Abstract
Objective
Although most frequently used in experimental osteoarthritis (OA) pain induction, intra-articular mono-iodoacetate (MIA) injection lacks concluded references for dose selection and timing of intervention. Herein, we aimed to compare the pain intensity of rats induced by different doses of MIA and explored the trajectory of pain.
Design
PubMed, Embase, and Web of Science were searched up to June 2021 for literatures involving MIA experiments investigating OA pain. Pain intensity was measured based on weightbearing distribution (WBD) and paw withdrawal thresholds (PWT), and the pain trajectory was constructed by evaluating pain intensity at a series of time points after MIA injection. A conventional meta-analysis was conducted.
Results
A total of 140 studies were included. Compared with saline, MIA injections caused significantly higher pain intensity for WBD and PWT. Dose-response relationships between different doses of MIA and pain intensity were observed (P-for-trend<0.05). A pronounced increase in pain occurred from day 0 to day 7, but the uptrend ceased between day 7 and day 14, after which the pain intensity continued to rise and reached the maximum by day 28.
Conclusions
Pain intensity after intra-articular MIA injection increased in a dose-dependent manner and the pain trajectory manifested a specific pattern consistent with the pathological mechanisms of MIA-induced pain, providing possible clues for proper dose selection and timing of specific OA pain interventions.
Introduction
Osteoarthritis (OA), a highly prevalent degenerative joint disorder, is a leading contributor to disability and economic burden worldwide.1,2 With pain as the predominant symptom of OA,3 -5 a growing emphasis of research and clinical practices is being put on pain management. Despite existing drugs such as acetaminophen, opioids, and non-steroidal anti-inflammatory drugs (NSAIDs) being widely used,6 -8 to date, there is no analgesic present for OA pain alleviation proven both safe and robustly effective, and the associated adverse effects (AEs) were reported to affect a spectrum of body systems.9 -11 Thus, a stringent need for new pain-relieving drugs for OA warrants further investigation in this field.
Mono-iodoacetate (MIA)-induced arthritis model, first described by Kalbhen 30 years ago,12 -14 is the most widely used animal model for elucidating underlying mechanisms and developing analgesics for OA pain with a wide range of doses used in previous studies.15 -19 However, how does pain intensity changes with different injected MIA doses remains undetermined, hindering the process of choosing the proper dose for experiments with a certain degree of desired pain intensity before intervention, as moderate-to-severe pain induction is needed to testify the pain-relieving effect of potential analgesics, whereas mild-to-moderate pain induction better serves the purpose of verifying pain-worsening effects.20 -22 Moreover, MIA is highly toxic, with potential lethal systemic effects, 12 thus making it necessary to find out the optimal dose with the highest pain-inducing efficacy, avoiding the use of unnecessary high doses.
In addition, the MIA model has a biphasic nature in pain mechanisms with an initial inflammatory phase and a following non-inflammatory one,23 -25 and the different phases of pain are reported to be responsive to specific treatments,26,27 thus making it necessary to clarify the beginning and endpoint of each phase. However, the current lack of an exact pain trajectory after MIA injection makes it difficult to decide the proper timing of potential interventions targeting different phases.
In an effort to fill in the aforementioned void of knowledge, this study intended to conduct a meta-analysis covering all available evidence to generate a unified and coherent analysis for all experimental studies featuring MIA injection. Moreover, we systematically evaluated the intensity of pain induced by different doses of MIA and the trajectory of pain after intra-articular injection, aiming to provide dose selection and timing reference for future OA pain investigations.
Methods
Protocol
This systematic review has been prepared according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 28 The protocol is registered in the International Prospective Register of Systematic Reviews (PROSPERO, reference number: CRD42021209220).
Literature Search
Systematic literature searches were undertaken using PubMed, Embase, and Web of Science up to June 2021. The search strategy was developed using the keywords “mono-iodoacetate” and “osteoarthritis” and was similar across all databases (Suppl. Appendix 1).
Inclusion/Exclusion Criteria
Two researchers (H.J. and X.L.) conducted the study selection independently. The titles and abstracts generated by the literature search were screened initially. Thereafter, reviewing of the full texts of eligible articles was performed. Studies matching the following criteria were included: (1) randomized controlled trials; (2) OA studies with enrolled rat strains; (3) studies including a healthy control group receiving intra-articular injection of saline instead of MIA; (4) studies including OA pain measurement; and (5) studies with any language. The following studies were excluded: (1) studies of secondary analyses; (2) studies with cross-over design; (3) studies of intra-articular injection of MIA combined with other drugs; (4) studies not involving knee OA; and (5) studies with abstract/title publication only (insufficient data).
Outcome Measures
To evaluate the relationships between intra-articular injection of different doses of MIA and the pain of OA, data reported on pain related to OA were extracted by three researchers (Y.Y., K.H., and C.D.) and an analysis of these data was conducted. Weightbearing distribution (WBD) and paw withdrawal thresholds (PWT), as the most commonly used index in pain detection, served as primary outcomes and were measured in rat knee joints. The effects of intra-articular injection of MIA or saline on weight distribution through the ipsilateral and contralateral knee were assessed using a Linton Incapacitance Tester,18,29 data were manifested as ipsilateral weight-bearing, which was calculated as a percentage of total hind limb weight-bearing.30 -32 PWT was evaluated using a dynamic plantar aesthesiometer or von Frey monofilaments.12,33 -35 The filament exerted a gradually increasing force on the plantar, starting below the threshold of detection and increasing until the stimulus became painful and the rats removed its paw. 36 The force required to produce a paw withdrawal response was recorded and measured in grams.
Risk of Bias Assessment
The quality of studies was independently evaluated by two reviewers (H.J., and X.L.) using the Systematic Review Centre for Laboratory animal Experimentation (SYRCLE) risk of bias tool for animal studies. 37 A total of nine domains were evaluated: sequence generation, baseline characteristics, allocation concealment, housing, blinding researchers, random outcome assessment, blinding outcome assessment, incomplete outcome data, and selective outcome reporting. Also, it relates to five types of bias: selection, performance, detection, attrition, and reporting bias. Meanwhile, we evaluated other possible methods from the Animals in Research: Reporting In Vivo Experiments (ARRIVE) guidelines for including animal experiments. 38 It contains four quality items: (1) ethical statement (ethical review permissions and national or institutional guidelines for the care and use of animals); (2) experimental procedures description (all procedures’ precise details); (3) experimental animal details (including the number of animal, strain, and age); and (4) financial conflicts of interest. 39 We classified each domain as having either “low,” “high,” or “unclear” risk of bias based on the information reported in each study.39,40
Statistical Analysis
A conventional meta-analysis was conducted to compare the pain after the intra-articular injection of saline and MIA. We analyzed the pain intensity of different MIA doses including 1, 2, 3, and 4 mg as they were commonly used in the studies. Also, we analyzed the pain trajectory after MIA injection and conducted an analysis by dose and time in order to see whether the pain trajectory differ by dose. The analysis was not carried out in MIA 4 mg group due to the insufficient sample size. To quantify the pooled effects of the primary outcome, we used standardized mean difference (SMD) with a 95% confidence interval (CI) according to the results of WBD (ipsilateral/total×100%) and PWT that were used to measure the pain. As there was significant heterogeneity regarding the methods used for scoring among the papers included in the meta-analysis, data were analyzed using a random-effect model. Moreover, we explored potential dose-response relationship between pain and MIA by using restricted cubic splines with three knots (25 50 75) in the dose-response regression model, and results from each study were then pooled together with the conventional meta-analysis.
All statistical analyses were conducted using Review Manager 5.3 software (RevMan 5.3, The Cochrane Collaboration, Oxford, UK) and STATA software (version 15.0; Stata Corp LP, College Station, TX).
Results
Study Selection and Characteristics
The search strategy identified a total of 1,379 studies, of which 394 studies were retrieved for full-text evaluation, and a final 140 studies which met the inclusion criteria were included. However, some of them included more than one dose of MIA, which were listed as different trials compared with saline group. Thus, a total of 153 trials were included. Figure 1 depicts the details of the selection process. The characteristics of the analyzed research are shown in Table 1 , and detailed information on the included studies is shown in Supplementary Appendix 2.
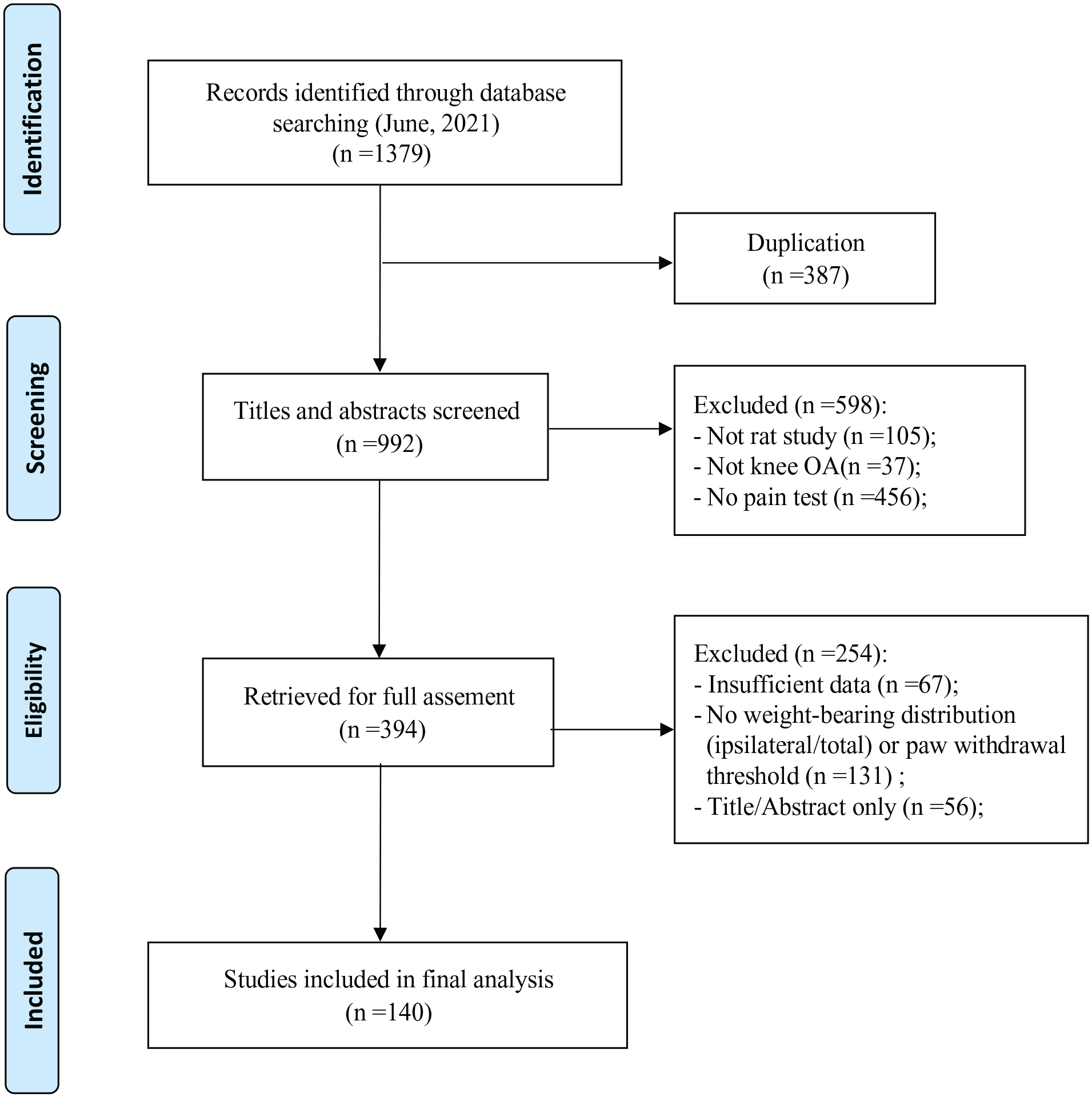
Selection process of included studies. OA = osteoarthritis.
Summary of Characteristics of the Analyzed Randomized Controlled Trials.
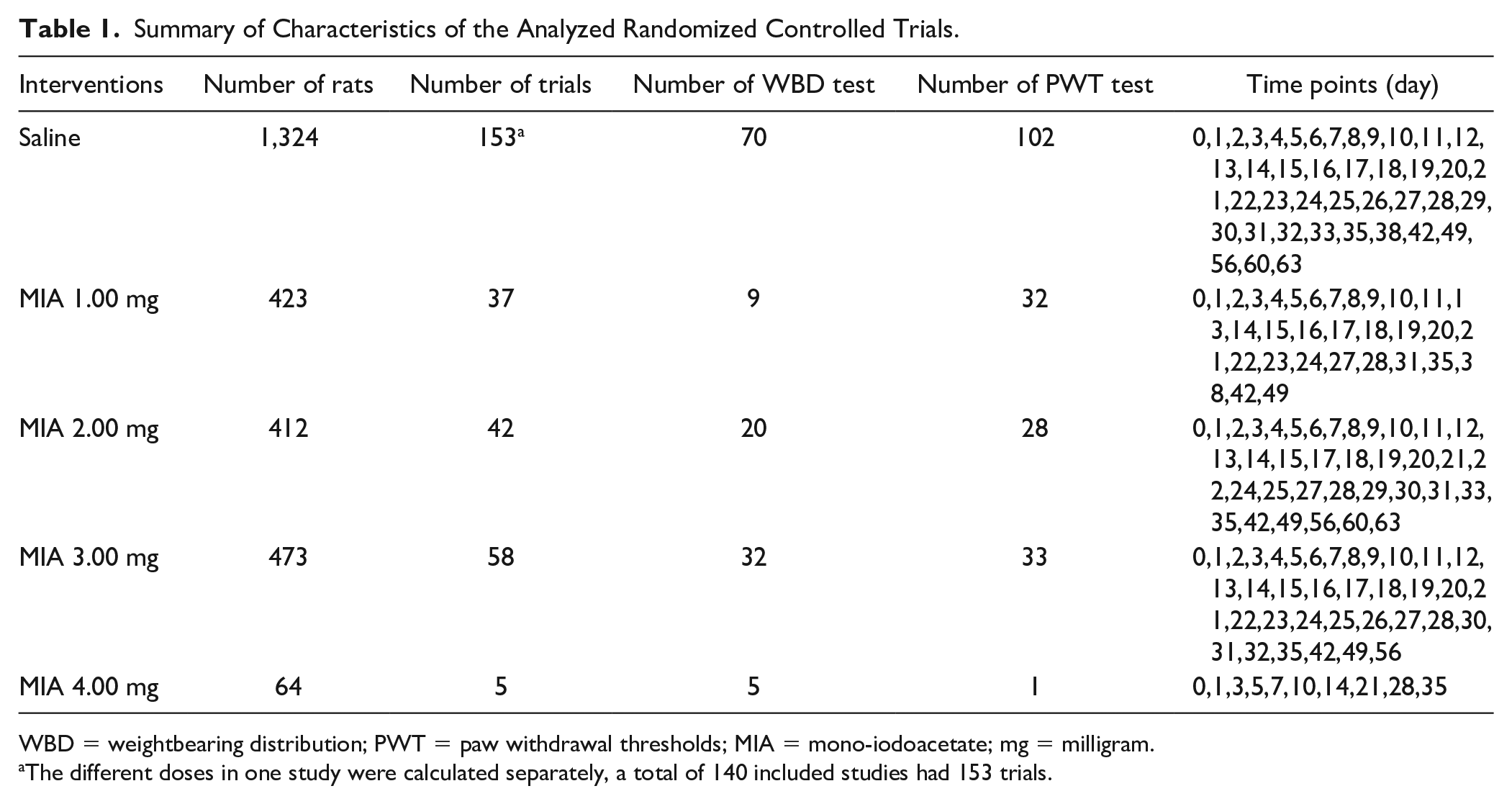
WBD = weightbearing distribution; PWT = paw withdrawal thresholds; MIA = mono-iodoacetate; mg = milligram.
The different doses in one study were calculated separately, a total of 140 included studies had 153 trials.
Risk of Bias Assessment
The majority of studies were judged at low risk of bias (Suppl. Appendix 3). Three of all included randomized controlled trials specifically described the method used to generate and conceal the assignment sequence; they were therefore classified as having a low risk of detection bias and selection bias. In total, 118 of the 140 studies reported measures used to house the animals within the animal room, 58 studies reported the baseline characteristics of rats included, and 7 studies did not report the details of the ethical statement. Moreover, outcome assessors were blinded in 22 studies; since some of the included studies did not specifically mention whether they were blinded, no definite conclusions can be drawn about detection bias. Fifty-five studies described the details of each main outcome, so these studies were at low risk of attrition bias. All studies described details of animal details, reported financial conflicts of interest and experimental procedures, they were at low risk of reporting bias.
Pain Intensity Following Intra-Articular Injection of Different Doses of MIA
Overall, intra-articular injection of MIA caused increased pain indicated by pain behavior tests. As shown in Table 2 and Supplementary Appendix 4, compared with saline, intra-articular injection of different MIA doses showed significantly higher pain intensity in terms of WBD and PWT. Moreover, Supplementary Appendix 5 shows the results of a dose-response meta-analysis. There was a positive dose-response relation between pain intensity and the dose of MIA in terms of WBD (P-for-trend=0.0031) and PWT (P-for-trend=0.0229) from 1 to 3 mg.
Pain Intensity for Different Doses of MIA Compared with Saline in Randomized Controlled Trials.
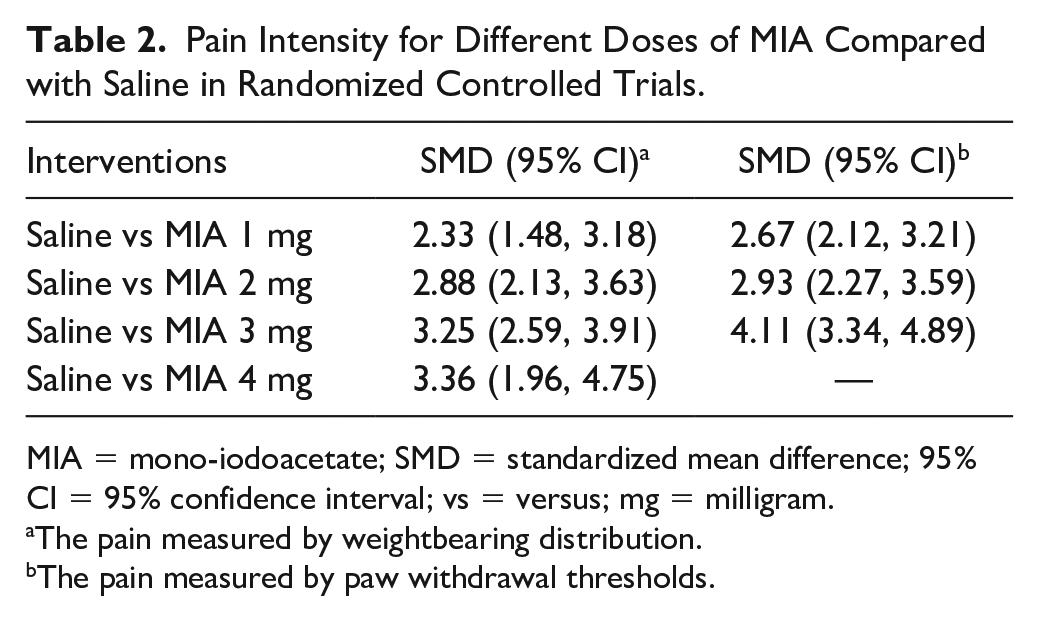
MIA = mono-iodoacetate; SMD = standardized mean difference; 95% CI = 95% confidence interval; vs = versus; mg = milligram.
The pain measured by weightbearing distribution.
The pain measured by paw withdrawal thresholds.
Pain Trajectory Following Intra-Articular Injection of MIA
Pain intensity at day 0, 3, 7, 14, 21, and 28 after the intra-articular injection of MIA was evaluated ( Fig. 2 , Table 3 , and Suppl. Appendix 6). In terms of both WBD and PWT, the trajectory of pain manifested a pronounced increase of pain intensity from day 0 to day 7, but the uptrend ceased between day 7 and day 14, after which the pain intensity continued to rise and reached the maximum by day 28. In addition, the result of an analysis by dose and time showed a faster pain development with a higher dose, but this trend was not obvious in WBD due to the insufficient sample size (Suppl. Appendix 7).
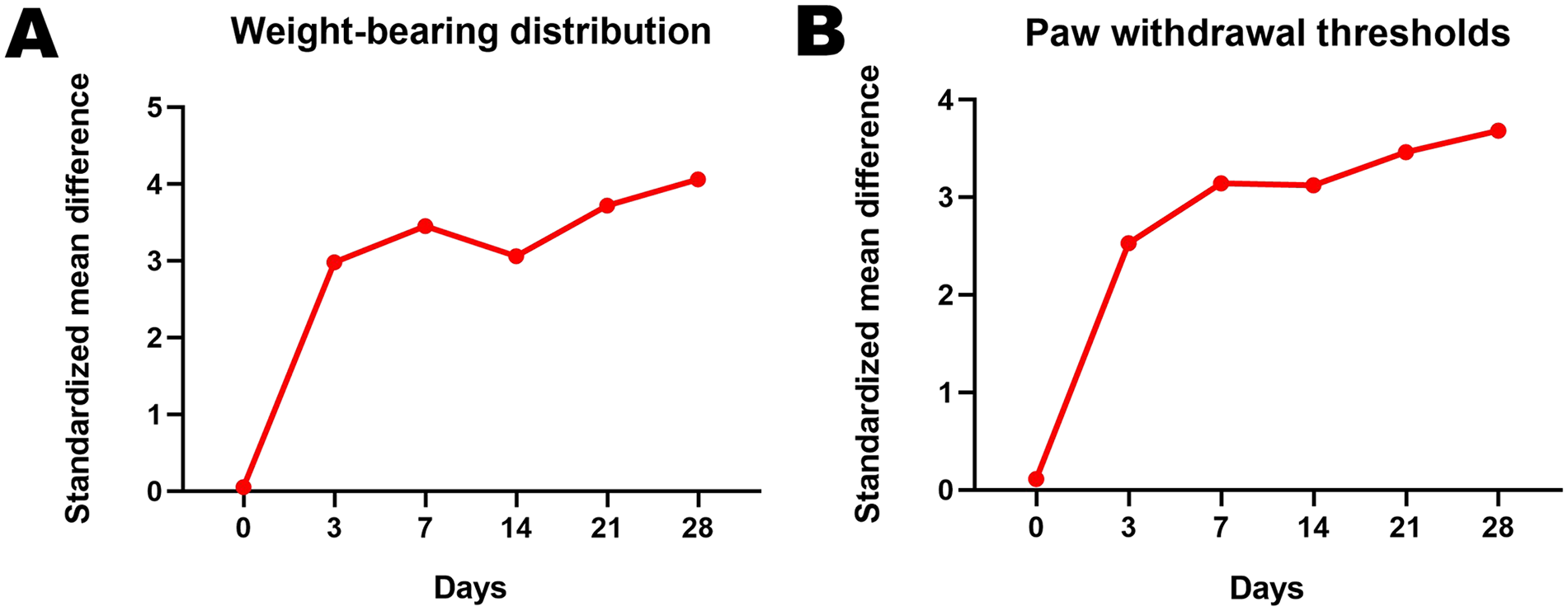
The trajectory of pain intensity based on WBD (
Pain Intensity at Different Time Points after Intra-Articular Injection of MIA in Randomized Controlled Trials.
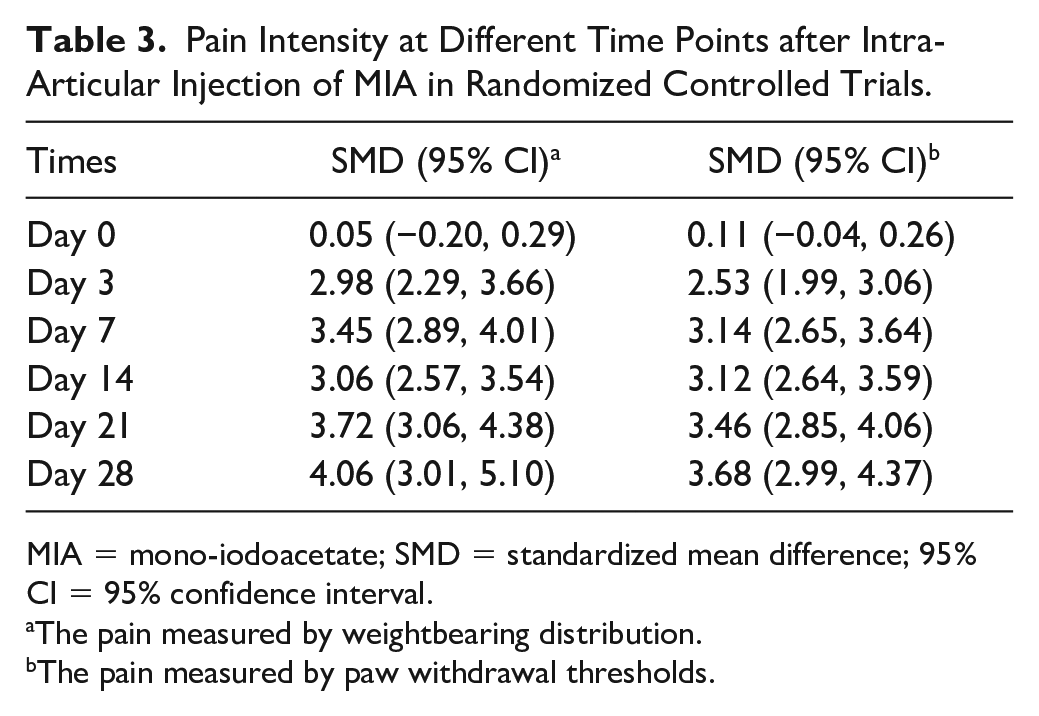
MIA = mono-iodoacetate; SMD = standardized mean difference; 95% CI = 95% confidence interval.
The pain measured by weightbearing distribution.
The pain measured by paw withdrawal thresholds.
Discussion
Principal Findings
To our knowledge, this is the first meta-analysis to directly compare the pain intensity between different doses of MIA and saline and to examine the pain trajectory after MIA injection. To comply with the current guidelines on high-quality conventional meta-analysis, we formulated a comprehensive search strategy alongside stringent inclusion criteria in our registered protocol and assessed the quality of the included literatures. The main findings were the following: (1) the pain intensity after intra-articular MIA injection increased in a dose-dependent manner from 1 to 3 mg; (2) the pain trajectory manifested a specific pattern: a pronounced increase of pain intensity occurred from day 0 to day 7, but the uptrend ceased between day 7 and day 14, after which the pain intensity continued to rise and reached the maximum by day 28.
Potential Explanations
MIA, an inhibitor of glyceraldehyde-3-phosphate dehydrogenase, could negatively impact the cellular aerobic glycolysis pathway and induce cell death and inflammatory responses.41,42 Histological evidence indicated that compared with lower doses, higher doses of MIA could lead to more severe synovial inflammation and subchondral bone exposure and deformation, hence greater pain. 43 In addition, as described in previous studies, higher doses of MIA in the knee joint evoked significant axonal injury to the dorsal root ganglia (DRG) cells, thus may inducing more severe pain. 44 In line with the above-mentioned investigations, according to our results, the MIA-induced pain increased in a dose-dependent manner from 1 to 3 mg, but the uptrend ceased from 3 to 4 mg. One possible explanation for this may be that 3 mg of MIA was already causing severe enough pathological changes, leaving little room for further deterioration. 44
The pain trajectory manifested a specific pattern in the present study, featuring 2 phases of continuing increasing pain intensity, namely from day 0 to day 7 and day 14 to day 28. This result is consistent with pathological changes described in previous literatures. Inflammatory infiltration within the synovium, characterized by recruitment of neutrophils, lymphocytes, and macrophages was reported to occur mainly in the first week after MIA induction,26,45 -47 followed by a transition period from day 7 to day 14.48,49 Thereafter, degenerative changes dominated and may fuel further increase in pain intensity, featuring characteristics of neuropathic pain perception.23,24,47
Research Implications
Choosing a proper dose of MIA when investigating OA pain is difficult due to the wide range of doses used in published literatures and a lack of concluded pain-inducing efficacy profile. According to the results of the present study, dose-response relationships between different doses of MIA used (1-3 mg) and pain intensity were observed, thus making it possible to choose the MIA dose in line with the desired pain intensity before intervention. Furthermore, given the fact that MIA is a lethal substance and the pain intensity (PWT) between 3 and 4 mg is very close, it is reasonable to speculate that 3 mg MIA would be a better choice compared with 4 mg, weighing the pain-inducing efficacy and overall safety.
During the 4 weeks after MIA injection, the pain trajectory of MIA manifested 2 phases of increasing pain intensity, which is in accordance with previous mechanistic illustrations of MIA-induced OA pain, providing potential timing reference to test interventions targeting inflammatory pain (day 0 to day 7) or pain caused by degenerative changes (day 14 to day 28) in MIA model.
Strengths and Limitations
Compared with previous investigations, this study systemically and quantitatively evaluates MIA-induced pain at different time points during the typical 4 weeks of observation. Moreover, we examined the dose-response relationship between different doses of MIA used and the pain intensity after intra-articular injection. Our results may assist future OA pain investigations in dose selection and timing of interventions.
However, there are several limitations to this study. First, the pain trajectory only took into account 4 weeks of time after MIA injection, as relatively few studies had longer period of pain observation; thus, further investigations are needed to conclude pain trajectory after day 28. Second, as the sample size of studies with both 4 mg of MIA injection and PWT results is relatively small, we were unable to include them in the current analysis, making it hard to conclude the exact pain-inducing profile of 4 mg MIA injection. Last, pain assessments are subjective, but when conducting the risk of bias assessment, it was found that some included articles did not describe whether the researchers were blinded when performing pain assessments, even though we believe this is an essential and fundamental requirement of experimental design. Researchers involved in studies in the future should make ensure that pain assessments were assayed in a blinded manner and methods of blinding are reported.
Conclusions
The findings of the current meta-analysis indicated that pain intensity after intra-articular MIA injection increased in a dose-dependent manner and the pain trajectory manifested a specific pattern consistent with the pathological mechanisms of MIA-induced pain, providing possible clues for proper dose selection and the timing of specific OA pain interventions.
Supplemental Material
sj-docx-1-car-10.1177_19476035221144748 – Supplemental material for Pain Intensity and Trajectory Following Intra-Articular Injection of Mono-Iodoacetate in Experimental Osteoarthritis: A Meta-Analysis of In Vivo Studies
Supplemental material, sj-docx-1-car-10.1177_19476035221144748 for Pain Intensity and Trajectory Following Intra-Articular Injection of Mono-Iodoacetate in Experimental Osteoarthritis: A Meta-Analysis of In Vivo Studies by Hongyu Jin, Yuanheng Yang, Guanghua Lei, Chao Zeng, Ke He, Yilun Wang, Caifeng Deng, Jie Wei, Xiaoxiao Li and Hui Li in CARTILAGE
Footnotes
Author Contributions
XL and HL are joint corresponding authors. HJ, YY, XL, and HL had full access to the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: CZ, GL, HL, and JW. Acquisition and interpretation of data: HJ, YY, CD, XL, and HL. Statistical analysis: HJ and XL. Drafting of the manuscript: HJ and YY. Critical revision of the manuscript for important intellectual content: CZ, HL, and XL. Study supervision: XL and HL. All authors have read, provided critical feedback on intellectual content, and approved the final manuscript.
Acknowledgments and Funding
Everyone who contributed significantly to the work has been listed. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grants 81902264, 81902265). No funding bodies had any role in study design, data collection, and analysis, decision to publish, or preparation of the manuscript. The funding source had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and the decision to submit the manuscript for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Availability of Data and Materials
The data sets analyzed during the current study are available from the corresponding author on reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
