Abstract
Objective
The identification of early-stage osteoarthritis (OA) is crucial for the deceleration of its progression; however, no reliable biomarker is available for this purpose. The current study aimed to determine the role of serum calprotectin in the detection of early-stage knee OA.
Design
In a case-control study, serum samples were collected from 84 patients with primary bilateral knee OA and 52 healthy controls. The radiographic grading of knee OA was performed using the Kellgren-Lawrence classification system. Serum concentrations of calprotectin were measured using an enzyme-linked immunosorbent assay.
Results
The mean serum calprotectin level was 2908 ± 2516 ng/mL in OA patients and 901 ± 875 ng/mL in healthy control subjects (P < 0.001). Mean serum calprotectin levels were significantly higher in the lower stages of OA: 3740 ± 2728 ng/mL in OA grade I, 3100 ± 2084 ng/mL in OA grade II, 2246 ± 1418 ng/mL in OA grade III, and 2035 ± 765 ng/mL in OA grade IV (P = 0.047). Serum calprotectin levels were significantly higher in patients with a disease duration <42 months compared with those with a disease duration >42 months (P = 0.043).
Conclusion
Serum calprotectin level increases significantly in the early stages of OA and shows a reverse association with disease severity. Therefore, it could be suggested as a promising blood-based marker for early-stage knee OA.
Introduction
Osteoarthritis (OA) is one of the leading causes of chronic pain and disability in the aged population and has significant economic and social impacts on health systems.1-3 The rate of OA increases with the advancement of the population age and with obesity; it is expected to grow 20% by 2030, meaning that the economic and health impact of OA will increase by 20% as well. 4 Therefore, OA prevention is a significant health priority.
Knee OA is estimated to account for more than 80% of the disease’s total burden. 5 While surgical intervention is the treatment of choice for end-stage OA, 6 early diagnosis will permit earlier nonsurgical interventions to modify the course of the disease, leading to reduced patient morbidity and lower associated costs. 7
Validated biomarkers capable of reflecting the dynamic and quantitative changes in joint remodeling and disease progression are of considerable importance in reducing the adverse impact of OA on global health. 8 Such biomarkers are also crucial for the early diagnosis and deceleration of OA progression through nonpharmacological interventions, including patient education and weight loss. However, to date, no single sufficiently validated biomarker has been recognized for systematic use in the early detection of OA. 9
Alarmins, or danger signals, are endogenous molecules that play important roles as initiators of the immune response. 10 Calprotectin, a member of the S100 protein family, is also an alarmin protein released from activated granulocytes and macrophages in a diverse range of physiological and pathophysiological processes. 11
Since alarmins, as their name implies, are released in the early stages of inflammation, I was hypothesized that they could be candidate markers for the identification of OA, particularly in its early stages. This study aimed to evaluate the role of serum calprotectin in the differentiation of OA patients from healthy controls and to assess its association with disease burden. To the best of our knowledge, no study to date has assessed the role of serum calprotectin as a biomarker of OA.
Patients and Methods
This study was approved by the ethics committee of our institute under the code of IR.IUMS.REC.1397.173, and participants provided written consent at the study entry. In a case-control study, serum samples were collected from patients who were referred to the knee clinic of our center and met the American College of Rheumatology classification criteria for knee OA. 12 Inclusion criteria were bilateral knee primary OA and the absence of evident OA in other joints such as the hip. Patients with conditions affecting the serum calprotectin level, including other inflammatory disorders, type 2 diabetes mellitus, cardiovascular diseases, metabolic syndrome, infections, and cancers, were excluded from the study. Patients who were taking anti-inflammatory medications to relieve OA pain were also excluded from the study. 13
From a total of 161 patients, 35 patients who had concurrent disorders and 42 patients who were taking anti-inflammatory medications were excluded. The remaining patients were included in the final study (n = 84). Radiographic grading of knee OA was performed by the Kellgren-Lawrence 5-grade classification system, in which grade 0 represents the absence of radiographic features of knee OA and grade 4 reveals the presence of the most severe form of knee OA. 14
The OA patients included 22 (26.1%) males and 62 (73.9%) females with a mean age of 49.3 ± 10.6 years. Kellgren-Lawrence grades I, II, III, and IV were identified in 32 (38.1%), 28 (33.3%), 13 (15.5%), and 11 (13.1%) patients, respectively. The mean disease duration was 43 ± 51 months.The demographic, clinical, and radiologic characteristics of the patients are presented in Table 1 .
Clinical and Demographic Characteristics of the Patients with Knee OA. a
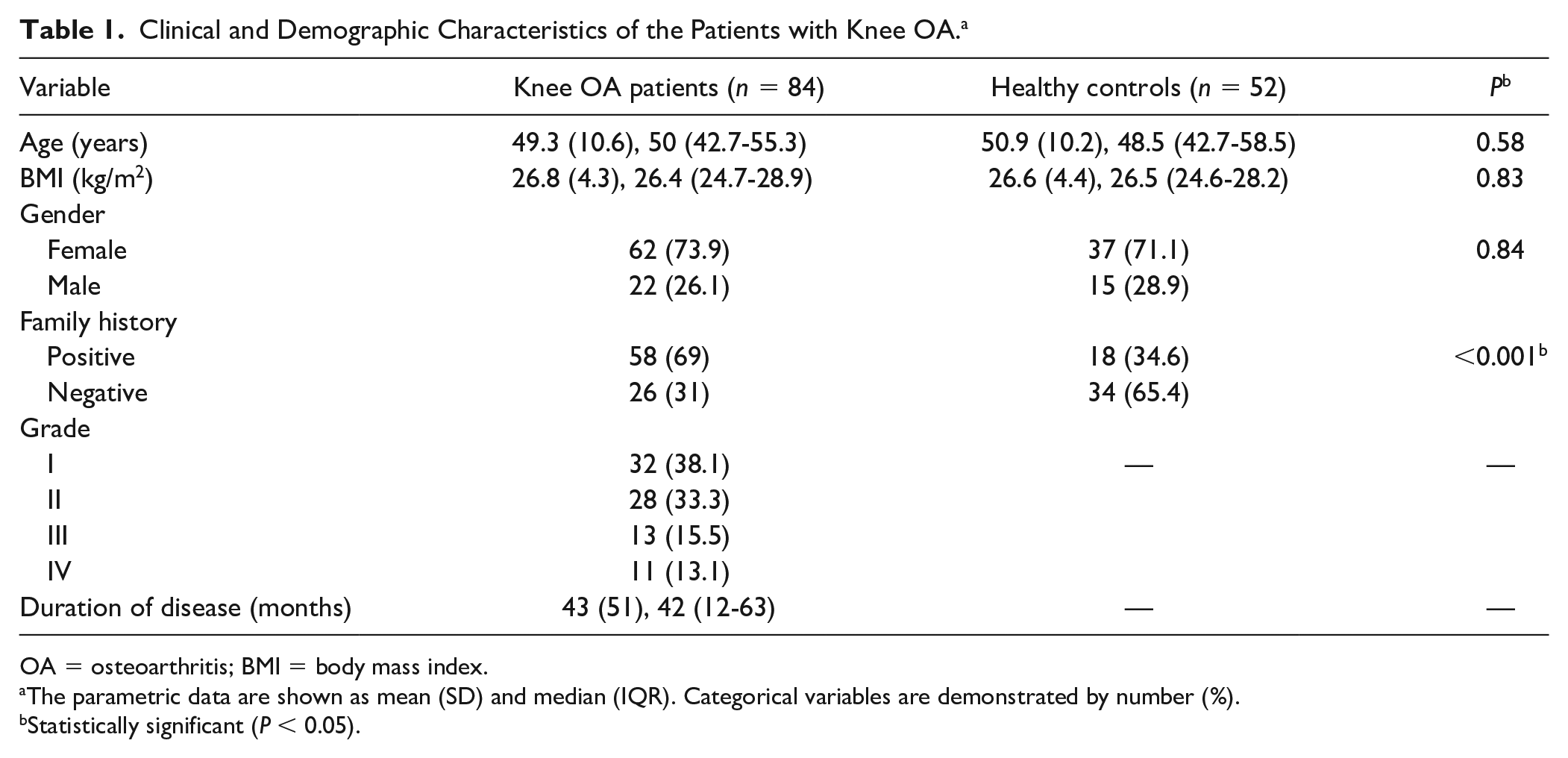
OA = osteoarthritis; BMI = body mass index.
The parametric data are shown as mean (SD) and median (IQR). Categorical variables are demonstrated by number (%).
Statistically significant (P < 0.05).
A total of 52 healthy controls that were group-matched for age, sex, and body mass index (BMI) were also enrolled in the study. The healthy control subjects were recruited from individuals who referred to the laboratory department of our center for a routine check-up. They were included if they had no underlying disorders, were taking no medications, and their inflammatory markers (including erythrocyte sedimentation rate, white blood cell count, and C-reactive protein) were in the normal range. To reduce the heterogeneity between the cases and control samples, all samples were taken while the subjects were fasting. The demographic characteristics of the control group are demonstrated in Table 1 .
Serum Calprotectin Analysis
Serum concentrations of calprotectin were measured using a commercial enzyme-linked immunosorbent assay (ELISA) kit (RD191217100R, BioVendor, Karasek, Czech Republic) and according to the manufacturer’s protocol. The detection method was based on a sandwich enzyme immunoassay and horse radish peroxidase–labeled antibody. Briefly, 100 µL of analytes were pipetted into the appropriate number of wells. The plate was then incubated at room temperature for 1 hour, while shaking in an orbital microplate shaker (300 rpm). After washing 5 times with washing solution, 100 µL of the conjugate solution was added to each well and incubated at room temperature for another 1 hour (shaking in an orbital microplate shaker, 300 rpm). After washing 5 more times with washing solution, 100 µL of substrate solution was added to each well and incubated for 10 minutes at room temperature, while the plate was covered with aluminum foil to avoid direct sunlight. Then 100 µL of stop solution was added, and absorbance was determined within 5 minutes using a microplate reader set to 450 nm (Hiperion MPR4++, Rödermark, Germany).
The assay range was 1 to 64 ng/mL. The intra-assay (within-run) coefficient of variation (CV) was 5%. Inter-assay (run-to-run) CV was 4.2%. The assay limit of detection was 0.22 ng/mL. The accuracy of measurement for spiked serum samples was 100.7%. The dilution linearity for serum samples was 96.8%.
Serum samples were kept at −20 °C until the date of examination. Just prior to the assay, samples were diluted 200× with dilution buffer. Each assay included 2 quality controls (High and Low), a negative control (Blank), and a standard curve with 7 concentrations. Serum samples were analyzed in duplicate. Sample measurement was regarded acceptable providing that the variation of the duplicates was <10%.
Statistical Analysis
SPSS for Windows version 16 (IBM, Armonk, NY) was used for the statistical analysis of the results. Descriptive information for parametric variables was demonstrated by mean, median, standard deviation, and interquartile range. Descriptive data for categorical variables was presented as number and percentage. Independent T test was used for the comparison of means, where appropriate. The 1-way analysis of variance test was used for the comparison of mean serum calprotectin levels at different stages of OA. Pearson’s or Spearman’s correlation coefficient test was used to evaluate potential correlations. The median split approach was used to categorize quantitative variables. The power of serum calprotectin in the differentiation of OA patients from healthy controls was estimated by depicting the receiver operating characteristic (ROC) curve, calculating the area under the curve (AUC), and computing the maximum point of the Youden index as the optimal cutoff value. A P value <0.05 was regarded as statistically significant.
Results
Serum calprotectin levels were evaluated in 84 OA patients and 52 healthy control subjects. The mean age, mean BMI, and sex distribution were not significantly different between the patients and healthy control subjects ( Table 1 ). The number of positive OA family histories was significantly higher in the OA group (P < 0.001).
The mean serum calprotectin level was 2908 ± 2516 ng/mL in OA patients and 901 ± 875 ng/mL in healthy control subjects ( Fig. 1a ). This difference was statistically significant (P < 0.001). After depicting the ROC curve, an AUC of 0.861 (95% CI = 0.793-0.929, P < 0.001) was obtained for serum calprotectin level in the differentiation of OA patients from healthy controls. The sensitivity and specificity of this differentiation at the cutoff value of 1270 ng/mL were 80% and 83.3%, respectively ( Fig. 2 ).
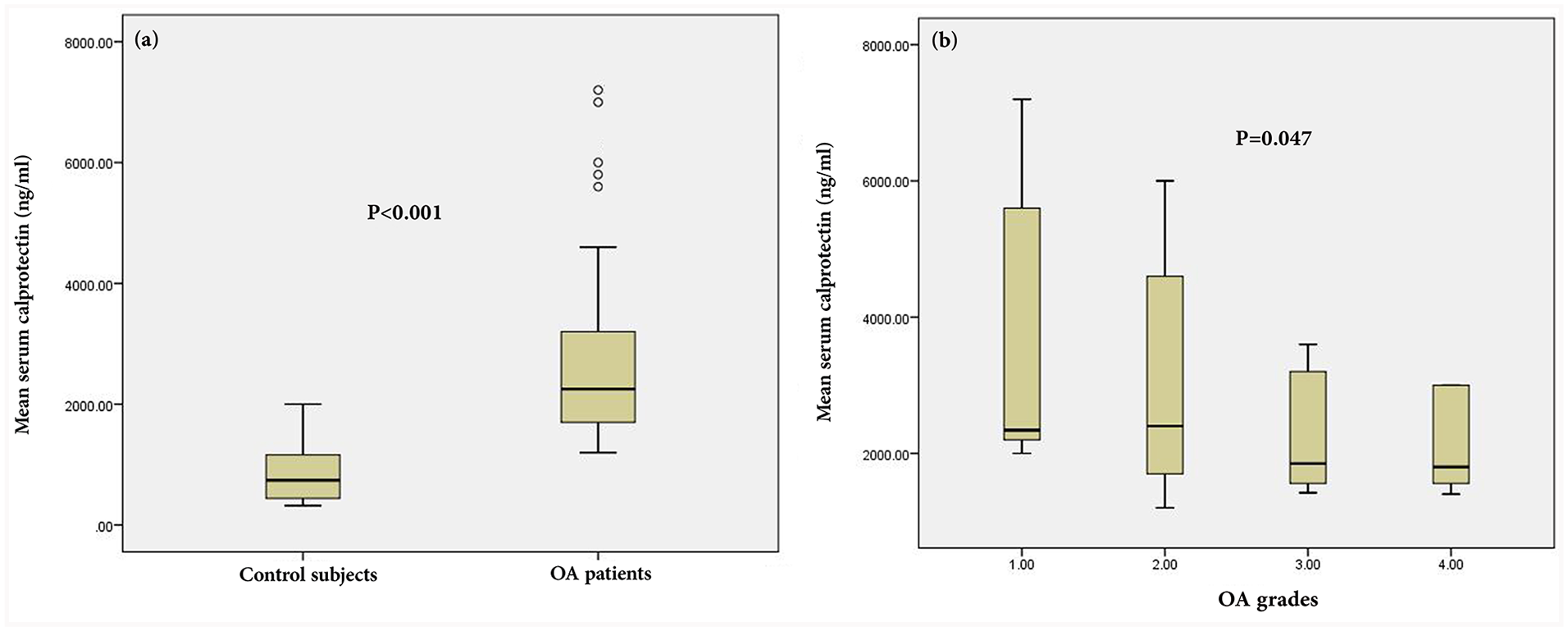
(
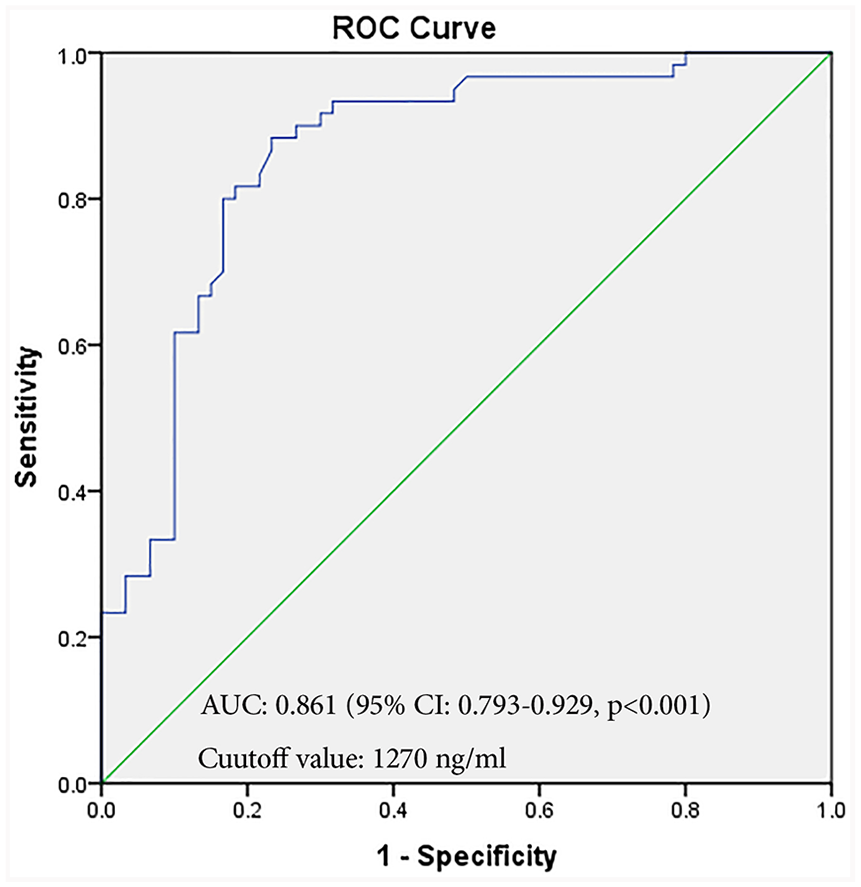
ROC curve showing the capability of serum calprotectin in discriminating osteoarthritis patients from healthy control subjects. ROC, receiver operating characteristic; AUC, area under the curve.
The mean serum calprotectin level was 3740 ± 2728 ng/mL in OA grade I, 3100 ± 2084 ng/mL in OA grade II, 2246 ± 1418 ng/mL in OA grade III, and 2035 ± 765 ng/mL in OA grade IV ( Fig. 1b ). Accordingly, the mean serum calprotectin level was significantly higher in OA grade I and significantly lower in OA grade IV (P = 0.047). Serum calprotectin levels were significantly higher in patients with a disease duration <42 months in comparison with those with a disease duration >42 months (P = 0.043). No significant association was found between the serum calprotectin level and the other clinical and demographic characteristics of the patients, such as age, gender, BMI, and history of OA ( Table 2 ).
Mean Serum Calprotectin Level in Different Subgroups of Patients with Knee OA. a
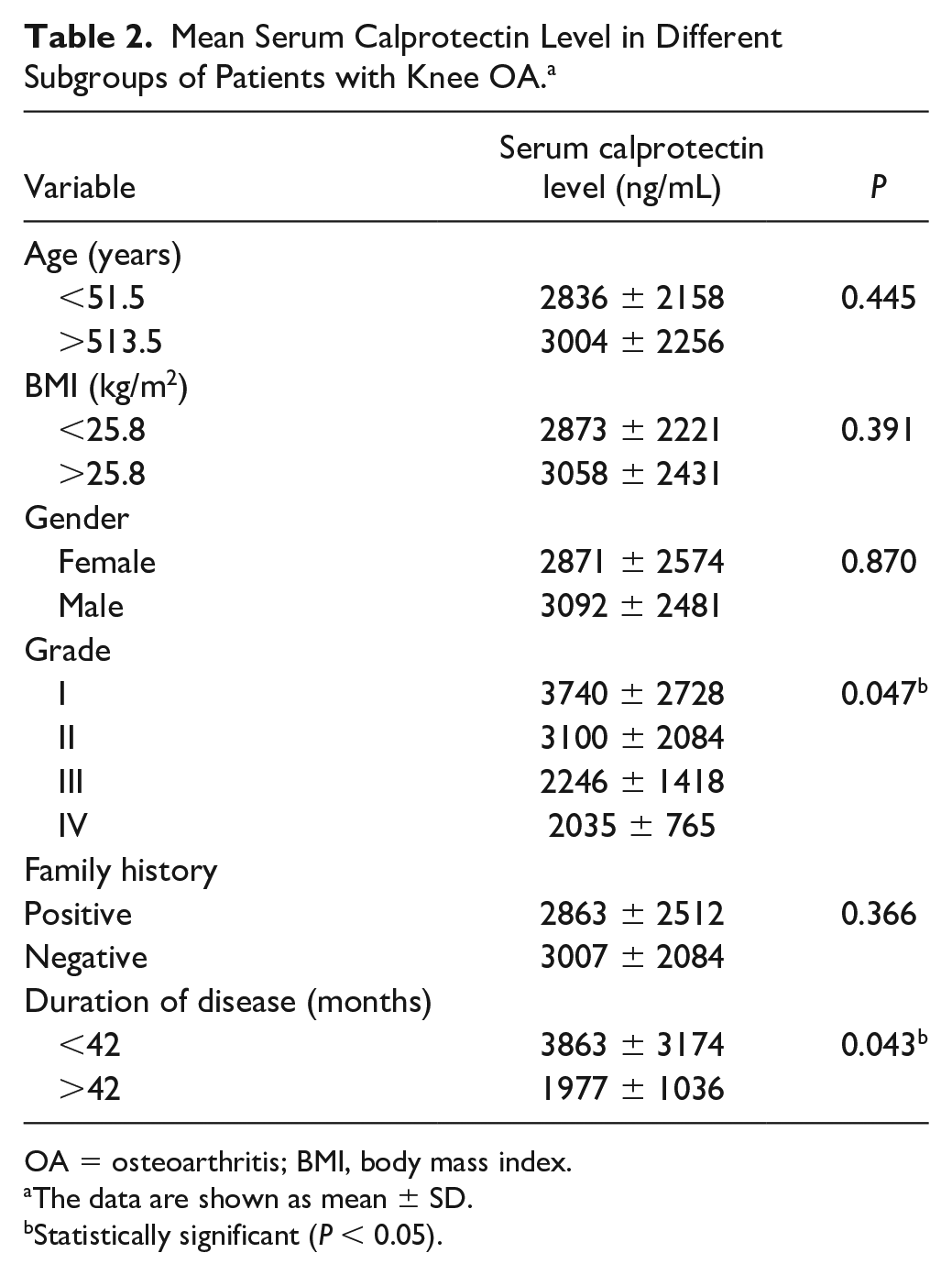
OA = osteoarthritis; BMI, body mass index.
The data are shown as mean ± SD.
Statistically significant (P < 0.05).
Serum calprotectin level was not significantly correlated with the age or the BMI of the participants; however, a significant negative correlation was found between serum calprotectin level and disease duration (r = −0.289, P = 0.009).
Discussion
The onset of OA comes long before its clinical presentation. However, radiologic evaluations are not sensitive enough to detect OA in the early stages. As the early diagnosis of OA may prevent and reverse OA development, the development of biomarkers capable of detecting OA in the early stages is of critical value. 15
The current study aimed to identify the role of an alarmin protein, namely calprotectin, as a blood-based biomarker for the detection of early-stage OA. The results showed that serum calprotectin levels were significantly higher in OA patients than in healthy control subjects. In addition, serum calprotectin levels were higher in the lower stages of OA; its concentration was highest in stage I and lowest in stage IV. Patients with a longer disease duration had a lower serum calprotectin level, which further supports the current findings, as these patients generally present with higher grades of OA.
The role of calprotectin as a biomarker has also been revealed in other diseases. As the most widely studied and applied biomarker in clinical practice, calprotectin is known to be a promising noninvasive biomarker of inflammatory bowel disease (IBD).16-18 Fecal calprotectin is reported to be strongly associated with disease activity indices and endoscopic activity in IBD. 16 Recently, serum calprotectin has also attracted attention as a novel blood-based biomarker of IBD, which correlates with endoscopic findings of the patients.19,20 It has also been suggested that serum calprotectin is associated with disease activity in rheumatoid arthritis.21,22 In contrast with the expression pattern of calprotectin in IBD, which is directly associated with the disease activity indices, the present study found a reverse association between serum calprotectin and OA severity. These findings could be interesting, as biomarkers generally increase with the severity of the disease. The elevation of serum calprotectin level in disease conditions other than OA suggests cautious interpretation of serum calprotectin levels in OA patients mainly through ruling out other potential disorders affecting the serum calprotectin level.
Zreiqat et al. 23 evaluated the changes of calprotectin in cartilage during the induction of OA in a murine model. They observed the increased chondrocyte mRNA expression of calprotectin heterodimers (S100A8 and S100A9) in early but not late mouse OA. Moreover, calprotectin significantly upregulated the expression of matrix metalloproteases (MMPs). They concluded that calprotectin might play a role in initiating cartilage degradation in OA, presumably by upregulating MMPs, while their declined expression in the late stages of OA suggests no ongoing role for calprotectin in cartilage destruction. 24 The current results are in accordance with those of Zreiqat et al., 23 in that the serum concentration of calprotectin declined in the late stages of OA. These results suggest that serum calprotectin levels could be used as markers for the detection of early-stage OA.
Calvet et al. 25 aimed to determine synovial calprotectin levels in 108 symptomatic patients with knee OA and evaluate its association with clinical and ultrasonographic features as well as other pro-inflammatory markers. Higher synovial calprotectin levels were associated with worse pain and function in patients. Patients with moderate to severe synovial effusion had higher synovial calprotectin levels in comparison with those with mild effusion. Synovial calprotectin revealed moderate correlations with intra-articular tumor necrosis factor-α, high-sensitivity C-reactive protein and a high correlation with synovial interleukin-6. The researchers concluded that calprotectin is directly associated with the severity of knee OA. 25 In contrast with the study of Calvet et al., 25 the results of the present study revealed a reverse association between calprotectin and OA severity.
In a progressing OA with gradual loss of the cartilage, the level of different cartilage-related products such as type II collagen will gradually lower, 26 which suggests tracing them as potential biomarkers of early-stage OA. Nevertheless, the majority of cartilage-related products are not released in blood circulation, 27 and therefore cannot be used as noninvasive OA biomarkers. Nonetheless, if a high level of serum calprotectin precedes the radiologic signs of OA by a substantial number of months or years, it could be considered a promising biomarker to prevent the progression of disease in its early stages. However, considering the conflicting results of earlier investigations, further studies are required to shed more light on the role of serum calprotectin as a biomarker of early-stage knee OA.
The present study was not without limitations. As the main limitation of this study, we included patients with a clinical and radiologic presentation of knee OA. However, an evaluation of serum calprotectin in subclinical knee OA would have better served our purpose, as the main goal of early-stage OA identification is preventing its clinical manifestation. In other words, an ideal biomarker should be able to identify early-stage knee OA before its clinical and radiologic manifestations. Therefore, future studies should focus on evaluating serum calprotectin levels in subclinical forms of knee OA. As another limitation, the control subjects of the current study were not checked for radiographic symptoms of OA, as it was not ethical to expose individuals with no clinical symptoms to unnecessary X-ray radiation. Hence, asymptomatic individuals with radiographic evidence of knee OA might have been included in the control group of this study.
Conclusions
Serum calprotectin levels are significantly higher in knee OA patients than in healthy control subjects. In patients with the clinical and radiologic presentation of knee OA, serum calprotectin level reveals a reverse association with the severity of the disease; serum calprotectin level is higher in lower stages of the disease, suggesting that serum calprotectin could be a promising blood-based marker of early-stage knee OA.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:This research was funded by the Iran University of Medical Sciences under the grant code of 33123.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the ethics committee of our institute under the code of IR.IUMS.REC.1397.173.
Informed Consent
All participants provided written consent at the study entry.
Trial Registration
Not applicable.
