Abstract
Objective
Up to 10 million Americans below the age of 65 years have symptomatic knee osteoarthritis (OA) and may not yet be candidates for arthroplasty. In response, a subcutaneous implantable shock absorber (ISA) that unloads the knee has been developed. The safety and effectiveness of ISA treatment were compared against a surgical unloading control, high tibial osteotomy (HTO).
Design
This was a prospective open-label cohort study with a historical control arm. Subjects underwent ISA placement or HTO. The primary endpoint was a composite variable combining pain, function, specific adverse events, integrity of implant or hardware, and conversion to subsequent surgery. Pain and function outcomes (Western Ontario and McMaster Universities Arthritis Index scores) were assessed through 24 months. Adverse events were tracked.
Results
The primary endpoint demonstrated superiority of the ISA arm versus the HTO arm, with 85.6% of ISA subjects meeting all criteria compared with 65.5% of HTO subjects. In addition, all 5 secondary endpoints showed superiority of ISA over HTO. At 24 months, the proportions of subjects considered responders were 95.8% (ISA) versus 87.9% (HTO) for pain and 91.7% (ISA) versus 81.3% (HTO) for function. The ISA procedure was well tolerated, with 13.4 days to full weightbearing status versus 58.0 days for the HTO arm.
Conclusions
Treatment with an ISA demonstrated noninferiority and superiority versus treatment with HTO in subjects aged 25-65 years who had OA of the medial knee. Treatment with ISA has high clinical benefit and is durable through at least 24 months.
Introduction
Knee osteoarthritis (OA) has a global prevalence estimated at 16%. 1 The initial diagnosis often comes during the working years, with a 9.3% risk of symptomatic knee OA by age 60. 2 Furthermore, 60% of arthritis sufferers may be of working age. 3 With estimates of up to 10 million cases in people below 65 years of age, symptomatic knee OA negatively impacts many people during the prime active years of their lives.4,5
Nonsurgical treatment of mild-to-moderate knee OA includes weight loss, analgesics, physical therapy, bracing, hyaluronic acid (HA) injections, and intra-articular corticosteroids. 6 These nonsurgical treatments have clinical and cost-effectiveness limitations 7 and are “high-touch” due to the relatively limited duration of treatment effect and repeated clinician visits. Arthroscopic debridement is not considered a durable solution for this population, 8 high tibial osteotomy (HTO) may be unattractive to patients, 9 and the majority of younger knee OA patients are ineligible for, or reject, total knee arthroplasty (TKA). 10
To address the clinical need of younger patients with symptomatic mild-to-moderate medial knee OA, an implantable shock absorber (ISA) was developed to unload the knee (Moximed, Inc, Fremont, CA) (see Fig. 1 ). The ISA is placed subcutaneously, outside the joint capsule, and superficial to the medial collateral ligament (MCL) during an outpatient-compatible procedure using surgical techniques familiar to orthopedic surgeons.11 -13 The cylindrical shock absorber is 0.8 cm × 5 cm and is fixed by 2.5 cm × 4.0 cm titanium plates to the distal femur and proximal tibia. The ISA reduces peak forces on the medial compartment by 32%. 14 This medial unloading is equivalent to HTO and does not transfer load to the lateral compartment. 15 Load management leads to symptom relief: because repetitive overload of the joint changes the knee’s mechanical environment and often initiates degeneration, interrupting this cascade via unloading may allow the knee to adjust to a new, less symptomatic environment. 16 Versions of an ISA with a polymer shock absorber have been used clinically since 2014. All generations have employed the same mechanism of action, a nearly identical physical footprint, and equivalent target unloading amount. Experience from earlier studies influenced the following evolution: exclusion of patients with excessive medial osteophytes or meniscal extrusion; refinement of surgical instrumentation for technique repeatability; and modification of material selection and dimensions of the implant for robustness and manufacturability.
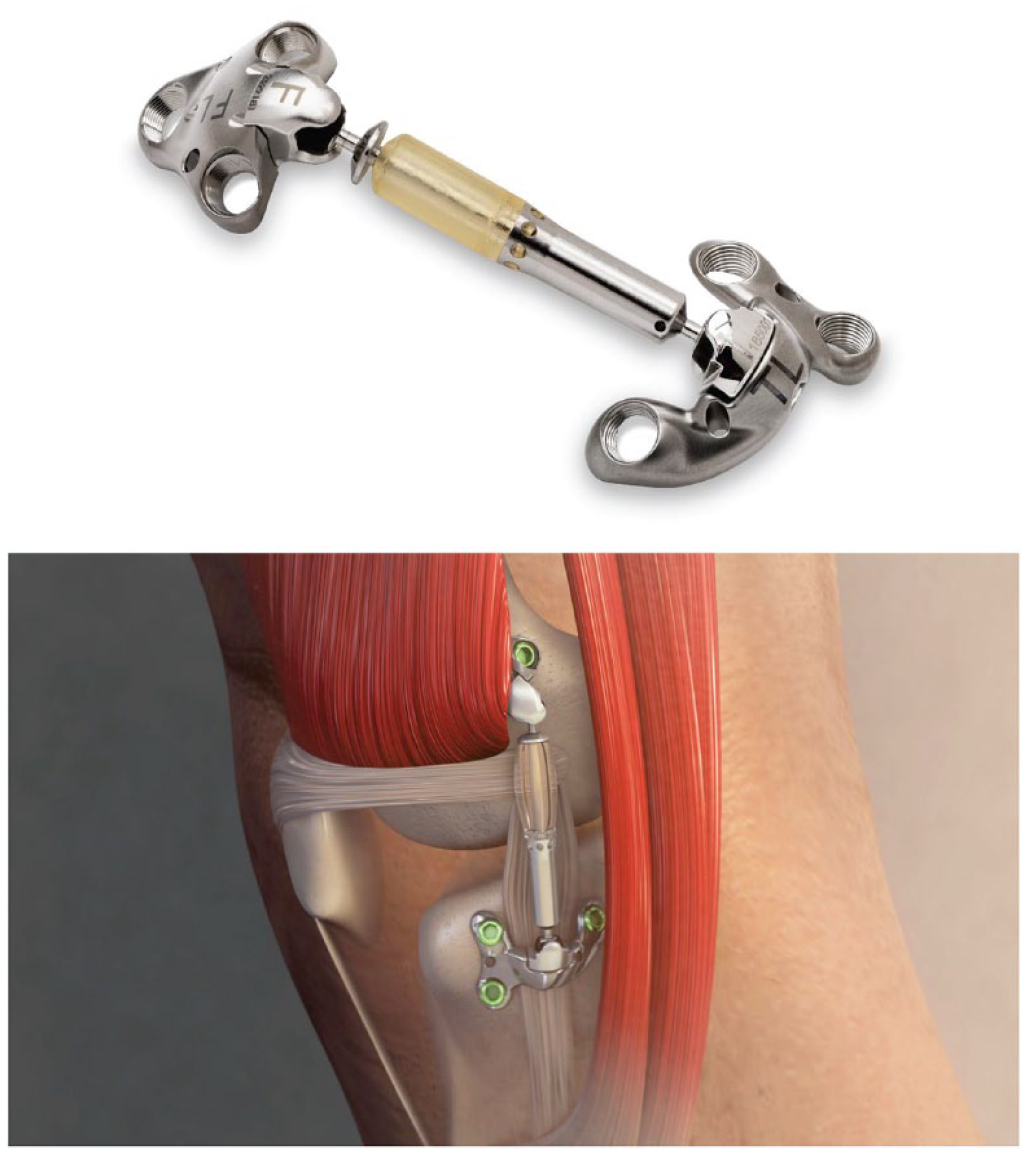
The assembled knee shock absorber (top) and its placement when implanted (bottom).
While arthroplasty is a gold standard for severe, end-stage OA, younger, moderately diseased patients have higher rates of dissatisfaction 17 and re-operation after TKA.18,19 In contrast, HTO unloading shares a similar patient profile with the ISA: disease isolated to the medial compartment, younger patient age, and higher activity demands.20,21 Modern HTO techniques report survivorship to at least 8-10 years,22,23 but the procedure is associated with a long recovery period that limits patient acceptance. This study evaluated the safety and effectiveness of the ISA in subjects with symptomatic, medial knee OA compared with a surgical control of HTO, in support of a US Food and Drug Administration (FDA) review of the ISA.
Methods
Study Design and Participants
This prospective, open-label cohort study was conducted in 10 centers in the United States and Europe. The study was conducted under an Investigational Device Exemption (IDE) from the FDA in accordance with Good Clinical Practice (GCP) and the Declaration of Helsinki. The study (clinicaltrials.gov; NCT03671213 and NCT03838978; results pooled) was approved by local institutional review boards (IRBs) and ministries of health (MOH) or ethics committees (ECs) as appropriate. Subjects granted their written informed consent prior to any study activities and were enrolled over 20 months between 2018 and 2020.
For the ISA arm, eligible subjects were between the ages of 25 and 65 years at the time of screening, with radiographic evidence of OA of the medial compartment of the study knee (Kellgren and Lawrence [KL] grade 24 1-4) and failure of ≥6 months of nonsurgical treatments. Study knee pain (minimum rating of 40 according to the Knee injury and Osteoarthritis Outcome Score [KOOS]-derived Western Ontario and McMaster Universities Arthritis Index [WOMAC] score) 25 was primarily localized to the medial aspect and generally exacerbated by weightbearing. Subjects with KL4 were allowed, except those with large marginal osteophytes in the medial compartment or bony erosion. An independent core laboratory performed all radiographic grading for screening eligibility.
Eighty-one subjects in the ISA arm were implanted with an ISA using standard orthopedic instruments and single-use, ISA-specific instruments. With the subject lying supine and the knee extended, the surgeon creates a medial incision beginning proximal to the femoral epicondyle and extending distally to the pes anserine. Following exposure of the superficial MCL as an anatomical reference, fluoroscopic guidance and ISA-specific instruments are used to confirm the femoral base location and trial functional performance of the device. Once confirmed, the implant is secured to bone in the extracapsular space using routine locking screw technique. No concomitant procedures, including arthroscopy, were performed in the ISA arm of the study to isolate the treatment effect. For these subjects, the average age was 51.2 years, 60.5% of subjects were male, the average body mass index (BMI) was 28.4 kg/m2, and the average KL grade was 2.5 (range 1-4). Notably, the ISA arm included some subjects with femoral and tibial lesions, medial meniscal posterior root tears, and evidence of subchondral insufficiency fracture (SIFK), but these details were not recorded in the HTO group.
The historical control HTO arm included subjects with knee OA who were treated with opening-wedge HTO according to the standard procedure as part of a nonrandomized prospective trial (NCT01610505). For inclusion in the HTO arm, subjects were aged 25-80 years, had knee OA of KL grade 1-4 (after rescoring of baseline radiographic images by the same independent core laboratory as the ISA group), and had failed ≥6 months of nonsurgical treatment. HTO was selected as a study control to satisfy FDA’s desire for a surgical comparator. At FDA’s request, a propensity score design was employed to ensure that the subjects included in the HTO arm were matched to subjects in the ISA arm across multiple demographic and baseline characteristics. In the arm, 81 propensity-matched subjects treated with HTO were included in the analysis. For HTO subjects, the average age was 52.5 years, 65.4% of subjects were male, the average BMI was 29.3 kg/m2, and the average KL grade was 2.8 (range: 1-4). Table 1 presents further baseline characteristics. No statistically significant differences in any variables existed between the 2 arms at baseline (all P values >0.05).
Subject Demographics and Baseline Characteristics.
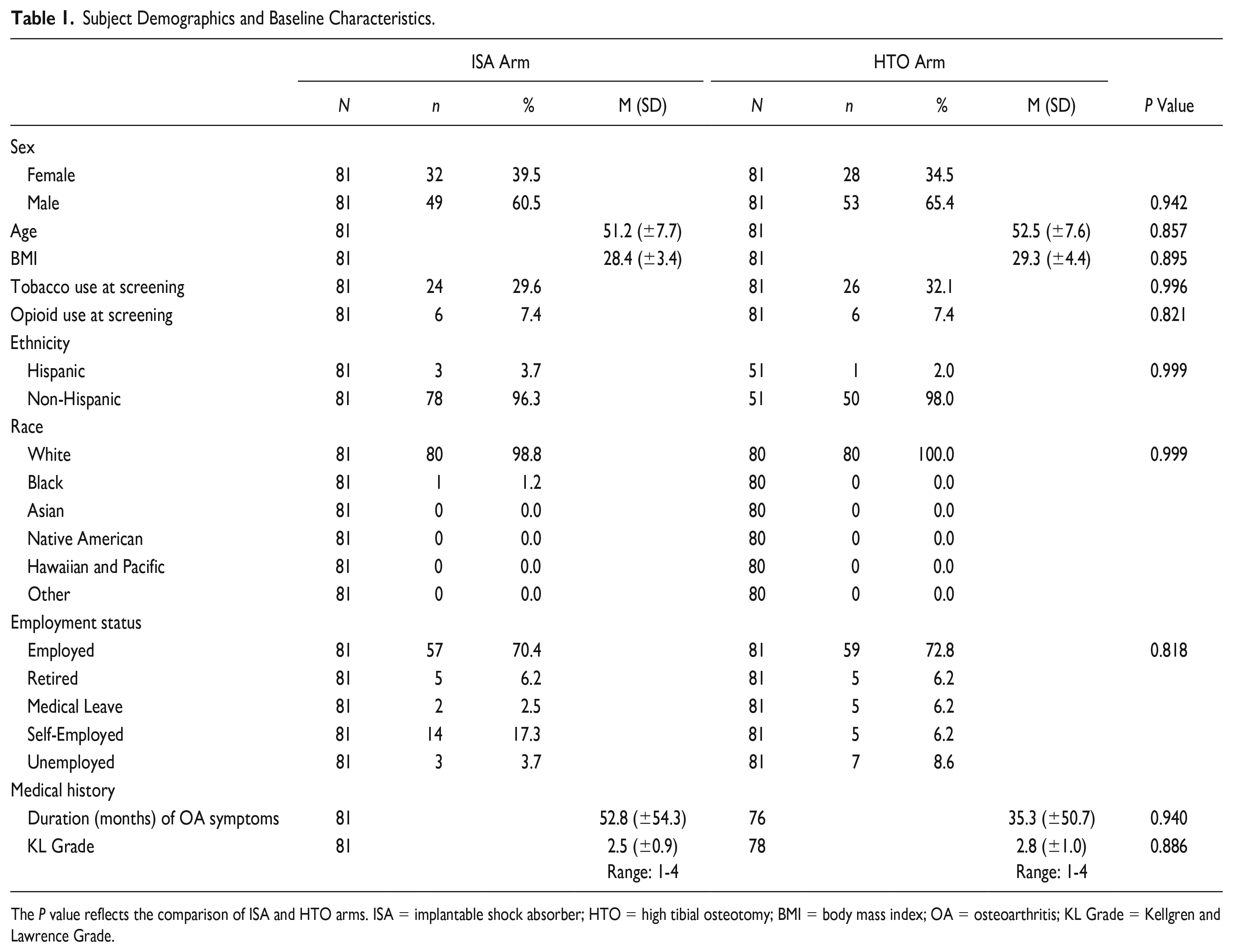
The P value reflects the comparison of ISA and HTO arms. ISA = implantable shock absorber; HTO = high tibial osteotomy; BMI = body mass index; OA = osteoarthritis; KL Grade = Kellgren and Lawrence Grade.
After the interventions, no weightbearing or range of motion restrictions were required, and there were no study-imposed limitations on return to activity. All subjects were encouraged to follow any surgeon-prescribed physical therapy.
Follow-Up and Outcomes
Subjects reported pain and function via KOOS questionnaires. 26 Responders were defined as those with at least 20% improvement from baseline, and an absolute change of ≥10 points, on the WOMAC pain or function (0-100 score, higher scores represent worse outcome) questions in the KOOS. In addition, subjects reported the number of days until full weightbearing after the procedure. Follow-ups were completed at 6 weeks and 3, 6, 12, 18 (ISA arm only), and 24 months after implantation. Intraoperative variables (operating time, blood loss, and length of hospital stay) were collected, and the integrity of the implant (ISA arm) and hardware (HTO arm) was collected. Concomitant medications and adverse events (AEs) were tracked throughout the study.
The primary endpoint was a composite variable, selected in consultation with FDA and supported by guidance documents,27,28 defined as the proportion of subjects in each arm that met all 5 criteria at 24 months: (1) responder for WOMAC pain; (2) responder for WOMAC function; (3) freedom from deep infection requiring surgical intervention, damage to adjacent neurovascular or ligament structures necessitating reconstruction, or HTO nonunion; (4) maintained the integrity of the implant or hardware; and (5) did not require conversion to arthroplasty or other joint-modifying surgery. Implant removal alone was not deemed a study failure for either arm.
The secondary endpoints were: (1) days to full weightbearing, (2) WOMAC pain at 3 months, (3) WOMAC function at 3 months, (4) WOMAC pain at 24 months, and (5) WOMAC function at 24 months.
Statistical Analysis
The statistical analysis plan was reviewed and accepted by the FDA. For the primary endpoint, an intent-to-treat analysis of all enrolled subjects was completed with multiple imputation for missing data and included noninferiority and superiority testing. A sample size of 80 was identified for a noninferiority margin of −0.10. For secondary endpoints, as-observed data were analyzed with no imputation for missing values. Secondary endpoints were evaluated correcting for multiplicity and based on the Hochberg (1988) approach (P < 0.025). Statistical analyses used the generalized linear model in SAS 9.4. The assumption of statistical significance required P < 0.05 using 2-way analysis of variance after adjusting for propensity score subclass. Descriptive statistics are presented as means, ranges, standard deviations (SDs), 95% confidence intervals (CIs), and percentages, unless otherwise stated.
Results
Primary Endpoint
Figure 2 shows subject disposition through the study. The clinical composite primary endpoint at 24 months was met by 85.6% of the subjects in the ISA arm and 65.5% of the subjects in the HTO arm (see Table 2 ). The propensity-score-adjusted group difference was 20.2%. Noninferiority and superiority of the ISA compared with HTO were confirmed with the lower bound of the 2-sided 90% CI at 6.7%.
Subject disposition for both arms.
Primary Endpoint Composite Variable (See Descriptions of Composite Variables in the Text).
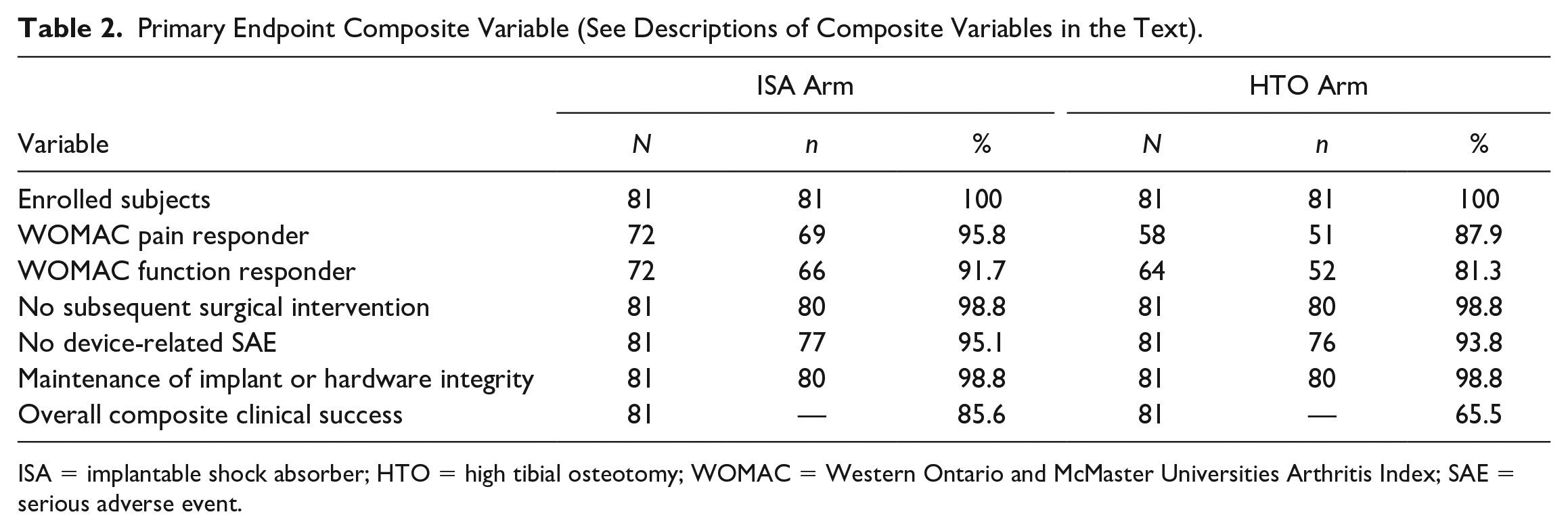
ISA = implantable shock absorber; HTO = high tibial osteotomy; WOMAC = Western Ontario and McMaster Universities Arthritis Index; SAE = serious adverse event.
Secondary Endpoints
All 5 secondary endpoints demonstrated statistical superiority of the ISA (see Table 3 ). The time to full weightbearing on the operated leg was 13.4 days (±10.12 [SD]) in the ISA arm and 58.0 days (±39.91) in the HTO arm (P < 0.001) (see Fig. 3 ). Percentage changes in pain and function relative to baseline at both 3 and 24 months were larger in the ISA arm than in the HTO arm (all P values <0.02).
Secondary Endpoints.
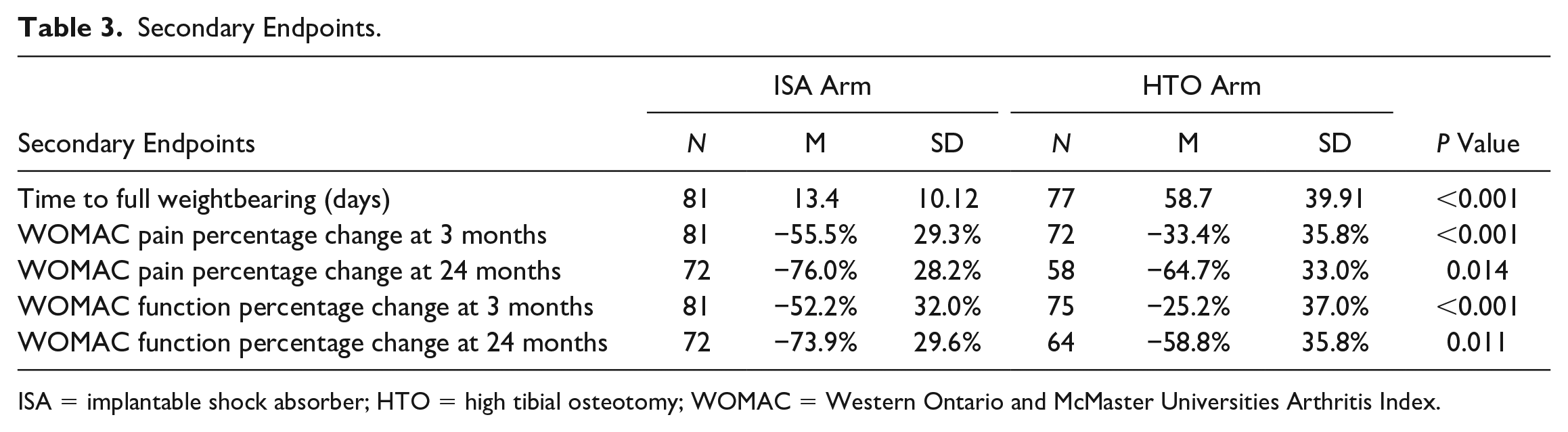
ISA = implantable shock absorber; HTO = high tibial osteotomy; WOMAC = Western Ontario and McMaster Universities Arthritis Index.
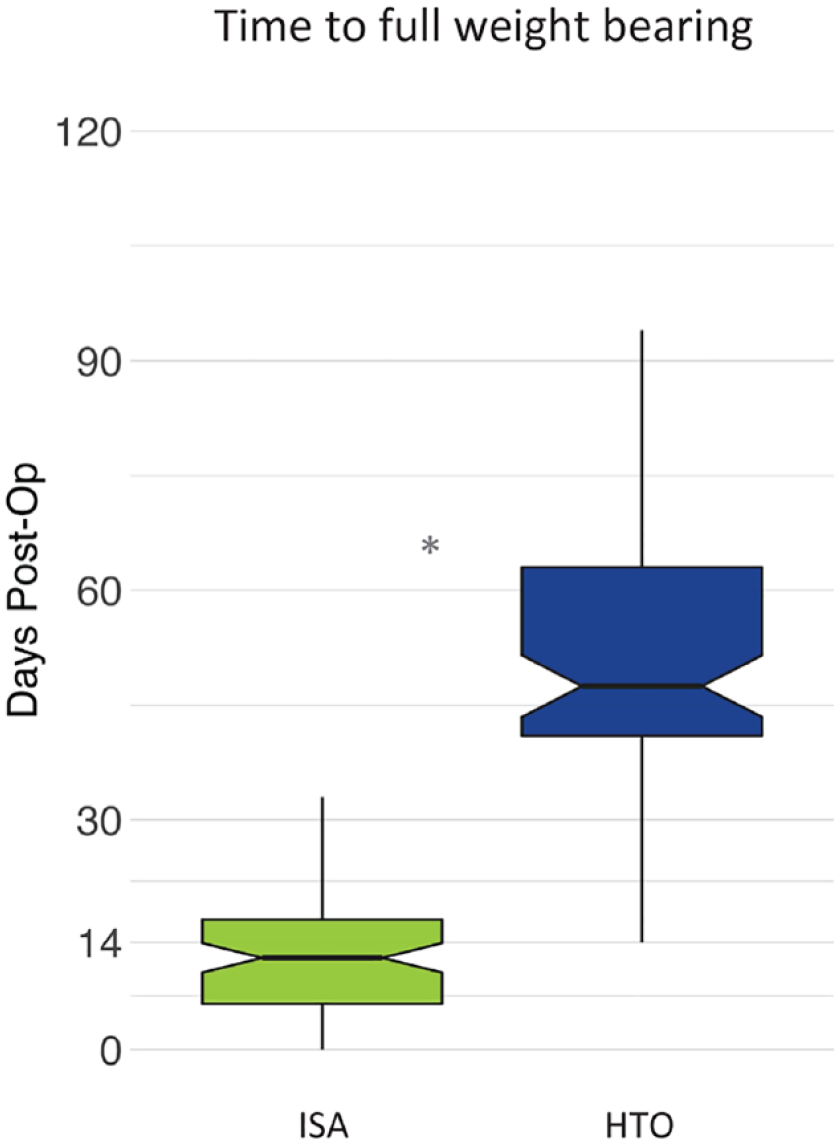
Days to full weightbearing in the ISA and HTO arms. Boxplot indicates median (notch) and interquartile range (IQR), with whiskers identifying the highest and lowest data point within 1.5x IQR, and * indicates a statistically significant difference between the arms. ISA = implantable shock absorber; HTO = high tibial osteotomy.
Pain and Function
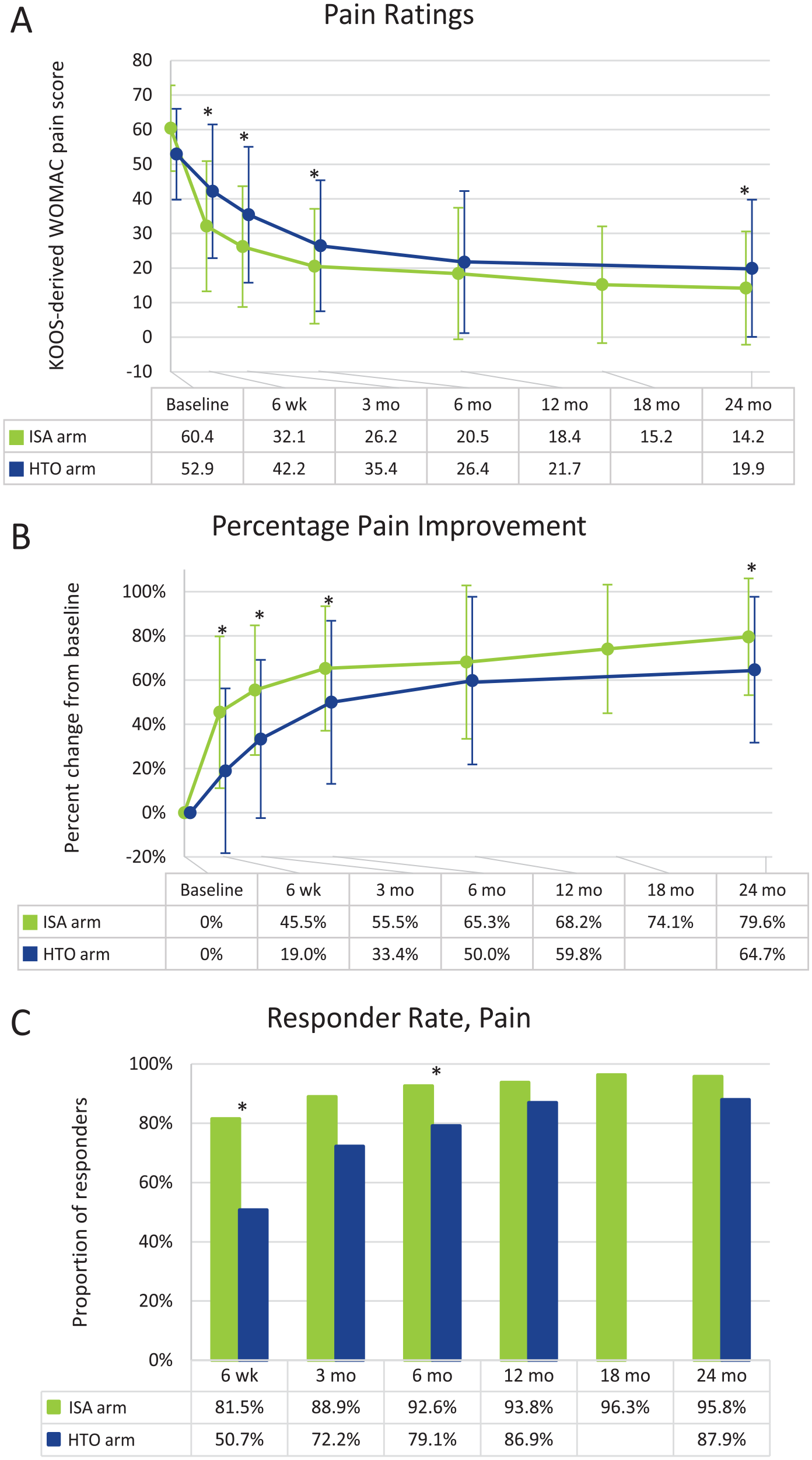
Pain ratings significantly decreased over time in both arms, and the magnitude of improvement was greater in the ISA arm than in the HTO arm. The mean WOMAC pain rating at baseline was 60.4 (±12.37 [SD]) and 52.9 (±13.1) in the ISA and HTO arms, respectively (P > 0.05). At all follow-up points except 12 months, pain ratings were statistically significantly lower in the ISA arm than in the HTO arm (all P values <0.005). At 24 months, the mean pain rating was 14.2 (±16.36), a 76.0% improvement, in the ISA arm and 19.9 (±19.84), a 64.7% improvement, in the HTO arm (see Fig. 4A and B ). The proportion of subjects considered responders at 24 months was 95.8% in the ISA arm and 87.9% in the HTO arm (see Fig. 4C ).
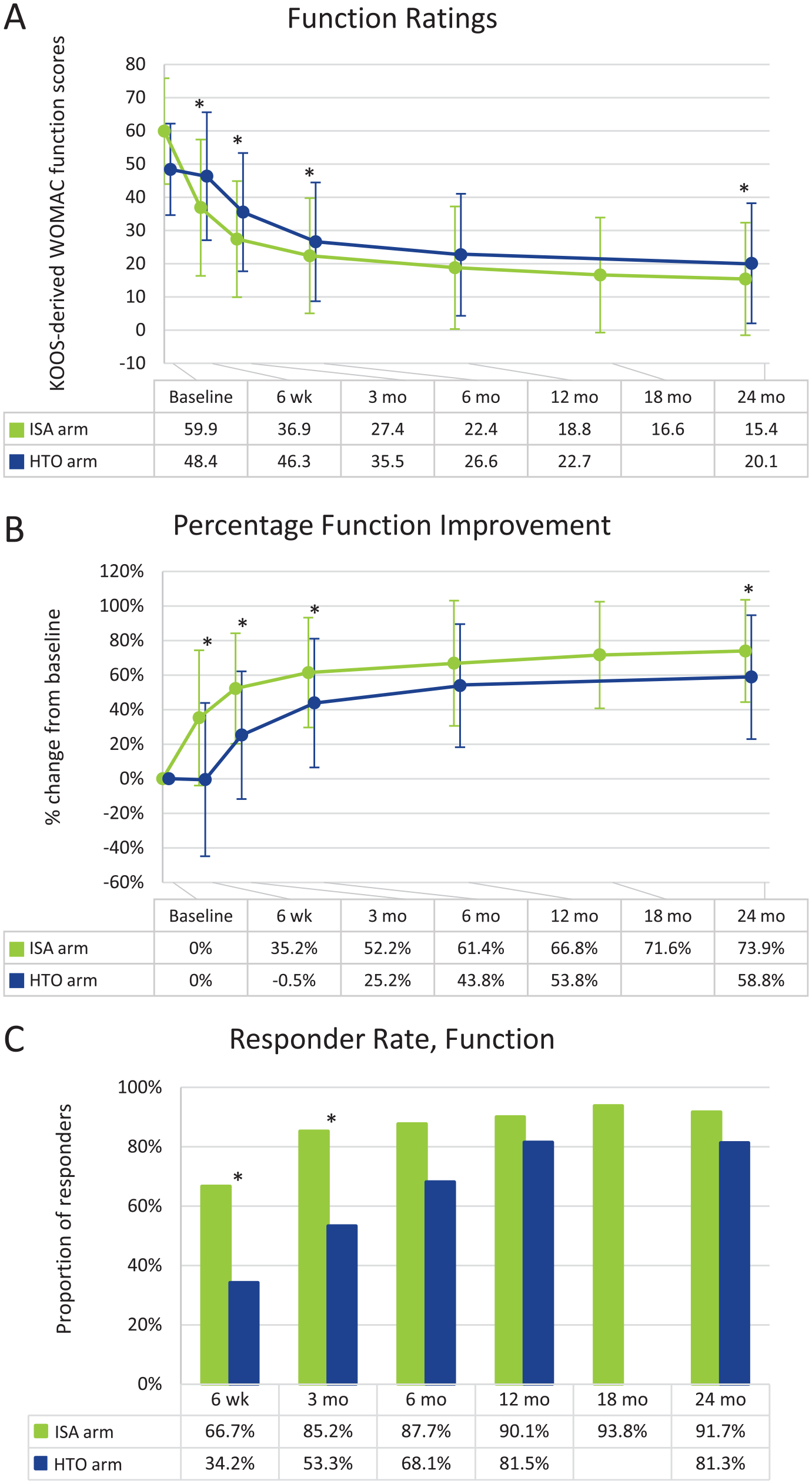
Function ratings significantly decreased (improved) over time in both arms, and the extent of improvement was greater in the ISA arm than in the HTO arm. The mean WOMAC function rating at baseline was 59.9 (±15.93) and 48.4 (±13.78) in the ISA and HTO arms, respectively (P > 0.05). At all follow-up points except 12 months, function ratings were statistically significantly lower in the ISA arm than those in the HTO arm (all P- values <0.02). At 24 months, the mean function rating was 15.4 (±16.93), a 73.9% improvement, in the ISA arm and 20.1 (±18.07), a 58.8% improvement, in the HTO arm (see Fig. 5A and B ). The proportion of subjects considered responders at 24 months was 91.7% in the ISA arm and 81.3% in the HTO arm (see Fig. 5C ).
(
Taken together, both pain and function improved more in the ISA arm than in the HTO arm (see Fig. 6 ).
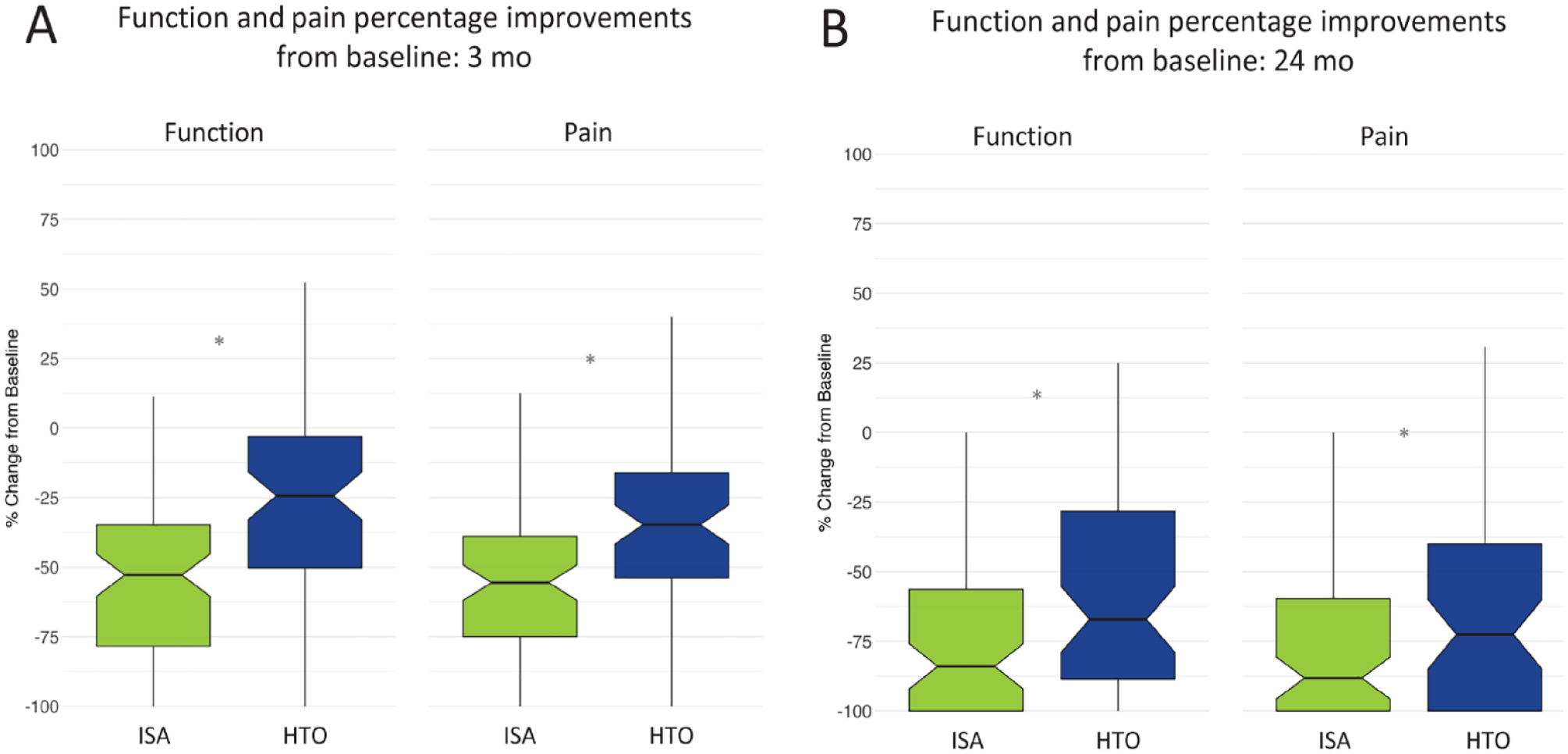
Percentage improvements relative to baseline in function and pain for the ISA and HTO arms at 3 months (
Intraoperative Variables
The operative time was 70.7 (±19.05) minutes for the ISA arm and 65.4 (±24.36) minutes for the HTO arm (P > 0.05). The estimated blood loss was 4.6 (±14.32) ml for the ISA arm and 42.4 (±85.89) m; for the HTO arm (P < 0.001). Length of hospital stay in the ISA arm was strongly influenced by location and regional clinical standard of care: 9.6 (±10.67) hours in the United States compared with 40.7 (±14.15) hours in Europe (P < 0.001). Overall, 90.5% of the subjects in the United States were treated on an outpatient basis.
Adverse Events
Classification of AEs was per the FDA IDE clinical protocol, and all serious adverse events (SAEs) were adjudicated by an independent clinical events committee. Overall, there were 15 device- and procedure-related SAEs in 13 subjects in the ISA arm (16.0%). In the HTO arm, there were 42 SAEs in 37 subjects (45.7%). The overall occurrence of SAEs was significantly higher in the HTO arm, with pain (reported by 4.9% in the ISA arm and 35.8% in the HTO arm) being the primary contributor to the group difference (P < 0.001) (see Table 4 ).
Device- and Procedure-Related SAEs.
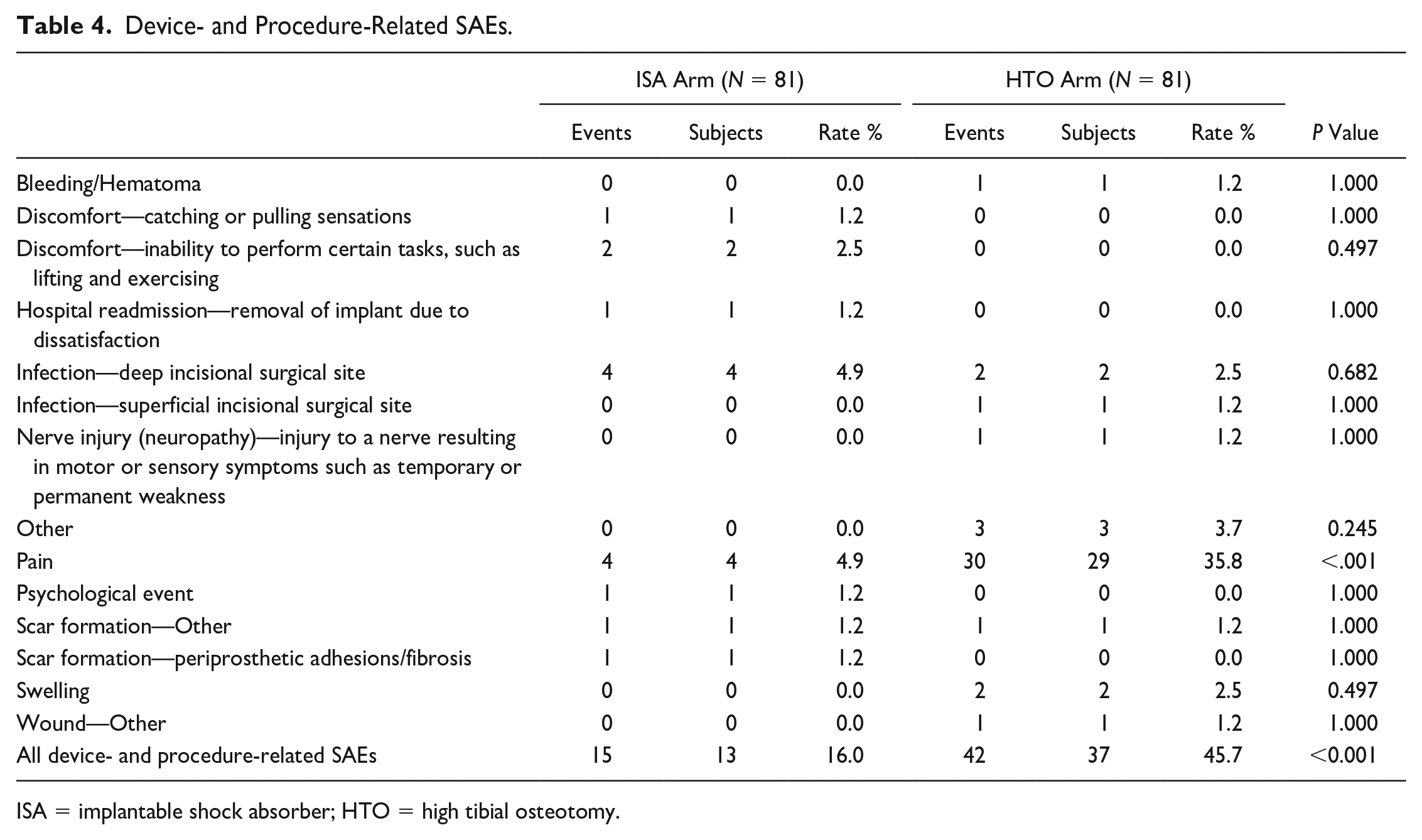
ISA = implantable shock absorber; HTO = high tibial osteotomy.
Implant and Hardware Integrity
There were zero mechanical device malfunctions in either arm. There was one instance of a screw partially backed out (ISA subject) and one instance of a screw breakage (HTO subject). The rate of secondary operations for implant removal in the ISA arm was significantly lower than that in the HTO arm. The ISA arm had 11 of 81 (13.6%) implant removals, of which 7 of 11 (63.6%) were elected by the subject. One subject in the ISA arm (1/81, 1.2%) was converted to unicompartmental knee arthroplasty (UKA). Reasons for removal were infection (n = 4), discomfort/catching-pulling sensation (n = 2), pain (n = 2), scar formation (n = 2), and dissatisfaction (n = 1). The HTO arm had 61 of 81 (75.3%) implant removals. One subject in this arm (1/81, 1.2%) underwent joint-modifying surgery after implant removal. Reasons for removal of the HTO hardware included: bone consolidation—may also be elective prevention of future hardware problems (30), pain (27), swelling (2), bone consolidation with plate pain (1), and nerve injury/neuropathy (1).
Discussion
The primary endpoint of the study was met and demonstrated superiority of the ISA arm versus the HTO arm. All secondary endpoints also met superiority. The ISA arm had clinically meaningful and statistically significant improvements in pain and function that were rapid, substantial, and sustained through the 2-year follow-up endpoint. Importantly, subjects treated with the ISA had a recovery time (return to full weightbearing status) that was 4 times shorter than that of the HTO arm.
The study population represents a high-demand patient with significant pain-related disability. Age and disease severity made the patients less suitable for arthroplasty, yet the stage and nature of their OA also made them poor candidates for cartilage restoration or other isolated biologic procedures. Mechanical overloading on the knee has been shown to contribute to defects in cartilage and subchondral bone in medial OA. 29 Unloading through bracing 30 or HTO 31 can improve outcomes, and this report indicates that outcomes with an ISA can meet or exceed those benchmarks. Patients who experience symptom relief but cannot comply with unloader brace wear may also be candidates for ISA treatment. TKA utilization in younger, less severely diseased patients has been increasing, despite data that arthroplasty can result in higher rates of suboptimal outcomes in this population. 32 Thus, ISA treatment may represent an important alternative.
Low AE rates were noted in both arms, but pain was reported more frequently in the HTO arm than in the ISA arm, which is consistent with reports of HTO recovery. 33 There were 4 infections in the ISA arm, 3 of which occurred at a single center. In 2 previous studies of a prior generation of the ISA (n = 90), zero infections were noted.13,34 No malfunctions of the ISA were observed. Eleven subjects had their ISA removed, compared with 61 subjects who had their HTO plates removed. Of the ISA removals, only 1 ISA subject had a conversion (UKA), while the remaining 10 had no subsequent implant. Of these 10 ISA subjects with removal, 9 had clinically meaningful improvement in pain and function scores at 24 months. Multiple studies of joint distraction showed a sustained benefit after relatively short (6-8 weeks) periods of complete joint unloading.16,35,36 Thus, a similar mechanism may be involved such that the unloading function of the ISA device provides continuing benefit to the joint even after removal. No intraoperative complications were observed during removal of the ISA device, suggesting that implant removability is feasible and safe. Importantly, both ISA and HTO hardware removal leave the articulating surfaces and structures intact, allowing implant removal without requiring immediate, subsequent arthroplasty.
Limitations of this study include its nonrandomized nature, although the historical control HTO arm was propensity-matched to the ISA-treated arm. The study provides evidence for the ISA performance versus HTO, but important research remains to determine the generalizability of results.
Currently, a gap in treatment options exists for younger patients with significant symptoms and mild-to-moderate OA. With a mean pain improvement of 79.6% at 24 months and 95.8% of subjects considered responders for pain, the results associated with ISA treatment are robust and durable. Moreover, the ISA allowed subjects to quickly mobilize, and the nearly 400% shorter recovery time versus HTO is valuable, as patients wish to avoid extended periods of non-weightbearing. The ISA offers a treatment option for medial OA patients who are not candidates for arthroplasty and have failed to gain symptom relief with conservative treatments.
Footnotes
Acknowledgments and Funding
The authors thank all investigators from the Calypso Study Group: Marcin Domzalski (Medical University of Łódź, Łódź, Poland), Andreas Gomoll (Hospital for Special Surgery, New York, New York), Mateusz Janik (Carolina Medical Center, Warsaw, Poland), Seth L. Sherman (Stanford University, Stanford, California), Konrad Slynarski (Slynarski Knee Clinic, Warsaw, Poland), Jacek Walawski (Lublin Hospital, Poland), and Urszula Zdanowicz (Carolina Medical Center, Warsaw, Poland). In addition, the authors thank Allison Foster, PhD, a consultant for Moximed, for editing support. Moximed, Inc. was the funder and sponsor of the study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors received institutional support from Moximed regarding this study. In addition, DRD is a consultant for Depuy Synthes and Osteocentric, has received royalties from Smith and Nephew, and has received institutional research support from Zimmer, Aesculap, and Donjoy. ASR is a consultant for Moximed, Enhatch, Conformis, Smith & Nephew, Anika, Bodycad, Xiros, NewClip, Ranfac, Marrow Cellution, and Cervos.
Ethical Approval
This prospective, open-label cohort study was conducted in 10 centers in the United States and Europe. The study was conducted under an Investigational Device Exemption (IDE) from the FDA in accordance with Good Clinical Practice (GCP) and the Declaration of Helsinki. The study (clinicaltrials.gov; NCT03671213 and NCT03838978; results pooled) was approved by local institutional review boards (IRBs) and ministries of health (MOH) or ethics committees (ECs) as appropriate.
