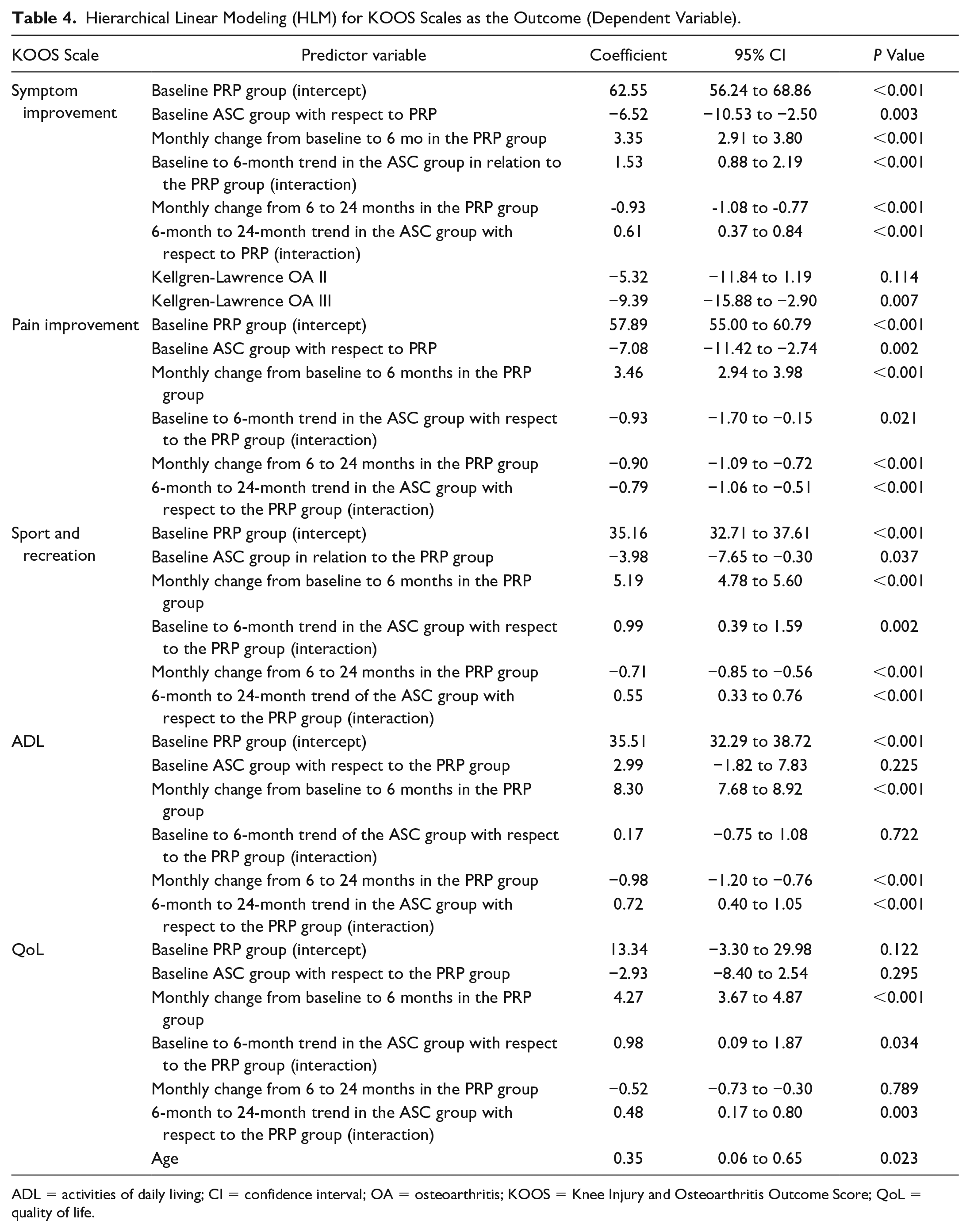
ADL = activities of daily living; CI = confidence interval; OA = osteoarthritis; KOOS = Knee Injury and Osteoarthritis Outcome Score; QoL = quality of life.
There were statistically significant baseline differences between the ASC and PRP groups in symptoms, pain, and sport-and-recreation subscales. The models allowed the adjustment for these differences. All subscales significantly improved at 6 months, but the ASC group significantly improved more than PRP at 6 months for symptoms, pain, and QoL subscales. After the 6-month follow-up, all KOOS subscales significantly deteriorated in the PRP group but not in ASC group. There were statistically significant differences in the progression afterward (statistically significant interaction terms) between the PRP and ASC groups in all KOOS subscales. Regarding covariables, age and the Kellgren-Lawrence OA III before injection were significantly associated with worse symptom subscale progression and worse evolution of the QoL subscale
The percentage of participants achieving an MICD is summarized
Minimal Clinically Important Difference at 24 Months in Comparison With Baseline Data for the 2 Groups.
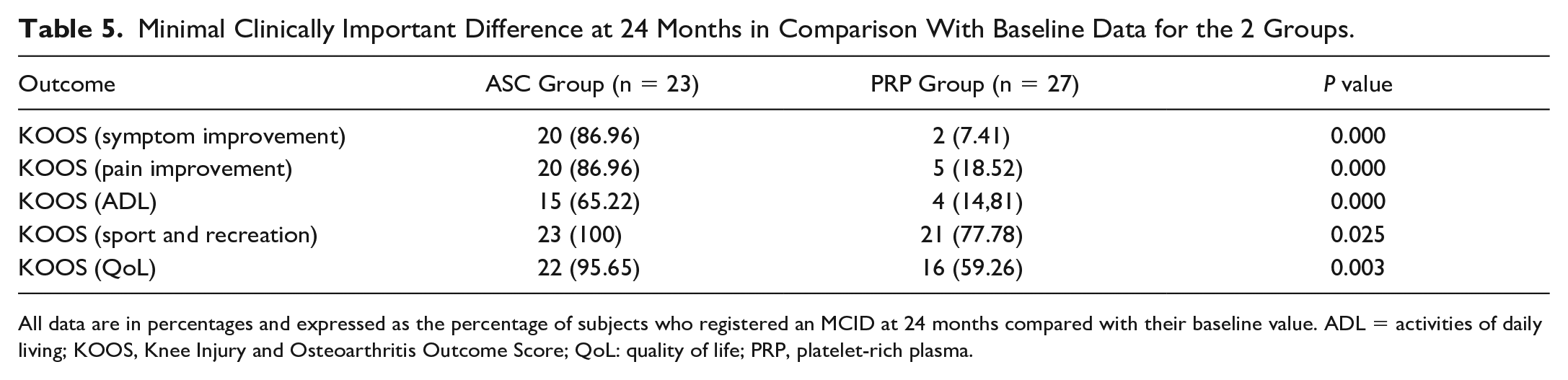
All data are in percentages and expressed as the percentage of subjects who registered an MCID at 24 months compared with their baseline value. ADL = activities of daily living; KOOS, Knee Injury and Osteoarthritis Outcome Score; QoL: quality of life; PRP, platelet-rich plasma.
MRI Outcomes
MOAKS assessment at 12 months revealed significantly better improvement in bone marrow lesion and articular cartilage pathology in the ASC group than in the PRP group (34.78% vs. 11.11 and 21.74% vs. 7.41%, P = 0.04, respectively). This superiority persisted at the 2-year follow-up in bone marrow lesions, articular cartilage pathology, and synovitis ( Table 6 and Figures 2 - 6 ).
Baseline Prevalence and Change in Global MOAKS at 12- and 24-Month Follow-Up.
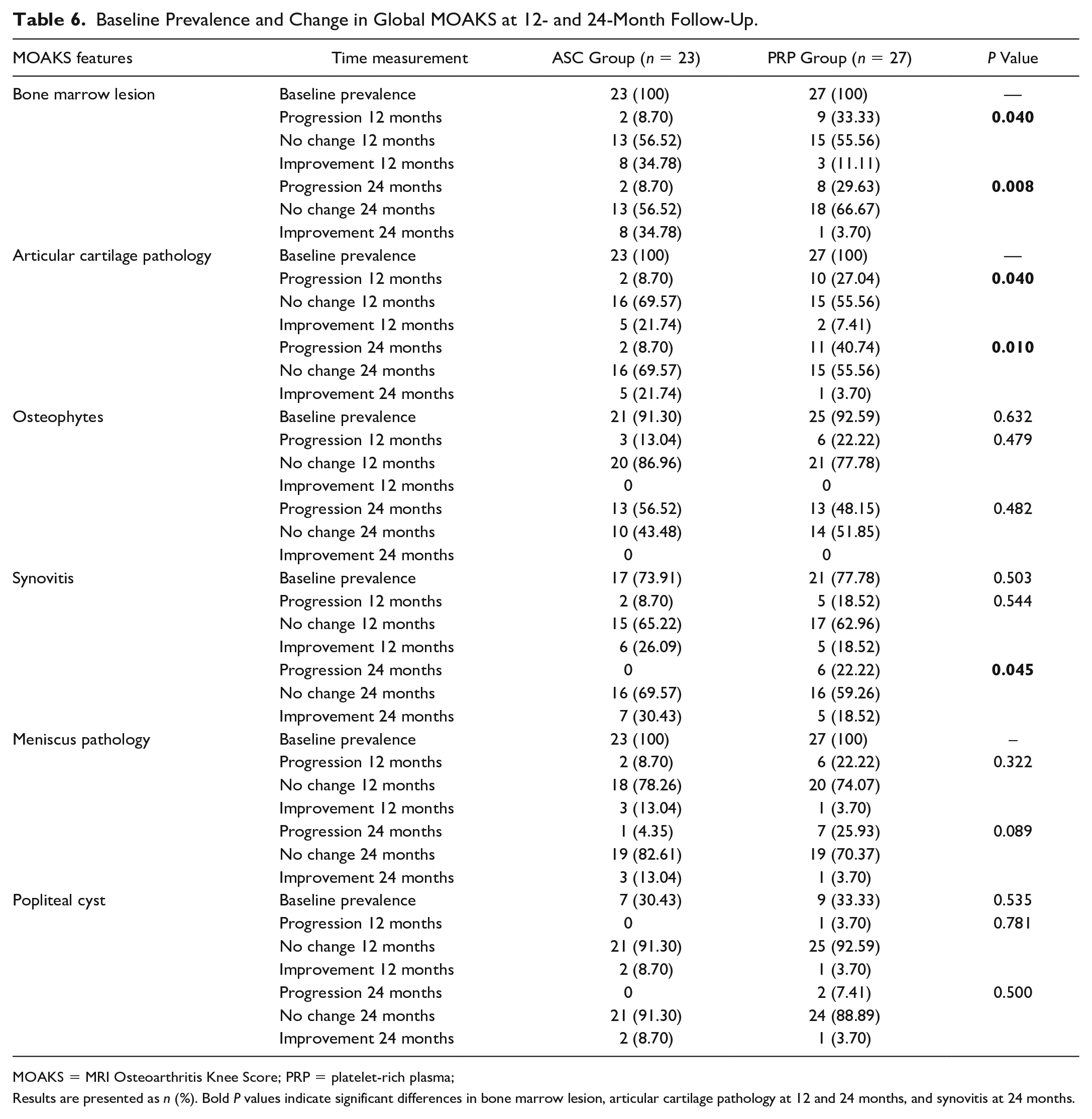
MOAKS = MRI Osteoarthritis Knee Score; PRP = platelet-rich plasma;
Results are presented as n (%). Bold P values indicate significant differences in bone marrow lesion, articular cartilage pathology at 12 and 24 months, and synovitis at 24 months.
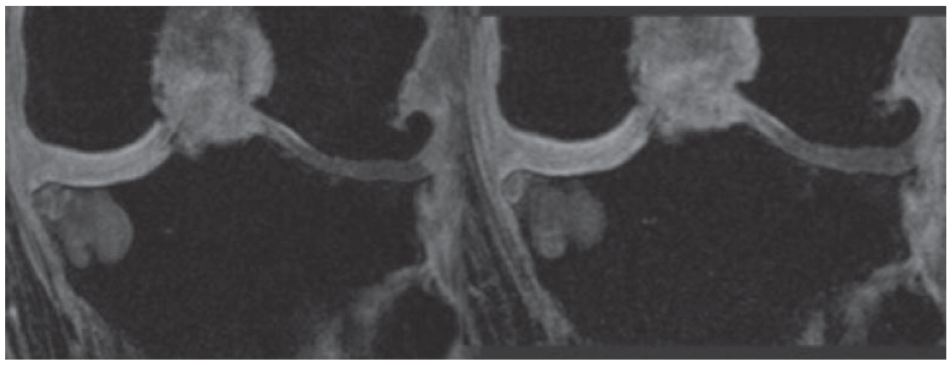
MRI Fat saturated coronal image BML, MOAKS grade 3 (left) and 2 (right) in the medial knee compartment (white arrow) and lateral MOAKS grade 3 osteophytes without modification (blue arrow) after 24 months ASCs injections.
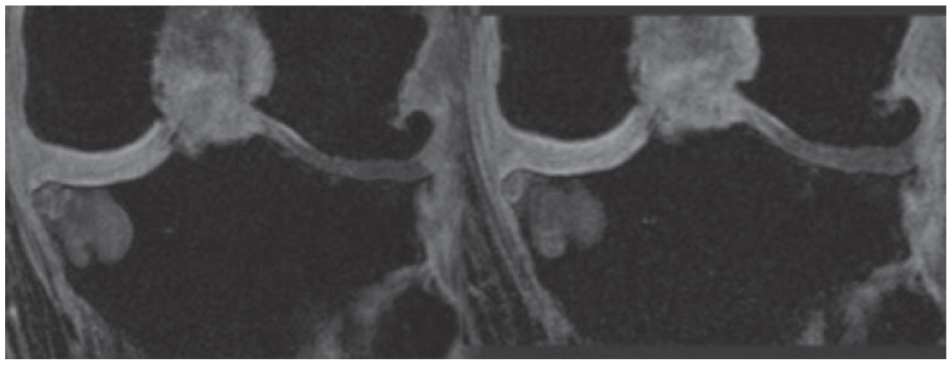
MRI fat-saturated sagittal image evaluating articular cartilage injury, MOAKS grade 2 (Left) and 0 (right) in medial knee compartment, weight-bearing region (white arrow) after 24 months ASCs injections.
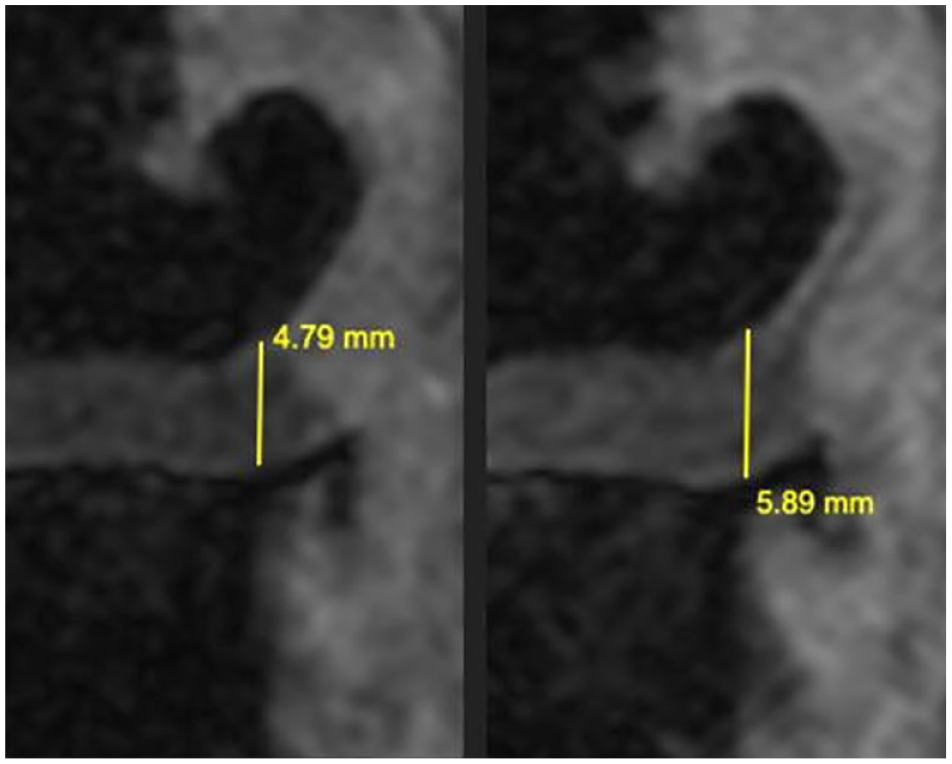
MRI fat-saturated signal coronal image evaluating the distance between femur and tibia. Notice the increase in distance after 24 months of ASC injection therapy. ASC = adipose-derived stromal cell.
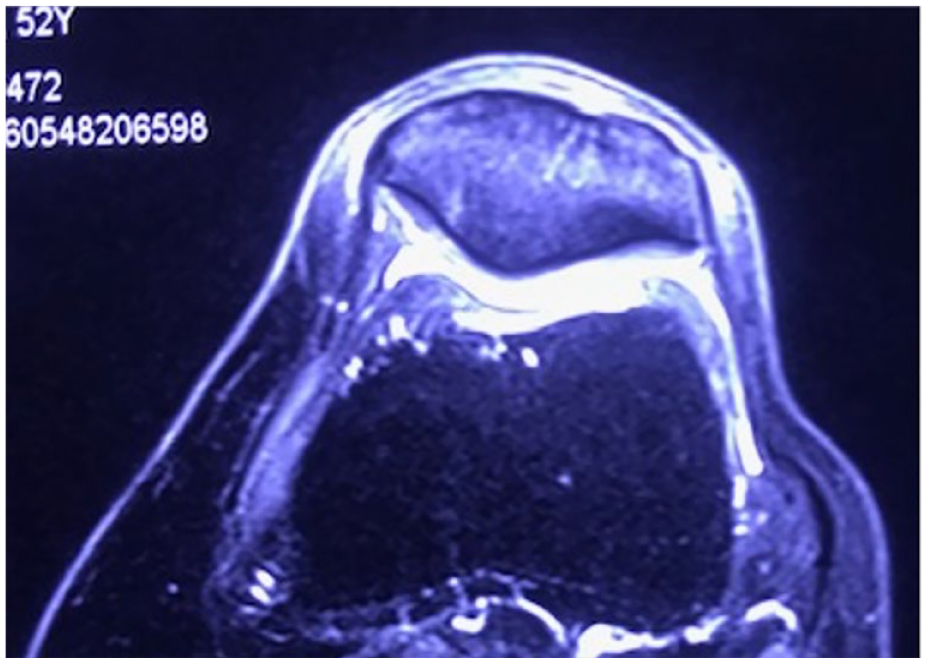
White arrow pointing at initial MOAKS grade 1 articular cartilage injury and bone marrow lesion in a PRP-treated patient. MOAKS = MRI Osteoarthritis Knee Score; PRP = platelet-rich plasma.
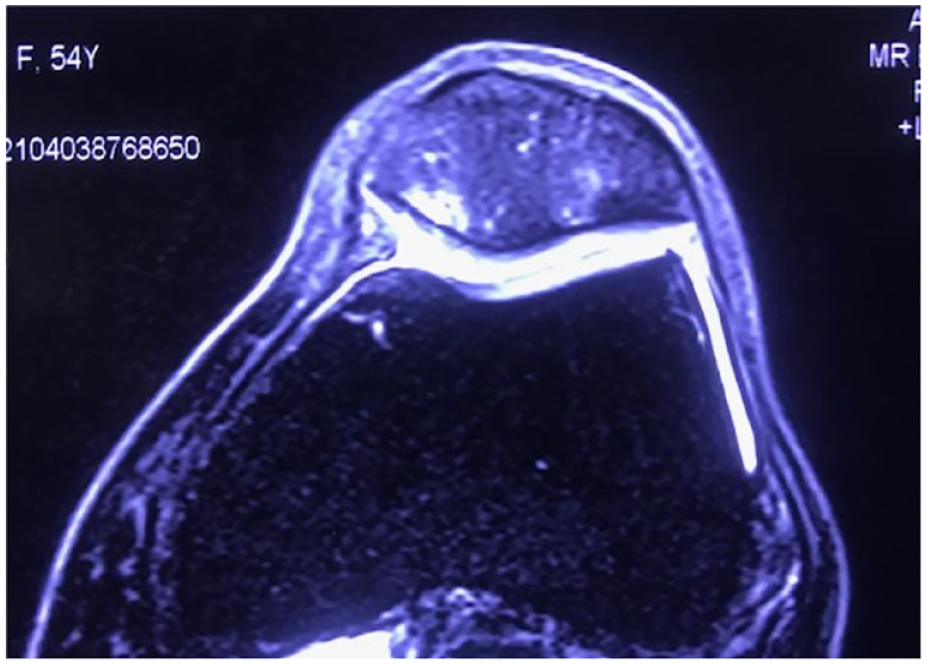
White arrow pointing at cartilage and bone marrow lesion in an MOAKS grade 2 injury. Progression of lesion in a PRP-treated patient. MRI at 24-month follow-up. MOAKS = MRI Osteoarthritis Knee Score; PRP = platelet-rich plasma.
Complications and Adverse Events
Five patients in the ASC group (18% of patients) and three in the PRP (13%) group experienced discomfort in 1 occasion and/or post-injection swelling. Only 1 patient experienced discomfort twice in 2 different injections in the ASC group. These adverse events (AEs) were self-limiting, requiring a period of unloading, analgesia, and/or oral anti-inflammatory medication. No related serious AEs were observed in any of the 2 groups’ participants. Minor discomfort and bruising were commonly noted in patients. Two patients, one of each group, developed a local induration on the injection site that resolved without further treatment at 2 months. Two patients chose to go for total knee replacement (the first after the second dose of PRP and the second after the first dose of ASCs) and were excluded from the analysis. They were not included in this series.
Discussion
Our study showed that (1) no serious AEs were observed in any patient; (2) ASC and PRP injections improved pain and symptoms in knee OA at 6 months post-treatment; and (3) ASCs subsequently outperformed PRP up to the second year post-treatment. ASCs showed significantly better NPRS, KOOS, and MRI features in bone marrow lesions, articular cartilage pathology, and synovitis at a 2-year follow-up. Particularly, bone marrow lesions have been considered as an important factor for knee deterioration in knee OA. 24
The evidence comparing PRP and ASC injection therapy for knee OA is scarce. A prospective study compared PRP, bone marrow aspirate concentrate, and micro-fragmented AT intra-articular injections in Kellgren-Lawrence I-III knee OA patients, respectively, showing no significant differences in International Knee Documentation Committee (IKDC), Knee score, and Function Knee Score between treatments at 12 months post-treatment. 25 A randomized controlled trial comprising 58 patients with symptomatic Kellgren-Lawrence I-IV knee OA showed comparable Visual Analog Scale (VAS) and KOOS scores after leukocyte-rich PRP and micro-fragmented AT intra-articular injections (3.56 ± 4.62 million nucleated cells/ml) at 6-month follow-up. 26
Similarly, Kaszyński et al., 27 in their randomized controlled trial including 60 patients with Kellgren-Lawrence I-III symptomatic knee OA, showed comparable VAS, KOOS, Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), and IKDC 2000 outcomes between intra-articular LP-PRP and autologous AT injections at 12 months. Similarly, Zaffagnini et al., 28 in a randomized controlled trial, showed no significant differences in IKDC, KOOS, and EuroQol 5-dimensions questionnaire (EQ-5D) between a single intra-articular injection of micro-fragmented AT and PRP at 12-month follow-up. In addition, no imaging differences were noticed using the Whole-Organ Magnetic Resonance Imaging Score between them.
However, the results of the cited comparative studies might be confounded due to the heterogeneous composition and doses of the PRP and MSC injections, allowing no fair comparisons between studies. In the previous studies, it should also be highlighted that the utilized AT contained cells without expansion. These inconsistencies in the reporting of orthobiologics interventions have been exposed by several authors, as has been the number of injections and presence of leukocyte cells in PRP preparations.29 -31 Using adipose-derived expanded MSC studies showed a dose-dependent effect of PRP and ASCs. 32 Bansal et al., 33 in a recent randomized controlled trial, found a cut-off dose of 10 billion platelets for sustained effects on knee OA, with significant IKDC and WOMAC score improvement at 1-year follow-up. Similarly, studies on ASC injections display a variety of dosages, ranging from single- to 3-injection protocols and 5 to 100 million cells/injection.12,17,18,34 Current data suggest that ASC injections with higher cell count and frequency (dose) correlate with better clinical and radiological outcomes. This may further support our results with 3-monthly intra-articular injections with an average of 14.6 million cells/injection.
MSC intra-articular knee injections have several proposed mechanisms of action. These include paracrine effects of secreted bioactive molecules such as anti-inflammatory, immunomodulatory, and chondroprotective mediators. This is achieved through molecular, organelle, or exosome transfer rather than direct cellular differentiation into chondrocytes. Murphy et al. 35 showed in a preclinical trial that there is no significant incorporation of labeled MSCs into regenerative cartilage tissue. De Windt et al. 36 reported that DNA short tandem repeated analysis delivered unique proof that the regenerated tissue contained patient DNA only. These findings support the hypothesis that allogeneic ASCs stimulate a regenerative host response. It has been suggested that rather than differentiating cells that integrate into the host tissue, MSCs stimulate the patient’s own cells to fill the defect and functions more as stimulatory (trophic) factors.36,37 ter Huurne et al., 38 in a model of experimental OA, showed that intra-articularly injected ASCs were found inside the cruciate ligaments and subintimal layers of the synovium in mice as early as 24 hours after injection. MSCs are engulfed by Ly6Clow monocytes, which then change T-cells to regulatory T-cells, which can account for long-term therapy mainly through a paracrine mechanism.
As reported by previous authors,12,17,18,34,39 -42 no severe AEs were observed in the current study. Only 3 incidences of mild knee discomfort and effusion in each group were seen and managed appropriately with a period of unloading and standard analgesia. Similarly, only minor bruises and 1 hematoma with tissue induration were seen in the ASC group.
The main limitation of this study is its retrospective nature and lack of randomization. Also, the structural assessment of this study was done through a semi-quantitative measure. Other techniques such as T2 mapping and delayed gadolinium-enhanced MRI of cartilage (dGEMRIC) have better sensitivity in evaluating articular cartilage. The mechanical axis was only measured for inclusion/exclusion purposes. Therefore, our results were not considered in relation to knee alignment. Most patients reported using analgesia and/or anti-inflammatory medication before enrolling in the study and were asked at every clinic visit to note any medication modifications. However, during the later stages of the trial study, visits were 12 months apart, and patient recall was poor. A different strategy to capture medication use in future trials, such as a patient diary, may enable a thorough analysis, although we are aware of the limitations associated with such procedures. Finally, our study did not measure serum, urine, or articular biomarkers, which if assessed in the future may contribute to a clearer understanding of the impact of cell therapy in this setting.
Conclusion
ASC and PRP injection therapies are safe and yield similar improved pain and symptoms in knee OA at 6-month follow-up. However, for long-term outcomes, ASCs outperformed PRP in clinical and radiological outcome measures such as NPRS, KOOS, and MRI features in bone marrow lesions, articular cartilage pathology, and synovitis up to the second year post-treatment.
Supplemental Material
sj-docx-1-car-10.1177_19476035231166127 – Supplemental material for Knee Osteoarthritis: Clinical and MRI Outcomes After Multiple Intra-Articular Injections With Expanded Autologous Adipose-Derived Stromal Cells or Platelet-Rich Plasma
Supplemental material, sj-docx-1-car-10.1177_19476035231166127 for Knee Osteoarthritis: Clinical and MRI Outcomes After Multiple Intra-Articular Injections With Expanded Autologous Adipose-Derived Stromal Cells or Platelet-Rich Plasma by Miguel A. Khoury, Karim Chamari, Montassar Tabben, Khalid Alkhelaifi, Emmanuel Papacostas, Theodorakys Marín Fermín, Markus Laupheimer and Pieter D′Hooghe in CARTILAGE
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval number RGC000227062016 by REGENERAR.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
