Abstract
Objective
Hyaline cartilage has limited innate healing abilities and hyaline cartilage loss is a hallmark of osteoarthritis (OA). Animal models can provide important insights into cartilage regeneration potential. One such animal model, the African spiny mouse (Acomys), is capable of regenerating skin, skeletal muscle, and elastic cartilage. This study aims to evaluate whether these regenerative abilities protect Acomys with meniscal injury from OA-related joint damage and behaviors indicative of joint pain and dysfunction.
Design
Acomys received destabilization of the medial meniscus (DMM) surgery (n = 11) or a skin incision (n = 10). Gait testing occurred at 4, 6, 8, 10, and 12 weeks after surgery. At endpoint, joints were processed for histology to assess cartilage damage.
Results
Following joint injury, Acomys with DMM surgery altered their walking patterns by increasing the percent stance time on the contralateral limb relative to the operated limb, thereby reducing the amount of time the injured limb must bear weight on its own throughout the gait cycle. Histological grading indicated evidence of OA-related joint damage in Acomys with DMM surgery; these changes were primarily driven by loss of structural integrity in the hyaline cartilage.
Conclusions
Acomys developed gait compensations, and the hyaline cartilage in Acomys is not fully protected from OA-related joint damage following meniscal injury, although this damage was less severe than that historically found in C57BL/6 mice with an identical injury. Thus, Acomys do not appear to be completely protected from OA-related changes, despite the ability to regenerate other wounded tissues.
Keywords
Introduction
Osteoarthritis (OA) is a degenerative joint disease characterized by cartilage breakdown and maladaptive joint remodeling, ultimately leading to pain and disability. 1 Traditionally, clinical OA diagnoses are based on radiographic evidence of joint space narrowing 2 ; however, there is discordance between the structural signs of OA and patient reports of painful symptoms. 3 Radiographs alone are poor predictors of pain and disability, as patients often report high levels of pain without severe radiographic evidence of OA-related pathological changes. 4 Ultimately, pain and loss of joint function drive patients to seek clinical care; thus, OA is holistically diagnosed based on patient history, pain experience, and physical examination, along with radiographic evidence of joint damage. 5
Hyaline cartilage loss is a hallmark of OA. Because hyaline cartilage is an avascular, alymphatic, and aneural tissue, it has limited innate healing abilities. 6 Once a joint injury or trauma initiates cartilage damage, the joint fails to replace the damaged cartilage with functional tissue, leading to the development of OA.7,8 Traditional cartilage repair strategies include marrow stimulation, autografts, and allografts 9 ; however, these techniques are limited, as the repaired tissue has inferior structural, chemical, and mechanical properties compared to healthy cartilage. 10 Furthermore, the repaired tissue often fails to fully integrate with the native cartilage and bone. 11 As such, current cartilage repair and regeneration strategies are not feasible to prevent and treat OA, creating a need for new approaches to advance cartilage repair and regeneration.
Animal models provide important insights into cartilage regeneration potential. For example, Murphy Roth Large (MRL/MpJ) mice regenerate properly aligned and functional elastic cartilage following small ear hole punches.12,13 Furthermore, following meniscal injury, MRL/MpJ mice are relatively protected from OA-related joint changes compared to C57BL/6 mice. 14 While the MRL/MpJ strain was produced through breeding, the African spiny mice (Acomys) have incredible natural wound-healing abilities. 15 Acomys fully close 4-mm ear hole punches, including the elastic cartilage; moreover, Acomys are capable of regenerating skin and skeletal muscle.16 -18 Acomys also have several human-like qualities (e.g., susceptibility to diabetes, 19 organogenesis before birth, 20 menstruation 21 ), making them exciting organisms to study within regenerative medicine. 15 It is unknown whether regeneration in Acomys extends to hyaline cartilage and whether Acomys are protected from OA damage following joint injury; however, their innate regenerative abilities suggest Acomys may be a new preclinical model to explore cartilage protection and repair.
Our study aims to evaluate whether Acomys with meniscal injury are protected from developing OA-related structural joint changes and behaviors indicative of joint pain and dysfunction. To achieve this objective, we evaluated gait changes and endpoint histological outcomes in Acomys following destabilization of the medial meniscus (DMM). In doing so, our study explores Acomys as a new model with the potential for cartilage protection and repair following joint injury.
Methods
Animals and Study Design
At 16 weeks old, 21 Acomys (n = 14 male, n = 7 female) were received from a breeding colony at the University of Florida. Animals were group-housed (2-5 per cage) with ad libitum access to food, water, and enrichment objects in an atmosphere-controlled room with a 12-hour light/dark cycle. Acomys were fed a standard rodent chow diet, supplemented with seeds, vegetables, and fruits. The experimental design for this study is shown in Supplementary Figure S1. Acomys were randomly distributed between surgical groups and received either DMM surgery or a skin-incision sham (n = 11 DMM, n = 10 skin incision, with males and females evenly distributed between groups). Gait was evaluated at baseline and at 4, 6, 8, 10, and 12 weeks after surgery. An a priori sensitivity analysis with 21 total samples, 2 groups, α = 0.05, and power = 0.8 determined an effect size (f) of 0.23 would be detectable for 6 measurements (G-Power). One animal was euthanized due to post-surgery complications and one animal was euthanized after developing diabetes; these 2 animals were eliminated from the study, resulting in an estimated detectable effect size (f) of 0.24. All methods described were approved by the University of Florida Institutional Animal Care and Use Committee and in accordance with the Association for Assessment and Accreditation of Laboratory Animal Care recommendations on animal research.
Animal Surgeries
Prior to surgery, Acomys were anesthetized in a 4% isoflurane sleep box. Animals were transferred to a 1% to 2% isoflurane mask to maintain anesthesia. Then, the right limb was aseptically prepared for surgery via 3 repeated washes of povidone-iodine and 70% alcohol, ending with a fourth application of povidone-iodine; animals were then transferred to a sterile field. For all surgeries, a medial parapatellar ligament incision was made on the right hind limb, and the fat pad was blunt-dissected to reveal the medial meniscus and the medial meniscotibial ligament. At this point, the surgeon was notified whether the procedure was a skin-incision sham or DMM surgery; for sham surgeries, the muscle and skin were closed with polydioxanone absorbable suture (PDS). For the DMM Acomys, the medial meniscotibial ligament was transected and the incisions were closed in the same way as the sham Acomys. During post-surgical inspection, VetBond (3M, cat. no. 1469SB) was applied upon veterinarian request to ensure the incisions remained closed. All Acomys received buprenorphine (0.05 mg/kg, subcutaneous) prior to surgery and then every 12 hours out to 48 hours post-surgery.
Gait Testing
With our custom gait analysis system, 22 spatiotemporal gait data were collected for all Acomys at 4, 6, 8, 10, and 12 weeks post-surgery. The setup consists of a 40-inch-long × 3.75-inch-wide arena resting on top of a transparent floor. A mirror angled 45° below the arena floor creates an unobstructed view of the lateral and ventral planes while an animal walks. At each timepoint, animals were individually placed in the gait arena to freely explore without incentives or external stimuli. High-speed videos (500 frames/s, Phantom Miro Lab 320) were recorded when animals walked across the arena with a consistent velocity. At each timepoint, the animal order for gait testing was random and a minimum of 10 gait trials were collected for each animal. Gait videos were processed to calculate spatiotemporal gait parameters (velocity, stance time, stride length, step width, temporal symmetry, and spatial symmetry). Gait data were screened for trials with velocities between 20 and 75 cm/s and at least 2 complete gait cycles, resulting in 1,016 trials for data analysis. Please see our group’s prior methodological review for further details on rodent gait characterization.23,24
Histology
At 12 weeks post-surgery, Acomys were euthanized via CO2 inhalation and decapitation, and hind limbs were collected for histology. Joints were fixed in 10% neutral buffered formalin for 24 hours, decalcified in 10% formic acid for 5 days, dehydrated in ethanol, and paraffin-embedded via vacuum infiltration. Sagittal sections were taken at 5 μm increments along the medial compartment of the knee, starting along the medial aspect through to the anterior and posterior cruciate ligaments. Sections were stained with safranin-O/fast green.
Using histological sections, the medial tibial compartment was evaluated with the modified Mankin grading scheme to assess degenerative joint changes. 25 In addition to these semi-quantitative grades, histological sections were evaluated by segmenting medial tibial cartilage and subchondral bone features to create binarized masks (Suppl. Fig. S2). These masks were used to calculate quantitative histological features including cartilage area, cartilage thickness, subchondral bone area, and bone marrow area. Sections were evaluated by blinded graders for all histological analyses. For each joint, 3 sections were graded and the section with the worst overall score was used for statistical analysis of modified Mankin scores. For quantitative histological features, the 3 sections per joint were averaged together for statistical analysis.
Statistical Analysis
Statistical analyses were conducted using R (version 4.0.4). Gait data were analyzed with linear mixed-effects models. For gait parameters that do not vary between limbs (velocity, stride length, step width, and symmetries), surgery and timepoint were treated as fixed effects and animal was treated as a random effect. For gait parameters that vary between limbs (stance time), surgery and limb were concatenated to create a surgery-limb factor. Velocity was also included as a fixed effect for velocity-dependent gait parameters (stance time, stride length, and step width). For linear mixed-effects models, type III analysis of variance (ANOVA) with Satterthwaite’s method was used to analyze the significance of fixed effects (P < 0.05 considered significant). When indicated by the ANOVA, Tukey’s post hoc comparisons of least squared means were used to evaluate specific differences. In addition, Bonferroni-corrected t tests were used to evaluate whether gait patterns were symmetric.23,24 For histological data, surgery and limb were concatenated to create 1 surgery-limb factor with 4 levels. The nonparametric modified Mankin scores were analyzed with pairwise comparisons using the Mann-Whitney U test and a Benjamini-Hochberg correction for multiple comparisons. Quantitative histological data were analyzed with a 1-way ANOVA and a Tukey’s HSD (honestly significant difference) post hoc test, when indicated.
Results
Velocity
There were no velocity differences between DMM and skin-incision Acomys, indicating surgery and subsequent OA progression did not affect walking speeds ( Fig. 1 ). Time after surgery was significant (P < 0.0001), with velocities increasing from baseline to week 12. This velocity increase over time is typical in rodents and has been previously observed in other rodent gait studies.26,27 Here, velocities increased from 39.64 ± 2.12 cm/s at baseline to 47.88±2.22 cm/s by week 12 (error presented as ±95% confidence interval).
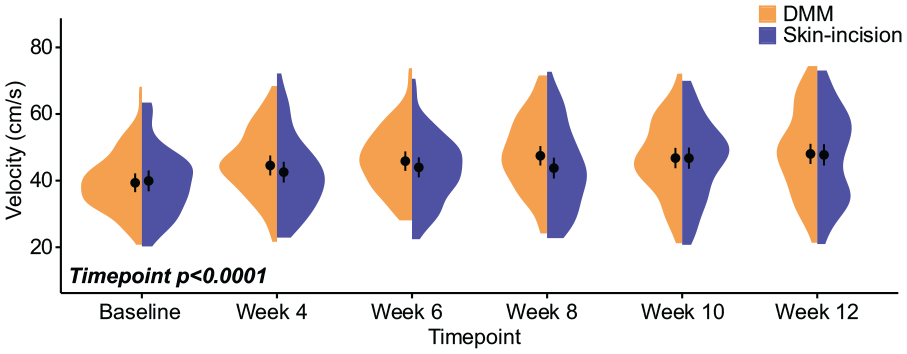
Walking velocities in Acomys following meniscal injury. Velocities for all animals are shown as half violin plots, where circles represent mean velocity and bars represent 95% confidence intervals. While velocity increased over time, there were no velocity differences between DMM and skin-incision animals. DMM = destabilization of the medial meniscus.
Percent Stance Time
While walking, Acomys with DMM surgery varied their stance times between the operated (right) and contralateral (left) limbs ( Fig. 2 ). In DMM Acomys, percent stance times (amount of time the limb is in ground contact during the length of the gait cycle) were greater on the contralateral limb compared to the operated limb (P = 0.0017), indicating DMM Acomys spent more time on the uninjured limb than the injured limb while walking. Across all animals, Acomys with DMM surgery had increased contralateral limb percent stances times compared to skin-incision animals (P = 0.012), indicating DMM Acomys also spent more time on the contralateral limb compared to sham control Acomys. Stance times generally decreased over time (P < 0.0001), although these shifts between timepoints were similar for each experimental group.
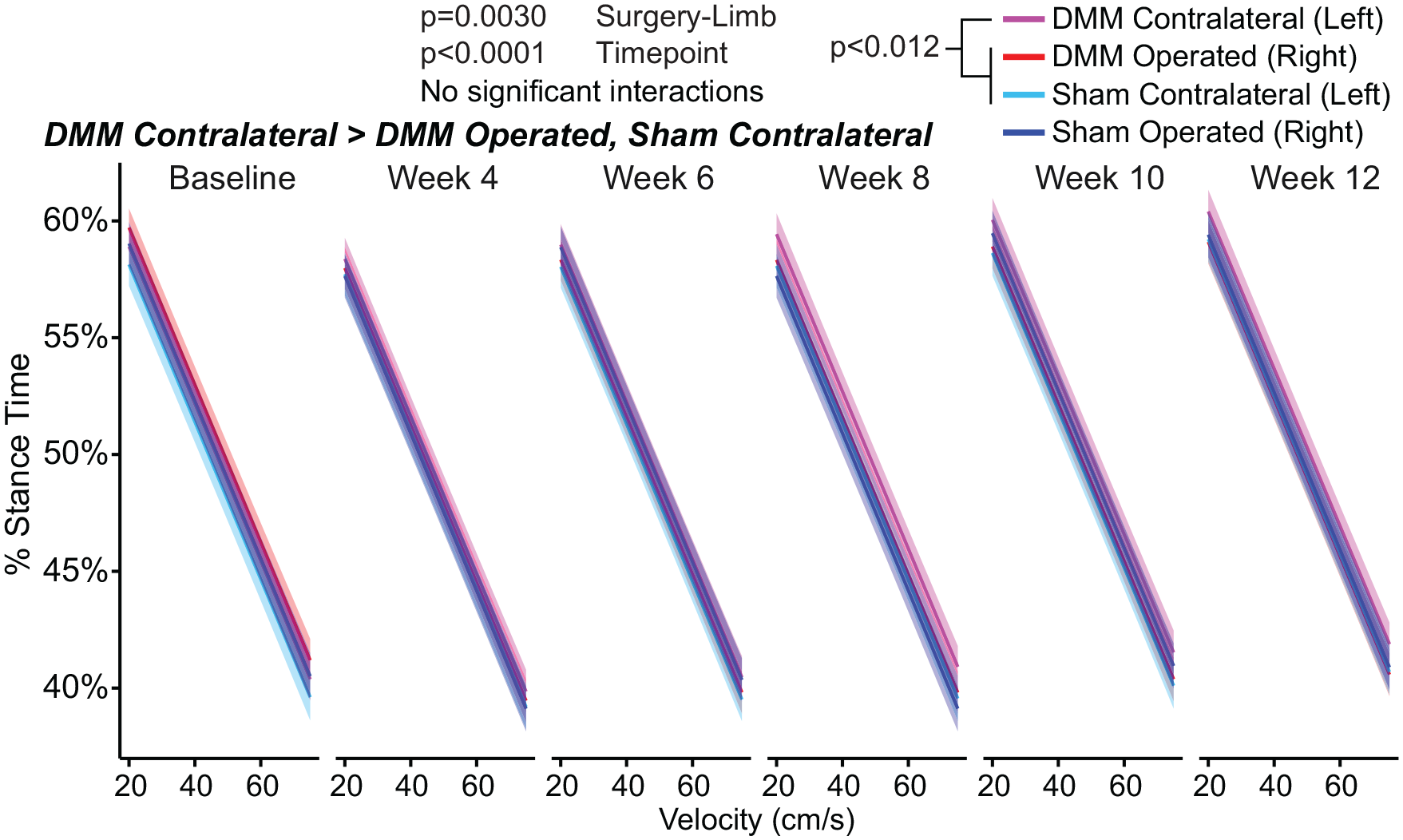
Stance times in Acomys following meniscal injury. Percent stance time describes the time the foot is in contact with the ground (stance time) during a complete gait cycle (stance time + swing time). Here, percent stance time is plotted against velocity with 95% confidence bands. Significant main effects for percent stance time include surgery-limb and timepoint. Percent stance time on the DMM contralateral (left) limb was greater than stance time on the DMM-operated (right) limb and the sham contralateral (left) limb. DMM = destabilization of the medial meniscus.
Foot Placement
At specific timepoints, stride lengths and step widths varied between DMM and skin-incision Acomys ( Fig. 3 ). At week 8, stride lengths were shorter for DMM Acomys compared to skin-incision Acomys (P = 0.0089, Fig. 3A ). At week 6, step widths were reduced for DMM Acomys compared to skin-incision Acomys (P = 0.030, Fig. 3B ). For all animals, stride lengths and step widths generally increased over time (P = 0.043 for stride length; P < 0.0001 for step width). However, again, shifts between timepoints were similar for each group.
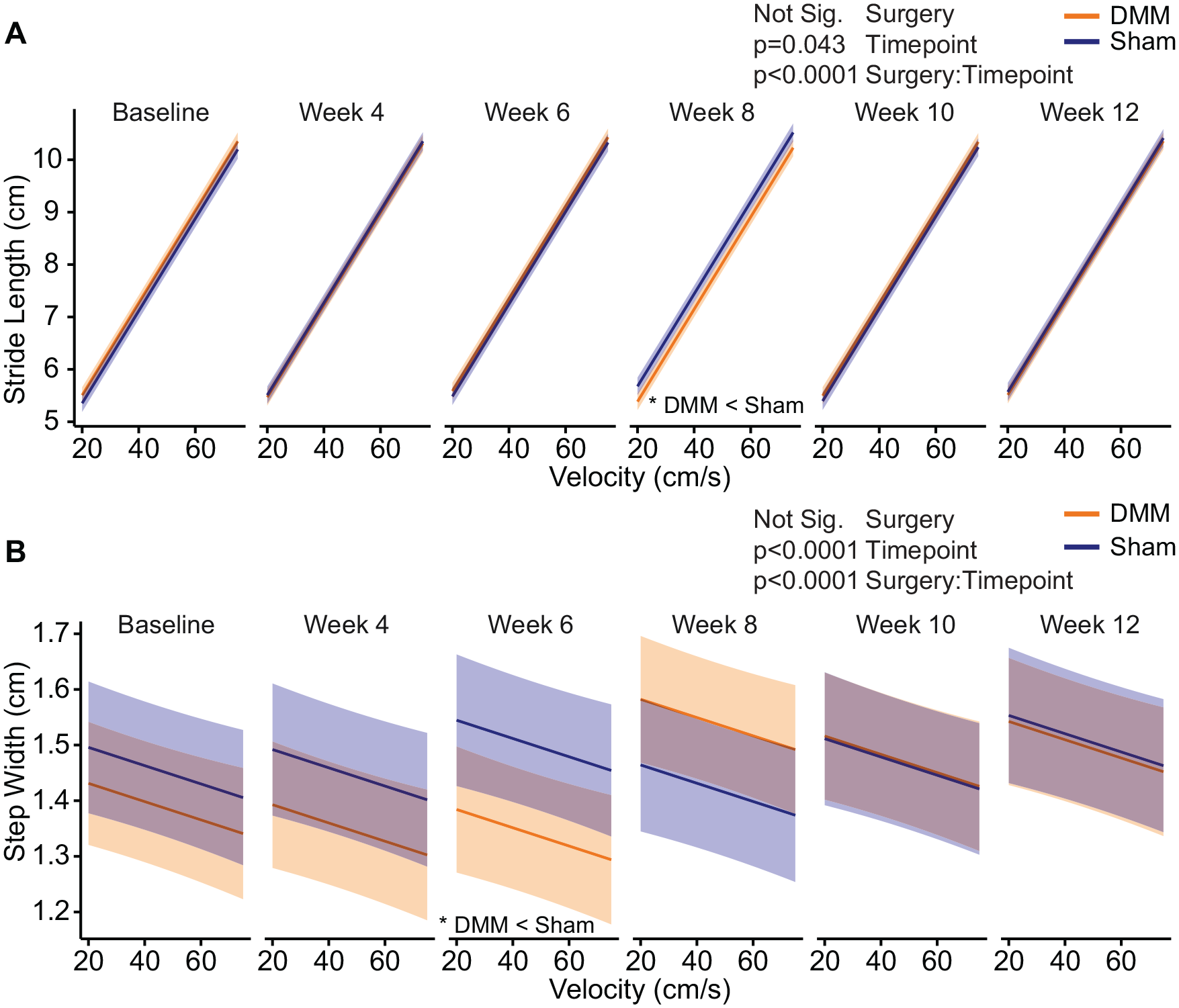
Stride lengths and step widths in Acomys following meniscal injury. Stride length (
Symmetry
Temporal symmetry measures the timing of foot-strikes throughout a gait cycle and spatial symmetry measures the spacing of footsteps. For both temporal and spatial gait symmetries, 0.5 is the mathematical definition of a symmetric gait pattern and values above or below 0.5 indicate an asymmetric gait pattern.23,24 Here, there were no instances of temporal or spatial asymmetry for either the DMM or skin-incision groups across all timepoints ( Fig. 4 ), indicating that distinct limps were not evident following meniscal injury. However, inspection of the distribution of data via half violin plots suggests that temporal symmetry is increasing above 0.5 in DMM Acomys at weeks 10 and 12.
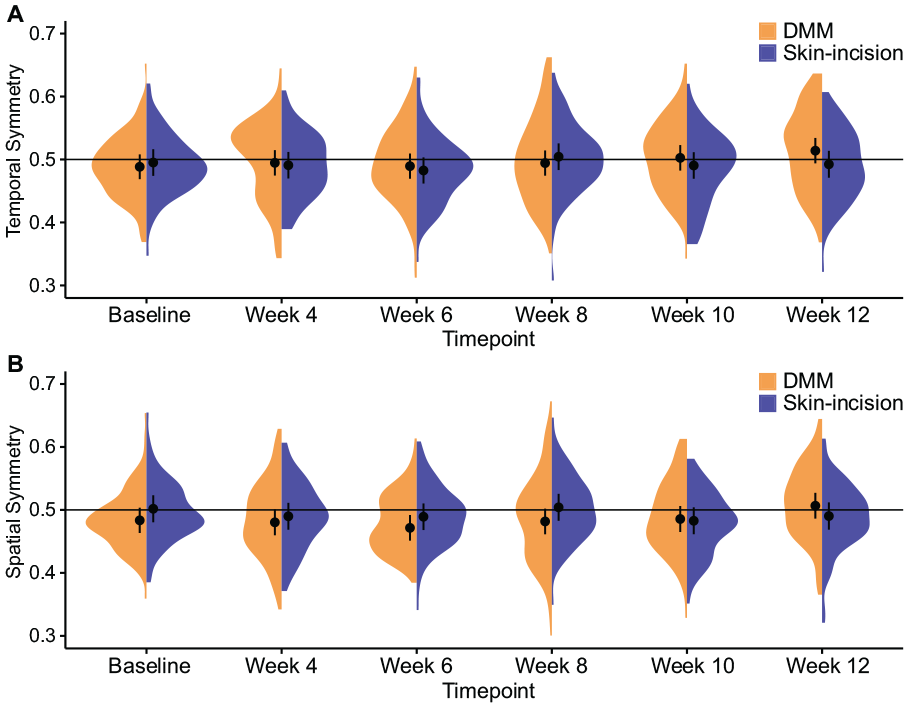
Gait symmetries in Acomys following meniscal injury. Temporal (
Histology
Representative histological images of the medial tibial compartment for the operated and contralateral knees are provided in Figure 5 ; modified Mankin scoring is provided in Figure 6 . In operated (right) DMM knees, some damage was observed in the tibial cartilage, including fissures and fibrillations along the surface. Mankin sums indicated more joint damage in DMM-operated knees compared to DMM contralateral knees (P = 0.049) and skin-incision right knees (P = 0.049, Fig. 6A ). When the Mankin sum was reduced to individual component scores, surgery-limb differences were primarily driven by cartilage structure (P = 0.0043, Fig. 6B ). Here, tibia cartilage structural damage in the DMM-operated knees was more severe than in the DMM contralateral knees (P = 0.011) and trending toward more severe compared to the skin-incision-operated and contralateral knees (P = 0.051 for both). For safranin-O staining scores (where higher scores indicate more proteoglycan loss), there were no significant differences for surgery-limb ( Fig. 6C ).
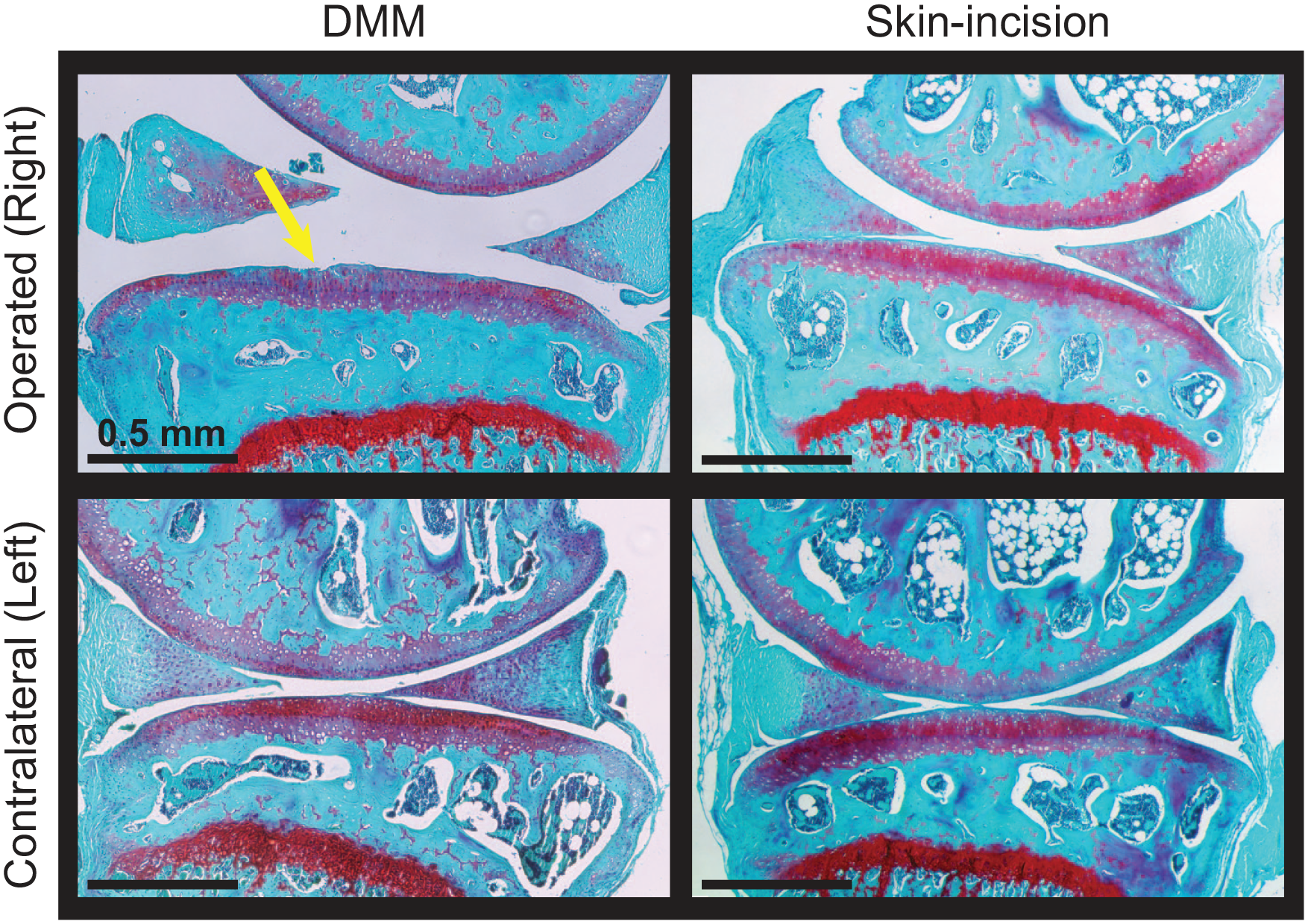
Representative histological images following meniscal injury in Acomys. Images are in the sagittal plane of the medial compartment for the operated (right) and contralateral (left) knees. Some cartilage damage, including small surface fissures, were observed in DMM right knees (yellow arrow). Scale bar = 0.5mm. DMM = destabilization of the medial meniscus.
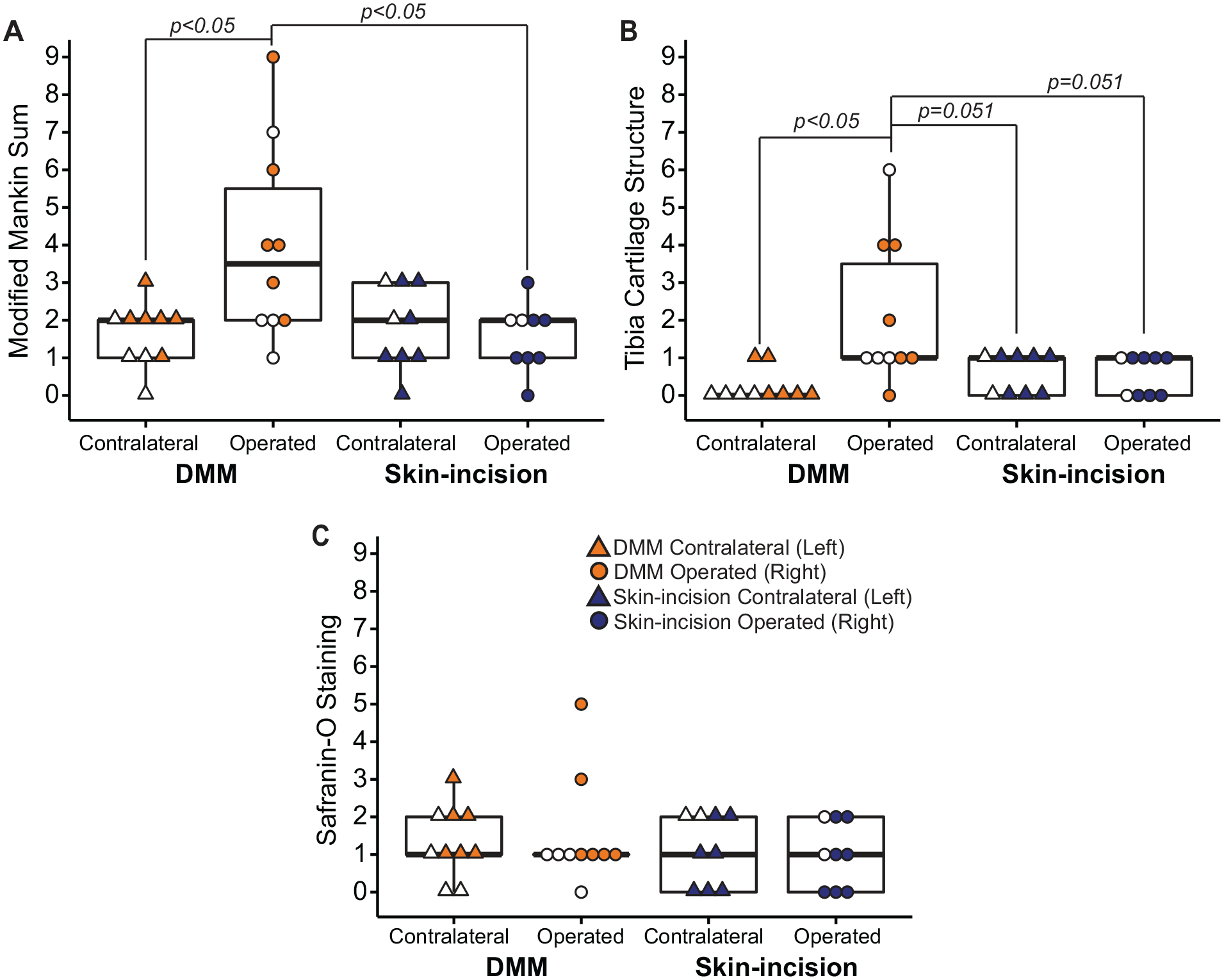
Modified Mankin scores of histological sections from Acomys following meniscal injury. The medial tibial compartment was graded for operated and contralateral knees. Relative to the contralateral knee and the skin-incision-operated knee, DMM-operated knees had more joint damage, as evidenced by higher Mankin sums (
In addition to semi-quantitative modified Mankin grading, quantitative histological grading is provided in Figure 7 . Quantitative histological grading showed no significant differences in tibial cartilage area across all knees, despite the presence of fibrillations and fissures in some DMM-operated knees ( Fig. 7A ). This indicates there was no significant cartilage loss in the DMM-operated knee, despite loss of structural integrity at the tibial cartilage surface. Verifying this observation, the total affected tibial cartilage surface was greater in DMM-operated knees compared to all other knees (P < 0.0005, Fig. 7B ). For average tibial cartilage thickness, the F test for the 1-way ANOVA was significant (P = 0.045); however, the follow-up Tukey’s HSD post hoc test was not significant for any pairwise comparison of means ( Fig. 7C ). Finally, there were no significant differences in bone marrow ratios (bone marrow area divided by total trabecular bone plus marrow area) across all knees ( Fig. 7D ). Here, bone marrow ratios closer to 0 would indicate more severe histological damage, as the increased bone density and decreased marrow space suggest the bone is sclerotic.
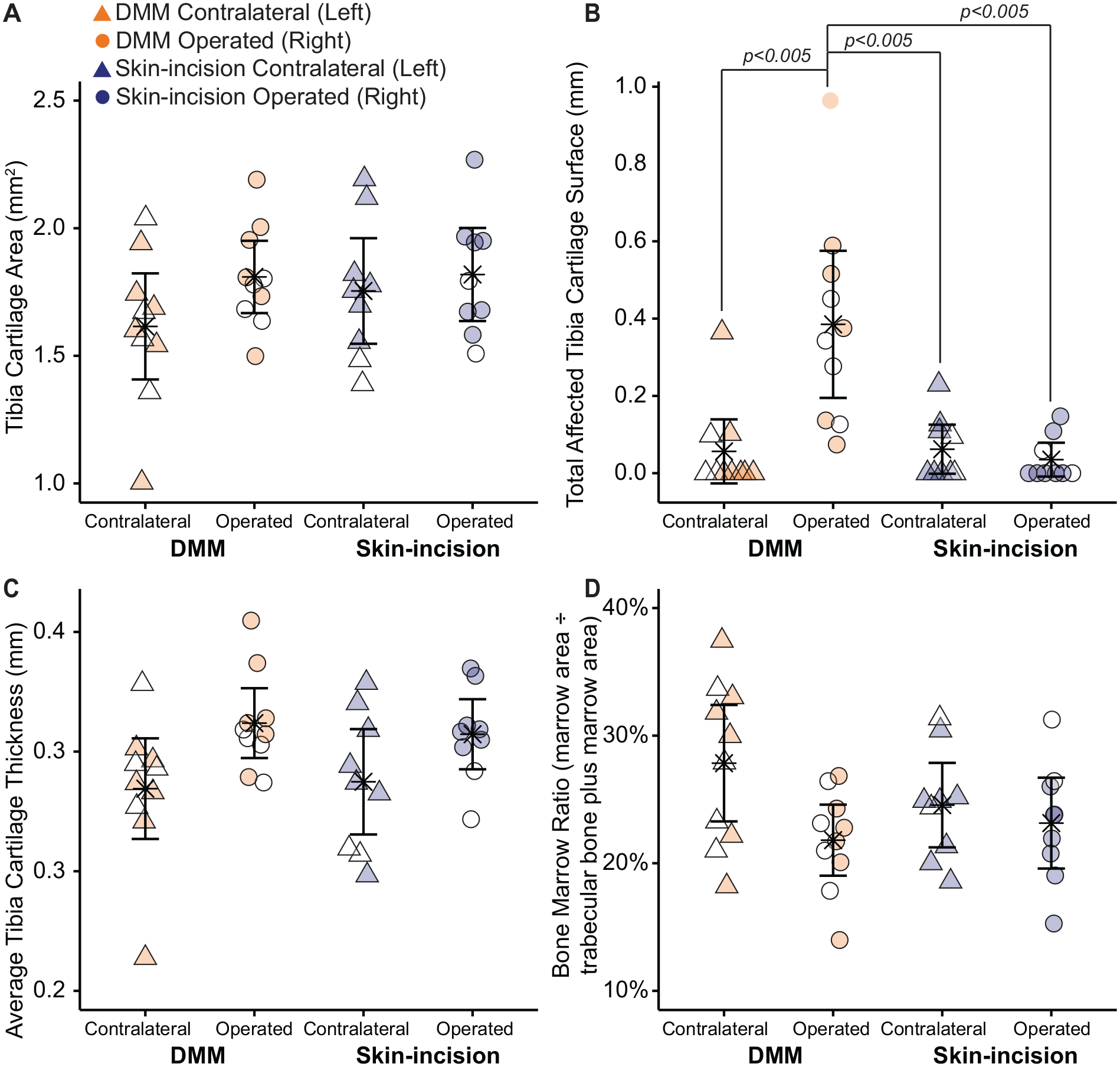
Quantitative histological grading following meniscal injury in Acomys. There were no significant differences in tibial cartilage area (
Discussion
The menisci are composed of highly aligned collagen fibers that attach to the bone via insertional ligaments. In the healthy knee, the menisci help absorb and distribute joint loads, in part by increasing the surface contact area in the tibiofemoral joint and reducing stresses on the tibial cartilage. The attachments to bone are crucial to meniscal function; these insertional ligaments prevent the menisci from extruding from the joint during axial loading and convert compressive loading into circumferential hoop stress. However, transecting the attachments (such as the meniscotibial ligament transection in the DMM model) inhibits the load distribution function of the meniscus, thus concentrating compressive loading on cartilage contact areas. Normally, cartilage develops regional thickness variations based on expected loading patterns; however, increased loading intensities following meniscal transection subject certain cartilage areas to higher loads than expected. In this study, we did not necessarily expect the transected meniscus to regenerate and reattach to the bone (though meniscal regeneration has not been studied in Acomys), yet we hypothesized that the hyaline cartilage would be protected from damage due to the regenerative abilities of Acomys. Moreover, we hypothesized Acomys would not develop OA-related behaviors indicative of joint dysfunction when compared to uninjured Acomys, in part due to an anticipated protection from cartilage damage. However, our data show Acomys did develop gait changes and did develop some histological evidence of OA following meniscus injury. To our knowledge, this is the first study evaluating OA protection following joint injury in Acomys.
While we did not observe complete hyaline cartilage protection in Acomys, these animals remain an emerging research organism for regenerative medicine. Initially, research interests in Acomys were related to the animals’ human-like qualities, such as sensitivity to developing diabetes when fed high-fat and high-sugar diets 19 and mostly complete organogenesis before birth. 20 More recently, Acomys have shown promise as a species for regenerative medicine research, with experiments demonstrating full regeneration of complex tissues (skin, ear tissue, and skeletal muscle) after extensive wounds. 15 The mechanisms of tissue regeneration in Acomys appear related to immune cells; when compared to traditional laboratory mice, Acomys have little evidence of M1 (proinflammatory) macrophages and an abundance of M2 (pro-reparative) macrophages in the wounded tissues.18,28 -31 These macrophage phenotype differences suggest the wounded tissue environment in Acomys promotes healing and repair. In OA, chronic, low-grade inflammation and an increasingly proinflammatory joint environment drive disease progression; this environment is generally associated with activation of M1 macrophages, although this was not directly assessed in this study. The unique immune environment and macrophage phenotype differences in Acomys suggest the regenerative abilities of Acomys may extend to protect hyaline cartilage in a destabilized joint. However, our data indicate that the hyaline cartilage of Acomys is not completely protected from post-traumatic OA-related damage.
To provide broad comparisons across the literature, we performed both semi-quantitative histological grading using the modified Mankin scheme 25 and quantitative grading using measurements that mirror the updated OARSI (Osteoarthritis Research Society International) recommendations.32,33 In the modified Mankin scheme, cartilage structure, tidemark duplication, safranin-O staining, fibrocartilage, chondrocyte clones in uncalcified cartilage, hypertrophic chondrocytes in calcified cartilage, and subchondral bone are graded on an ordinal scale based on the extent of joint damage. These individual scores are summed to calculate the total Mankin score, with greater sums indicating more severe histological evidence of OA-related joint damage. Our histology data indicate more severe OA-related changes in the injured knees of DMM Acomys compared to the contralateral knees in DMM Acomys and the operated knees in skin-incision controls. Moreover, reducing the total sum into individual scores indicates that the higher overall scores are primarily driven by cartilage structure changes. Nevertheless, DMM-operated Acomys knees did not show severe fibrillations or cartilage defects extending past the tidemark. In contrast, the same DMM model conducted in C57BL/6 mice and 129S6/SvEv mice shows cartilage erosion, fibrillations, and proteoglycan loss after 8 to 10 weeks of injury progression, with cartilage lesions often extending past the tidemark and into the calcified cartilage34,35 Moreover, other OA-related changes, such as loss of safranin-O staining (i.e., proteoglycan loss), were not significant in Acomys. As such, Acomys appear to develop changes to cartilage surface integrity, but not necessarily proteoglycan content, after 12 weeks of meniscal injury progression, and these structural changes may be less severe than previous reports for disease progression in traditional laboratory mice following DMM surgery.
Quantitative histological data support similar findings with regard to cartilage surface integrity. The total affected tibial cartilage surface was significantly greater in DMM-operated knees compared to all other knees. This finding relates to the higher Mankin scores for cartilage surface, further showing Acomys develop cartilage surface changes following meniscal injury progression. However, quantitative measures of tibial cartilage area, tibial cartilage thickness, and subchondral bone and bone marrow do not show differences across knees. This indicates DMM surgery in Acomys may not result in the concomitant damage to other joint tissues that is common in other rodent models after 12 weeks of DMM progression. Combined, our histological data could be interpreted as slowed or delayed OA progression in Acomys, as both our modified Mankin grading and quantitative histology data suggest Acomys did not develop severe evidence of OA following 12 weeks of meniscal injury. However, some caution is necessary with this interpretation. There is no consensus on an appropriate control species for Acomys, and while historically Acomys were grouped with mice in the Murinae subfamily, recent biological and immunological data reveal Acomys differ from common laboratory mice.36,37 In fact, molecular and evolutionary evidence indicate Acomys may be more closely related to gerbils and should be classified within the Gerbillinae or Deomyinae subfamilies. To our knowledge, data on DMM surgery in this subfamily are unavailable; thus, conclusions related to the rate of OA progression, particularly relative to laboratory mice, should be tempered.
OA-related joint damage progresses at different rates in male versus female C57BL/6 mice following DMM surgery. However, it is unknown whether these sex differences related to the rate of OA progression extend to other species. In this study, we were unable to control for or evaluate sex differences in Acomys due to animal availability. Acomys have long gestation periods and relatively small litters (discussed further in limitations), making it challenging to build a robust Acomys colony; moreover, there is a high demand for Acomys among the existing colony at the University of Florida. Here, we plotted the histological data to show male versus female differences, and overall the male and female data points appear to be randomly distributed (
Symptomatically, DMM Acomys adopted temporal gait patterns that deviate from those of uninjured animals. Healthy animals typically walk with balanced gaits and spend equal time on both hind limbs; as such, percent stance time does not normally vary between the right and left limbs of a limb pair. However, DMM Acomys increased percent stance times on the uninjured, contralateral limb relative to the injured limb. This compensation reduces the amount of time the injured limb must bear weight on its own during a gait cycle. Typically, this left and right limb stance time imbalance coincides with temporally asymmetric gait sequences. Although injured Acomys gaits were not temporally asymmetric based on a statistical test, visual inspection of the distribution of temporal symmetry measures in Figure 4 suggests the distribution of data begins to skew at weeks 10 and 12, with DMM Acomys trending toward temporally asymmetric gaits. Combined, our data suggest a protective mechanism whereby injured Acomys reduce weight-bearing on the affected limb throughout the gait cycle.
Following DMM surgery in C57BL/6 mice, mice showed increased contralateral hind print areas relative to the operated limb as OA developed (using the CatWalk system).35,38 Moreover, our group has shown altered gait in male and female C57BL/6 mice following injury, with male mice developing a shuffling gait. 39 Thus, the gait changes following meniscal injury in Acomys are somewhat similar to those in other rodents.
Finally, an important distinction must be made related to our results and our experimental design. In our experiment, the joint destabilization is not repaired and remains throughout the study; thus, our data do not necessarily demonstrate Acomys are unable to heal cartilage in a stable joint. Prior work in the MRL/MpJ strain evaluated cartilage regeneration and integration following full and partial-thickness lesions in the trochlear groove of hyaline cartilage. 40 While these studies are related, the chronic instability following DMM surgery is a clear confounding factor. Whether Acomys could resist cartilage damage or increase the quality of cartilage repair in an osteochondral defect model remains unanswered. Future studies would be needed to explore this potential.
While there are still exciting OA-related research questions to explore in Acomys, there are several considerations and challenges for future work.41,42 In addition to lack of consensus on an appropriate control species, Acomys colonies are currently maintained by individual researchers and institutions, limiting their widespread availability for research purposes. Moreover, Acomys must reach 4 to 6 months of age for successful breeding, have a longer gestation period (40 days), and have small litters of only 1 to 4 pups; this makes it difficult to acquire enough Acomys for a robust experimental design, even within an institution that maintains a colony. Acomys also require larger housing spaces, as they engage in territorial behaviors such as chasing, fighting, and other forms of aggression to establish dominance within a cage. Finally, Acomys skin is designed to tear easily as an avoidance mechanism from predators, along with their remarkable climbing and jumping abilities; this necessitates special handling practices and techniques. Acomys skin is so fragile that they cannot be easily scruffed. For example, here, the incision on the knee was not made with a scalpel, as the simple act of placing pressure on both sides of the knee produced a skin tear over the patella. These factors should be considered when conducting future work with Acomys.
In conclusion, these data indicate Acomys are not fully protected from OA-related structural joint changes or gait changes following a simulated meniscal injury. Here, Acomys developed mild evidence of OA that appears to be driven by the loss of structural integrity in the hyaline cartilage. Alongside this damage, Acomys developed gait compensations that reduced stance times on the injured limb while walking. Taken together, this study shows Acomys are not completely protected from OA-related changes following a meniscal injury, despite the promising regenerative capabilities of other wounded tissues in Acomys.
Supplemental Material
sj-pdf-1-car-10.1177_19476035221149146 – Supplemental material for African Spiny Mice (Acomys) Exhibit Mild Osteoarthritis Following Meniscal Injury
Supplemental material, sj-pdf-1-car-10.1177_19476035221149146 for African Spiny Mice (Acomys) Exhibit Mild Osteoarthritis Following Meniscal Injury by Kiara M. Chan, Nat A. Thurlow, Malcolm Maden and Kyle D. Allen in CARTILAGE
Supplemental Material
sj-pdf-2-car-10.1177_19476035221149146 – Supplemental material for African Spiny Mice (Acomys) Exhibit Mild Osteoarthritis Following Meniscal Injury
Supplemental material, sj-pdf-2-car-10.1177_19476035221149146 for African Spiny Mice (Acomys) Exhibit Mild Osteoarthritis Following Meniscal Injury by Kiara M. Chan, Nat A. Thurlow, Malcolm Maden and Kyle D. Allen in CARTILAGE
Footnotes
Author Contributions
K.M.C., M.M., and K.D.A. conceptualized and contributed to the study design. M.M. provided the animals and contributed important insights with regard to working with Acomys. K.D.A. performed the animal surgeries. K.M.C. managed day-to-day animal care, collected rodent gait data, and prepared samples for histology. N.A.T. performed the quantitative histological data and assisted with the quantitative histological data analysis. K.M.C. analyzed and interpreted histology and gait data with K.D.A. The manuscript was prepared by K.M.C. with critical feedback from M.M. and K.D.A. All authors agree with the content and interpretation of the data as presented.
Acknowledgments and Funding
The authors would like to thank Jessica Aldrich, Markia Bowe, Carlos Cruz, and Jacob Griffith for their help with the modified Mankin histological grading. This work was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS) of the National Institutes of Health under grants R01AR071431 and R01AR071335-01 and by the WM Keck Foundation. This work was also supported by a graduate student fellowship from the Herbert Wertheim College of Engineering and the J. Crayton Pruitt Family Department of Biomedical Engineering at the University of Florida.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All methods described were approved by the University of Florida Institutional Animal Care and Use Committee and in accordance with the Association for Assessment and Accreditation of Laboratory Animal Care recommendations on animal research.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
