Abstract
Objective
The medial femoral condyle of the knee exhibits some of the highest incidences of chondral degeneration. However, a dearth of healthy human tissues has rendered it difficult to ascertain whether cartilage in this compartment possesses properties that predispose it to injuries. Assessment of young, healthy tissue would be most representative of the tissue’s intrinsic properties.
Design
This work examined the topographical differences in tribological, tensile, and compressive properties of young (n = 5, 26.2 ± 5.6 years old), healthy, human medial femoral condyles, obtained from viable allograft specimens. Corresponding to clinical incidences of pathology, it was hypothesized that the lowest mechanical properties would be found in the posterior region of the medial condyle, and that tissue composition would correspond to the established structure-function relationships of cartilage.
Results
Young’s modulus, ultimate tensile strength, aggregate modulus, and shear modulus in the posterior region were 1.0-, 2.8-, 1.1-, and 1.0-fold less than the values in the anterior region, respectively. Surprisingly, although glycosaminoglycan content is thought to correlate with compressive properties, in this study, the aggregate and shear moduli correlated more robustly to the amount of pyridinoline crosslinks per collagen. Also, the coefficient of friction was anisotropic and ranged 0.22-0.26 throughout the condyle.
Conclusion
This work showed that the posteromedial condyle displays lower tensile and compressive properties, which correlate to collagen crosslinks and may play a role in this region’s predisposition to injuries. Furthermore, new structure-function relationships may need to be developed to account for the role of collagen crosslinks in compressive properties.
Keywords
Introduction
Articular cartilage degeneration usually occurs at specific regions of the distal femur. 1 The patellofemoral groove and the lateral and medial condyles are exposed to different strains that contribute to characteristic lesion patterns.2 -6 Grade IV chondral defects are found predominantly in the medial femoral condyle,2,3 and radiological studies have identified 60% of osteoarthritic lesions to be in the medial condyle, followed by the lateral condyle and the patellofemoral groove. 7 Chondral defects, moreover, show topographical patterns; an examination of 127 distal femora not only confirmed higher incidence of lesions in the medial condyle compartment, but specifically identified a majority to localize in the posteromedial region. 8 The higher incidences of degeneration in the posteromedial condyle may be due to topographical differences in biochemical and biomechanical properties, although this has yet to be verified due to the paucity of young, healthy specimens for characterization studies.
The mechanical properties of articular cartilage differ throughout the knee. For example, the adult equine posteromedial condyles showed significantly lower aggregate and shear modulus values than the anterior region. 9 Tensile Young’s modulus and ultimate tensile strength (UTS) values were also found to be lower in the posteromedial condyle when compared with the anterior region in the sheep. 10 Finally, in 1- to 3-month-old calves, some of the highest coefficients of friction were observed in the posterior part of the medial condyle, whereas the anteromedial condyle showed the lowest values. 11 Thus, one would expect to find that the human posteromedial condyle, a site of frequent degeneration, may also exhibit inferior compressive and tensile properties.
In contrast to the animal data presented above, only scant data exist on the human femoral condyle, especially of young, healthy tissue. A study that compared the femoral condyle cartilage across multiple species determined that, in the human knee, the aggregate modulus of the posterior region of the medial condyle was 16% lower than the modulus of the anterior region of the lateral condyle. 12 However, no comparisons within the femoral condyles were included. 12 Likewise, studies have compared the tensile properties of medial and lateral condyles, associating them to biochemical changes that follow aging and chondral degeneration, but have not included comparisons within the condyles.13,14 Finally, human tribological data appear to be less available, with one study comparing the coefficient of friction of tissue-engineered constructs to a human native tissue value of ~0.25. 15 Additional studies on human cartilage condyle can be found, mostly covering the compressive properties on the condylar compartment.16,17 Still, the topographical determination of mechanical properties and their correlation to biochemical properties for the human medial condyle would represent information that has been long absent from the field despite the posterior medial condyle’s predisposition for pathologies.
The lack of studies on human samples may be due to the scarcity of healthy, undamaged, young joint samples. This is relevant, because age affects the properties of cartilage, even if considered healthy. For example, in a study that examined the tensile properties of healthy and diseased human cartilage, the authors noted that even their control (i.e., non-degenerative) samples, with a mean age of 68 ± 2 years, showed age-related damages such as surface roughness and fibrillation. 13 Most human tissues found in other characterization studies were derived from donors ranging 40- to 75-years-old, on which signs of wear and susceptibility to osteoarthritis are expected.13,18,19 Clinically “silent” injuries, in which the cartilage morphology appears undisturbed but exhibits diminished functional properties, also increase with age. 20 Thus, an updated characterization of the mechanical properties of a young adult cohort with healthy human femoral cartilage tissue is needed.
In lieu of the limited characterization of the human condyle thus far, and the significance of the material properties of the medial condyle, the goal of this study was to provide a topographical characterization of healthy articular cartilage of the human medial condyle. This work includes the characterization of 3 locations of the articular cartilage of the medial femoral condyle: anterior, central, and posterior regions. For all, histological, biochemical, and mechanical tests were performed. Corresponding to clinical incidences of pathology, it was hypothesized that the lowest mechanical properties would be found in the posterior region of the medial condyle, and that the biochemical composition and biomechanical properties would follow the well-established structure-function relationships for articular cartilage.
Methods
Sample Collection
This research is exempt from institutional review board approval because it does not constitute as human subject research. Human articular cartilage samples were obtained from leftover allograft tissue provided by commercial entities (JRF Ortho [Englewood, CO] and MTF Biologics [Redlands, CA]) to the University of California, Irvine Medical Center. Discarded tissues, resulting from the unused portions of allografts that were used in surgeries, were collected and analyzed. The allografts, upon harvest, were kept in proprietary preservation solutions for 2-6 weeks at 4°C based on JRF Ortho and MTF Biologics protocols until donor matching and transplantation surgery. During the allograft transplantation surgery, the patient would receive a 15-22.5 mm diameter core allograft taken from the donor condyle. The remaining tissue was immediately processed for testing; specimens were separated into 3 regions (anterior, central, and posterior), and for each, samples were obtained for histological, biochemical, and mechanical testing ( Fig. 1A ). 21 The specimens used in this study, collected over a period of 18 months, include 4 male and 1 female donors, with an age of 26.2 ± 5.6 years (Fig. 1B).
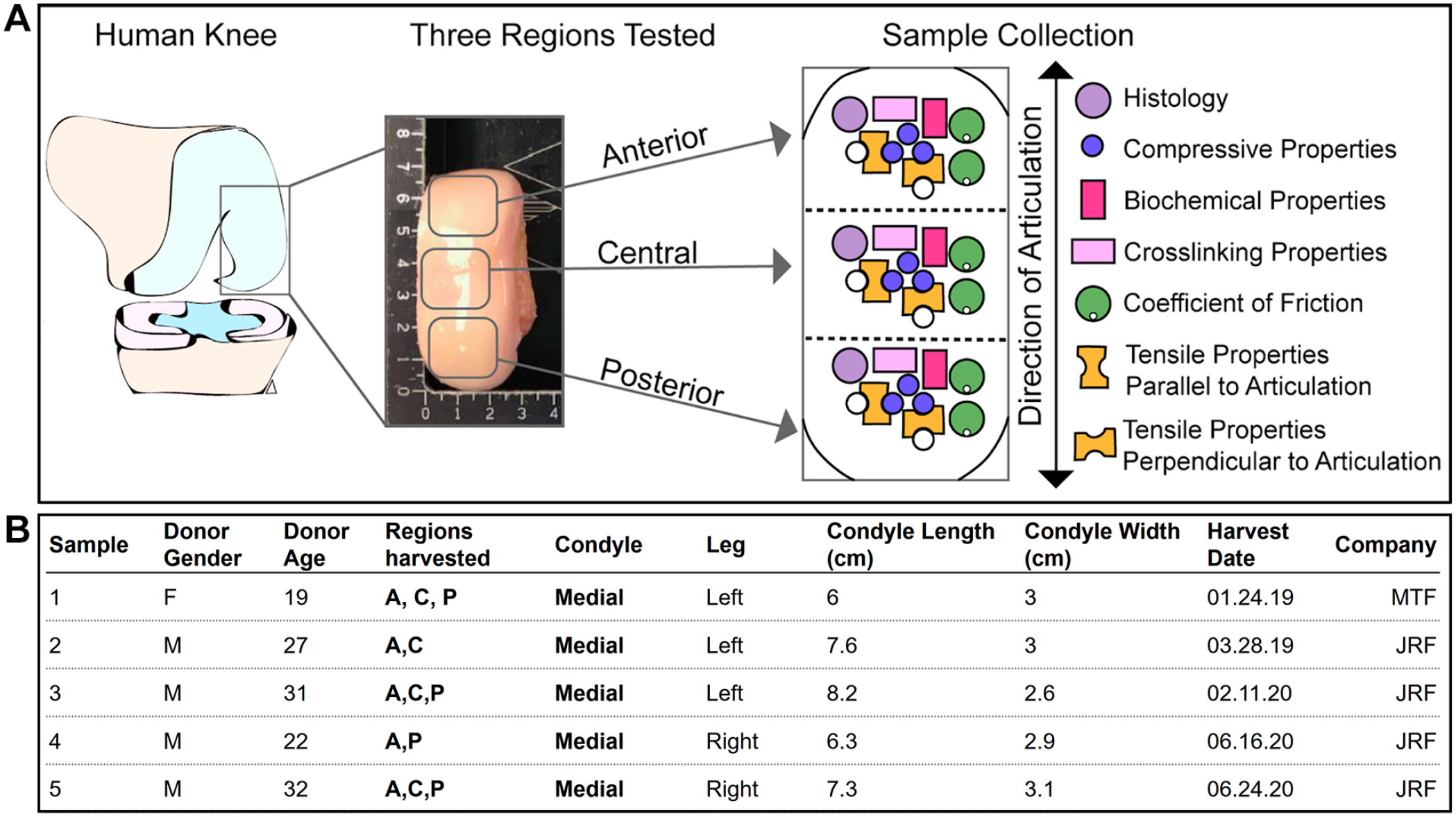
Experimental design for the characterization of the human medial femoral condyle.
Histology
Samples (5-mm-diameter punches) were fixed in 10% neutral-buffered formalin, embedded, and sectioned at a thickness of 6 µm for histological evaluation. The sections were subsequently processed and stained with hematoxylin and eosin (H&E), safranin-O and fast green, and picrosirius red using standard protocols. Sections where further analyzed using OARSI grading system to assess the quality of the samples. 22
Biochemical Properties
Cartilage samples (two 1 mm punches) were trimmed to a similar size, excluding any calcified tissue. The samples were then weighed to obtain wet weight (WW), lyophilized for 3 days, and weighed again to obtain dry weight (DW). Lyophilized samples were digested in 125 μg/mL papain (Sigma), 5 mM N-acetyl-L-cysteine, and 5 mM ethylenediaminetetraacetic acid (EDTA) in phosphate buffer pH 6.5 for 18 hours at 60°C. DNA content was quantified with a Picogreen assay (ThermoFisher Scientific). Sulfated glycosaminoglycan (GAG) content was quantified using a Blyscan Glycosaminoglycan Assay kit (Biocolor, Newtownabbey, Northern Ireland). Total collagen content was quantified using a modified chloramine-T hydroxyproline assay using Sircol 0.5 mg/ml acid-soluble bovine collagen as a standard (Biocolor). 23
For the quantification of collagen PYR crosslinks, separate samples (~0.2-0.6 mg) were weighed, lyophilized, and acid-digested for 12 hours in 6N HCl at 105°C. After evaporation, dried hydrolysates were resuspended in a 75%/25% (v/v) solution of 0.1% formic acid and acetonitrile, filtered, and transferred to liquid chromatography–mass spectrometry (LCMS) autosampler vials. Samples were measured via liquid chromatography mass spectrometry using a Cogent diamond hydride high-performance liquid chromatography (HPLC) column (2.1 mm × 150 mm, particle size 2.2 μm, pore size 120Å, MicroSolv) and a pyridinoline standard (BOC Sciences). 24
Tribological Properties
Tribology was assessed using a custom pin-on-disk tribometer under boundary lubrication conditions. 11 For every donor tissue, 2 India ink-labeled 3 mm punches were obtained and glued to the pin of the tribometer parallel or perpendicular to the direction of articulation to obtain measurements in both orientations. All samples were immersed in phosphate-buffered saline (PBS), allowed to reach equilibrium for 2 minutes, and then sheared against the test surface for 5 minutes with velocity set to 0.5 mm/s and a compressive normal force applied by a 200 g mass. The measured coefficient of friction describes the interaction between the articular cartilage sample and the underlying glass plate immersed in PBS. Data were collected at a rate of 50 Hz.
Tensile Properties
Uniaxial tension was performed in an Instron model 5565 (Instron, Canton, MA). Dog bone-shaped samples were obtained from every donor sample, parallel and perpendicular to the direction of articulation, with a gauge length of 1.55 mm. For each direction, the top 1mm layer and full-thickness samples were prepared. 25 All samples were photographed to measure thickness and width using ImageJ. The ends of the dog bone were fixed to paper tabs with cyanoacrylate outside the gauge length and pulled at a strain rate of 1% of the gauge length/second until failure. Using the cross-sectional area, Young’s modulus was obtained from the linear region of the stress-strain curve and the UTS was defined as the maximum stress obtained.
Compressive Properties
The aggregate modulus and shear modulus were obtained using a semi-analytical, semi-numerical, linear biphasic model followed by a finite element optimization. 26 Briefly, 3 mm punches were obtained from each donor tissue sample and trimmed to a height of 1 mm approx. Each sample was photographed, then submerged in PBS until equilibrium, and indented with a flat porous 1-mm-diameter tip perpendicular to the surface of the sample. A tare mass of 0.5 g was applied until equilibrium was achieved (≥600 s), and a test mass of 7.5-12 g was applied during testing (≥1800 s), which corresponded to ~10% strain. During the test, deformation is measured over time. A porous platen is used to allow for fluid to exude from the sample at the platen contact site. By fitting the stress versus time data with finite element analysis using the biphasic model, the aggregate modulus, Poisson ratio, and permeability of the sample were determined. 27
Relaxation modulus, instantaneous modulus, and coefficient of viscosity were calculated using a standard linear solid viscoelasticity model and an Instron model 5565 (Instron, Canton, MA). 28 For height detection, the 3 mm punches were submerged in PBS and compressed until reaching a 0.2 N force at a rate of 0.025 mm/second, and compared with a reference value set prior to testing. For testing, samples were preconditioned with 10 cycles at 5% strain and a 10% strain rate of the sample height/second, and incremental stress-relaxation were performed at 10% (≥900 s), and 20% strain (≥1200 s).
Statistical Analysis
All quantitative biochemical and biomechanical tests were performed using a samples size of n = 4-5. All data were presented as mean ± standard deviation. A single factor 1-way analysis of variance was used to assess differences among experimental groups. Multiple comparisons were performed using Tukey’s post hoc test. Letters depict the connecting letters report, and different letters over bars indicate statistical difference between groups (P < 0.05). All statistical analyses were performed using GraphPad Prism version 8.4.1 for Windows (GraphPad Software, San Diego, CA). The correlation analyses were also performed in GraphPad Prism using a 2-tailed parametric Pearson correlation.
Results
Histological Evaluation
Histologic staining was used to visualize tissue morphology and distribution of collagen and glycosaminoglycan ( Fig. 2 ). Per the companies providing the tissues for allograft transplantation, all donors’ medical and social history were screened for medical conditions or diseases that could contraindicate the donation of tissues, in compliance with the U.S. Food and Drug Administration (FDA) regulations published in 21 CFR Part 1271. 29 Moreover, and as stated by JRF Ortho (which performs a “Comprehensive inspection criteria to detect allograft imperfections, tissue and cartilage quality”), a physical assessment was performed by the companies in order to provide tissues that consist in mature, viable hyaline cartilage matrix, and proprietary processing methods for prolonged storage were used (e.g., JRF uses proprietary processing methods shown to maintain 80% absolute cartilage viability). Thus, considering the source of the samples and their intended use, all chondral tissues were considered to be healthy (e.g., with no signs of chondral degeneration, morphological abnormalities, or physical/chemical damage) after a gross morphological examination.
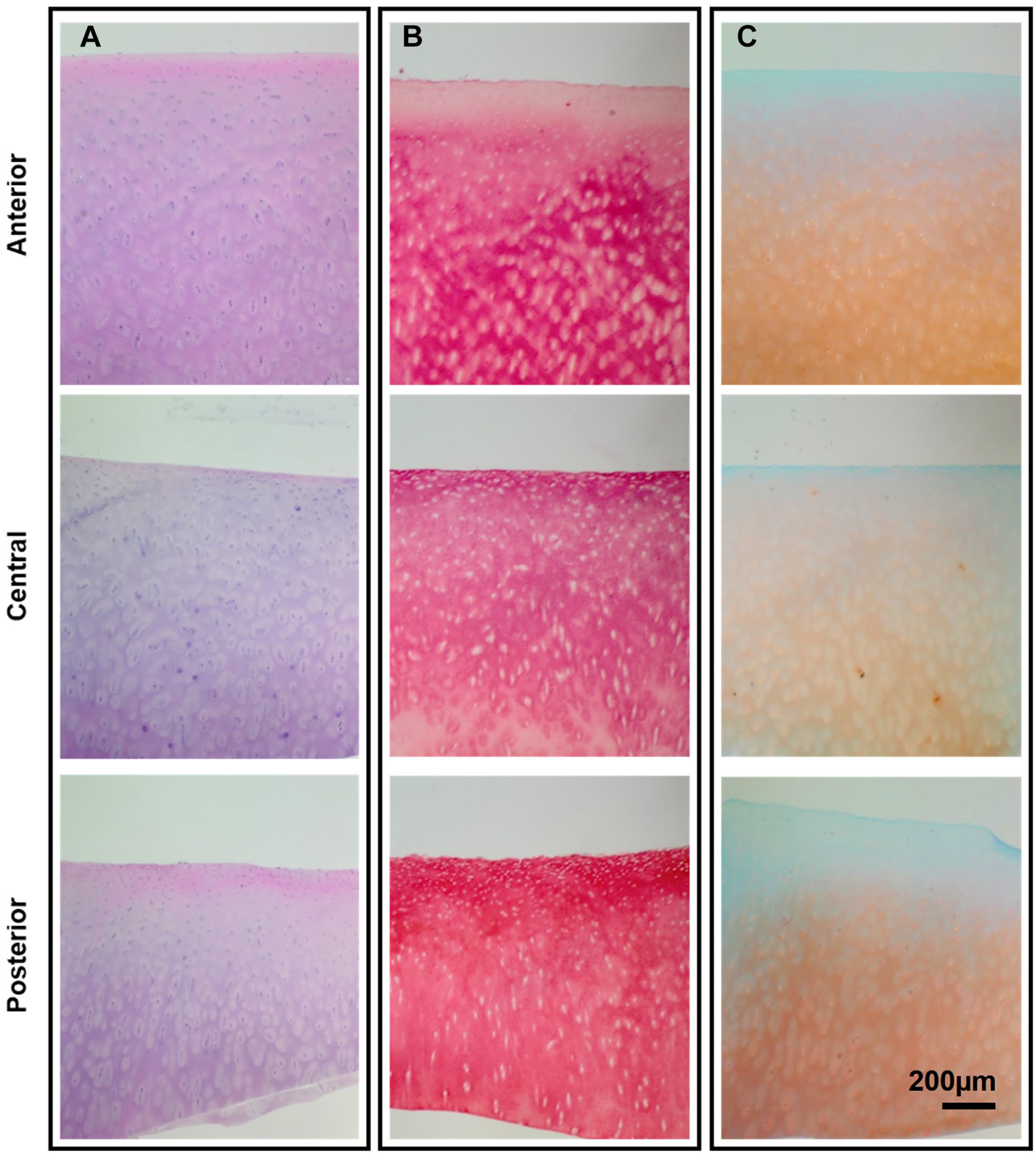
Histological analysis of the medial condyle.
The adult cartilage H&E staining revealed a predominant basophilic stain, and an eosinophilic surface layer toward the anterior and posterior regions. Cell lacunae were observed in all regions of the medial condyle ( Fig. 2A ). Multiple isogenous groups and vertically arranged cells were also found toward the deep zone in all regions. The picrosirius red stain, to visualize total collagen distribution, showed a high intensity throughout the tissue and an increased intensity on the superficial zone toward the posterior region ( Fig. 2B ). The safranin-O stain, with a fast green counterstain, showed that GAG is primarily concentrated in the deep zone of cartilage, as reflected by the intensity of the stain ( Fig. 2C ). Finally, and toward corroborating the healthy state of the samples, the sections were analyzed and scored using the OARSI grading system, 22 which confirmed that all the samples showed a normal architecture and possessed a grade of 0, consistent with undamaged normal hyaline cartilage, and with no discontinuity or fibrillation observed in any of the samples
Biochemical Properties
Glycosaminoglycan, collagen, and pyridinoline content of the native articular cartilage in the 3 topographical locations of the condyle are shown in Figure 3 . Glycosaminoglycan content was normalized to both wet weight and dry weight ( Fig. 3A ). There were no significant differences among the regions in terms of glycosaminoglycan normalized to wet weight, with an averaged content ranging between 5% and 6.8%. Likewise, there were no significant differences when normalized to dry weight; the anterior region contained 20.9% ± 3.7% glycosaminoglycan per dry weight, whereas the central and posterior regions contained 18.8±7.4 and 18.2±2.6% glycosaminoglycan per dry weight, respectively. Similarly, no significant differences the collagen content per wet weight showed, albeit with an increase in the posterior region ( Fig. 3B ). This difference is significant when normalizing the collagen content per dry weight; the posterior region showed significantly higher amount of collagen (78.4% ± 11.8%) compared with the anterior (52.3% ± 11.2%) and central (47.1% ± 22.2%) regions. Finally, no significant differences among the regions were found when comparing the quantification of pyridinoline crosslinks content per wet weight ( Fig. 3C ). However, the pyridinoline content per collagen content revealed a significant increase of the collagen crosslinking toward the central and the anterior region.
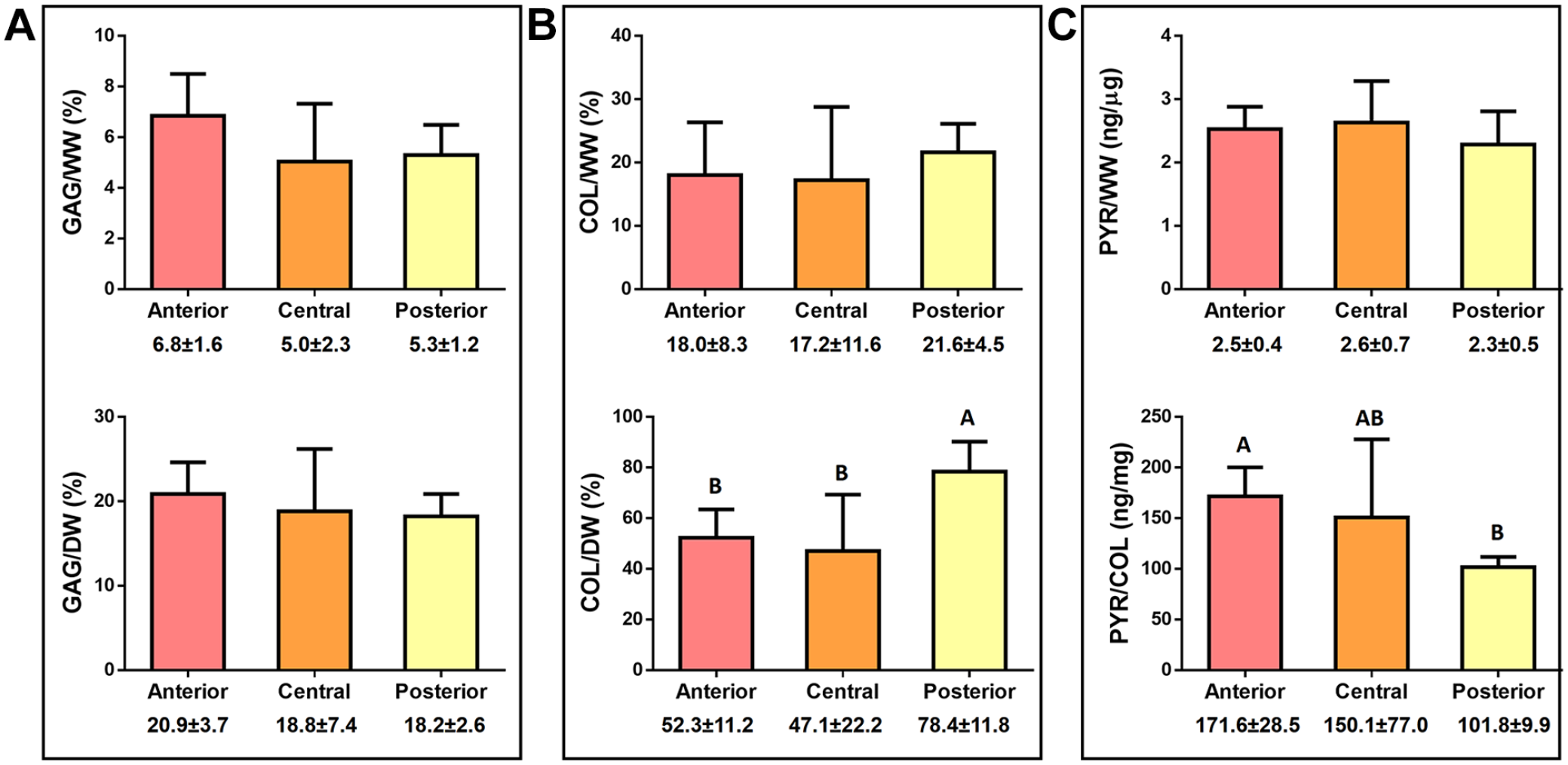
Biochemical properties of the human medial femoral cartilage.
Tribological Properties
The coefficients of friction for each region were obtained in 2 directions: (1) in the direction of articulation and (2) perpendicular to articulation ( Fig. 4 ). Interestingly, there were no significant differences in the coefficient of friction when comparing directionality. Additionally, there were also no significant differences among the coefficient of friction of the 3 regions. For all regions, regardless of the direction tested, the coefficient of friction varied between 0.22 and 0.26 ( Fig. 4 ).
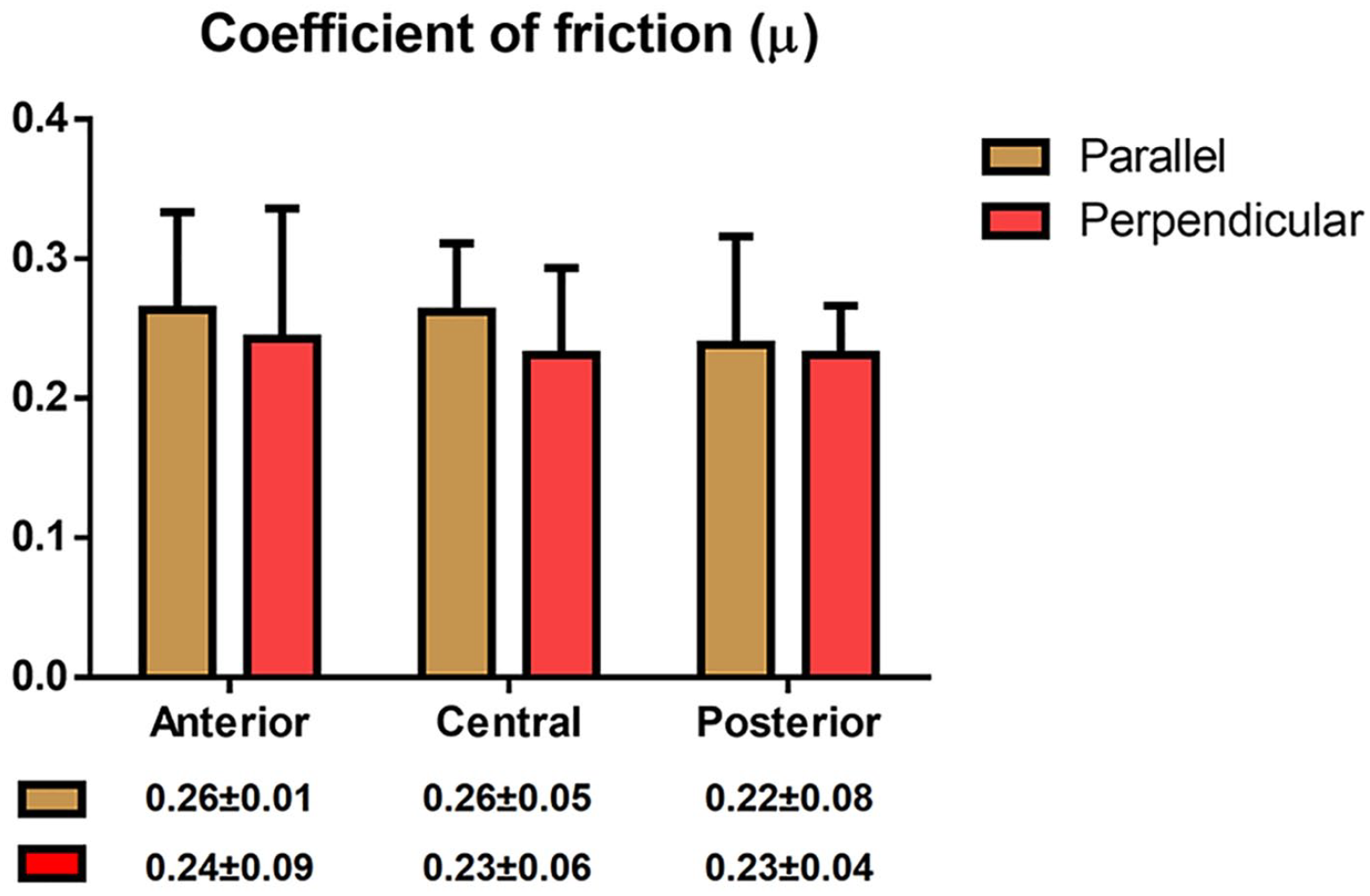
Coefficient of friction of the articular cartilage surface. Three distinct regions of human femoral cartilage were tested in 2 directions: anteroposterior and mediolateral. The coefficient of friction was isotropic and homogeneous throughout the medial condyle in young, healthy tissues.
Tensile Properties
Young’s modulus and UTS of the human femoral condyle were characterized in the direction parallel to articulation ( Fig. 5A ). The anterior region possessed significantly higher Young’s modulus values for both top 1 mm layer and full-thickness tissues (32.5 ± 11.7 and 22.9 ± 7 MPa, respectively) compared with top 1 mm layer and full-thickness tissues of the central region (12.4 ± 4.5 and 7.0 ± 0.5 MPa, respectively). The posterior region was also less stiff compared with the anterior region, albeit only significant for the full-thickness tissue (11.6 ± 3.3 MPa). In terms of the UTS, the anterior region of the condyle had a significantly higher strength compared with the central and posterior regions, regardless of the layer measured. Finally, and although the stiffness of the top 1 mm layer trended higher than the stiffness of the full-thickness tissue, the comparison between both measurements showed no significant differences in this study.
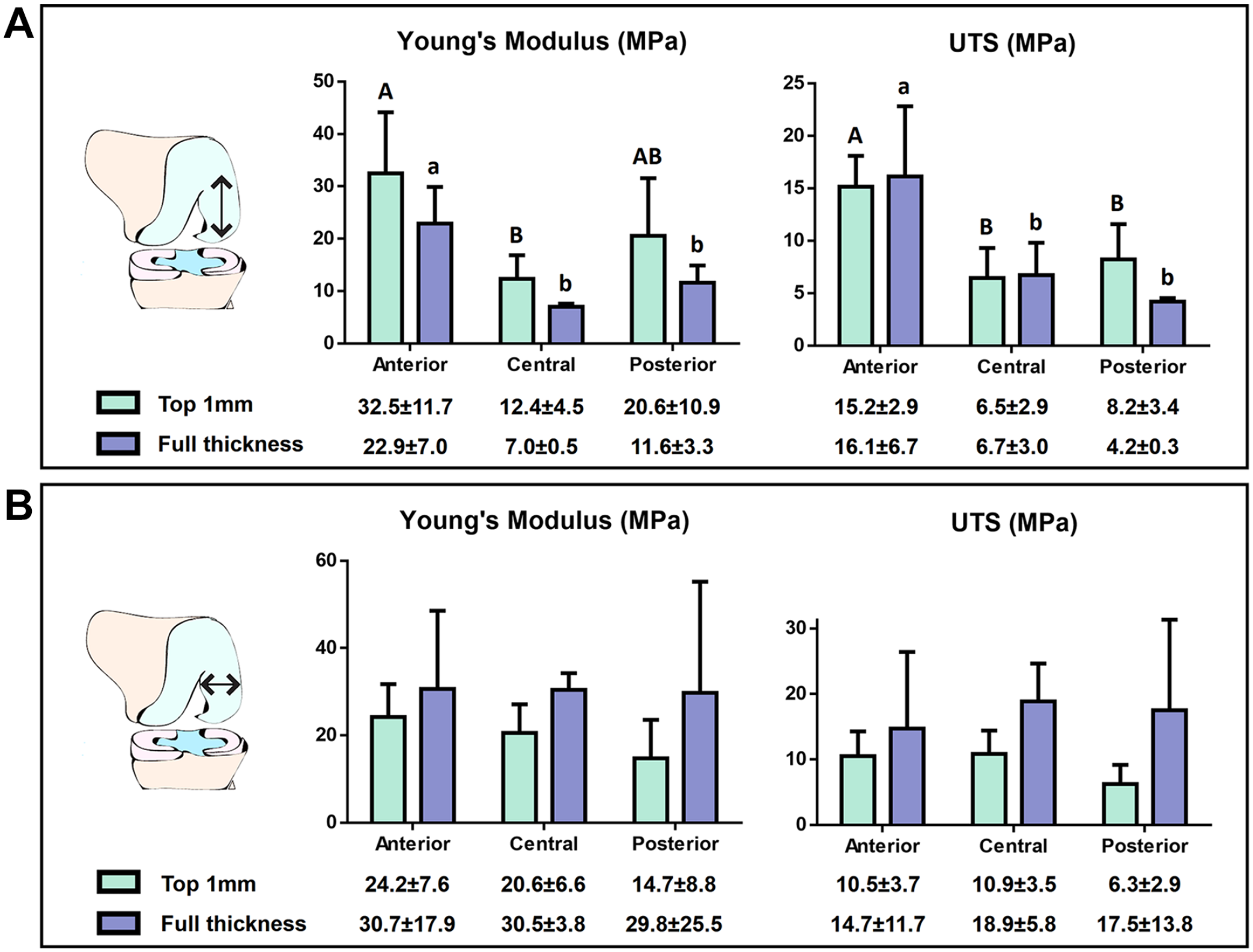
Tensile properties of the human medial femoral condyle.
The tensile properties of the human femoral condyle were also characterized in the direction perpendicular to articulation, as indicated in the schematic shown in Figure 1A . No regional differences of Young’s modulus or the UTS were observed when testing the tissue in this direction ( Fig. 5B ).
Compressive Properties
Aggregate modulus obtained from creep indentation varied significantly between the anterior and posterior regions of the medial condyle. The aggregate modulus of the anterior region was 777 ± 245 kPa, whereas for the posterior region 379 ± 94 kPa. The central region, with an aggregate modulus of 602 ± 171 kPa, represents a transition region ( Fig. 6A ). Shear modulus values also varied significantly following the same pattern observed for the aggregate modulus. The shear modulus values for the anterior, central, and posterior regions of the human femoral condyle were 442 ± 132 kPa, 349 ± 116 kPa, and 223 ± 62 kPa, respectively, with the anterior and posterior regions being significantly different ( Fig. 6A ).
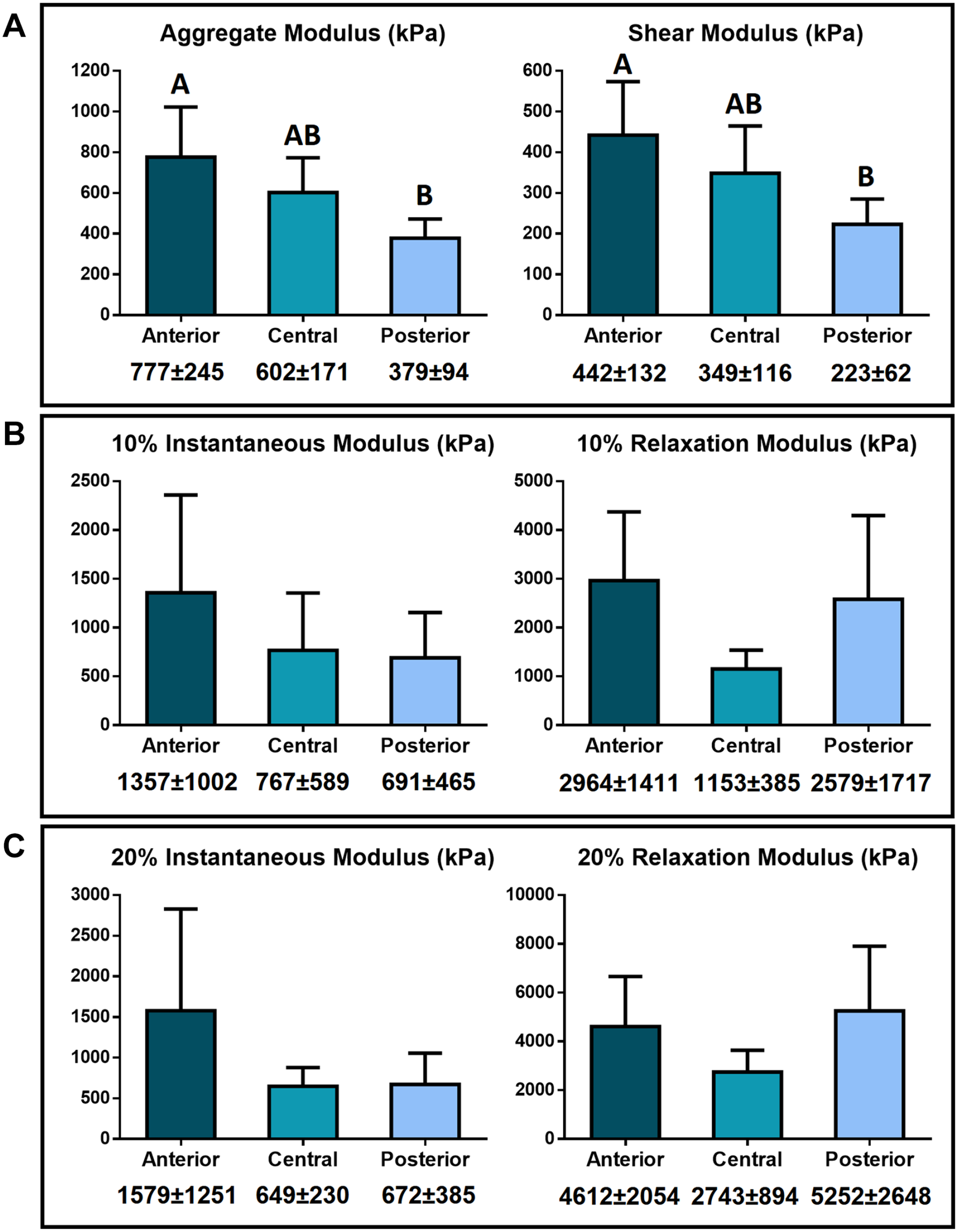
Compressive properties of 3 distinct regions of human femoral cartilage.
Compressive stress–relaxation testing showed no significant differences among the medial condyle regions (
Fig. 6B
). The average instantaneous modulus at 10% strain was 1357 ± 1002kPa, 767 ± 589kPa, and 691 ± 465 kPa for the anterior, central, and posterior regions, respectively. The average relaxation modulus at 10% strain was 2964 ± 1411 kPa, 1153 ± 385 kPa, and 2579 ± 1717 kPa for the anterior, central, and posterior region, respectively. At 20% strain, the average instantaneous modulus was 1579 ± 1251 kPa, 649 ± 230 kPa, and 672 ± 385 kPa for the anterior, central, and posterior region, respectively, and the average relaxation modulus was 4612 ± 2054 kPa, 2743 ± 894 kPa, and 5252 ± 2648 kPa for the anterior, central, and posterior region, respectively (
Fig. 6C
). No significant differences were observed in the coefficient of viscosity (
A high correlation was found between the compressive properties and the PYR/COL content, for both the aggregate modulus (R2 = 0.98) and the shear modulus (R2 = 0.98) (
Discussion
Toward elucidating whether topographical differences in cartilage properties exist in the medial femoral condyle and whether such differences inform the incidences of pathology, this study applied histological, biochemical, and mechanical assays to the anterior, central, and posterior regions of young (mean age of 26.2±5.6 years), healthy human medial condyles. Based on clinical incidences of pathology, the limited data currently available on human tissues, and animal characterization studies,9,12 it was hypothesized that the posterior region of the medial condyle would exhibit the lowest mechanical properties, and that tissue composition would follow the established structure-function paradigm for articular cartilage. Our results indicate that, in contrast to the tribological properties, both compressive and tensile properties vary according to location within the medial condyle, with the posteromedial compartment displaying the lowest properties. Interestingly, the aggregate modulus, shear modulus, and UTS strongly correlate with the PYR crosslink content normalized to collagen content, suggesting new function-structure relationships. Overall, this work’s findings are significant for providing (1) tribological properties of young, healthy human cartilage in multiple directions, (2) evidence of new structure-function relationships between collagen crosslinks and compressive properties in healthy, young samples, and (3) an assessment of healthy human articular cartilage of the medial condyle toward understanding degenerative processes.
With regard to the healthy medial condyle cartilage, Young’s modulus and UTS showed topographical variation when measured in the direction of articulation. For the full-thickness samples, the anterior region showed the highest values, which were 3.3 and 2.0 times greater than the central and posterior regions for Young’s modulus, and 2.4 and 3.8 times greater than in the central and posterior regions for UTS. The compressive properties also varied within the medial condyle. The aggregate modulus of the anterior region was 1.4 and 2.1 times greater than the central and posterior regions, and the shear modulus was 1.3 and 2.0 times greater than in the central and posterior regions. Collagen content and PYR crosslink per collagen content also showed topographical variations. Importantly, the collagen crosslink content was correlated with UTS (R2 = 0.73), but this correlation was much stronger with the compressive properties (R2 = 0.98). In addition, although the tribological characteristics of bovine, equine, porcine, and ovine cartilage have been previously investigated, 30 to the best of our knowledge, this study is the first to provide a detailed description of the tribological properties of human femoral condyle articular cartilage, including topographical and directional data. It was found that, for the young, healthy tissues examined, tribological properties did not significantly differ when testing in the direction of articulation or perpendicular to articulation, indicating frictional isotropy.
Limited tribology data have been collected from human articular cartilage.15,30 In this study, characterization of young, healthy human medial condyle cartilage showed that the coefficient of friction under boundary lubrication appeared isotropic and ranged between 0.22 and 0.26, with no significant differences among topographical regions. This finding is significant because it provides a target value for the coefficient of friction of engineered implants intended to repair or replace human cartilage. For comparison, a wide range of coefficient of friction (from 0.002 to 0.5) has been reported for animal cartilages in the past depending on the tissue and loading configuration used,11,31 -34 and, of these, only 1 reported topographical differences, showing that the posteromedial condyle had the highest value. 11 It should be noted that the coefficient of friction varies from study to study due to differences in tissue age, testing configuration (boundary vs. dynamic lubrication), and the presence of PBS or synovial fluid.35,36 In a recent review, 37 the only human tribological data provided was a coefficient of friction of 0.013 for the human hip, highlighting the dearth of information on this topic. The lack of additional evidence of human articular cartilage coefficient of friction values highlights the significance of the tribological results obtained in this work.
No significant differences in the coefficient of friction were observed when testing parallel or perpendicular to the direction of articulation, in spite of reports of anisotropy in bovine articular cartilage samples. 21 A possible cause for the isotropy observed, aside from species-specific variations, may be the age of the donors used in this study. With an average age of 26.2 ± 5.6 years and all tissues considered healthy, the cartilage examined might have been exposed to considerably less age-related wear, a factor known to affect tissue properties.14,37 The observation that young, healthy tissue does not display anisotropy in its frictional properties may indicate that implants, including tissue-engineered neocartilage, would not have to be designed to have directional coefficients of friction.
This study found that tensile properties of the medial condyle vary according to location. The posterior region showed significantly lower Young’s modulus and UTS values compared with the central and anterior regions, when the testing was performed anteroposteriorly. In contrast, the collagen content was significantly larger in the posterior region compare with the central and anterior regions. In agreement to the known structure-function relationships of articular cartilage that correlate collagen content with tensile properties 38 or GAG content with compressive properties, 39 one would infer that the collagen content in the posterior region would be lower as well. While the collagen content values in this study were on par with literature, 40 collagen did not correlate with tensile properties. This may be due to several reasons, such as variable collagen organization between regions due to the different stresses to which different regions are exposed. 41 Our results showed that the PYR crosslink per collagen correlates with the tensile properties, especially the UTS (R2 = 0.73). Differences in crosslinks may better explain the increase in the tensile properties, as the formation of covalent PYR crosslinks between individual collagen fibers stabilizes the formation of heterotypic fibrils and the tissue extracellular matrix. 42 It is, thus, suggested that the PYR crosslink content is an important contributor to tensile properties.
The tensile properties in the top layer as well as the full-thickness of articular cartilage were also quantified. Young’s modulus values consistently trended higher in the top 1 mm layer samples, when testing in the direction of articulation. This is in agreement with prior work showing that the superficial zone (top 250-500 µm) has a higher tensile stiffness than the full-thickness tissue. 43 However, the tensile properties did not differ significantly with respect to directions. This could be because in this study we tested the top 1 mm layer, which includes the superficial zone and part the middle zone. More work needs to be done on characterizing the tensile properties of young, healthy knee cartilage, especially to establish zonal effects.
The compressive properties of the medial condyle also decrease in the anterior-posterior direction. The aggregate and shear moduli of the posterior region were 2.1 and 2.0 times lower than those of the anterior region. Similar results were observed for the 10% instantaneous modulus, however, the high variability observed in the stress-relaxation data calls for a larger number of samples. In terms of the known relationship between GAGs and compressive properties,
44
our results showed correlations between GAG/DW and aggregate modulus (R2 = 0.93) and shear modulus (R2 = 0.86) (
The scarcity of young, healthy tissues is an important limitation toward the determination of the biomechanical and biochemical properties of articular cartilage tissue. While multiple studies exist in which osteoarthritic samples have been quantified, a handful have employed healthy tissues. The work presented by Ebrahimi et al., 16 in which healthy cartilage was considered, mentions the presence of full-thickness degradation in several medial condyles of total knee replacements and healthy knees. In other cases, the authors have pooled samples with a certain degree of damage, such as cartilage with an OARSI grade 1, as healthy tissue. 17 In this work, the limited number of donors and samples per group was compared with the results reported in other studies so as to determine the accuracy of our results. Using the aggregate modulus, it was determined that the mean and variance of the results shown in this work are comparable to those of the results reported by others.12,15 -17 In addition, we performed an a posteriori power analysis for those measurements that did not show statistical differences, with the objective of identifying the appropriate sample size needed to show significance in the properties. This post hoc power analysis indicated that one would need to employ an unrealistic sample size to show significance, supporting our findings. Thus, and in spite of the limiting availability of young, healthy human cartilage, the results of this work are in agreement with the scant literature on healthy human articular cartilage.
The lower tensile and compressive properties identified in the posteromedial condyle of young, healthy donors, together with the higher contact pressure and strains reported in the posteromedial condyle compartment, may be associated with the higher prevalence of injury and tissue degeneration in the posterior region.8,45,46 Our results show that UTS, aggregate modulus, and shear modulus have a strong and positive correlation to the degree of collagen crosslinking (PYR/Col). Thus, articular cartilage’s predisposition to pathologies may be better predicted by the degree of PYR crosslinks. This new emerging relationship between PYR crosslinks and strength and stiffness of the articular cartilage matrix may also be linked to degenerative diseases in the knee joint.
The biochemical and mechanical properties of the human condyle, especially from young and healthy donors, are yet to be fully determined. In this study, the topographical analysis of the medial condyle showed isotropy and homogeneity of the coefficient of friction. The tensile and compressive properties, instead, varied according to location. Also, new structure-function relationships emerged. Specifically, UTS, aggregate modulus, and shear modulus were shown to have a strong correlation with the PYR crosslink per collagen content. Finally, the posterior region of the condyle, which has been shown to be the region with the highest incidence of chondral degeneration, was identified as the region with the lowest tensile properties, compressive properties, and PYR/collagen, indicating that these tissue properties may predispose the cartilage to injuries and degeneration.
Supplemental Material
sj-docx-1-car-10.1177_19476035221141421 – Supplemental material for Topographical Characterization of the Young, Healthy Human Femoral Medial Condyle
Supplemental material, sj-docx-1-car-10.1177_19476035221141421 for Topographical Characterization of the Young, Healthy Human Femoral Medial Condyle by Evelia Y. Salinas, Gaston A. Otarola, Heenam Kwon, Dean Wang, Jerry C. Hu and Kyriacos A. Athanasiou in CARTILAGE
Footnotes
Author Contributions
E.Y.S., G.A.O., D.W., J.C.H., and K.A.A. conceived of and designed this study; E.Y.S. and G.A.O. acquired the data, E.Y.S., G.A.O., D.W., J.C.H., and K.A.A. reviewed and interpreted the data; and E.Y.S., G.A.O., J.C.H., and K.A.A. wrote and edited the paper. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge support from the National Institutes of Health (NIH) R01 AR067821 and Fulbright Chile Scholarship. This funding source did not play a role in the collection, analysis, or interpretation of the data presented.
Ethical Approval
This study does not constitute as human subject research and is exempt from institutional review board approval.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
