Abstract
Objective
Single-surgery variants of autologous chondrocyte implantation to repair cartilage are emerging, but practical data on such procedures are scarce. We set out to describe a 1-stage autologous chondrocyte co-implantation procedure and include quantitative characteristics of the biopsy tissues collected and of the cells obtained from those tissues in the operating room.
Design
Data concerning patient age, articular cartilage lesion size and location, as well as measurements of cartilage biopsy mass, bone marrow aspirate volume, and the cell yields harvested from those biopsies were intraoperatively collected for 141 patients.
Results
The mean patient age was 35.7 ± 9.8 years, and the mean total lesion size was 4.0 ± 3.1 cm2. Cartilage biopsy mass ranged from 0.19 to 2.0 gram and provided a mean yield of 1.23 × 106 chondrocytes/gram. Bone marrow aspirate volume ranged from 7.2 to 62 milliliters and provided a mean yield of 7.18 × 106 mononuclear cells/mL. The cell yields did not correlate with patient age, which ranged from 12 to 57 years. The mean primary chondrocyte supply was 272 thousand per cm2 of lesion, ranging from 10.4 thousand to 1.07 million per cm2. The total cell supply was kept at 9 million cells per cm2 of lesion by adding mononuclear cells to the chondrocytes. The mean tissue processing time was 100 ± 19 minutes, which was frequently used to perform concurrent interventions.
Conclusion
Single-surgery co-implantation of clinically relevant numbers of autologous primary articular chondrocytes and bone marrow cells is feasible for a wide range of ages and lesion sizes.
Introduction
The articular cartilage of synovial joints has a poor capacity to heal. If an injury extends into the subchondral bone, repair tissue will grow, but it is of a fibrous quality and prone to wear. 1 If an injury involves only the cartilage and does not penetrate into the bone marrow, no apparent repair response is initiated. 2 Surgical intervention may become necessary if the injury becomes symptomatic. For articular cartilage lesions that exceed an area of 2 cm2, matrix-assisted autologous chondrocyte implantation (MACI) is considered appropriate. 3
Treatment of knee cartilage lesions with MACI provides sustained improvement of patient-reported outcome measures and of magnetic-resonance-image-based scores, both at mid-term 4 and at long-term. 5 Moreover, MACI outperforms microfracture in the treatment of lesions larger than 3 cm2.6,7 Nevertheless, the 2-stage approach of MACI does have drawbacks. The patient has to undergo 2 invasive procedures. The transport of human tissues (the cartilage biopsy and, a few weeks later, the cell-seeded implant) needs to be registered and traceable, imposing transport costs and risks. Expensive laboratory work is necessary for the expansion of chondrocytes and membrane seeding, which must comply with Good Manufacturing Practices and Good Laboratory Practices. Incidentally, a cell culture may get infected. Adding to these challenges is the biological fact that the primary articular chondrocytes (PCs) isolated from the cartilage biopsy will shift their phenotype during the in vitro monolayer expansion. 8 The resulting daughter chondrocytes are often described as “dedifferentiated,” referring to their more fibroblastic phenotype. These drawbacks stimulated the exploration of single-stage variants of autologous chondrocyte implantation in the past decade and a half.
Research into the dissociation of tissue resulted in “short-isolation” protocols for cells, including protocols as brief as 30 minutes for the isolation of chondrocytes, which previously required isolation times of 16 to 22 hours. 9 The yields of such dissociation protocols are roughly 1 million cells per gram of tissue. Within autologous chondrocyte implantation (ACI) practices, the supply of cultured chondrocytes gravitates to around 1 million cells per 1 cm2 of lesion. 10 Adhering to that practice, a single-stage variant of ACI implementing a short-isolation protocol would require the harvest of a cartilage biopsy of, for example, 4 grams to treat a lesion of 4 cm2. The harvest of such amounts of articular cartilage is too detrimental to the articular donor surface. A solution to this problem was found with the discovery of coculture effects.11,12 When chondrocytes are cocultured with cells of a stromal type, the glycosaminoglycan output per coculture or per in vivo co-implantation construct is frequently greater than that of a 100%-chondrocyte culture. In addition, chondrocyte proliferation is enhanced in cocultures and co-implants, and gene expression of, amongst others, collagen type II, aggrecan, and SOX9 is upregulated.13,14 These findings formed the basis of a single-stage autologous chondrocyte co-implantation strategy in which the chondrocyte supply is partly supplanted with cells of a stromal, and possibly multipotent, phenotype. To date, 2 clinical case series explored this strategy: the INSTRUCT trial (NCT01041885) and the IMPACT trial (NCT02037204).
In the INSTRUCT trial, which started in January 2010, autologous PCs were co-implanted with autologous mononuclear bone marrow cells (MNCs) to treat knee cartilage lesions, using a load-bearing osteochondral scaffold to house the cells. 15 In March 2013, the IMPACT trial started, in which autologous primary articular chondrons—chondrocytes with pericellular matrix—were co-implanted with allogeneic multipotent mesenchymal stromal cells to treat knee cartilage lesions.16,17 For both trials, statistically and clinically relevant improvements in patient-reported outcome measures were reported, indicating the potential of single-stage autologous chondrocyte co-implantation.
Single-stage autologous chondrocyte co-implantation is relatively new, and its use is not widespread. It is important to report on the basic characteristics of performing a single-stage autologous chondrocyte-based treatment in the operating room (O.R.). Here, we present quantitative data collected in the O.R. on the input (tissues) and output (cells) of a commercially available service for the single-stage co-implantation of autologous articular chondrocytes with MNCs. We report data on the collection of articular cartilage and bone marrow aspirate, the encountered variation in cell yields harvested intraoperatively from those tissues, how those yields relate to patient age, the composition of the cell mixture that was implanted, the scaffold types used, and the duration of the procedure. A reflection on the methodology of this single-stage autologous chondrocyte co-implantation and how its cell output compares to chondrocyte-based alternatives is provided in the Discussion.
Methods
Patients and Intraoperative Procedure
The single-stage autologous chondrocyte co-implantation method under study (CartiONE Service, Cartilage Repair Systems, LLC, New York, NY, USA) was used in a selection of hospitals throughout Europe for the treatment of 141 patients between February 2010 and May 2022. The initial 40 patients were part of the INSTRUCT trial (NCT01041885). In 3 of the 141 patients, the isolated cells were used to seed a partial-meniscus substitute scaffold (Actifit, Orteq Sports Medicine, London, UK). In the other 138 patients, the cells were used in the treatment of lesioned articular cartilage—including the INSTRUCT trial patients.
The O.R. procedure is as follows. The patient is provided with general or spinal anesthesia. During surgery, the patient lies in supine position. The orthopedic surgeon first arthroscopically diagnoses the lesion or lesions and determines suitability of the treatment. The treatment is indicated for symptomatic articular cartilage lesions that have defined borders and are of grade 2 or 3 according to the system provided by the International Cartilage Regeneration & Joint Preservation Society.
18
An entirely arthroscopical approach is possible when the type of scaffold and the size and location of the lesion allow for it. Otherwise, the surgeon proceeds with open-knee surgery shortly before scaffold placement and cell implantation. The cell isolations are performed in the O.R. under sterile laminar airflow by trained and qualified technicians with experience in tissue culture, cell culture, and cell counting (
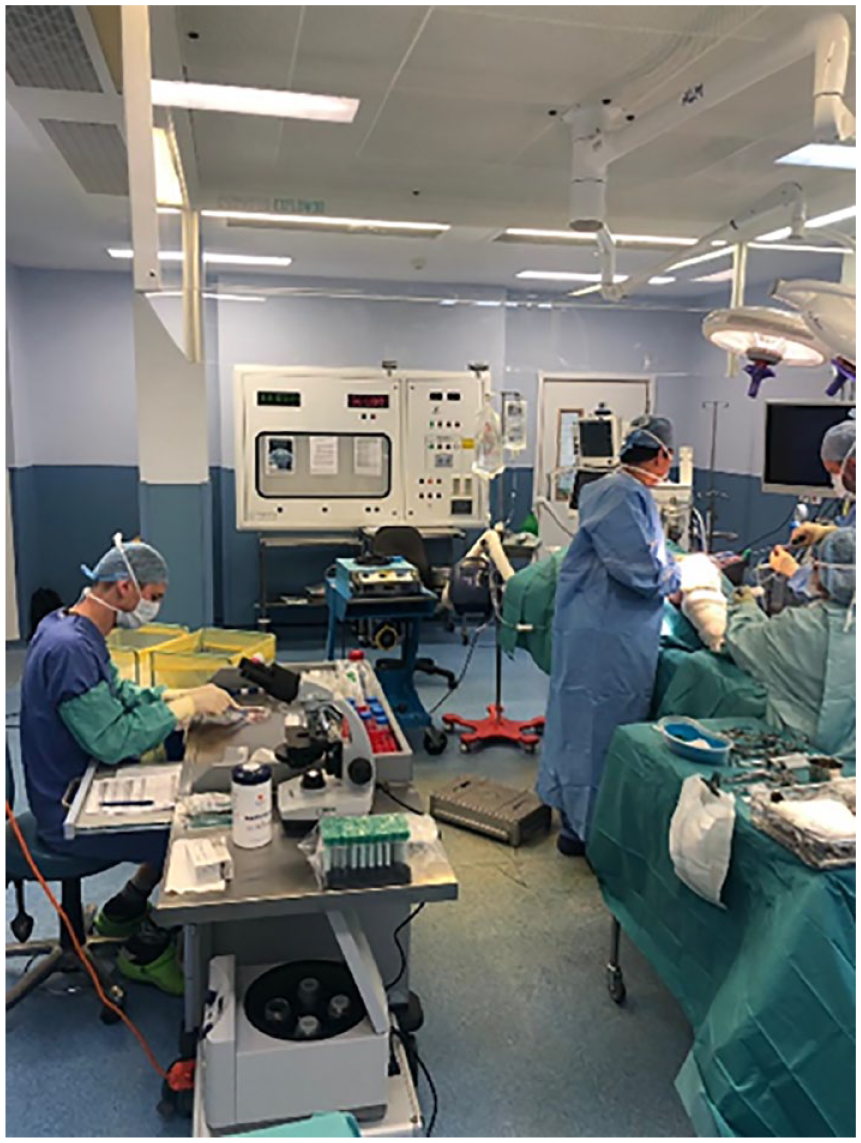
Intraoperative cell-isolation set-up. The workstation is positioned under sterile laminar airflow.
The technicians operate at a fully equipped workstation, which includes an incubator, a centrifuge, and a phase-contrast light microscope. The surgeon debrides the articular cartilage lesion so that its borders are stable and perpendicular to the subchondral bone. The surgeon also collects a small biopsy of healthy-appearing less-load-bearing cartilage that is added to the debridement (see “The Use of Primary Articular Chondrocytes and Chondrons” section). This less-load-bearing cartilage is commonly taken from the intercondylar notch or the linea terminalis. The cartilage tissue is weighed and manually minced with scalpels. The cartilage particulate is digested at 37 °C for 35 minutes with enzymes sourced from the bacterium Clostridium histolyticum. The digestate is strained and the isolated PCs are washed and counted. After cartilage collection, the surgeon proceeds with aspiration of bone marrow. Although aspiration from the iliac crest is advised by the technicians, the surgeon decides on the aspiration site and may opt for aspiration from, for example, the distal femur. The bone marrow aspirate is incubated with an erythrocyte-lysing buffer, and the remaining cell isolate is washed and counted. The number of primary MNCs that is added to the PCs depends on the lesion size: The final cell mixture is specified to supply the surgeon with a total of 9 million cells per cm2 of lesion. The minimum PC supply is specified as 60.3 thousand cells per cm2 of lesion. Should a cell yield fall below the specification, the technicians communicate this with the surgeon together with the notion that the insufficient cell number potentially negatively impacts the efficacy outcome. The surgeon decides whether or not to use the cells that are available. The surgeon uniformly seeds the PC + MNC cell mixture dropwise onto or into the cut-to-shape scaffold.
Data Collection and Results Presentation
During the intraoperatively performed cell isolations, the CartiONE Service technicians recorded data concerning, a.o., lesion size, lesion location, cartilage biopsy mass, bone marrow aspirate volume, and the cell yields as assessed with phase-contrast light microscopy. Cell viability was measured for the first 62 cases only, using the Trypan Blue dye exclusion test. 19 As in these 62 cases the viability of both the primary chondrocytes and the primary MNCs was never lower than 90%, it was decided to stop viability measurement and administration to save surgery time. During the initial 40 cases (the INSTRUCT trial cases), the technicians took samples of the supernatant of the final PC + MNC cell suspension that was to be implanted. The samples were tested for sterility in accordance with European Pharmacopeia 2.6.27 by an independent laboratory (Bactimm B.V., Nijmegen, The Netherlands).
The Results are mainly descriptive statistics. Means are given with the standard deviation in parentheses. Box-whiskers graphs plot, from top to bottom, the maximum, 3rd quartile, median, 1st quartile, and minimum value.
Chondron-Marker Stain of the Primary Articular Chondrocytes
Outside of the 141 clinical cases, an immunohistochemical stain of collagen type VI was done on PCs that were isolated according to the clinical procedure. Collagen type VI is prominently present in the pericellular matrix of articular chondrocytes. 20 Together with its pericellular matrix, a single chondrocyte or group of chondrocytes forms a functional unit, called a chondron. 21 Cartilage was harvested from the excised knee parts of 2 female and 1 male donor who received total-knee replacements. Care was taken to avoid harvest of yellowed, calcified, or fibrous cartilage, but osteoarthritic changes within this harvested cartilage were likely. However, the main pathophysiological change to the distribution of collagen type VI in osteoarthritic cartilage is an increased presence of this collagen in the interterritorial matrix, 22 backed up by an increase in mRNA expression. 23 The chondrocytes isolated from the knee cartilage were washed twice. We therefore considered the presence of any extracellular matrix positive for collagen type VI around the double-washed chondrocytes to be the original pericellular matrix. The chondrocytes were resuspended at a concentration of 0.250 × 106 mL–1. This cell suspension was spun (100 µL/slide) at 80 g for 5 minutes at room temperature. Slides were washed 3 times with phosphate-buffered saline (PBS) and fixed with a 5-minute submersion in ice-cold 99.0% acetone. For the immunostain, slides were first blocked with 0.3% H2O2 in methanol, washed with 0.1% Tween 20 in PBS, and blocked further with 2% bovine serum albumin (BSA) in PBS. The primary antibody, goat anti-type VI collagen (SouthernBiotech, Birmingham, AL, USA), was incubated for 1 hour at a 1:10 dilution in 2% BSA in PBS. Slides were washed with 0.1% Tween 20 in PBS before incubation for 1 hour with rabbit anti-goat IgG (heavy chain + light chain) with horseradish peroxidase conjugate (SouthernBiotech, Birmingham, AL, USA) at a 1:100 dilution in 2% BSA in PBS. Slides were washed again with 0.1% Tween 20 in PBS and incubated for 8 minutes with 3,3’-diaminobenzidine (Bright-DAB), rinsed with tap water, and counterstained with hematoxylin Gill No. 3. Blocking, rinsing, and staining were done at room temperature.
Results
The sterility samples collected during the initial 40 cases all tested negative for microbiological contamination, indicating sterility of the final cell mixture suspension. Furthermore, there were no reports of surgical-site infection for any of the subsequent cases.
The mean age of the patients treated with the cell service was 35.7 (±9.8) years, ranging from 12 to 57 years (n = 139; no record for 2 patients;
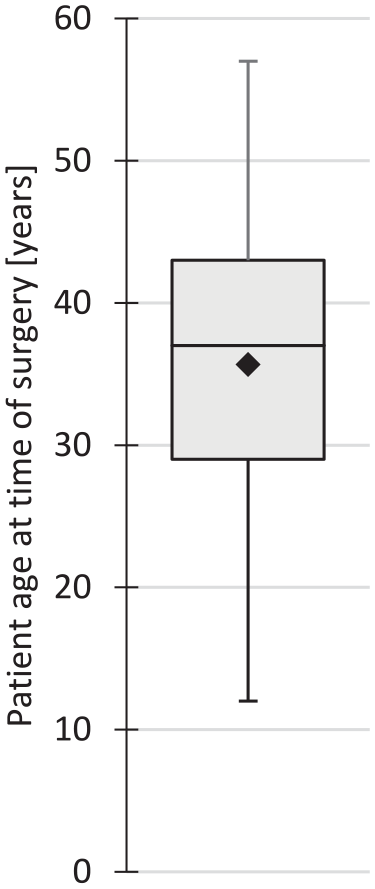
Box-whiskers plot of patient age at time of surgery (n = 139). Black diamond indicates the mean.

Box-whiskers plot of the total area of the articular cartilage lesion or lesions treated with the cell mixture per patient (n = 138). Black diamond indicates the mean.
Debridement cartilage for the chondrocyte isolation was available in 135 of the 141 cases. In the 3 Actifit-plus-cells cases, only a biopsy of healthy-appearing less-load-bearing cartilage was used. In 2 cases, there was no cartilage to debride, and in 1 case the debridement was of a quality deemed too poor to process further. Healthy-appearing less-load-bearing cartilage was collected in 126 of 140 cases (no record for 1 case). Separate measurement of its mass was not always possible as the surgeon frequently presented the technicians with the combined cartilage collection. The mean mass of debridement cartilage collected was 0.54 (±0.38) g (n = 100), and the mean mass of less-load-bearing cartilage collected was 0.20 (±0.18) g (n = 92). The mean mass of the total amount of articular cartilage collected was 0.74 (±0.36) g, ranging from 0.19 to 2.01 g (n = 140; no record for 1 case;
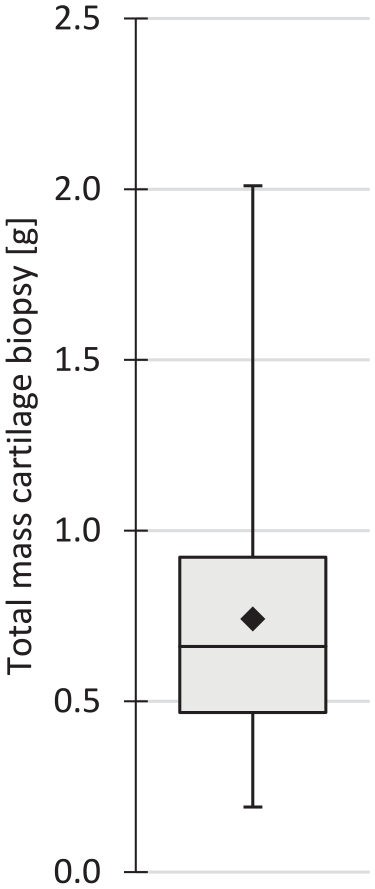
Box-whiskers plot of the total mass of collected articular cartilage material per patient (n = 140). Black diamond indicates the mean.
The mean PC yield was 1.23 (±0.75) × 106 g–1, ranging from 0.07 to 4.38 × 106 g–1 (n = 139; 1 datum unavailable and 1 outlier of 9.7 × 106 g–1;
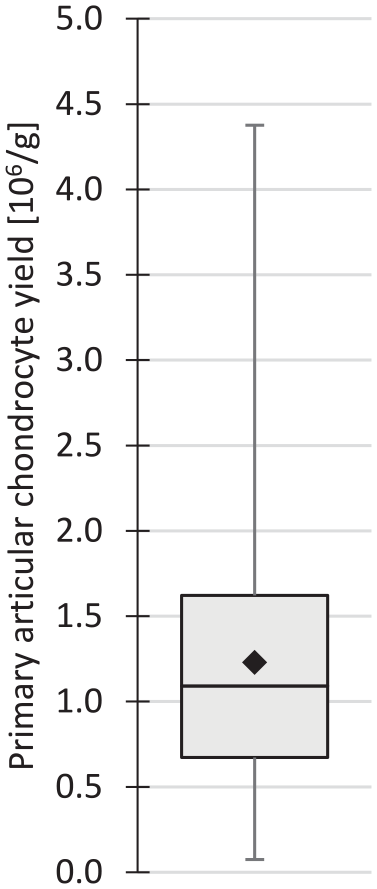
Box-whiskers plot of the primary articular chondrocyte yield (n = 139). Black diamond indicates the mean.
The number of PCs isolated per gram of cartilage did not correlate with patient age (n = 139;
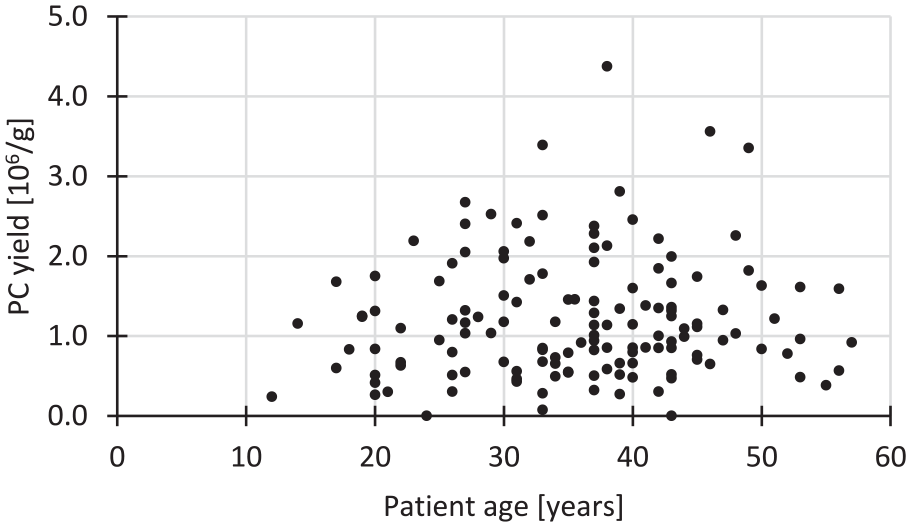
Scatter plot of the primary articular chondrocyte (PC) yield per gram (g) of digested cartilage over patient age (n = 139). The coefficient of determination R 2 = 0.0055 for a linear correlation.
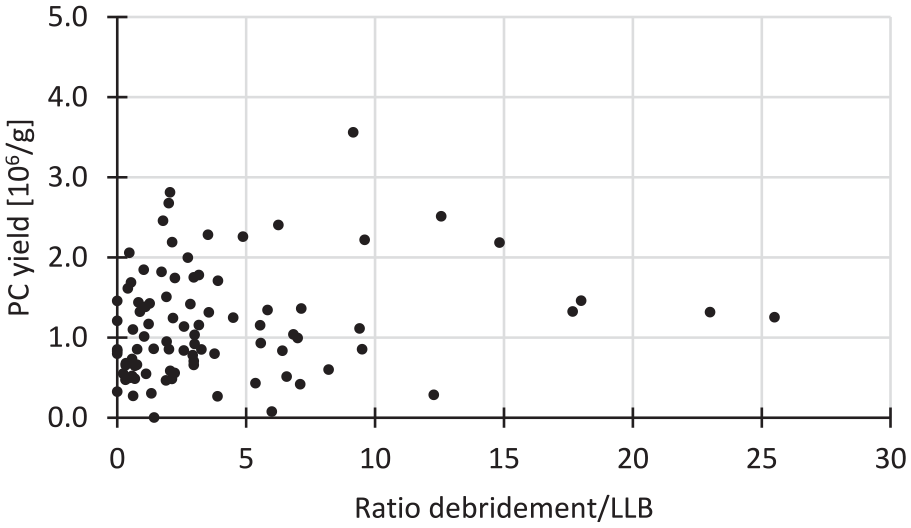
Scatter plot of the primary articular chondrocyte (PC) yield per gram (g) of digested cartilage over the debridement/less-load-bearing (LLB) cartilage ratio (n = 92). The x-axis has been cut off at 30 for clarity purposes. In 1 case, the ratio of debridement/LLB was 56 and the accompanying PC yield was 1.0 × 106 g–1. The coefficient of determination R 2 = 0.0108 for a linear correlation.
Bone marrow was aspirated mostly from the anterior iliac crest (n = 118). The posterior iliac crest was chosen in 9 cases, the distal femur in 8 cases, the proximal tibia in 3 cases, and a combination of the distal femur and the proximal tibia was used in 2 cases. In 1 case, no record of the aspirate location is available. The volume of the aspirate depended on the total lesion area, with more bone marrow aspirated for larger lesions. As a general guideline, the technicians requested a minimum volume of 15 mL, if available. The mean volume of bone marrow aspirate drawn was 22.8 (±8.2) mL, ranging from 7.2 mL to 62.0 mL (n = 141;
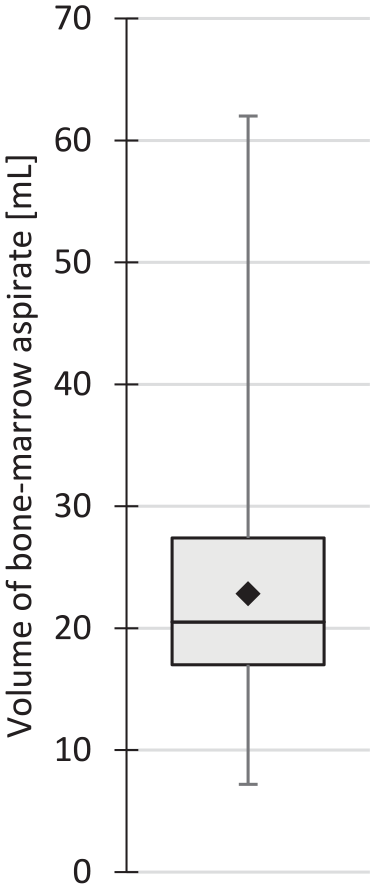
Box-whiskers plot of the volume of bone marrow aspirated per patient (n = 141). Black diamond indicates the mean.
The mean primary MNC yield was 7.18 (±5.94) × 106 mL–1, ranging from 0.76 to 46 × 106 mL–1 (n = 141;

Box-whiskers plot of the primary mononuclear bone marrow cell (MNC) yield per mL of aspirate (n = 141). Black diamond indicates the mean.
The number of MNCs isolated per milliliter of aspirate did not correlate with patient age (n = 139;
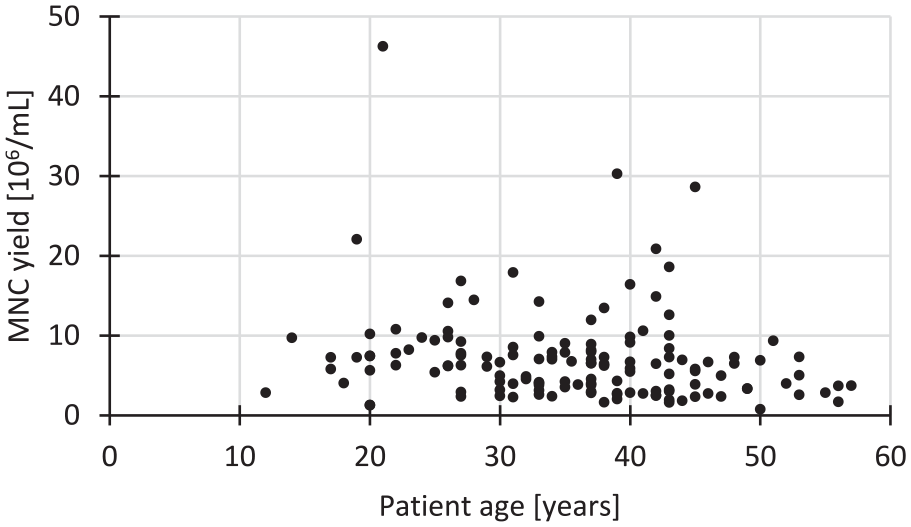
Scatter plot of the primary mononuclear bone marrow cell (MNC) yield per milliliter (mL) of aspirated bone marrow over patient age (n = 139). The coefficient of determination R 2 = 0.0307 for a linear correlation.
After isolation, washing, and counting of the PCs and MNCs, the technicians prepared a cell mixture that consisted of all isolated PCs supplemented with a controlled number of MNCs to provide the surgeon with a total cell supply of 9 million cells per square centimeter of lesion. Thus, not all isolated MNCs were necessarily added to the PCs. The mean percentage of PCs in the cell mixture was 2.9 (±2.1)% (n = 140; 1 outlier of 33%). The mean areal PC supply was 272 (±196) × 103 cm−2 of lesion, ranging from 10.4 × 103 to 1.07 × 106 cm−2 of lesion (n = 137;
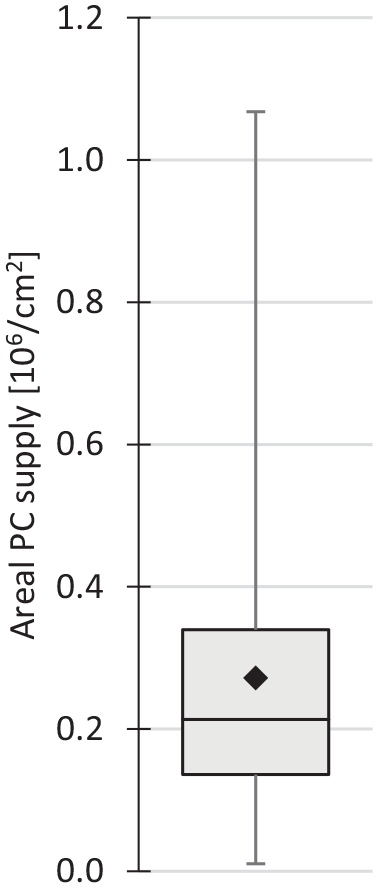
Box-whiskers plot of the areal supply of primary articular chondrocytes (PC) that was available to the surgeon (n = 137). Black diamond indicates the mean.
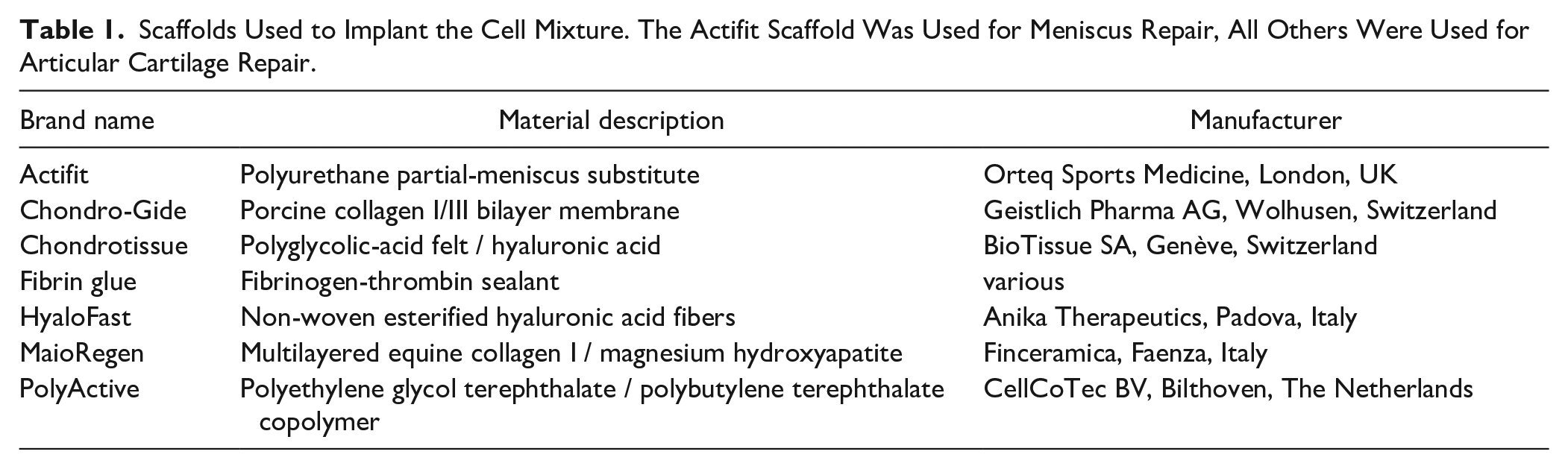
The seeding of the cell mixture onto or into the scaffold was done either in situ, in which case the scaffold had already been positioned into the lesion, or was done on a sterile table. Cell seeding was done dropwise with a low-dead-volume 1-mL syringe. The choice of scaffold depended on on-site availability and the preference of the surgeon. An overview of the types of scaffold used is given in Table 1 . Completely arthroscopical procedures were limited to the use of Actifit, Chondrotissue, fibrin glue, or HyaloFast, and a lesion size and location the surgeon felt comfortable with to treat arthroscopically.
Scaffolds Used to Implant the Cell Mixture. The Actifit Scaffold Was Used for Meniscus Repair, All Others Were Used for Articular Cartilage Repair.
The mean duration of the cell mixture preparation was 100 (±19) minutes, ranging from 65 to 159 minutes (n = 131; no record for 10 patients). The preparation time started when all cartilage biopsy material had been collected, and it ended when the ready-for-use cell mixture was given to the surgeon. The mean surgery time, that is, the time between first incision and final suture, was 152 (±31) minutes, ranging from 95 to 220 minutes (n = 77; no record for 64 patients). Frequently, concurrent interventions were performed during the cell isolations. These interventions included reconstruction of the anterior cruciate ligament, reconstruction of the medial patellofemoral ligament, tibial-tubercle osteotomy and transportation, trochleoplasty, partial meniscectomy, partial resections of synovial plica, and microfracture of small (≤1 cm2) cartilage lesions.
The immunohistochemical stain of cytospun cells that were isolated from human articular cartilage according to the clinically used short-isolation protocol revealed that many cells and cell clusters were still wrapped in their pericellular matrix (
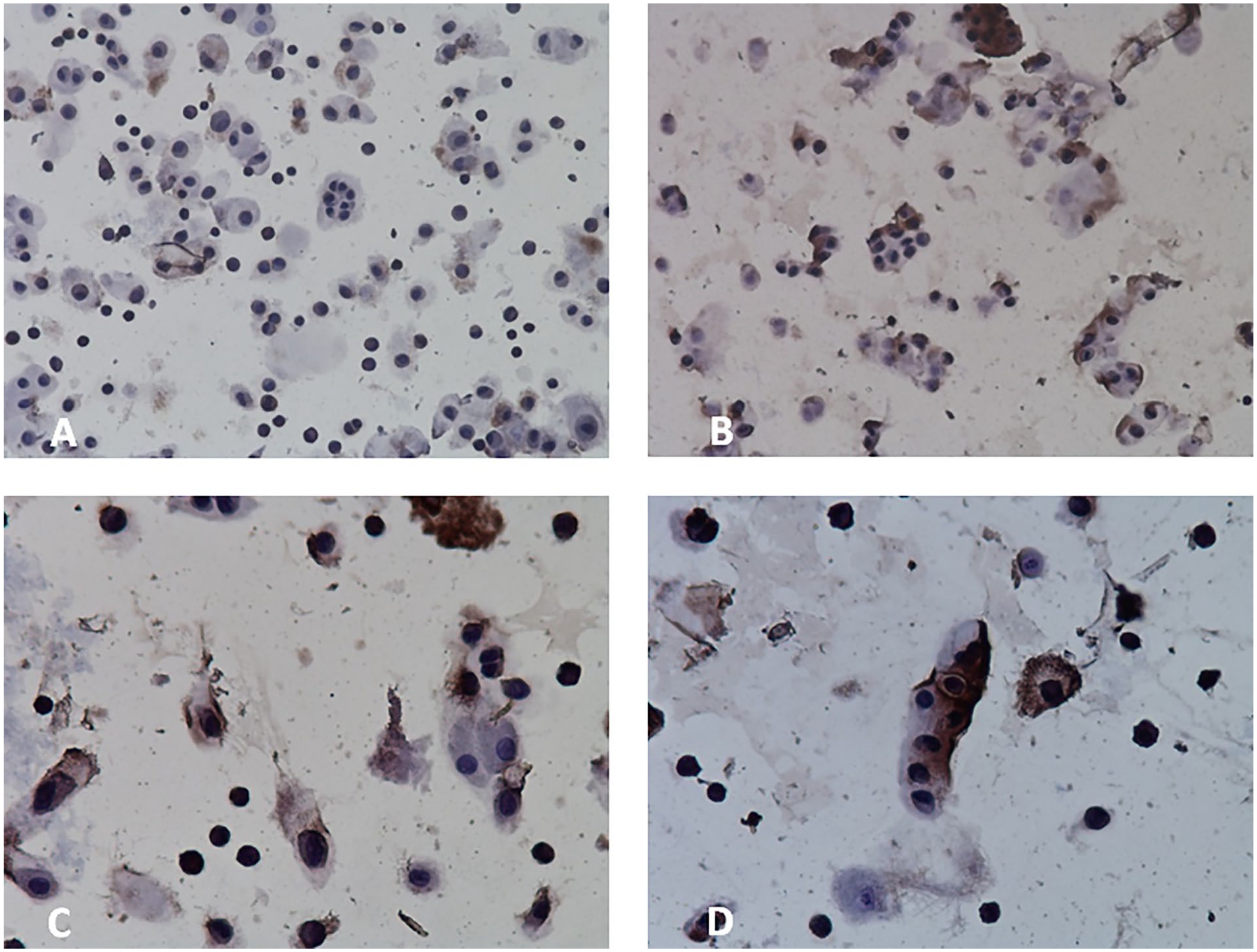
Representative microphotographs of the suspension of cells isolated from human articular cartilage with the short-isolation protocol. The cell suspension was stained for collagen type VI (brown) and counterstained with hematoxylin. Note that many cells and cell clusters are still encapsulated in pericellular matrix. Pericellular matrix was stained brown inhomogeneously. Magnification: A and B 200x, C and D 400x. No control cell-suspension stains were available.
Discussion
The Chondrocyte Supply in a Single-Stage Approach
The encountered cell yields appeared to have no relation to the age of the patients and allowed for a mean areal primary articular-chondrocyte supply of 0.272 million/cm2 of lesion. This supply is lower than what is adhered to in 2-stage ACI. For the original ACI generation, Brittberg et al. 24 implanted 2.6 to 5.0 million culture-expanded chondrocytes into lesions ranging 1.6 to 6.5 cm2, which calculates into a chondrocyte supply range of 0.4 to 3.1 million/cm2. For MACI (Vericel Corporation, Cambridge, MA, USA), culture-expanded chondrocytes are pre-seeded on a porcine collagen type I/III membrane at an areal density of 0.5 to 1.0 million/cm2. The pre-seeded membrane has to be manipulated by the orthopedic surgeon to cut it to shape and to position it into the lesion, which is estimated to result in a net supply of viable chondrocytes of 0.361 million/cm2. 25 With Spherox (CO.DON AG, Teltow, Germany), 10 to 70 spheroids of chondrocytes are provided per square centimeter of lesion. Each spheroid is created by seeding 0.2 million cells in a hydrogel-coated well culture. 26 Excluding chondrocyte proliferation within the spheroid, Spherox would then provide a culture-expanded chondrocyte supply in the range of 2.0 to 14 million/cm2. The primary chondrocyte supply of 0.272 (±0.196) million/cm2 we report is on the order of magnitude of MACI after surgeon handling. The minimum number of chondrocytes, primary or expanded, that needs to be supplied per square centimeter of lesion to start up a viable chondrogenic process of which the outcome would be a regenerated articular cartilage surface is unknown.
The Use of Primary Articular Chondrocytes and Chondrons
In the cell service described in this article, as well as in the INSTRUCT and IMPACT trials, PCs and chondrons were used, rather than in vitro expanded daughter chondrocytes. The monolayer culture necessary for chondrocyte proliferation in vitro alters the cells’ phenotype: They become more primitive and less specialized in terms of collagen synthesis and the cell surface marker proteins that are present on their plasma membranes. 27 With each monolayer passage, chondrocytes produce less hyaline-cartilage-specific extracellular matrix. 28 Primary chondrocytes, however, are “fresh” and have not been exposed to phenotype-shifting in vitro culture conditions. Yet, some concern may rise about the viability and the chondrogenicity of the primary chondrocytes isolated from debrided cartilage, as its quality can vary. Based on observations during harvest of, for example, any fibrousness or the presence of calcifications, the surgeon makes the final decision on suitability of the debridement for chondrocyte harvest. It should be kept in mind though that in vitro chondrocytes isolated from debridement cartilage can synthesize as much collagen type II and proteoglycans as chondrocytes isolated from healthy hyaline cartilage from the same individual.29,30 Not using debridement may therefore be wasteful. In the intraoperative cell isolation procedure under study here, healthy-appearing less-load-bearing cartilage was added to the debridement to mitigate the risk of a low chondrocyte yield and to lessen any variation in chondrocyte viability. In 5 of the 138 cartilage lesion cases, the primary chondrocyte yield was below specification. In all, the surgeon should consider the advantage of inclusion of some healthy-appearing low-load-bearing cartilage versus the disadvantage of the comorbidity this inclusion causes, while taking the apparent qualities of the debridement into account.
The IMPACT trial and its preclinical research report on the implantation of chondrons rather than chondrocytes. In vitro, chondrons produce more collagen type II and proteoglycans than chondrocytes, both in pellet culture 31 and in alginate-bead culture. 32 The short-isolation protocol used in the treatment under study here is very close to the protocol published by Bekkers et al., 33 who reported that they isolated chondrons. In addition, the cartilage cell isolate obtained using the method under study stained positive for collagen type VI, a marker collagen type for pericellular matrix around chondrocytes. Although no control cell suspensions were stained, the available stain results and the appearance of the cells and cell clusters in the isolate indicate that the cells that were isolated from the articular cartilage at least partly consisted of chondrons.
Co-Implantation
The PCs/chondrons were supplemented with an abundance of freshly isolated autologous MNCs. A total cell supply of 9 million cells per cm2 was adhered to. MNC yields were lower when aspirate was collected from the distal femur or the proximal tibia. It therefore appears advisable to adhere to aspiration from the iliac crest. The addition of MNCs not only increased the total cell density in the lesion, but also allows for paracrine signaling and direct cell-cell contact between the 2 cell types in situ. The anti-inflammatory, glycosaminoglycan-production-enhancing, and chondrogenic-marker-gene-upregulating effects observed in vitro11-14 may be assumed to take place in the treated articular cartilage lesion in vivo as well. 34 It is not known how the regenerative performance compares between lesions treated with in vitro expanded chondrocytes and lesions treated with PCs or chondrons mixed with stromal cells. As is true for the 100%-chondrocyte approach: The optimal areal supply in a co-implantation approach remains elusive.
The Short-Isolation Method for Chondrocytes/Chondrons
The short protocol to free chondrocytes/chondrons from the cartilage biopsy involved manual mincing and a 35-minute digestion at 37 °C using an enzyme solution sourced from C. histolyticum. Medical application of Clostridial collagenases is not new. Injections of Clostridial collagenases are used as a non-surgical treatment of Dupuytren’s disease, in which case the enzymes are injected into the cords of fibrous tissue that cause contractures of the joints of the metacarpus and fingers. 35 Clostridial collagenases are also used to debride necrotic tissue from wounds. 36 Any negative influence of the enzymes on the chondrocytes and chondrons must be extremely limited as the chondrocyte viability was on average 97.9 (±1.9)%. In addition, the pronounced localization of collagen type VI around chondrocytes and chondrons may lessen an influence of the Clostridial collagenases on the cells as collagen type VI resists degradation by bacterial collagenases in the absence of a reducing agent. 37
The yields of the short isolation varied, with an average of 1.23 (±0.75) × 106 PCs per gram of digested cartilage. Besides individual and pathophysiological causes for cell-yield variation, the efficacy of reconstituted enzymes may vary somewhat per lot as well. Theoretically, it is possible to increase the chondrocyte yield, as the current yield is only a fraction of what is available in the cartilage material. 38 To shorten the surgery time, fundamental research is required to further shorten the isolation time of PCs without losing cell yield.
Limitations of the Cell-Isolation Service
The intraoperative cell-isolation method has 2 main limitations: (1) an appointment with the company that provides the service has to be scheduled, thereby preventing ad hoc implementation and (2) the cell isolations cost a substantial amount of operating room time. The duration of the intraoperative tissue processing and cell isolations was 100 minutes on average, although a duration of 65 minutes was attainable. The bare cartilage dissociation protocol itself lasts 35 minutes and the work around it 30 minutes, if without hurdles. The primary cause of longer procedures was the volume of cartilage to be minced. Mincing the cartilage manually allows for care and precision and avoids the high stresses and impact energy that can be expected with automated mincers. It also allowed for cartilage harvest from loose bodies with a calcified core. A secondary cause for longer durations were difficulties encountered with aspiration of bone marrow, such as blocking of the needle or difficulty with needle repositioning. When bone marrow aspiration took longer than expected, the subsequent isolation and washing of the MNCs could logistically interfere with the washing of the PCs. Normally, the MNCs were washed and counted and ready for use before the first washing step of the PCs. However, working at a mobile workstation necessitates the use of a single centrifuge and can cause logistical interference between concurrent steps of the process. Another limitation of the intraoperative cell-isolation method is the space the workstation requires underneath the laminar airflow. Before surgery, the O.R. logistics must therefore be planned carefully between the O.R. personnel and the cell technicians.
Conclusions
The intraoperative isolation of articular chondrocytes and MNCs and their combined use to single-surgically treat lesions of the articular cartilage provided a total cell supply of roughly 9 million autologous cells per cm2 of lesion and a mean of 0.272 million primary autologous chondrocytes, both with and without pericellular matrix, per cm2 of lesion. The mean intraoperative cell yields were 1.23 million chondrocytes per gram of cartilage and 7.18 million MNCs per milliliter of bone marrow aspirate. Single and multiple lesions could be treated within the single surgery. The surgery time was frequently used to perform concurrent interventions. Continued research is necessary to further shorten isolation protocols for PCs.
The short-isolation protocol allows for an all-autologous articular-chondrocyte-based treatment to be performed within 1 surgical intervention. This approach spares the patient a second invasive intervention, cancels out laboratory work, provides articular chondrocytes whose phenotype has not been shifted by in vitro expansion, and instead provides the implanted cell population with an in vivo and an in situ environment to proliferate in and start up a repair response.
Footnotes
Acknowledgments and Funding
The authors thank Roderick Polak and Wilte Olijve for their constructive comments on the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Both authors have consulting agreements with Cartilage Repair Systems, LLC, who own CartiONE.
Ethical Approval
The data presented in this paper were collected during delivery of a commercial cell-isolation service under the supervision of the attending orthopedic surgeon. Collection of these data did not involve review of an Ethics Committee. Patient consent, after informing, was given for the use of biopsy materials for the collagen-type-VI stain.
