Abstract
Objective
Evaluate serum and urine biomarker panels for their capabilities in discriminating between individuals (13- to 34-years-olds) with healthy hips versus those with developmental dysplasia of the hip (DDH) prior to diagnosis of secondary hip osteoarthritis (OA).
Design
Urine and serum were collected from individuals (15-33 years old) with DDH, prior to and following diagnosis of hip OA, and from age-matched healthy-hip controls. Samples were analyzed for panels of protein biomarkers with potential for differentiation of hip status using receiver operator characteristic curve (area under curve [AUC]) assessments.
Results
Multiple urine and serum biomarker panels effectively differentiated individuals with DDH from healthy-hip controls in a population at risk for developing secondary hip OA with the best performing panel demonstrating an AUC of 0.959. The panel comprised of two serum and two urinary biomarkers provided the highest combined values for sensitivity, 0.85, and specificity, 1.00, while a panel of four serum biomarkers provided the highest sensitivity, 0.93, while maintaining adequate specificity, 0.71.
Conclusion
Results of this study indicate that panels of protein biomarkers measured in urine and serum may be able to differentiate young adults with DDH from young adults with healthy hips. These data suggest the potential for clinical application of a routine diagnostic method for cost-effective and timely screening for DDH in at-risk populations. Further development and validation of these biomarker panels may result in highly sensitive and specific tools for early diagnosis, staging, and prognostication of DDH, as well as treatment decision making and monitoring capabilities.
Level of Evidence:
III
Introduction
Developmental dysplasia of the hip (DDH) is a significant risk factor for early-onset hip osteoarthritis (OA). 1 Importantly, the progression of DDH to secondary hip OA can be mitigated when timing and accuracy of diagnosis allow for preventative interventions to be effectively implemented.2 -4 Currently, DDH is often overlooked and screening diagnostics including infant hip palpation and ultrasonography are associated with a misdiagnosis rate of at least 14%, such that hip OA secondary to DDH is a growing healthcare concern. 5 As such, improved methods for screening and early diagnosis for DDH are critically needed. Serum, urine, and synovial fluid protein biomarkers have been reported to have utility for disease screening, diagnosis, and staging for several joint disorders6 -9 suggesting that they may have potential for addressing this need for individuals at risk for DDH. In particular, serum and urine biomarkers are attractive for development of clinically applicable DDH diagnostics based on the capabilities for obtaining these fluids in a minimally or non-invasive manner as standard of care at routine clinical appointments throughout an individual’s lifetime.10 -12
Previous research has identified panels of serum and urine protein biomarkers to discriminate between dogs with healthy hips and those with hip dysplasia, prior to the onset of secondary osteoarthritis. 13 Initial data comparing individuals with DDH, prior to and following diagnosis of hip OA, and from age-matched healthy-hip controls delineated significant differences in mechanistic serum and urine biomarkers between hip-status cohorts. 14 Taken together, these data provide a foundation for development and validation of biomarker panels as a clinically applicable and cost-efficient method for screening, diagnosis, and staging for DDH to avoid misdiagnosis and improve patient outcomes. Therefore, this study was designed to evaluate serum and urine biomarker panels in discriminating between healthy-hip and DDH status in 13- to 34-year-old patients. It was hypothesized that one or more biomarker panels could effectively differentiate individuals with DDH from those with healthy hips in a population at risk for developing secondary hip OA.
Methods
With IRB approval (#2012192) and informed consent, blood and urine were collected from two cohorts of human subjects:
DDH cohort (n = 32): Young adult (13–34 years old) patients with physician confirmed DDH, prior to clinical or radiographic signs of secondary hip OA.
Healthy-hip controls (n = 35): 13- to 34-year-old volunteers with no clinical or radiographic signs of DDH.
Exclusion criteria included incompetent adults, prisoners, patients with cancer or received cancer treatment within the past 6 months, patients who had any surgery within the last 6 months, previous hip surgeries at any time, recent joint trauma, metabolic disorders, rheumatoid arthritis, corticosteroid injections within the last 6 months, currently taking oral corticosteroids, serious organ diseases or failures, are pregnant or lactating, or syndromic diseases (e.g. cystic fibrosis or multiple sclerosis).
Diagnosis of DDH was determined based on physical examination by a board-certified orthopedic surgeon in conjunction with diagnostic imaging assessments of the hips, including anteroposterior, Dunn, false-profile, and lateral radiographic views. In addition, computed tomography (CT) was utilized to assess acetabular version and femoral torsion. Measurements of left center-edge angle, anterior center-edge angle, Tönnis angle, acetabular extrusion index, alpha angle, head sphericity, acetabular version, and femoral torsion were utilized for diagnosis. Hips were categorized by definitive diagnosis by clinical exam for those with DDH, while the control cohorts were composed of volunteers with self-reported healthy hips, based on routine physical examinations and lack of any symptoms related to the hips.
Sample Collection and Storage
Blood and urine samples were collected from participants in who were diagnosed with DDH through clinical examination and radiological assessment prior to surgical intervention. Whole blood (2-6 ml) was collected by aseptic peripheral venipuncture in a Vacutainer Serum Tube (Becton, Dickinson and Company, Franklin Lakes, NJ), and urine (>4 ml) was collected by voluntary micturition. After sample collection, the fluids were immediately transported to an on-site laboratory for processing. Whole blood samples were allowed to clot and then centrifuged (1200 x g, 10 min), followed by serum collection. Serum and urine samples were aliquoted and stored at -80°C for subsequent analyses.
Protein Analysis
Based on previous data for application in canine hip dysplasia 13 and DDH, 14 clinically relevant biomarkers with mechanisms related to joint health and disease were selected for testing. To measure dynamics of collagen degradation and synthesis, cross-linked C-telopeptide of type I collagen (CTX-I), and cross-linked C-telopeptide type II collagen (CTX-II), procollagen I C-terminal propeptide (PICP) and procollagen II C-terminal propeptide (PIICP) using commercially available enzyme-linked immunosorbent assays (ELISA) assays (ABclonal; Woburn, MA). The concentration of hyaluronan (HA), a disintegrin-like and metalloproteinase domain with thrombospondin motifs 4 (ADAMTS4) and ADAMTS5, receptor activator of NF-kappa B ligand (RANKL), and Cartilage Oligomeric Matrix Protein (COMP) in the samples were assessed using DuoSet ELISA assays according to the manufacturer’s protocol (R&D Systems; Minneapolis, MN). The concentration of matrix metalloproteinase (MMP)-1, MMP-2, MMP-3, MMP-7, MMP-8, MMP-9, and MMP-13 was determined using Human multiplex Luminex immunoassay (R&D System, Minneapolis, MN). The concentration of dickkopf-related protein 1 (DKK-1), osteoprotegrin (OPG), osteocalcin (OC), osteopontin (OPN), sclerostin (SOST), and parathyroid hormone (PTH) using the Human Bone Metabolism multiplex Luminex immunoassay (MilliporeSigma, Burlington, MA). The concentration of fibroblast growth factor (FGF)-2, Fractalkine, interferon (IFN)-γ, growth-regulated oncogene (GRO)-α, interleukin (IL)-1β, IL-1-receptor antagonist(RA), IL-4, IL-6, IL-10, and IL-13, monocyte chemoattractant protein (MCP)-1 and MCP-3, platelet derived growth factor (PDGF)-AA and PDGF-AB/BB, macrophage inflammatory protein (MIP)-1α and MIP-1β, regulated upon activation, normal T expressed and secreted (RANTES), tumor necrosis factor (TNF)-α, vascular endothelial growth factor (VEGF) using the Human Cytokine/Chemokine multiplex Luminex immunoassays (MilliporeSigma, Burlington, MA). The concentration of tissue inhibitor of metalloproteinases (TIMP)-1, TIMP-2, TIMP-3, and TIMP-4 was determined using the Human TIMP multiplex Luminex immunoassay bead panel 2 (MilliporeSigma, Burlington, MA). The urine creatinine concentration was measured with the creatinine colorimetric assay (Cayman Chemical Company; Ann Arbor, MI), and was used to standardize the urinary concentrations obtained for the other assays.
Statistical Analysis
Biomarker data were summarized, and non-normality was determined using Shapiro-Wilk normality tests. The data were not normally distributed, so a Mann-Whitney U test was performed to determine significant differences in the serum and urine biomarker concentrations between patients with diagnosed DDH and their healthy controls. Significance was determined by a two-sided P < 0.05 and Bonferroni corrections for multiple comparisons were made following the pairwise comparison.
Statistically significant different biomarkers with the largest median differences were considered for the biomarker panels. Utilizing a logistic regression model panels were first created within biomarker types (e.g., inflammatory/anabolism, bone-related, degradative) for the top four biomarkers for each fluid type and a combined fluid panel with two biomarkers from each fluid type. The biomarkers with the lowest p-values for each type of biomarkers were selected and utilized in a logistic regression model with DDH as the outcome of interest (e.g., general linear model using the log link function). A Hosmer-Lemeshow test was used to determine model overfitting, with p<0.05 indicating overfitting. Logistic regression models were used for ROC curve analyses and the area under the curve (AUC) obtained. The optimal threshold for each ROC curve was calculated to maximize, while equally weighting, both sensitivity and specificity using a previously described process. 15 All analyses were performed in R version 4.1.2 (R Core Team, 2021), figures were produced using the ggplot2 package (v3.3.5; Wickham H, 2016), and ROC analysis using pROC (v1.18.0; Robin X, 2021).
Results
The DDH cohort had an average age of 22.8 and a median age of 21 ranging from 15 to 31 years of age. The healthy-hip controls had an average age of 23.8 and a median age of 23 ranging from 15 to 33 years of age. The DDH cohort had an average body mass index (BMI) of 25.6 with a median BMI of 25 ranging from 19 to 35. The healthy-hip controls had an average BMI of 25 with a median BMI of 24.4 ranging from 20 to 37. There was no significant difference between group age or BMI ( Table 1 ).
Average, Median, and Range of Patient Group’s Age and BMI.
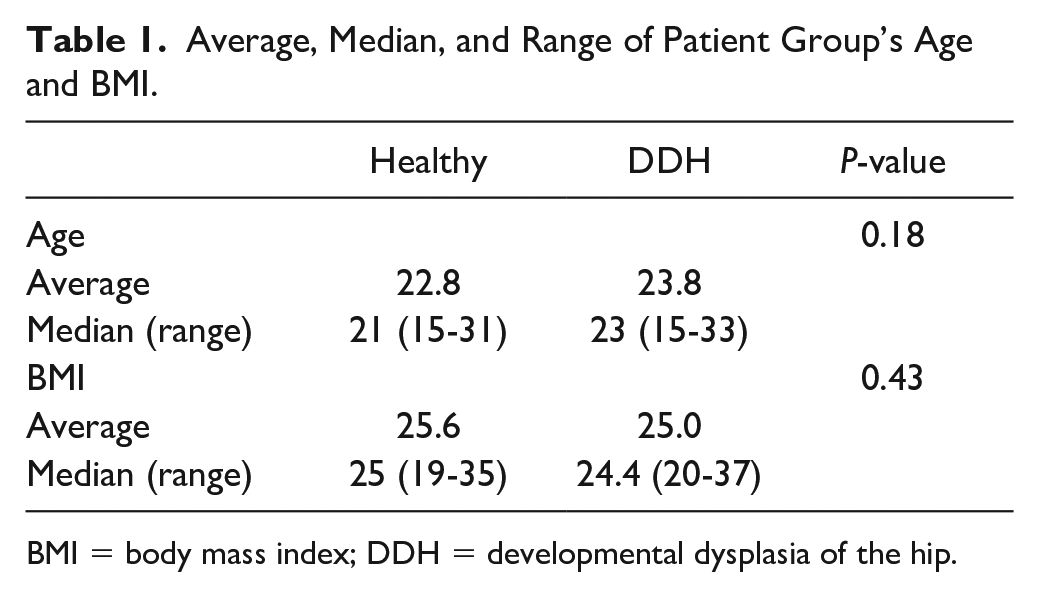
BMI = body mass index; DDH = developmental dysplasia of the hip.
Differences in Protein Biomarker Concentrations Related to Hip Status
In the urine (
Table 2
), DDH patients had significantly increased median concentrations of PIICP (P = 0.037) (
Fig. 1A
), FGF-2 (P = 0.03) (
Fig. 1B
), IL-10 (P = 0.037) (
Fig. 1C
), IL-1RA (P = 0.002) (
Fig. 1D
), and MIP-1
Median Urine Biomarker Concentrations and Interquartile Ranges. P-Values Are From a Mann-Whitney U Test With Bonferroni Correction.
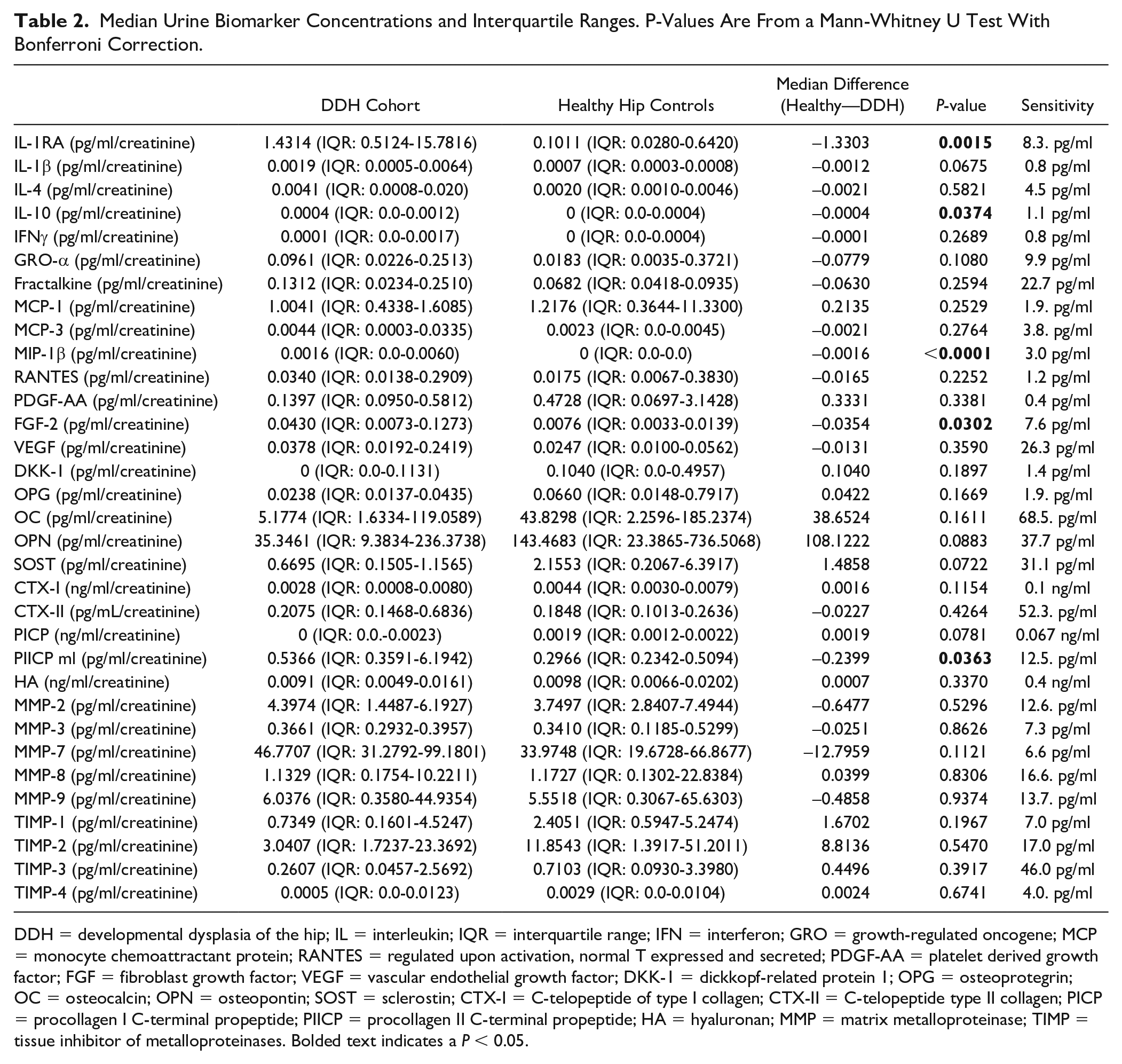
DDH = developmental dysplasia of the hip; IL = interleukin; IQR = interquartile range; IFN = interferon; GRO = growth-regulated oncogene; MCP = monocyte chemoattractant protein; RANTES = regulated upon activation, normal T expressed and secreted; PDGF-AA = platelet derived growth factor; FGF = fibroblast growth factor; VEGF = vascular endothelial growth factor; DKK-1 = dickkopf-related protein 1; OPG = osteoprotegrin; OC = osteocalcin; OPN = osteopontin; SOST = sclerostin; CTX-I = C-telopeptide of type I collagen; CTX-II = C-telopeptide type II collagen; PICP = procollagen I C-terminal propeptide; PIICP = procollagen II C-terminal propeptide; HA = hyaluronan; MMP = matrix metalloproteinase; TIMP = tissue inhibitor of metalloproteinases. Bolded text indicates a P < 0.05.
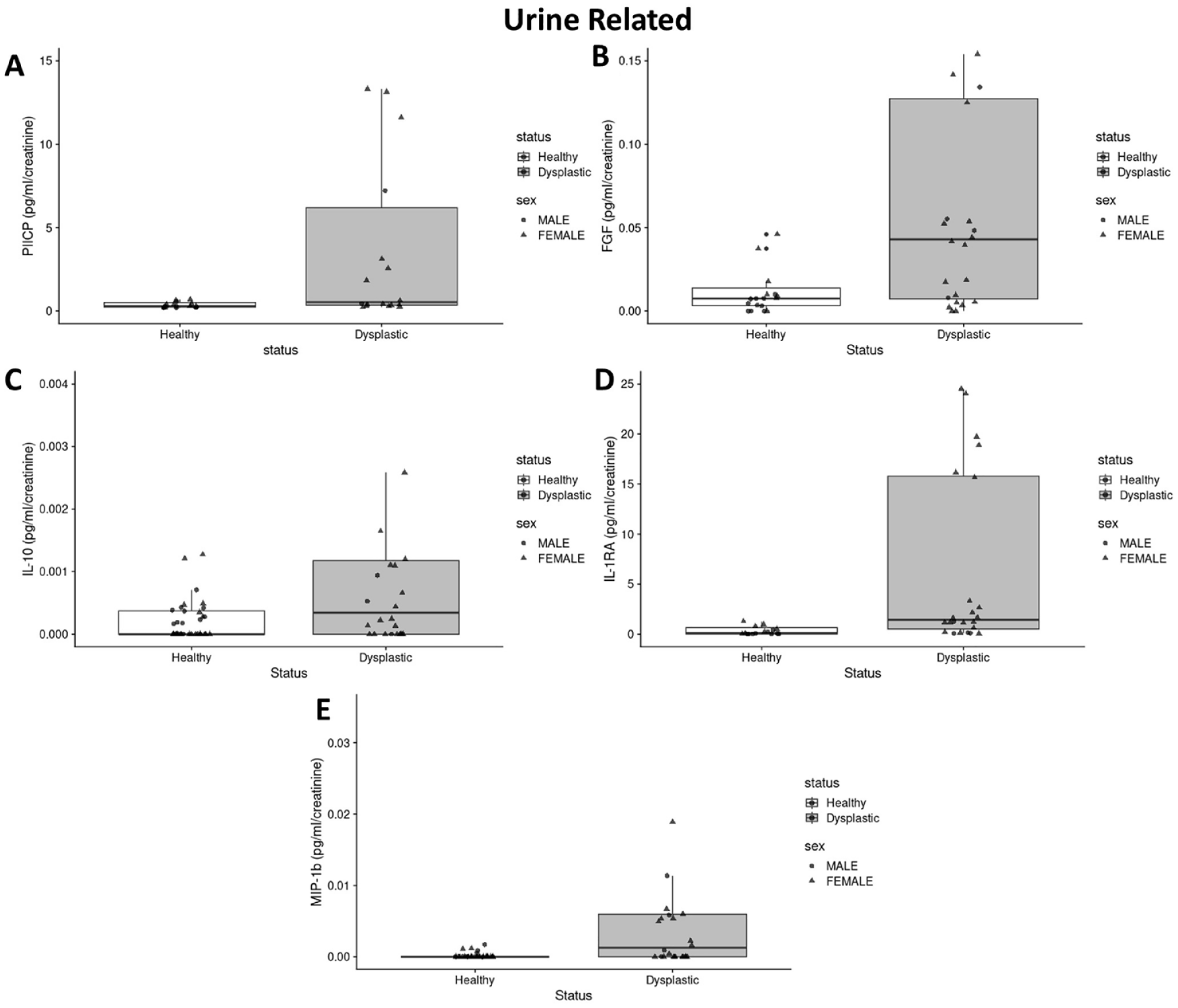
Boxplots of significantly different median urine biomarker concentrations between the DDH cohort and healthy controls (P < 0.05). Median and the interquartile range are denoted by the middle and end points, respectively, of the “box” in the boxplot. (A) PIICP, (B) FGF-2, (C) IL-10, (D) IL-1RA, and (E) MIP-1ß.
In the serum (
Table 3
), DDH patients had significantly decreased median concentrations of DKK-1 (P = 0.003) (
Fig. 2A
), OC (P < 0.001) (
Fig. 2B
), SOST (P < 0.001) (
Fig. 2C
), MMP-8 (P = 0.003) (
Fig. 2D
), TIMP-3 (P = 0.02) (
Fig. 2E
), TIMP-4 (P = 0.049) (
Fig. 2F
), MCP-1 (P < 0.001) (
Fig. 2G
), RANTES (P < 0.001) (
Fig. 2H
), GRO-
Median Serum Biomarker Concentrations and Interquartile Ranges. P-Values Are From a Mann-Whitney U Test With Bonferroni Correction.
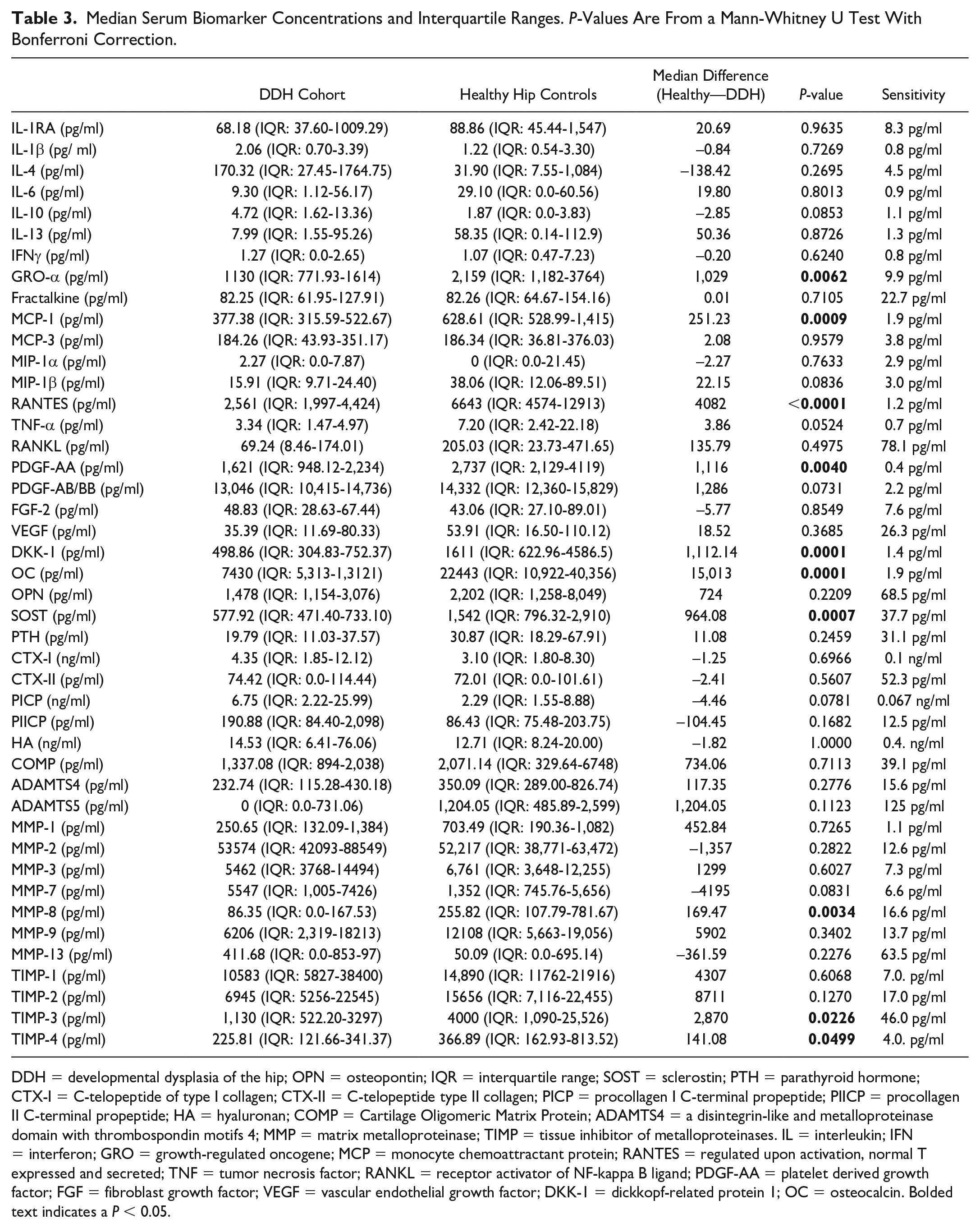
DDH = developmental dysplasia of the hip; OPN = osteopontin; IQR = interquartile range; SOST = sclerostin; PTH = parathyroid hormone; CTX-I = C-telopeptide of type I collagen; CTX-II = C-telopeptide type II collagen; PICP = procollagen I C-terminal propeptide; PIICP = procollagen II C-terminal propeptide; HA = hyaluronan; COMP = Cartilage Oligomeric Matrix Protein; ADAMTS4 = a disintegrin-like and metalloproteinase domain with thrombospondin motifs 4; MMP = matrix metalloproteinase; TIMP = tissue inhibitor of metalloproteinases. IL = interleukin; IFN = interferon; GRO = growth-regulated oncogene; MCP = monocyte chemoattractant protein; RANTES = regulated upon activation, normal T expressed and secreted; TNF = tumor necrosis factor; RANKL = receptor activator of NF-kappa B ligand; PDGF-AA = platelet derived growth factor; FGF = fibroblast growth factor; VEGF = vascular endothelial growth factor; DKK-1 = dickkopf-related protein 1; OC = osteocalcin. Bolded text indicates a P < 0.05.
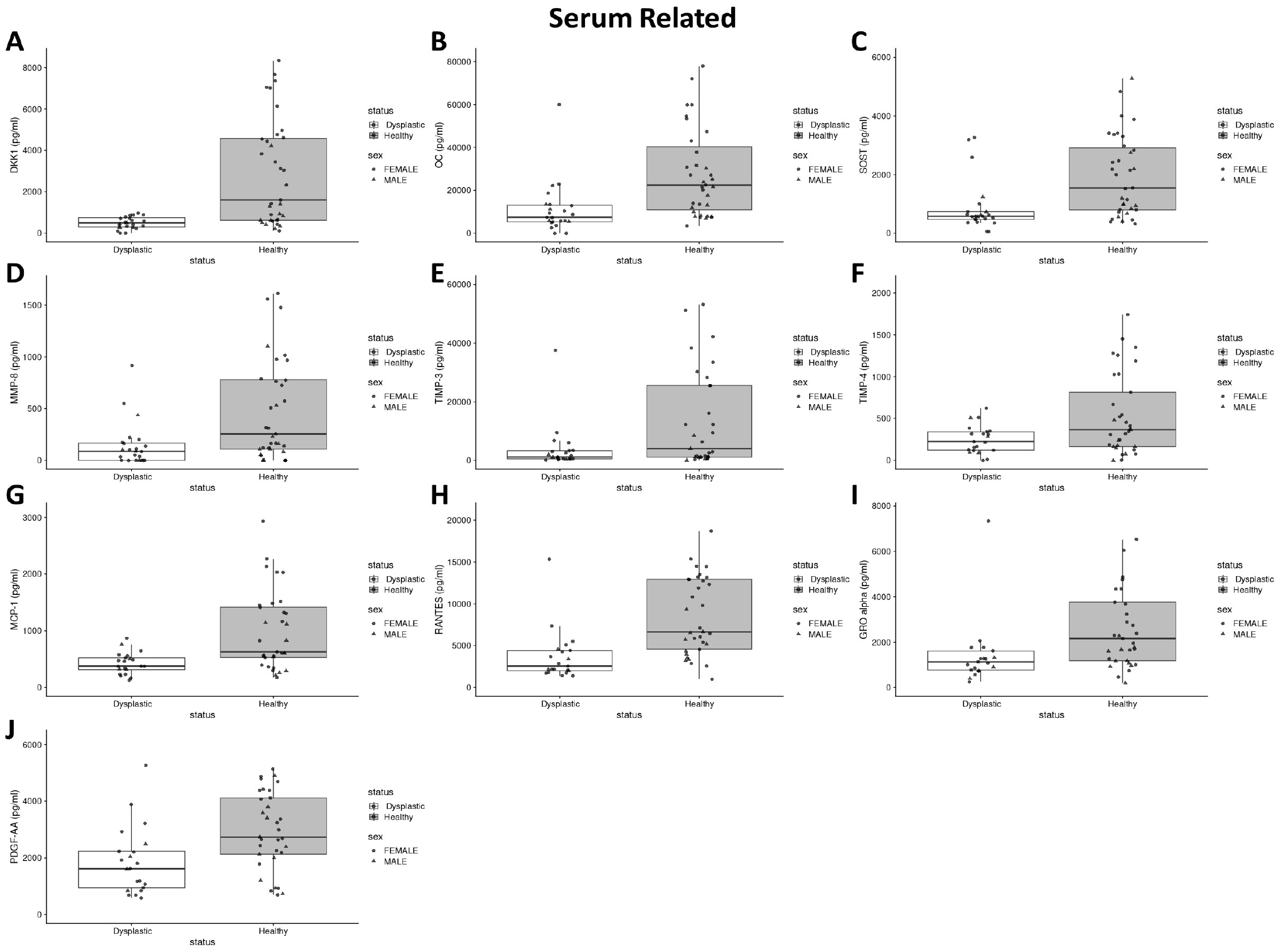
Boxplots of significantly different median serum biomarker concentrations between the DDH cohort and healthy controls (P < 0.05). Median and the interquartile range are denoted by the middle and end points, respectively, of the “box” in the boxplot. (A) DKK-1, (B) OC, (C) SOST, (D) MMP-8, (E) TIMP-3, (F) TIMP-4, (G) MCP-1, (H) RANTES, (I) GRO-α, and (J) PDGF-AA.
All other biomarkers measured in the serum and urine were not significantly different between DDH patients and healthy controls.
Diagnostic Biomarker Panels
The following panels are listed with the biomarkers contained within them, their AUC, H-L value, sensitivity, and specificity (
Table 4
). A urinary inflammatory/anabolism-related biomarker panel including IL-10, IL-1RA, and MIP-1
Biomarker Panels With Their Respective Biomarker Makeup, Area Under the Curve (AUC), Hosmer-Lemeshow Goodness of Fit (H-L), and Threshold for Specificity and Sensitivity.
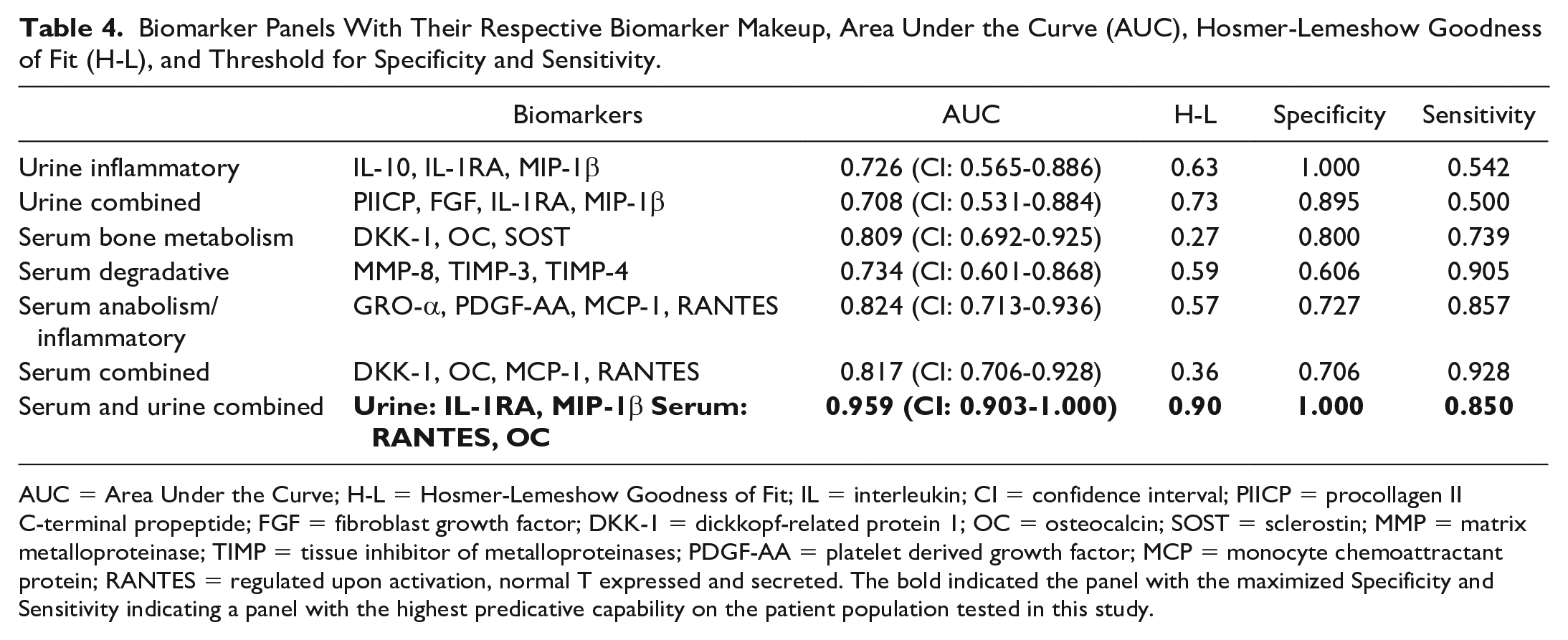
AUC = Area Under the Curve; H-L = Hosmer-Lemeshow Goodness of Fit; IL = interleukin; CI = confidence interval; PIICP = procollagen II C-terminal propeptide; FGF = fibroblast growth factor; DKK-1 = dickkopf-related protein 1; OC = osteocalcin; SOST = sclerostin; MMP = matrix metalloproteinase; TIMP = tissue inhibitor of metalloproteinases; PDGF-AA = platelet derived growth factor; MCP = monocyte chemoattractant protein; RANTES = regulated upon activation, normal T expressed and secreted. The bold indicated the panel with the maximized Specificity and Sensitivity indicating a panel with the highest predicative capability on the patient population tested in this study.
A serum bone-related biomarker panel including DKK-1, OC, and SOST had an AUC of 0.809 (CI: 0.692-0.925) with no evidence of overfitting and a specificity of 0.800 and sensitivity of 0.739. A serum degradative-related biomarker panel including MMP-8, TIMP-3, TIMP-4 had an AUC of 0.734 (CI: 0.601-0.686) with an no evidence of overfitting and a specificity of 0.606 and sensitivity of 0.905. A serum inflammatory/anabolism-related biomarker panel including GRO-
A panel including the two urine (IL-1RA and MIP-1
Discussion
The results from this study suggest that one or more bodily fluid biomarker panels can effectively differentiate patients with DDH from healthy-hip controls in a population at risk for developing secondary hip OA. Using ROC curve analyses, multiple serum and/or urine biomarker panels were able to provide good to excellent discrimination between young (13-34 years old) individuals with self-reported healthy hips and those with DDH. A panel comprised of the two urine biomarkers (IL-1RA, MIP-1β) and the two serum biomarkers (RANTES, OC) that had the strongest statistically significant differences in concentrations between cohorts provided the most robust discrimination of hip status (sensitivity of 0.85, specificity of 1). Based on relative ease, invasiveness, and feasibility for routine sample collection, in conjunction with the rapid method of analysis, this four-biomarker panel could be readily incorporated into clinical practice as a screening tool for detection of DDH prior to the onset of secondary hip OA in this at-risk patient population. However, prospective longitudinal studies assessing a larger and more diverse population of patients are required to validate this biomarker panel for clinical application. 16
In the present study, DDH patients had increased urinary concentrations of biomarkers related to type II collagen production (PIICP), catabolism (FGF-2), and inflammation (IL-1RA, IL-10, and MIP-1β) when compared to healthy-hip controls. Higher levels of IL-1RA and IL-10 in the urine of dysplastic patients suggest attempted regulation of inflammatory processes to counteract progression to secondary hip OA as both of these interleukins have been reported to mitigate inflammatory pathways that lead to OA.17 -20 MIP-1β, a macrophage recruiting protein, has been reported to be produced by articular chondrocytes in response to stimulation with IL-1β and correlated with severity of knee OA.21 -23 While defined as a growth factor, FGF-2 has been reported to induce catabolic effects on human articular cartilage and, in the setting of DDH, may be involved in dysplastic and remodeling processes that occur in the affected hip prior to the development of secondary OA.24,25 In conjunction with these inflammatory and catabolic processes, articular chondrocytes often upregulate collagen production for tissue repair and remodeling, explaining the increases in PIICP in this population of DDH patients.26,27
Young adult DDH patients had decreased serum concentrations of biomarkers related to bone metabolism (DKK-1, OC, and SOST), degradation (MMP-8, TIMP-3, and TIMP-4), inflammation (GRO-α, MCP-1, and RANTES), and anabolism (PDGF-AA) when compared to healthy-hip controls. Lower levels of the bone metabolism biomarkers, DKK-1, OC, and SOST, may be indicative of altered bone development in dysplastic hips related to abnormal remodeling and distorted joint loading.28 -34 Similarly, PDGF-AA can promote bone formation and healing by guiding osteogenic differentiation of mesenchymal stem cells, such that the lower serum concentrations in DDH patients may be related to aberrant bone development and remodeling.35 -37 The lower serum concentrations of degradation-related biomarkers, MMP-8, TIMP-3, and TIMP-4, in DDH patients suggest an imbalance in regulation of articular tissue homeostasis in dysplastic hips.38 -40 Similarly, the lower serum concentrations of inflammation-related biomarkers, GRO-α, MCP-1, and RANTES, suggest a dysregulation of inflammatory pathways during this phase of DDH.41 -44
Taken together, urine and serum protein biomarker differences suggest that pre-OA DDH hips are mechanistically distinct from healthy hips in terms of inflammatory and remodeling processes that typically lead to secondary OA if left untreated. When considering these mechanistic differences in regard to developing effective and clinically applicable diagnostic biomarker panels, ROC curve analyses were used to consider both sensitivity and specificity in determining the fewest number of biomarkers to include in a panel that maintained high discriminatory capabilities. For application to DDH, sensitivity was prioritized to ensure a low false negative rate, intended to reduce misdiagnosis while capturing all potential DDH cases to allow for further assessment and monitoring. As such, a panel of 4 serum biomarkers, DKK-1, OC, MCP-1, and RANTES, with sensitivity of 0.93 and specificity of 0.71 and a panel of 2 urine, IL-1RA and MIP-1
There are several limitations to this study that should be considered when interpreting and applying the results. The number of subjects in each cohort was relatively small. In addition, patient cohorts were considered only based on age range (13–34 years), such that potentially confounding factors including comorbidities, body mass index, medications, and others were not controlled for with respect to patient inclusion or analyses. However, this experimental design was intentional such that the translational application of biomarker panels to “real life” patient populations was legitimate. In addition, the study involved a cross-sectional analysis of individuals such that the biomarker panels cannot be considered to have prognostic implications. Furthermore, the panels developed in this study were not tested against an independent data set, and the study only included patients with clinically diagnosed DDH. Therefore, it is not known if the developed panels will have the same sensitivity and specificity characteristics when assessed on an independent patient population, or in a patient population where DDH is undiagnosed and asymptomatic. Future studies should include longitudinal sampling in a larger population of participants over a broad demographic spectrum with correlation of biomarker data to clinical, diagnostic, and functional measures of disease severity and progression. In addition, other proteins reported to be upregulated during the developmental stages of DDH, including HtrA1 and vimentin, should be evaluated for inclusion in an optimized biomarker panel. 45
The results of the present study expand the evidence for serum and urine protein biomarkers to characterize mechanisms of disease that define DDH prior to progression to secondary hip osteoarthritis and indicate that panels of protein biomarkers measured in the urine and serum may be able to differentiate young individuals with DDH from those with healthy hips. Of the panels developed and assessed, a panel comprised of two serum and two urinary biomarkers provided the highest combined values for sensitivity and specificity, while a panel of four serum biomarkers provided the highest sensitivity while maintaining adequate specificity. Taken together, these data suggest that there is potential for clinical application of a routine diagnostic method for cost-effective and timely screening for DDH in this at-risk population. Further development and validation of these biomarker panels may result in highly sensitive and specific tools for early diagnosis, staging, and prognostication of DDH, as well as treatment decision making and monitoring capabilities, filling a critical unmet need in healthcare.
Footnotes
Authors’ Note
Portions of these data were presented at the annual conference of the Orthopedic Research Society Annual Meeting, February 8 to 11, 2020, Phoenix, Arizona, submitted to present at the Orthopedic Research Society Annual Meeting, February 4 to 8, 2022, Tampa, Florida.
Author Contributions
All authors were instrumental in the study design, acquisition, and analysis of study data, manuscript drafting and final approval of all manuscript-related items.
Acknowledgment and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Preston N. Wolfe is a full-time employee with Arthrex, Inc. • Brett D. Crist is a board or committee member for AO Trauma North America: Board or committee member; receives IP royalties from Globus Medical; is a board or committee member for International Geriatric Fracture Society; is on the editorial or governing board of the Journal of Hip Preservation and Journal of Orthopedic Trauma; is a paid presenter or speaker for KCI; has stock or stock options for Orthopedic Implant Company; is a board or committee member for Orthopedic Trauma Association; is an unpaid consultant for Osteocentric; has stock or stock options for RomTech; is on the editorial or governing board for SLACK Incorporated; receives publishing royalties, financial or material support from Springer; is a paid consultant and receives research support from Synthes. • Aaron M. Stoker receives IP royalties from Musculoskeletal Transplant Foundation • Emily Leary is on the editorial or governing board for JISAKOS and The Journal of Knee Surgery • James L. Cook is a paid consultant, paid presenter or speaker and receives research support from Arthrex, Inc; is a paid consultant for Bioventus; receives research support from the Coulter Foundation; receives research support from DePuy, A Johnson & Johnson company; receives research support from GE Healthcare; is on the editorial or governing board of the Journal of Knee Surgery; receives research support from Merial; is a board or committee member for Midwest Transplant Network; is a board or committee member, receives IP royalties and research support from Musculoskeletal Transplant Foundation; receives research support from National Institutes of Health (NIAMS and NICHD); receives research support from Purina; receives publishing royalties, financial or material support from Thieme; is a paid consultant for Trupanion; and receives research support from the U.S. Department of Defense. Chantelle Bozynski has no conflict of interest(s) to disclose.
Ethical Approval
This study was approved by the University of Missouri Institutional Review Board (IRB #2012192) and informed consent was documented for all patients involved.
