Abstract
Objective
A complete understanding of the glenohumeral joint anatomy is crucial for osteochondral allograft (OCA) transplantation and prosthetic design. However, existing data on the cartilage thickness distribution are not consistent. This study aims to describe the cartilage thickness distribution at both the glenoid cavity and humeral head in males and females.
Design
Sixteen fresh cadaveric shoulder specimens were dissected and separated to expose the glenoid and humeral head articular surfaces. The glenoid and humeral head were cut into 5-mm coronal sections. Sections were imaged and cartilage thickness was measured at 5 standardized points on each section. Measurements were analyzed based on age, sex, and regional location.
Results
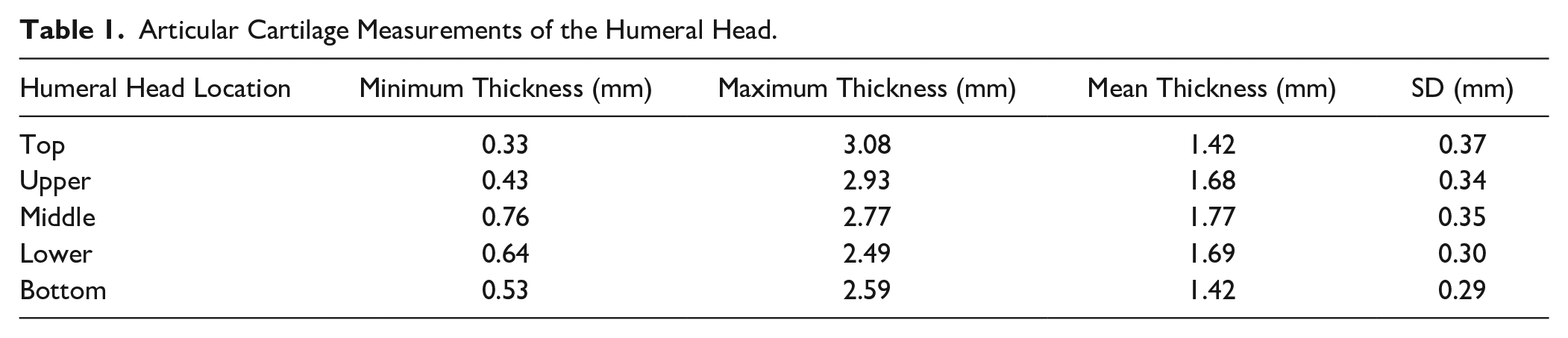
For the humeral head, cartilage was thickest centrally (M = 1.77 ± 0.35 mm) and thinnest superiorly and inferiorly (M = 1.42 ± 0.37 mm, 1.42 ± 0.29 mm). At the glenoid cavity, cartilage was thickest in the superior and inferior areas (M = 2.61 ± 0.47 mm, 2.53 ± 0.58 mm) and thinnest centrally (M = 1.69 ± 0.22 mm). Males were found to have thicker cartilage at both the humeral head and glenoid (P = 0.0014, P = 0.0133).
Conclusions
Articular cartilage thickness distribution of the glenoid and humeral head is nonuniform and reciprocal in nature. These results can be used to further inform prosthetic design and OCA transplantation. We noted a significant difference in cartilage thickness between males and females. This suggests that the sex of the patient should be taken into consideration when matching donors for OCA transplantation.
Keywords
Introduction
Complication rates for revision shoulder arthroplasty are notably higher compared with primary arthroplasty.1,2 Similarly, it is imperative that anatomical prosthetic implants replicate original anatomy as closely as possible to restore shoulder kinematics. Currently, most prosthetic designs and placements are focused on obtaining an anatomical lateral humeral offset, neck-shaft angle, humeral head height, and humeral head size as defined by the center of rotation.3 -5 However, prosthetic sizing is often based on bony anatomy and does not account for cartilage thickness, providing a source for potential size mismatch. It has been reported that a humeral component that is 5 mm too thick compared with a perfect anatomical match can result in a decrease in the range of motion by 23° to 30°. 1 In addition, it has been shown that shortening the humeral component by 5 mm will reduce the surface arc of the prosthetic by 24° and limit range of motion. 6 Increased knowledge regarding glenohumeral articular cartilage thickness will help to inform future prosthetic design.
Successful osteochondral allograft (OCA) transplantation relies on donor tissue that conforms to the shape of the recipient’s articular surface. The geometry of the humeral head and glenoid articular surfaces has been well defined.7 -10 Iannotti et al. 8 found the humeral head to be spherical at the center, but the peripheral radius was 2 mm less in the axial compared with coronal plane. Soslowsky et al. 10 found the average radius of curvature of the humeral head in the coronal plane to be 24 mm, which was supported by Iannotti et al. 8 However, Iannotti et al. 8 reported that the average radius of curvature in the coronal plane of the glenoid is 2.3 mm greater than that of the humeral head. Knowledge of the cartilage thickness and potential sex-based differences at the glenohumeral joint will positively inform selection of donor tissue for OCA transplantation to ensure articular congruency and osseous integration.
Previous studies on the glenohumeral joint have employed a variety of modalities to evaluate the glenoid and humeral head cartilage thickness, but have reported a wide range of results.10 -14 Based on our review, no prior studies have evaluated cartilage thickness directly using fresh, unfrozen, and unprocessed cadaveric shoulders. The aim of this study is to evaluate the regional articular cartilage thickness distribution of the glenohumeral joint in fresh, unprocessed cadavers. This is to further inform prosthetic design and OCA transplantation. We hypothesized that the cartilage thickness of both the glenoid cavity and humeral head will be nonuniform in nature.
Methods
Sixteen fresh, unprocessed cadavers were used in this study: 7 males with an average age of 73.6 (range, 61-85) years and 9 females with an average age of 81.6 (range, 70-95) years. Shoulders were separated from the medial scapular border to the middle third of the humeral shaft. Skin and soft tissue around the glenohumeral joint was removed via sharp dissection until the entire proximal humerus and scapula were visible. The tendons and ligaments were severed carefully, to prevent articular cartilage damage. The humerus and glenoid were separated and inspected for pathologic abnormalities, including gross arthritic changes, deformities, or previous fractures. Specimens with apparent abnormalities were removed ( Fig. 1 ).
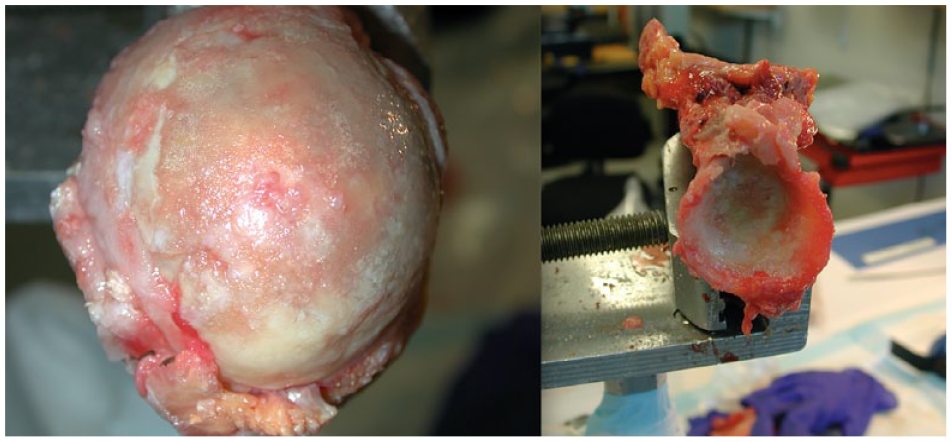
A humeral head (left) and glenoid specimen (right) that were excluded due to the presence of arthritic changes and abnormalities.
After dissection, each humeral head specimen was promptly sectioned using a sagittal saw. Each humeral head was cut into 5 mm sections in the coronal plane and then shortened at the anatomical neck. The glenoid was similarly prepared into 5-mm coronal sections and shortened. These sections were then individually photographed on both the anterior and posterior aspects using a digital camera. A metric ruler was photographed alongside each specimen for scale.
Measurement of the articular cartilage of each section was performed using the Image J program. For each humeral head section, a point corresponding to the center of the section was identified. A line was then drawn from the center of the section to the superior- and inferior-most articular surfaces ( Fig. 2 ). Division of the resulting angle by 4 and production of corresponding lines to the articular surface generated 5 points of measurement, namely, a superior, upper ¼, central, lower ¼, and inferior point ( Fig. 2 ). At each point, humeral cartilage thickness was measured perpendicularly to the tangential cartilage surface to the nearest 1/100th of a millimeter.

Identification of 5 measurement locations on the humeral head. Lines drawn from the superior and inferior most points of articular cartilage to the best fit center of the humeral head can be observed along with equal divisions creating three subsequent points.
For each glenoid section, the superior- and inferior-most points of articular cartilage were identified. A line that was parallel to the tangential plane at the deepest point of the section was constructed beginning and ending with the 2 points previously identified. The length of the line was then divided by 4 to yield the locations of measurement for the final 3 points ( Fig. 3 ). At each location, the cartilage thickness was again measured to the nearest 1/100th of a millimeter.
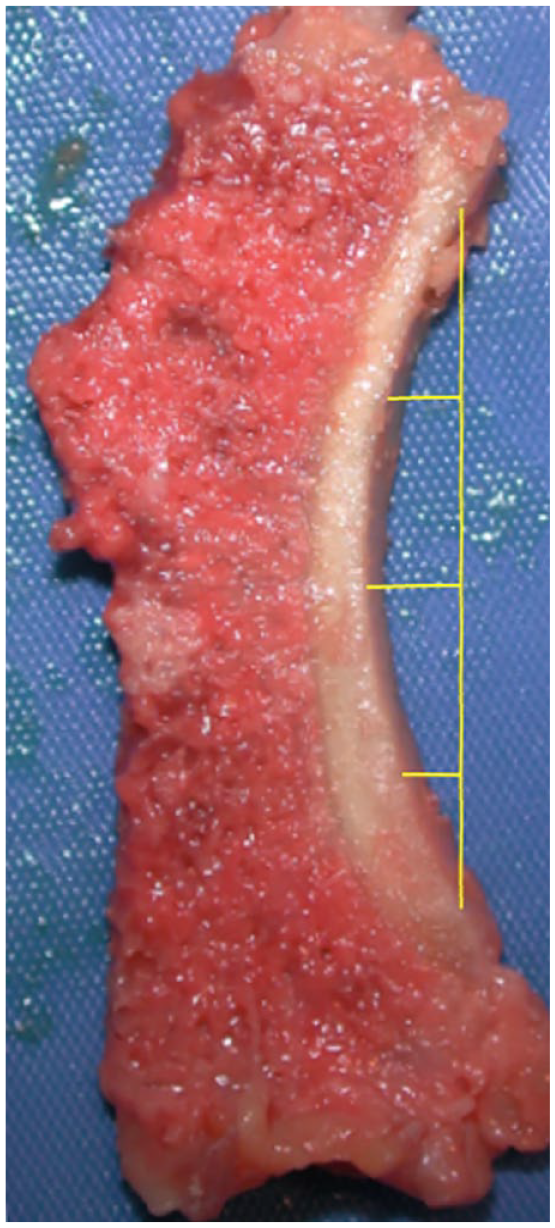
Identification of 5 measurement locations on the glenoid. A line drawn from the superior and inferior most points of articular cartilage can be observed. Equal divisions of its length creating three subsequent points can also be observed.
A waiver of approval was obtained from the University at Buffalo Institutional Review Board prior to starting the study.
Statistical Analysis
A 1-way analysis of variance (ANOVA) along with Tukey’s multiple comparisons test was performed to determine the differences present in cartilage thickness across the 5 points measured at the glenoid cavity and humeral head. An unpaired t test was performed to compare the mean cartilage thickness values of the glenoid cavity and humeral head between males and females and subjects aged 61-78 (n = 7, M = 70.29) and 79-95 (n = 9, M = 84.11) years. All statistical analyses were performed using GraphPad Prism (Version 9.0; GraphPad Software, San Diego, CA).
Results
An average of 5.38 sections (4-6) and 3.38 sagittal sections (2-4) were obtained from each humeral head and glenoid, respectively. In total, 80 data points were generated for each humeral head and each glenoid.
Humeral Head
The 1-way ANOVA revealed that the mean cartilage thickness values differed across the 5 locations measured on the humeral head—F(4, 75) = 3.702, P = 0.0084. The average thickness of the humeral head articular cartilage was 1.59 ± 0.36 mm. The thickest cartilage was found centrally (M = 1.77 ± 0.35 mm; Table 1 , Fig. 4 ), while the thinnest cartilage was found superiorly and inferiorly (M = 1.42 ± 0.37 mm, 1.42 ± 0.29 mm; Table 1 , Fig. 4 ). The average cartilage thickness was 1.74 ± 0.33 mm and 1.48 ± 0.35 mm for males and females, respectively. Tukey’s multiple comparisons test showed significant differences in cartilage thickness when comparing the superior and inferior regions to the middle (P = 0.0364, P = 0.0343; Table 2 ). There was no difference observed for cartilage thickness between subjects aged 61-78 (n = 7, M = 70.29) and 79-95 years (n = 9, M = 84.11) at the humeral head (P = 0.8277).
Articular Cartilage Measurements of the Humeral Head.
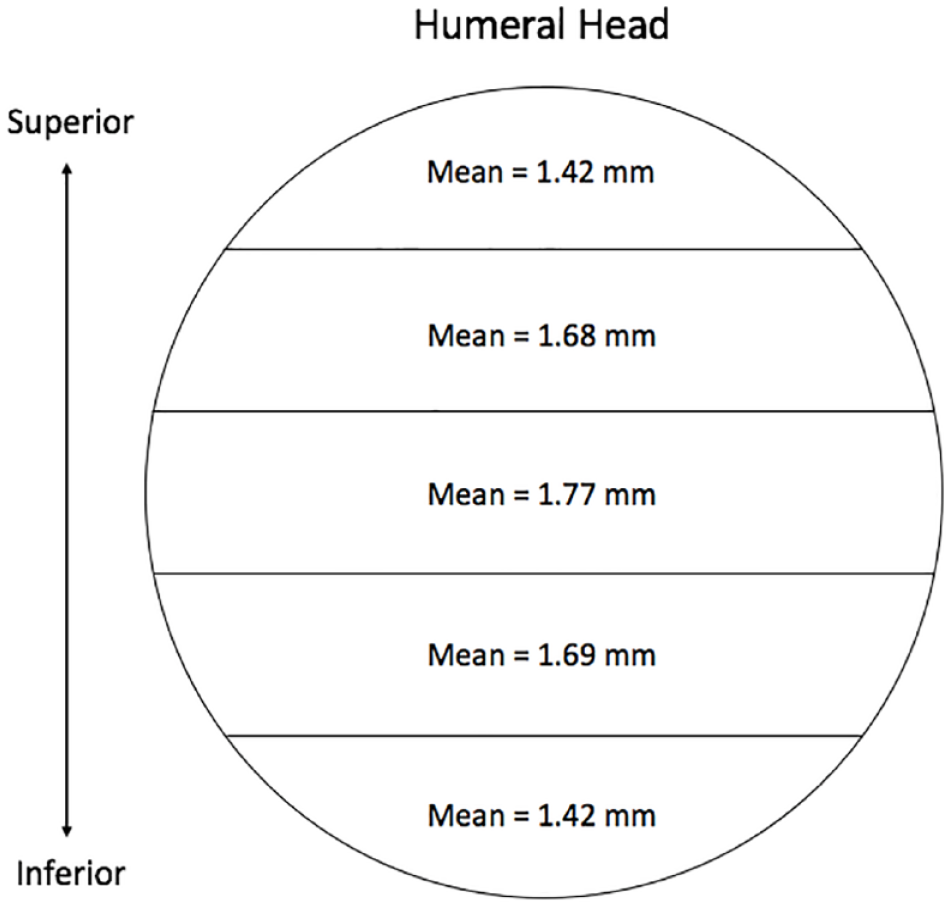
Anatomical depiction of cartilage thickness findings at the humeral head.
Significance (Lower Left-Hand Corner) and Mean Difference (mm) of Cartilage Thickness (Upper Right-Hand Corner) Between the 5 Locations of Measurement at the Humeral Head (n = 16 Humeral Heads).
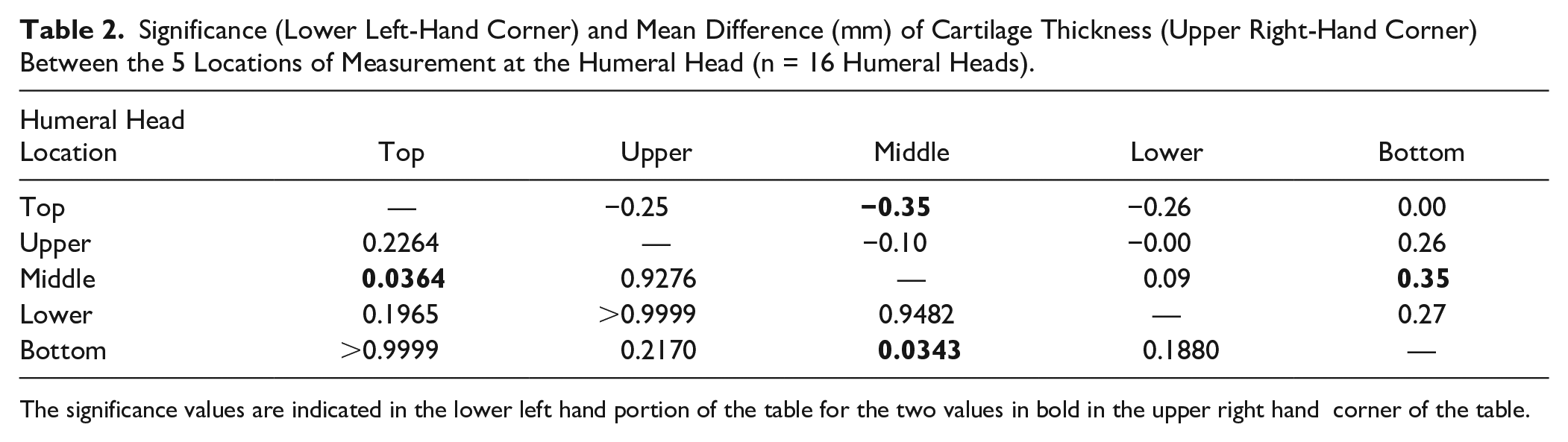
The significance values are indicated in the lower left hand portion of the table for the two values in bold in the upper right hand corner of the table.
Glenoid Cavity
The 1-way ANOVA revealed that mean cartilage thickness values differed across the 5 locations measured on the glenoid cavity—F(4, 75) = 13.96, P < 0.001. The average thickness of the glenoid articular cartilage was 2.14 ± 0.56 mm. The thickest cartilage was found in the superior and inferior areas (M = 2.61 ± 0.47 mm, 2.53 ± 0.58 mm; Table 3 , Fig. 5 ), while the thinnest cartilage was found centrally (M = 1.69 ± 0.22 mm; Table 3 , Fig. 5 ). The average cartilage thickness for males was 2.31 ± 0.54 and 2.00 ± 0.54 mm for females. Tukey’s multiple comparisons test showed significant differences when comparing the superior and inferior areas to those areas in between on the glenoid cavity (P ranging from <0.001 to 0.0026; Table 4 ). There was no difference observed for cartilage thickness values between subjects aged 61-78 (n = 7, M = 70.29) and 79-95 years (n = 9, M = 84.11) at glenoid cavity (P = 0.2366).
Articular Cartilage Measurements of the Glenoid Cavity.
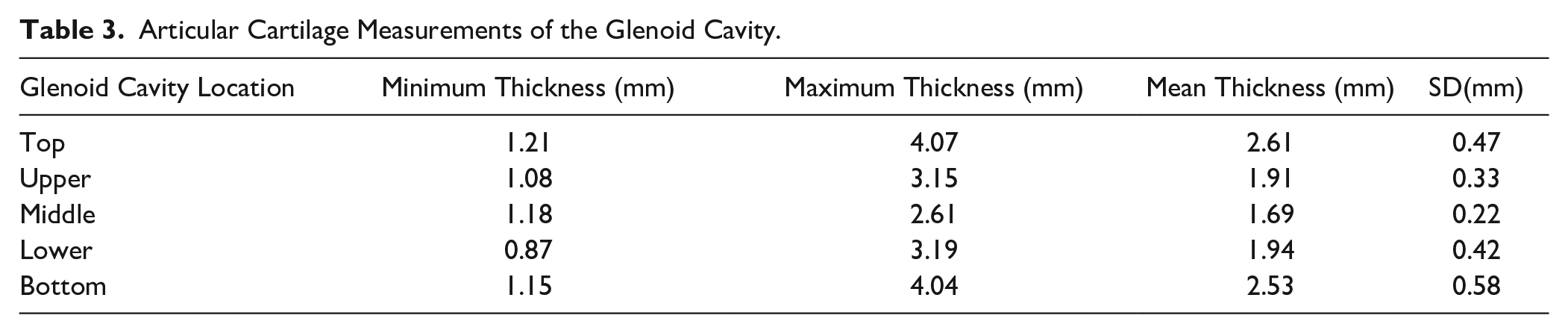
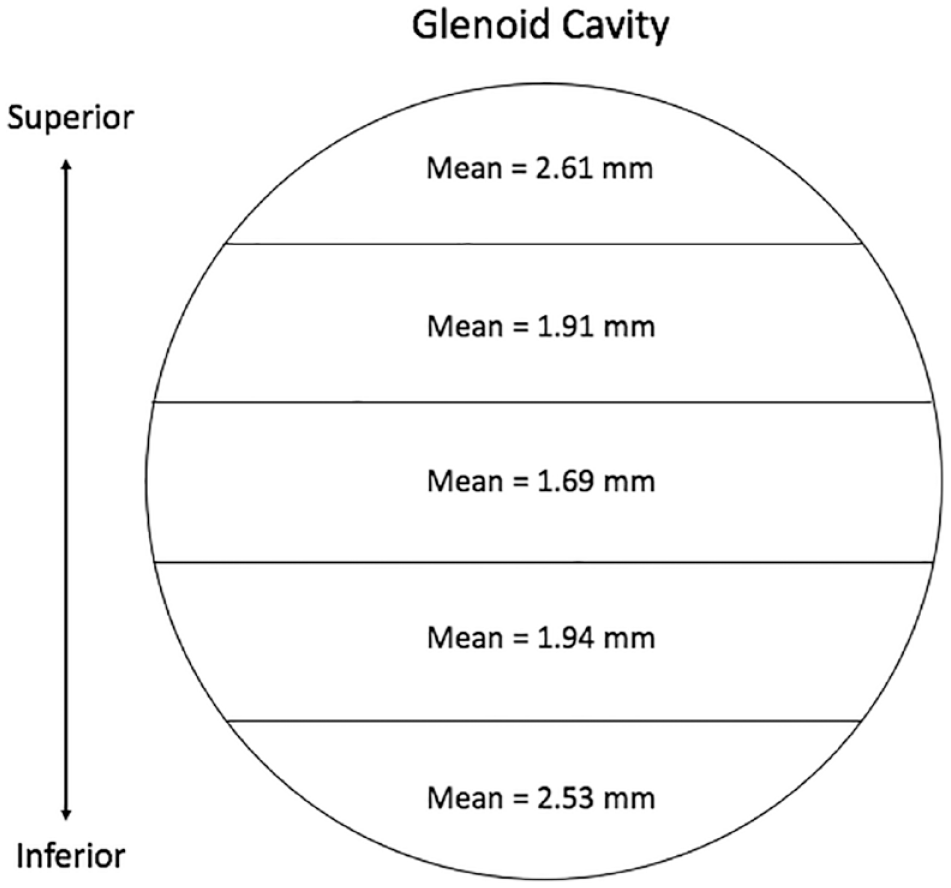
Anatomical depiction of cartilage thickness findings at the glenoid cavity.
Significance (Lower Left-Hand Corner) and Mean Difference (mm) of Cartilage Thickness (Upper Right-Hand Corner) Between the 5 Locations of Measurement at the Glenoid Cavity (n = 16 Glenoid Cavities).
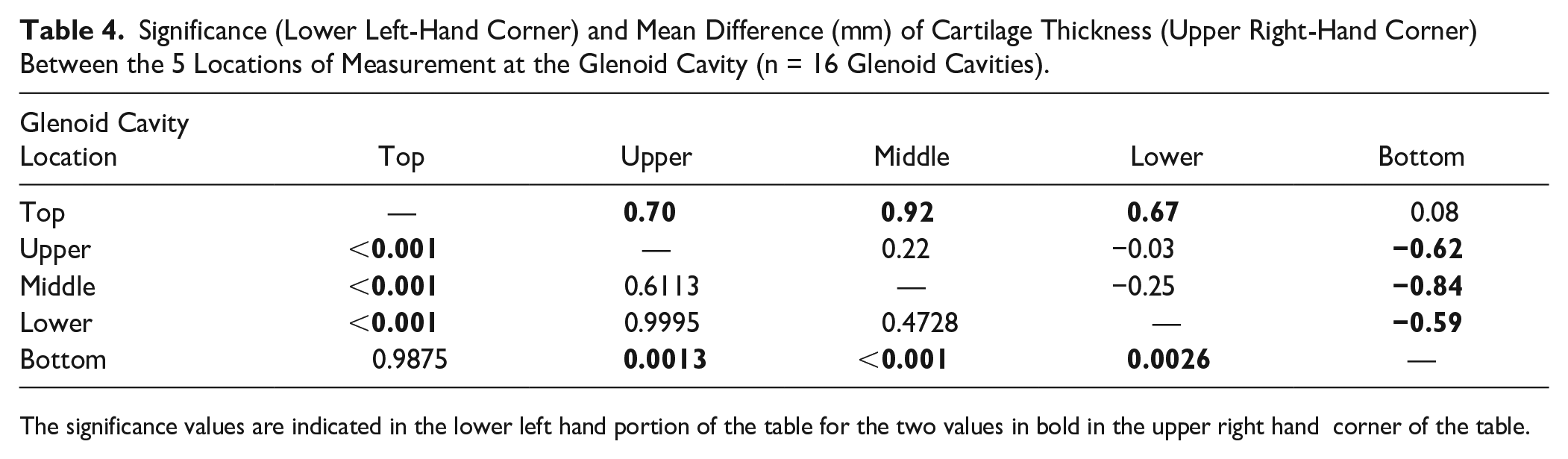
The significance values are indicated in the lower left hand portion of the table for the two values in bold in the upper right hand corner of the table.
After comparing regional cartilage thickness values between the humeral head and the glenoid cavity, a reciprocal pattern was discovered. Humeral head cartilage is thickest centrally and is decreased superiorly and inferiorly. By comparison, the thickest cartilage of the glenoid is found superiorly and inferiorly and is decreased centrally. The range in thickness from minimum to maximum, however, was larger in the glenoid than the humeral head ( Tables 1 and 3 ). The cartilage of the humeral head increased, on average, only 0.35 mm from its superior and inferior aspects. The change in thickness of the glenoid was significantly larger, progressively increasing 0.88 mm, on average, from its central to superior and inferior aspects.
Males versus Females
Finally, we compared the average thickness of the humeral head and glenoid cartilage between males and females, and discovered a significant difference. Although the patterns of distribution were similar between the two, specimens from males were found to have thicker cartilage in both the humeral head and glenoid (P = 0.0014, P = 0.0133).
Discussion
A thorough understanding of glenohumeral joint anatomy is critical for prosthetic design and successful shoulder arthroplasty. Multiple studies have previously described components of this relationship, including the role of articular cartilage thickness, producing a range of results.10 -14 Considering the inconsistencies of previously reported values, we sought to define glenoid and humeral cartilage thickness in fresh, nonarthritic cadaveric specimens. Our study is the first to assess the cartilage thickness of both the humeral head and glenoid in corresponding, fresh, unfrozen, and unprocessed specimens by direct measurement. These measurements are more accurate than other techniques such as MR arthrography, which have been shown to either overestimate or underestimate thickness.15,16
The results of our study demonstrate that in the humeral head, the thickest cartilage is found in the central area and decreases progressively as you move away from this point. The opposite pattern is seen in the glenoid, which has thicker cartilage overall along with a greater range of thickness compared with the humeral head. Cartilage in male specimens was also found to be significantly thicker than that in female specimens for both components.
Fox et al. 11 previously used an anatomical sectioning process similar to ours to study humeral head cartilage in 16 embalmed specimens. They reported an average thickness of 0.89 mm, with the thickest cartilage found in the central portion. 11 This was significantly thinner than prior results reported by Soslowsky et al., 10 who, using stereophotogrammetry, found the average thickness to be 1.44 mm and 2.16 mm for the humeral head and glenoid, respectively. Despite similarities in methodology, the results of our study suggest that the humeral head cartilage is significantly thicker than that described by Fox et al. 11 and support the average thickness reported by Soslowsky et al. 10 This discrepancy is possibly a product of differences in specimen processing. Fox et al. 11 generated sections from embalmed specimens after acrylic potting and staining with methylene blue, whereas in this study, fresh cadaveric specimens were sectioned and measured immediately after dissection.
Graichen et al. 12 conducted a validation study of quantitative MRI cartilage thickness measurements against those of A-mode ultrasound. They reported a mean cartilage thickness by quantitative MRI of 1.2 and 1.7 mm for the humeral head and glenoid cavity, respectively. 12 However, they found mean cartilage thickness values by A-mode ultrasound of 1.4 and 2.2 mm at the humeral head and glenoid, respectively. Schleich et al. 13 also investigated cartilage thickness at the joint using MRI and found mean thickness values ranging from 1.2 to 1.5 mm. Our results of mean cartilage thickness at both the humeral head and glenoid (1.59 and 2.14 mm) are in disagreement with those obtained from MRI by these 2 studies. However, our results and those presented by Soslowsky et al. 10 are in agreement with the results presented by Graichen et al. 12 using A-mode ultrasound. Zumstein et al. 14 investigated cartilage thickness at the joint using computed tomography (CT) and reported mean cartilage thickness values of 1.74 and 1.93 mm for the humeral head and glenoid cavity, respectively. Their results show a thicker humeral head cartilage and thinner glenoid cartilage compared with the results presented in this study along with those by Soslowsky et al. 10 and the A-mode ultrasound results from Graichen et al. 12
Our finding that maximal cartilage thickness is centrally located on the humeral head is in agreement with Fox et al. 11 and supported by Zumstein et al., 14 who used CT to identify the superocentral portion of the humeral head and the anteroinferior portion of the glenoid as areas of maximal thickness. Together these results support the conclusion that the articular cartilage thickness of the glenoid and humeral head is nonuniform, reciprocal in nature, and complementary in creating joint articulation.
Our data are the first to explicitly suggest significant sex-based differences in glenohumeral cartilage thickness, which were not described by Fox et al. 11 and found to be statistically insignificant by Zumstein et al. 14 However, Merrill et al. 17 noted significant gender-related differences in other aspects of glenoid anatomy, while Tackett and Ablove 18 described scale differences in other parameters between male and female shoulders. Additional studies are necessary to make a definitive assessment, but overall these findings support further exploration of sex-specific criteria in evaluation of joint health and prosthetic design. Furthermore, our reported sex-based differences in cartilage thickness suggest that donors should be matched to recipients based on sex for OCA transplantation among other factors.
We observed no statistical difference in cartilage thickness values between subjects aged 61-78 (n = 7, M = 70.29) and 79-95 years (n = 9, M = 84.11) for both the glenoid cavity and humeral head (P = 0.2366, P = 0.8277). This finding gives us further confidence in our reported sex-based difference in cartilage thickness at both locations when the average age of males and females is taken into account (M = 73.6 and 81.6). Our report of no difference based on age is in agreement with the results presented by Meachim 19 who found no significant cartilage thickness difference at the humeral head in subjects ranging from 25 to 75 years old. Interestingly, they found a nonsignificant increase in cartilage thickness in specimens presenting with early degenerative changes of the articular surface. 19 The results presented by Meachim et al. are further supported by Petersson 20 who found no changes in cartilage thickness with age when looking at 51 shoulders ranging in age from 18 to 92 years. It is important to note that Petersson did find an increasing frequency of degenerative changes with age. 20 This finding by Petersson is further supported by Kobayashi et al. 21 who found a prevalence of osteoarthritis in those younger than 65 years to be 11.1% and in those older than 65 years to be 20.3%. However, cartilage degeneration in primary osteoarthritis most commonly affects the central aspect of the humeral head and posterior aspect of the glenoid.22 -24 We would have expected for this to be evident in our data if our specimens had signs of osteoarthritis.
Although our data are consistent with multiple prior results, the anatomical nature of our study has several limitations. Despite findings by Meachim 19 and Petersson, 20 there is concern that our sample (average age 78 years) may not be truly representative of the population as a whole. In addition, laterality was not taken into consideration for our measurements on both the glenoid and humeral head specimens. Similarly, our data correspond only to the cartilage thickness values observed superiorly and inferiorly along both anatomical locations. Finally, although specimens with gross evidence of arthritis were excluded, we consider unidentified degenerative processes a potential source of error.
Conclusion
Cartilage thickness of the humeral head and the glenoid has a complementary relationship. Humeral head cartilage is thickest in its central-most portion and becomes symmetrically thinner superiorly and inferiorly. Reciprocally, the glenoid cartilage is thinnest in its central portion. Glenoid cartilage is thicker than humeral head cartilage and varies more widely from maximum to minimum. These relationships are maintained in both males and females, although the cartilage in males is significantly thicker overall irrespective of the age. These results can be used to further inform prosthetic design and OCA transplantation.
Footnotes
Authors’ Note
Location of investigation: Department of Orthopaedics, University at Buffalo, Buffalo, NY 14215, USA.
Acknowledgments and Funding
We would like to thank Craig Howard for his assistance in the sectioning and imaging of shoulder specimens. The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was granted by the University at Buffalo Institutional Review Board (Federalwide Assurance ID#: FWA00008824).
