Abstract
Objective
This study was conducted to compare therapeutically relevant properties of platelet-rich plasma (PRP), a commonly used autologous intra-articular treatment for osteoarthritis (OA), with those of a novel placental tissue particulate, PTP-001, which is in development as a regulated biologic treatment for knee OA.
Design
Quantitative immunoassays were performed to determine the content of key growth/regulatory biofactors in PTP-001, and in leukocyte-rich (LR)-PRP or leukocyte-poor (LP)-PRP. An anti-inflammatory bioassay was used to evaluate the effects of each treatment on pro-inflammatory cytokine (tumor necrosis factor (TNF)-α) production in a macrophage cell culture system. Gene expression experiments were conducted using a co-culture system of human synoviocytes (pre-stimulated with interleukin (IL)-1β) and articular chondrocytes, with quantitative polymerase chain reaction analyses of the separate cellular compartments.
Results
The concentrations of several biofactors (e.g., basic fibroblast growth factor, tissue inhibitor of metalloproteases-3, interleukin-1 receptor antagonist) representative of diverse disease-relevant mechanisms of action were significantly higher for PTP-001 relative to LR-PRP or LP-PRP. PTP-001 and PRP preparations were able to reduce TNF-α production in macrophage cell cultures; however, greater variability was observed for PRP in comparison with PTP-001. In the chondrocyte/synoviocyte co-culture experiments, PTP-001 and LR-PRP (but not LP-PRP) significantly reduced chondrocyte MMP13 expression in cultures containing IL-1-pretreated synoviocytes. In addition, ADAMTS5 expression was reduced in the chondrocyte compartment following treatment with PTP-001 relative to PRP.
Conclusion
These findings support evidence of a potent, multifactorial mechanism of action for a consistently manufactured biologic (PTP-001), which may be of greater therapeutic benefit in comparison with more heterogeneous preparations of PRP which may be generated at the time of treatment.
Introduction
Effective nonsurgical options for the treatment of osteoarthritis (OA) continue to represent an urgent unmet medical need.1,2 While no verified disease-modifying therapies have yet been identified, clinical treatments intended to manage pain and improve function include injectable therapies such as hyaluronic acid, platelet-rich plasma (PRP), and, more recently, micronized placental tissue allografts. PRP, which is prepared from a patient’s own (autologous) blood, has been the subject of considerable study; however, there remain few well-controlled trials for PRP, and comparative characterization and compositional data for different PRP formulations are sparse.3,4 In particular, it should be noted that methods and materials used for the preparation of PRP, as well as patient variability, can be sources of considerable heterogeneity in the molecular constitution and potential efficacy of PRP. While there are proposed classification schemes specifying platelet concentrations relative to whole blood, there are no generally accepted “standard” criteria defining PRP in terms of growth factor content and numbers of cells (leukocytes, red blood cells, etc.). 3 In contrast, PTP-001 is a novel placental tissue particulate (injectable), which is rigorously characterized using validated, quantitative growth factor and potency assays as required for its development as a biologic for the treatment of pain and impaired function in patients with OA of the knee.
PTP-001 is manufactured as a lyophilized (dry) particulate, which can be resuspended in sterile saline for administration. Our recent data indicate that PTP-001 represents a consistent source of beneficial growth factors, and can display a multifactorial mechanism of action to reduce inflammatory and catabolic processes which can contribute to the progression of OA. 5 In addition, we found that treatment with PTP-001 can significantly decrease pain responses as well as cartilage degeneration in a preclinical rat model of surgically induced OA. 5 Studies which evaluated the effects of PRP in preclinical animal models of OA have recently been the subject of a systematic review. 6 Although some of these studies reported a favorable effect of PRP on disease modification and symptomatic outcomes, the overall risk of bias and low quality of studies were noted, and there were few reports of the effects of leukocytes on PRP performance in the animal models.
With respect to clinical efficacy in human patients, a recent randomized controlled trial comparing PRP with placebo (saline) treatment for mild to moderate OA demonstrated no significant differences in symptoms (pain) or joint structure observed between cohorts after 12 months. 7 Notably, the PRP utilized in that study was classified as being “leukocyte poor” (LP), and there has been considerable debate concerning the relative merits of such LP-PRP versus “leukocyte rich” (LR)-PRP.8,9 Of particular concern are the potential deleterious effects associated with elevated pro-inflammatory cytokine levels in LR-PRP which may contribute to increased tissue damage and a reduced capacity for repair. Interestingly, however, a more recent preclinical study comparing LP-PRP and LR-PRP treatment in a mouse model of surgically induced OA demonstrated reduction of pain responses following injection of LR-PRP (but not LP-PRP). 10 In the current study, we performed quantitative analyses of protein biofactor content, and assessed bioactivity in cell-based assays to compare PTP-001 with LR-PRP and LP-PRP prepared from multiple donors.
Methods
Preparation of PTP-001 and PRP
Individual PTP-001 lots (and eluates thereof) were prepared using gifted placental tissues from consented donors as described. 5 In brief, eluates were prepared by suspending and mixing the lyophilized tissue particulates in PBS, PBS containing 0.2% (w/v) collagenase type I (Thermo Fisher Scientific, Waltham, MA), or serum-free media, followed by centrifugation to recover the supernatant (eluate). The concentration of PTP-001 tested (50 mg dry tissue weight per ml) was chosen to emulate a potential clinical dose. For the PRP preparations, healthy donor blood samples were collected by venipuncture into 15% acid citrate dextrose tubes, and equal volumes of the same sample from individual donors were used to prepare both LR-PRP and LP-PRP using a single-spin or double-spin centrifugation technique, respectively (Zen-Bio, Durham, NC). LR-PRP was also prepared by using a commercially available system (GPS® III; Zimmer Biomet, Warsaw, IN). PRP preparations were activated by incubation for 1 hour at 37 °C with 45 mM CaCl2 and the liquid phase was retained for further experiments.
Protein Biofactor Analysis
PTP-001 eluates and LR-PRP or LP-PRP samples (N = 10 individual donors for each) were assayed using ELISAs from R&D Systems (Minneapolis, MN) for basic fibroblast growth factor (bFGF) and interleukin-1 receptor antagonist (IL-1Ra), and from RayBiotech (Peachtree Corners, GA) for tissue inhibitor of metalloproteases-3 (TIMP-3).
Human Macrophage Cell Culture (Anti-Inflammatory) Bioassay
PTP-001 eluates and LR-PRP or LP-PRP samples were combined with culture media at a 1:5 ratio to emulate dilution into the synovial fluid volume of aspirated human knee joints, 11 and tested in a macrophage anti-inflammatory assay as previously described. 5 Briefly, human THP-1 monocytes were differentiated to a macrophage phenotype and treated with lipopolysaccharide, in the absence or presence of PTP-001 eluates or PRP preparations for 24 hours. Dexamethasone (Dex; 500 nM; Sigma-Aldrich, St. Louis, MO) was utilized as a positive inhibitory control. Pro-inflammatory cytokine content (tumor necrosis factor (TNF)-α) of the culture media was assessed by ELISA (R&D Systems), and the results were normalized to viable cell numbers quantified using a CellTiter cell proliferation assay (Promega, Madison, WI).
Human Synoviocyte/Chondrocyte Co-Culture Bioassay
PTP-001 eluates and PRP preparations were tested in a co-culture system comprising human articular chondrocytes and synoviocytes (non-OA donor tissues). Human condylar cartilage and synovial membrane samples from the knee joints of 5 individual donors were acquired from MTF Biologics (Edison, NJ) and underwent standard collagenase digestion to obtain single cell suspensions. P0 synoviocytes were plated for 48 hours in high-glucose Dulbecco’s Modified Eagle’s Medium (DMEM) containing 10% fetal bovine serum (FBS), 25 mM (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES)), penicillin (100 U/ml), and streptomycin (100 μg/ml) to allow for stabilization. Synoviocytes were lifted with Accumax (Innovative Cell Technologies, Inc., San Diego, CA) and replated in a 12-well plate in DMEM+10% FBS medium for 24 hours.
Synoviocytes were then rinsed and treated with or without 2 ng/ml human recombinant IL-1β (Life Technologies Corporation, Carlsbad, CA) for 24 hours in Opti-MEM. Synoviocytes were rinsed and chondrocytes that had been cultured in Ham’s F-12 containing 10% FBS, 25 mM HEPES, penicillin (100 U/ml), streptomycin (100 µg/ml), ascorbic acid (50 µg/ml), and α-ketoglutaric acid (30 µg/ml) for 48 hours were added to establish co-cultures in transwell chambers. The synoviocyte compartment was plated on the bottom of the tissue culture well, with the chondrocyte compartment seeded on the membrane insert (pore size 0.4 µm; Millipore, Burlington, MA). Co-cultures were treated for 48 hours with Opti-MEM with or without 1:5 dilutions of PTP-001 eluate (50 mg/ml in Opti-MEM), or 1:5 dilutions of either LR-PRP or LP-PRP (the PTP-001 or PRP donor samples used for this assay had generated approximately equal results in the macrophage anti-inflammatory assay, and are indicated by the yellow-filled circles in Fig. 2 ).
Gene Expression Analysis
Synoviocytes or chondrocytes were lysed in the separate transwell compartments, and total RNA was isolated using an RNeasy Mini Kit (QIAGEN, Hilden, Germany). Purity and concentration of the RNA were assessed by UV microspectrophotometry (NanoDrop 1000 Spectrophotometer: Thermo Fisher Scientific). Real-time quantitative polymerase chain reaction (PCR) was performed using the TaqMan One-Step RT-PCR technique with all samples run in duplicate (ViiA 7 Real-Time PCR System: Applied Biosystems, Foster City, CA).
Gene expression quantification was performed for MMP13 and ADAMTS5. PCR primers and probes were obtained from Thermo Fisher Scientific’s proprietary human-specific gene expression assay database. The total copy number of mRNAs was determined using absolute quantitative PCR derived from a standard curve developed for each gene at the time of analysis, and these values were normalized to 18S rRNA.
Statistical Analysis
Data from the ELISAs and macrophage bioassay were analyzed with GraphPad Prism (GraphPad Software, San Diego, CA) using a one-way analysis of variance, respectively, with Tukey’s multiple comparisons test. Data from the gene expression analysis were analyzed with Minitab (Minitab LLC, Chicago, IL) using a least squares model with Tukey’s post hoc test.
Results
Protein Biofactor Content of PTP-001 and PRP
The levels of 3 key bioactive factors illustrative of diverse potential mechanisms of action were significantly higher for PTP-001 in comparison with LR-PRP or LP-PRP ( Fig. 1 ). More specifically, the mean bFGF levels in both of the PRP preparations were <1% of that present in PTP-001. TIMP-3 levels in either of the PRP preparations were approximately 10% of that found in PTP-001, and IL-1Ra concentrations in LR-PRP and LP-PRP were approximately 5% and 1%, respectively, of the PTP-001 levels.
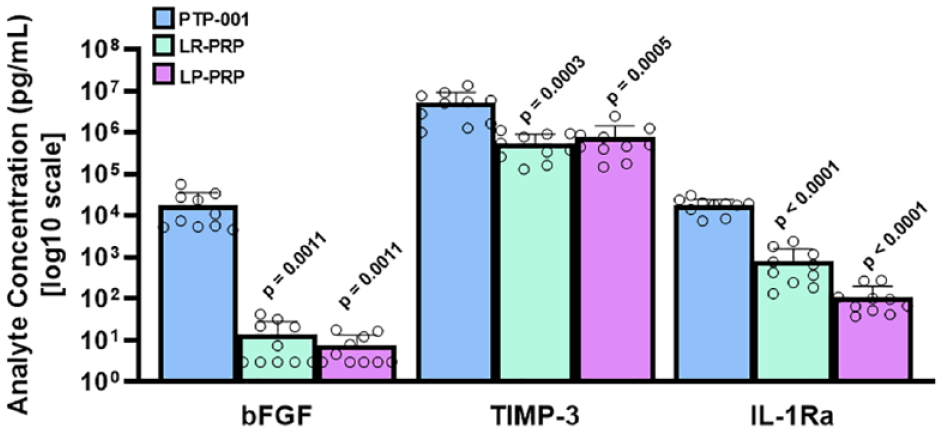
Levels of key biofactors in PTP-001 relative to PRP preparations. Protein biofactor concentrations in PTP-001 eluates or PRP preparations were quantitatively measured by ELISAs specific for bFGF (pro-anabolic factor), TIMP-3 (anti-catabolic factor), and IL-1Ra (anti-inflammatory factor). Biofactor levels are shown (on a log10 scale) in the bar graphs as mean and standard deviation for N = 10 donors (indicated by individual circles). Data were analyzed by one-way ANOVA, and P-values are indicated with the PTP-001 group as comparator. ANOVA = analysis of variance; LR = leukocyte-rich; LP = leukocyte-poor; PRP = platelet-rich plasma; TIMP-3 = tissue inhibitor of metalloproteases-3; IL-1Ra = interleukin-1 receptor antagonist; bFGF = basic fibroblast growth factor; ELISA = enzyme-linked immunosorbent assay.
Effects of PTP-001 and PRP on Macrophage Pro-Inflammatory Cytokine Production
In the macrophage bioassay, treatment with steroid (dexamethasone) inhibited TNF-α levels by >90%, and PTP-001 treatment also resulted in a significant (approximately 80%) reduction in TNF-α production ( Fig. 2 ). Interestingly, a similar effect was observed for LR-PRP, although LP-PRP was much less effective in this assay, with a mean inhibition value of around 37%. In addition, the results for LP-PRP treatment were much more variable, with 2 of the LP-PRP donor preparations actually causing an increase in TNF-α production in the assay.
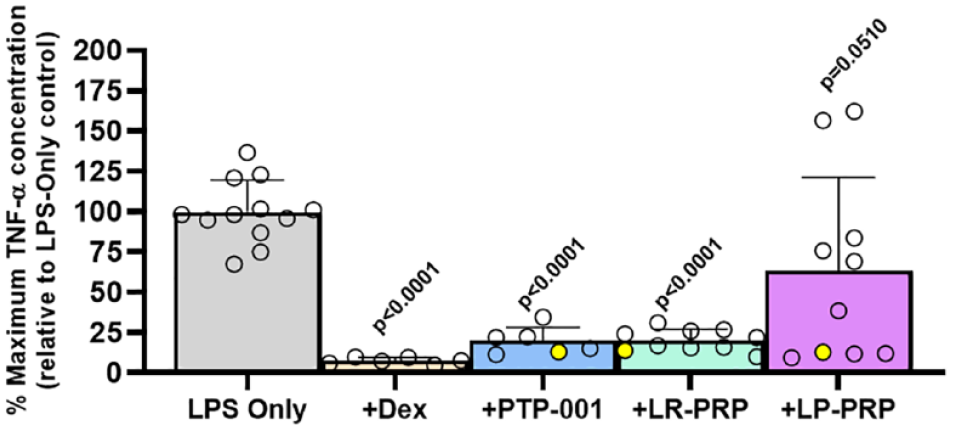
Inhibition of TNF-α production in macrophage cell cultures by PTP-001 in comparison with PRP preparations. THP-1 macrophages were cultured in the presence or absence of LPS and PTP-001 or PRP preparations diluted 1:5 in serum-free culture media. Levels of TNF-α in culture media were measured by ELISA, normalized to viable cell numbers, and expressed relative to maximum levels in LPS-only-treated cultures. Data shown are mean and standard deviation for N = 12 cultures (LPS-only), N = 6 cultures (Dex and PTP-001), and N = 10 cultures (LR-PRP and LP-PRP). Data for separate cultures/donors (PTP-001 and PRP) are indicated by individual circles (donors indicated with filled yellow circles were used for the gene expression experiments). Data were analyzed by one-way ANOVA and P-values are indicated with the LPS-only group as comparator. Dex = dexamethasone; ANOVA = analysis of variance; LPS = lipopolysaccharide; LR = leukocyte-rich; LP = leukocyte-poor; PRP = platelet-rich plasma; ELISA = enzyme-linked immunosorbent assay; TNF-α = tumor necrosis factor-α.
Effects of PTP-001 and PRP on Gene Expression in a Synoviocyte/Chondrocyte Co-Culture System
PTP-001 and PRP preparations were tested in a co-culture system comprising human synoviocytes (pre-treated for 24 hours with IL-1β) and articular chondrocytes. A significant increase in chondrocyte MMP13 mRNA levels associated with synoviocyte IL-1β pre-treatment was observed compared with control co-cultures with unstimulated synoviocytes, and this effect was significantly reduced by PTP-001 and LR-PRP (but not LP-PRP; Fig. 3A ). Chondrocyte ADAMTS5 expression was not significantly increased in association with synoviocyte IL-1β pre-treatment; however, expression of this gene was significantly lower for cultures treated with PTP-001 relative to LR-PRP ( Fig. 3B ).
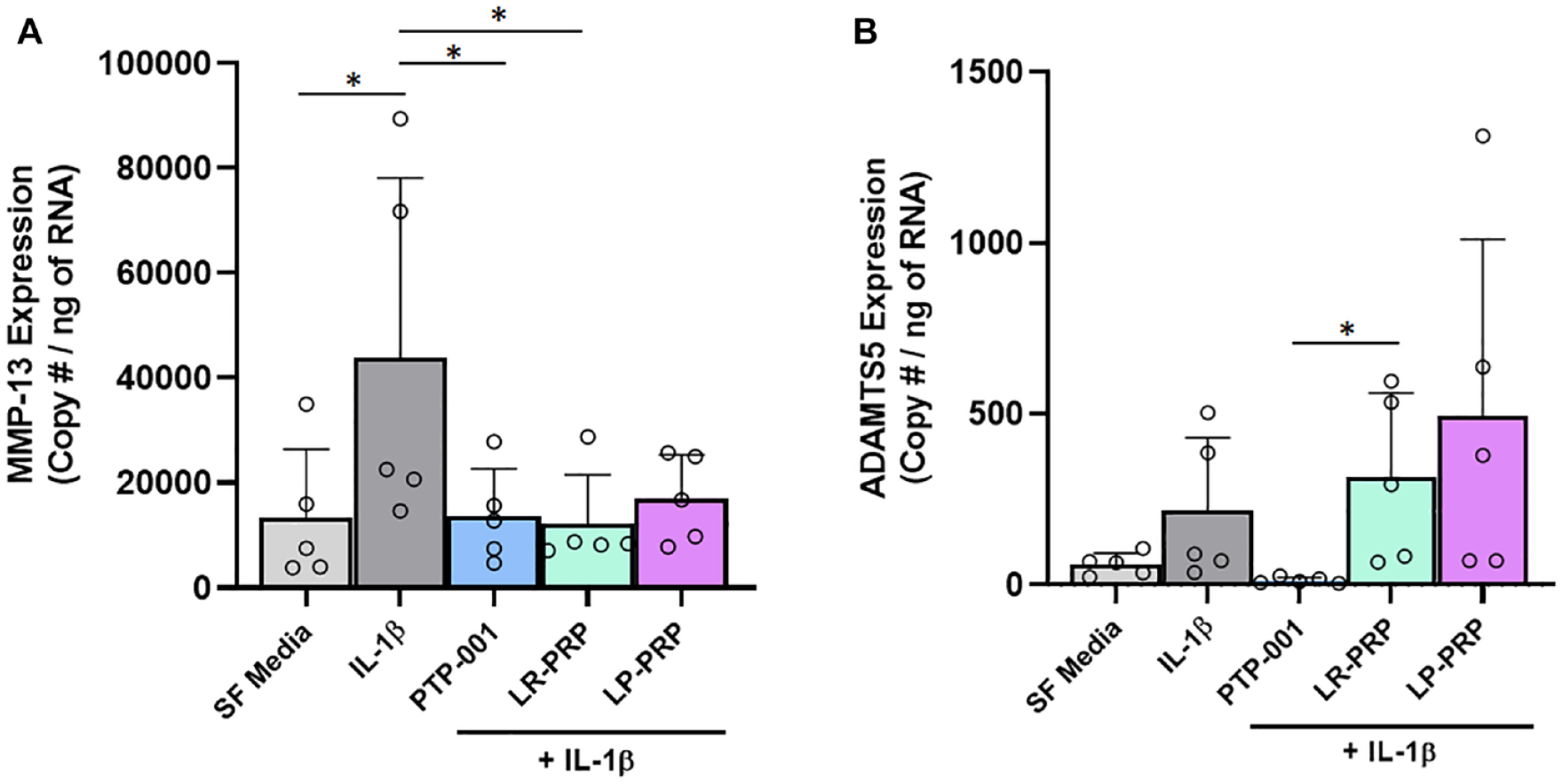
Gene expression levels of (
Discussion
The application of biologics for the treatment of musculoskeletal indications such as OA has the potential to represent a paradigm shift in alleviating these notoriously intractable pathologies. Whereas autologous treatments such as PRP have shown the promise of efficaciousness, there remains both a lack of rigorous clinical evidence and the challenge of substantial compositional heterogeneity associated with patient variability and PRP preparation techniques. Collectively, the findings reported here support the promise for the use of rigorously validated biologics, such as PTP-001, as potential symptom- and disease-modifying treatments for OA. Thus, PTP-001 is being developed as a well-characterized, consistently manufactured biologic, and in contrast to PRP would be available as an “off-the-shelf” product. PTP-001 is a non-cellular biologic which is prepared from readily available, donated tissues from medically screened donors. Its particulate nature renders PTP-001 highly amenable for suspension in saline and intra-articular injection. In the current study, significantly higher mean values were observed for the protein biofactor content of PTP-001 relative to the different PRP preparations ( Fig. 1 ). Furthermore, the concentrations of bFGF and IL-1Ra that we measured for PRP are in good alignment with previously reported values,12,13 which indicates that the differences observed for PTP-001 compared with PRP were not a by-product of obtaining lower than normal concentrations for PRP in our study. It should be noted, however, that the in vitro nature of this study may not necessarily reflect clinical effects. Thus, the interplay of additional biofactors in the joint could contribute to therapeutic effects observed in vivo. Future preclinical studies using animal model(s) of OA may be helpful in further comparing PTP-001 with other treatments such as PRP.
PTP-001 performed as previously observed in the anti-inflammatory (macrophage) bioassay by significantly inhibiting TNF-α production in a consistent manner 5 ; however, the different PRP preparation techniques yielded variable results in this assay ( Fig. 2 ). Interestingly, in this culture system LR-PRP appeared to exert a greater anti-inflammatory effect in comparison with LP-PRP, whereas in a previous publication LR-PRP appeared to induce the expression of NF-κB and downstream (inflammation-associated) responsive genes. 14 Interestingly in this regard, it has been shown that NF-κB/p65 has the potential to promote Sox9 expression and promote early chondrogenic differentiation, 15 suggesting that acute, controlled inflammation could be beneficial, at least at an early stage, in the regenerative process. Continued (chronic) inflammation, however, could lead to osteophyte formation during OA, as well as propagating painful and degenerative sequelae.
In the synoviocyte/chondrocyte co-culture experiments, differences between the effects of PTP-001 and the PRP preparations were also observed ( Fig. 3 ). The reduction of MMP13 gene expression by chondrocytes in response to PTP-001 aligns with our previous data demonstrating a significant reduction in MMP13 protein levels in cultures of human OA chondrocytes 5 ; future work expanding on the current findings which utilized non-OA cartilage and synovium could also include the use of OA tissues, which may better represent the cellular phenotype associated with knee OA.
In summary, PTP-001 appears to represent a more consistent source of beneficial growth factors, and may have a greater capacity to reduce inflammatory and catabolic processes, in comparison with PRP preparations. Development of PTP-001 as a regulated biologic product may represent a novel approach for the clinical treatment of OA and other musculoskeletal indications with high unmet medical need.
Footnotes
Ethical Approval
Ethical consent was obtained for all donated tissues.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funding from Bioventus.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
