Abstract
Objective
The surgical management of nasal septal defects due to perforations, malformations, congenital cartilage absence, traumatic defects, or tumors would benefit from availability of optimally matured septal cartilage substitutes. Here, we aimed to improve in vitro maturation of 3-dimensional (3D)-printed, cell-laden polycaprolactone (PCL)-based scaffolds and test their in vivo performance in a rabbit auricular cartilage model.
Design
Rabbit auricular chondrocytes were isolated, cultured, and seeded on 3D-printed PCL scaffolds. The scaffolds were cultured for 21 days in vitro under standard culture media and normoxia or in prochondrogenic and hypoxia conditions, respectively. Cell-laden scaffolds (as well as acellular controls) were implanted into perichondrium pockets of New Zealand white rabbit ears (N = 5 per group) and followed up for 12 weeks. At study end point, the tissue-engineered scaffolds were extracted and tested by histological, immunohistochemical, mechanical, and biochemical assays.
Results
Scaffolds previously matured in vitro under prochondrogenic hypoxic conditions showed superior mechanical properties as well as improved patterns of cartilage matrix deposition, chondrogenic gene expression (COL1A1, COL2A1, ACAN, SOX9, COL10A1), and proteoglycan production in vivo, compared with scaffolds cultured in standard conditions.
Conclusions
In vitro maturation of engineered cartilage scaffolds under prochondrogenic conditions that better mimic the in vivo environment may be beneficial to improve functional properties of the engineered grafts. The proposed maturation strategy may also be of use for other tissue-engineered constructs and may ultimately impact survival and integration of the grafts in the damaged tissue microenvironment.
Keywords
Introduction
Surgical reconstruction of nasal cartilage defects may be approached by prefabricated flaps and tissue expanders. However, surgeons often need use of a limited number of donor sites in the body, such as costal or auricular cartilage, which (1) may not be adequate to fulfill the defect, (2) may not have the same functional characteristics as the target cartilage tissue, and (3) may be associated with significant morbidity risks. 1 Therefore, a number of tissue engineering strategies are being explored for cartilage regeneration, mostly at the preclinical level.2-5 Among these, 3-dimensional (3D) bioprinting of cartilage substitutes presents the advantage of being tailor-made for each patient’s defect, thus providing as yet another example of personalized medicine.6-15 Of note, it is relevant to consider criteria such as the size, shape, and biological and mechanical properties of the nasal cartilage for successful (i.e. functional) tissue reconstruction. 16 Among many possible materials to be used as a scaffold for nasal cartilage reconstruction, polycaprolactone (PCL) seems suitable as it is an absorbable and porous material which has been used in numerous clinical applications, 17 including rhinoplasty.18,19 Furthermore, PCL-based scaffolds have tunable properties, 20 and their biocompatibility and long-term efficacy and safety in humans are well known. 21 Cell-free materials could theoretically be used to restore damaged cartilage tissue and provide a scaffold for endogenous cell proliferation onto focal defects. However, most studies on the in vivo performance of cell-free scaffolds have yielded unsatisfactory results. 22 For these reasons, the field is now focused on the development of diverse types of cell-laden constructs for cartilage repair.
On the cellular side, many studies have focused on proving biocompatibility of available scaffolds, with no particular attention to chondroinductive potential of used materials. 23 However, it is well known that chondrocytes dedifferentiate in expansion culture, 24 and therefore, use of alternative cell sources (for instance, articular cartilage progenitor cells) 25 or strategies for re-differentiation of chondrocytes prior to transplantation 26 seem advisable. Since the pioneering studies of Hollander comparing nasal and articular chondrocytes for tissue engineering applications, 27 the field has advanced a great deal and diverse cell sources have been explored for cartilage regeneration. 28 Most studies have focused on the use of mesenchymal stromal cells (MSCs) of diverse origins.29,30 However, MSC multipotency entails the possibility of alternative adipose or osteogenic differentiation. 31 Other research groups have used culture-expanded nasal32-34 or auricular 35 chondrocytes or have compared diverse heterotopic chondrocyte sources. 36
In this study, we aimed to improve rabbit chondrocyte-laden, PCL-based scaffolds maturation and test their in vivo performance in a rabbit auricular cartilage model. To this end, we exposed the scaffolds to prochondrogenic media in vitro. As cartilage is avascular and presents a low oxygen tension microenvironment, we tested hypoxic versus normoxic culture conditions. To simulate the septal cartilage environment, a rabbit ear surgery protocol was selected due to the low morbidity and high reliability of the structure to simulate the clinical conditions to test the scaffold. We further analyzed the in vitro and in vivo behavior of the scaffolds, their mechanical properties, and their chondrogenic potential in vivo.
Methods
Animal Welfare
Twenty adult 2.0- to 2.5-kg New Zealand white rabbits were included. They were housed with access to water and nourishment ad libitum under pathogen-free conditions in barrier facilities of the Biodonostia Health Research Institute. Rabbits were housed 1 animal per cage under diary observation to assess their welfare during the study period (12 weeks).
Scaffold Design and Synthesis
Scaffolds were designed using Creo Parametric 3D Modeling Software v4.0 (PTC products). The first layer was composed of 9 cylinders of 10.35 mm length and 0.15 mm thickness separated 1 mm between them. The second layer presented 11 cylinders of 8.25 mm length and 0.15 mm thickness separated 1 mm between them, transversal to the underlying layer. To provide an elliptic form, the outermost 2 cylinders at each side were shortened in curvature. Scaffolds with 4, 8, and 16 layers were designed by copy-pasting the 2-layered construct. PCL scaffolds were 3D-printed using fused deposition modeling (FDM) technology on a Biobots 1 bioprinter (Allevi, Philadelphia, PA). PCL polymer (Polycaprolactone Bioink Kit, Allevi) was introduced into the bioprinter syringe, melted by heating to 100 °C and subsequently dispensed through a 27G metal needle, using a pressure of 100 PSI and a deposition speed of 0.1 mm/seg. Printed scaffolds were sterilized by soaking in 70% ethanol for 1 hour and exposed to ultraviolet (UV) lamp irradiation for 30 minutes.
Rabbit Chondrocyte Isolation and Expansion
Briefly, rabbit ear cartilage was obtained post mortem after removing hair, skin, and perichondrium of the tissue. Cartilage fragments were sliced in small pieces and transferred onto multiwell plates where they were rinsed with washing medium (Hank’s balanced salt solution [HBSS, Gibco], supplemented with 2% Penicillin/Streptomycin [P/S] and 2% Fungizone™ [Gibco]). Tissue fragments were digested in digestion medium (Dulbecco’s Modified Eagle Medium [DMEM, Sigma-Aldrich] containing 1.5 mg/ml of Collagenase type II [Gibco] and 2% P/S plus 2% Fungizone™) overnight at 37 °C, with gentle shaking. Cells were filtered through a 70-μm nylon cell strainer (Corning) in order to get rid of undigested tissue fragments and aggregations. Cells were centrifuged at 1,500 rpm for 5 minutes and washed 3 times with washing medium. The cells were then resuspended in expansion medium (low glucose DMEM supplemented with 10% fetal bovine serum [FBS; Gibco™] and 1% P/S). Viable cells were determined using trypan blue dye exclusion technique. Isolated chondrocytes were cultured until confluence at 37 °C in a humidified CO2 incubator, replacing the medium every 3 days.
In Vitro Cell-Laden 3D Scaffold Maturation
A total amount of 50,000 auricular chondrocytes were seeded on top of the scaffolds allowing them to adhere and grow for 1 week in expansion medium in a humidified CO2 incubator, changing medium twice a week. At this specific point, half of the scaffolds were kept in expansion medium and normoxic (21% O2) atmosphere, while the other half were cultured under hypoxic conditions (1% O2) with prochondrogenic medium (High Glucose DMEM [Sigma-Aldrich], 5% FBS [Gibco], 1x Insulin-Transferrin-Selenium [ITS; Gibco], 100 nM dexamethasone, 100 μg/ml ascorbic acid, 1% P/S, 400 mM glutamine [Sigma-Aldrich], 10 ng/ml transforming growth factor-beta 2 [TGF-β2; Peprotech]) for 3 weeks. Samples were taken at days 7, 14, and 21 of culture. The matured scaffolds in both conditions were implanted into rabbits at day 21 of culture.
Biochemical Evaluation of Neocartilage Formation and Quantification of DNA Content and Cell Viability
Three samples of each condition and time were digested with 4 U/ml Papain at 60 °C overnight in phosphate buffered saline (PBS, Gibco) containing 6 mM of
In Vivo Assay
For rabbit ear surgery, after the skin incision, a perichondrium pocket was designed. A circle shape biopsy of elastic cartilage of 1 cm of diameter was extracted without penetrating the lateral skin. The generation of perichondrium pocket allowed us to simulate the cartilage environment. In the case of the defect group (Control-Ct; N = 5 ears), the wound was closed without any kind of insert or scaffold. In the rest of the cases, the gap generated in the tissue was refilled with acellular scaffolds (Acellular-Ac; N = 5), scaffolds with chondrocytes cultured in non-prochondrogenic conditions in normoxia (Normoxia-No; N = 5), and scaffolds with chondrocytes cultured in prochondrogenic conditions in hypoxia (Hypoxia-Hy; N = 5). To control sepsis all along the experiment, early markers of sepsis and inflammation were monitored every 14 days. Thus, blood samples were collected from the jugular vein of anesthetized rabbits and sent to the Biochemistry Service of Donostia University Hospital to determine C-reactive protein (CRP) and procalcitonin serum levels. At study end point (12 weeks), rabbits were sacrificed and grafted tissues were extracted.
Histological Assessments: Staining and Immunofluorescence
Tissue cryosections (7 μm thick) were sliced in a cryostat and fixed for staining. Glycosaminoglycan (GAG) content was visualized by Alcian Blue-Safranin O staining. For immunohistochemistry, tissue sections were blocked with FBS (Gibco) and bovine serum albumin (BSA, Sigma). Sections were incubated with primary antibodies anti-type I (AB90395, Abcam) and anti-type II (1320-01, Southern Biotech) collagens. Secondary antibodies Alexa Fluor 488 and 555 were added at 1:500 dilution and incubated for 1 hour at room temperature. Sections were analyzed using a Fluorescence Microscope Nikon 80i.
RNA Extraction and Reverse Transcription-Quantitative Polymerase Chain Reaction (RT-qPCR) Analysis
For the in vitro experiments, total RNA was isolated from the scaffolds of each condition at days 7, 14, and 21 of culture. For scaffolds analyzed after extraction from the in vivo setting, tissues from surgical areas of each condition were recovered (with 0.5-1 cm of margin around the inserted scaffold/cartilage/defect) from the ears of the sacrificed rabbits at study end point. Tissues or scaffolds were frozen and lysed with buffer RLT (Qiagen) containing β-mercaptoethanol. RNA extractions were carried out with RNeasy Mini Kit and RNeasy Plus Micro Kit (Qiagen) for the in vivo and in vitro experiments, respectively. A total of 1 ng of RNA was reverse transcribed into complementary DNA by using High-Capacity RNA to DNA kit (Applied Biosystems). Then, cDNA was pre-amplified using TaqMan® PreAmp Master Mix according to the manufacturer instructions. Quantitative real-time PCR was performed using TaqMan® Expression Master Mix and the following TaqMan assay primers: ACAN (Oc06726465_m1), COL1A1 (Oc03396073_g1), COL2A1 (Oc03396134_m1), COL10A1 (Oc04097225_s1), and SOX9 (Oc04096872_m1). The reaction was carried out in a CFX384 Touch Real-Time PCR Detection System with the following cycling parameters: 50°C for 2 minutes, 95°C for 10 minutes for polymerase activation, and 40 cycles of 15 seconds at 95°C for denaturation plus 1 minute at 60°C for annealing. Results were analyzed using 2–ΔCt method relative to GAPDH housekeeping gene primers (Oc03823402_g1), in the case of in vitro samples, or 2–∆∆Ct method (relative to control group), in the case of in vivo samples.
Scanning Electron Microscopy (SEM)
A Philips XL30 CP (Amsterdam, The Netherlands) SEM was used to observe the scaffolds and cells morphology with an acceleration voltage of 15 kV and different magnifications. Briefly, scaffolds were washed 3 times with Dulbecco’s phosphate-buffered saline (DPBS) and fixed with glutaraldehyde 2.5% plus paraformaldehyde 2% in PBS for 90 minutes at room temperature. Samples were then dehydrated in graded ethanol and air-dried after their immersion in hexamethyldisilane (HMDS, Sigma-Aldrich) for 10 minutes. Finally, they were coated with a few nanometers of Palladium (SC7620 Mini Sputter Coater).
Mechanical Analysis by Uniaxial Compression Tests
Uniaxial tests were carried out on a ZwickiLine Z1.0 testing machine (Zwick/Roell, Ulm, Germany). The load cell used was a 50 N Zwick/Roell Xforce P (Zwick/Roell). The software for controlling the machine and recording data was the Zwick/Roell testXpert III v1.4 with all patches installed up to the sx406-218-5. The toll used for performing the compression tests was homemade and consists of 2 aluminum cylinders. From these experiments, the Young’s modulus was calculated for each sample.
Statistical Analysis
IBM SPSS Statistics 24 (Armonk, NY) was used. After running the Shapiro-Wilk test, in case of the normal distribution of the results, 1-way analysis of variance (ANOVA) tests were used. For the post hoc analysis, Bonferroni was applied in the case of homoscedasticity; on the contrary, Games-Howell non-parametric test was used. In cases of non-parametric data, for the ANOVA, the Kruskal-Wallis test was performed.
Results
PCL Scaffold Printing and Rabbit Chondrocyte Seeding
To develop 3D-printed PCL scaffolds, parallel meshed structures composed of 2, 4, 8, and 16 layers were designed and their printability at 100 °C was tested, excluding the possibility of printing with live cells (
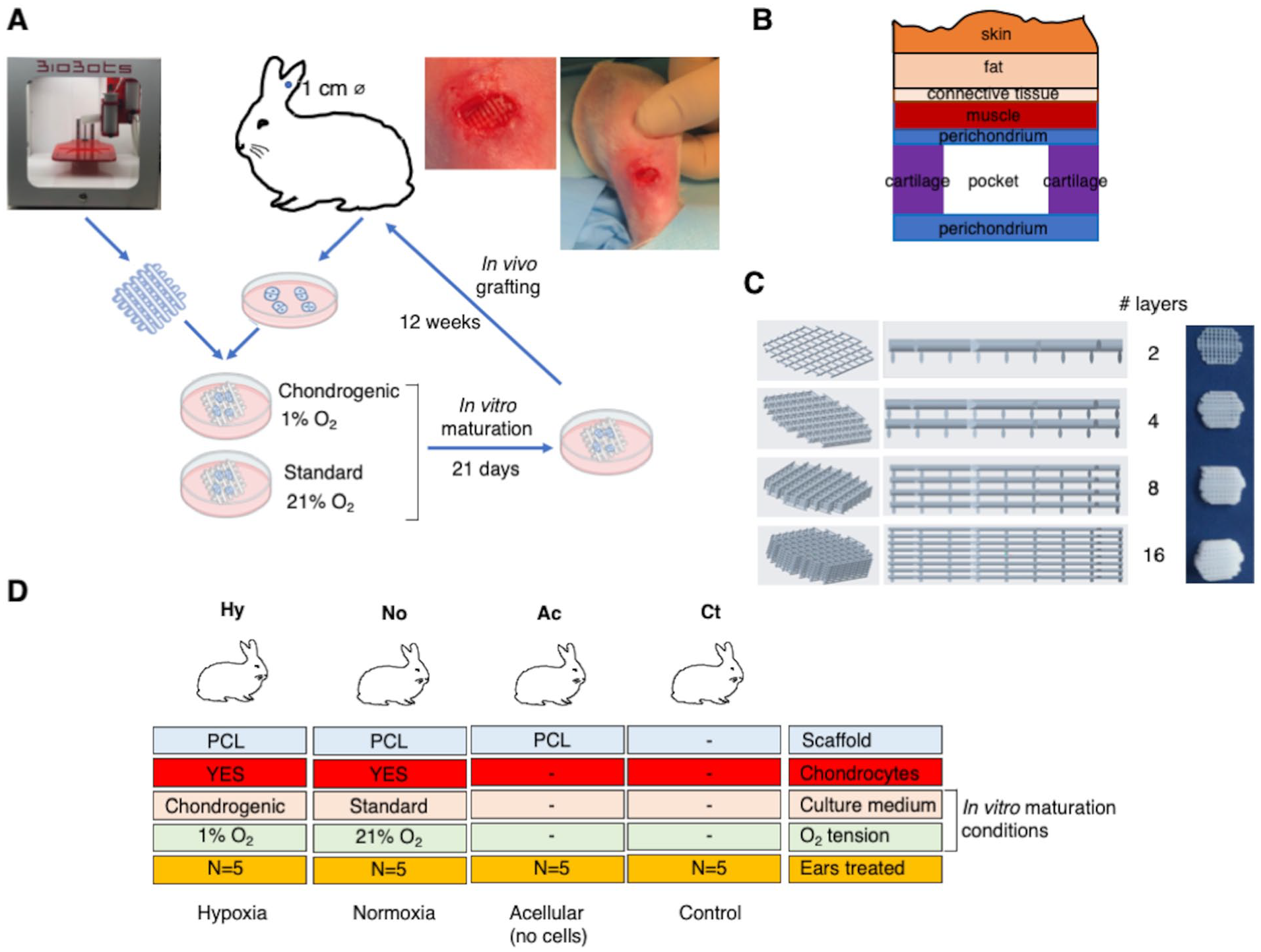
Experimental strategy and in vivo study groups. (
3D Culture and In Vitro Chondrogenic Differentiation
Biochemical characterization of neocartilage formation
To test biocompatibility of the PCL scaffolds with rabbit chondrocytes and to determine the best incubation period and culture conditions to induce cartilage regeneration, a live/dead assay was performed at days 7, 14, and 21 of culture (
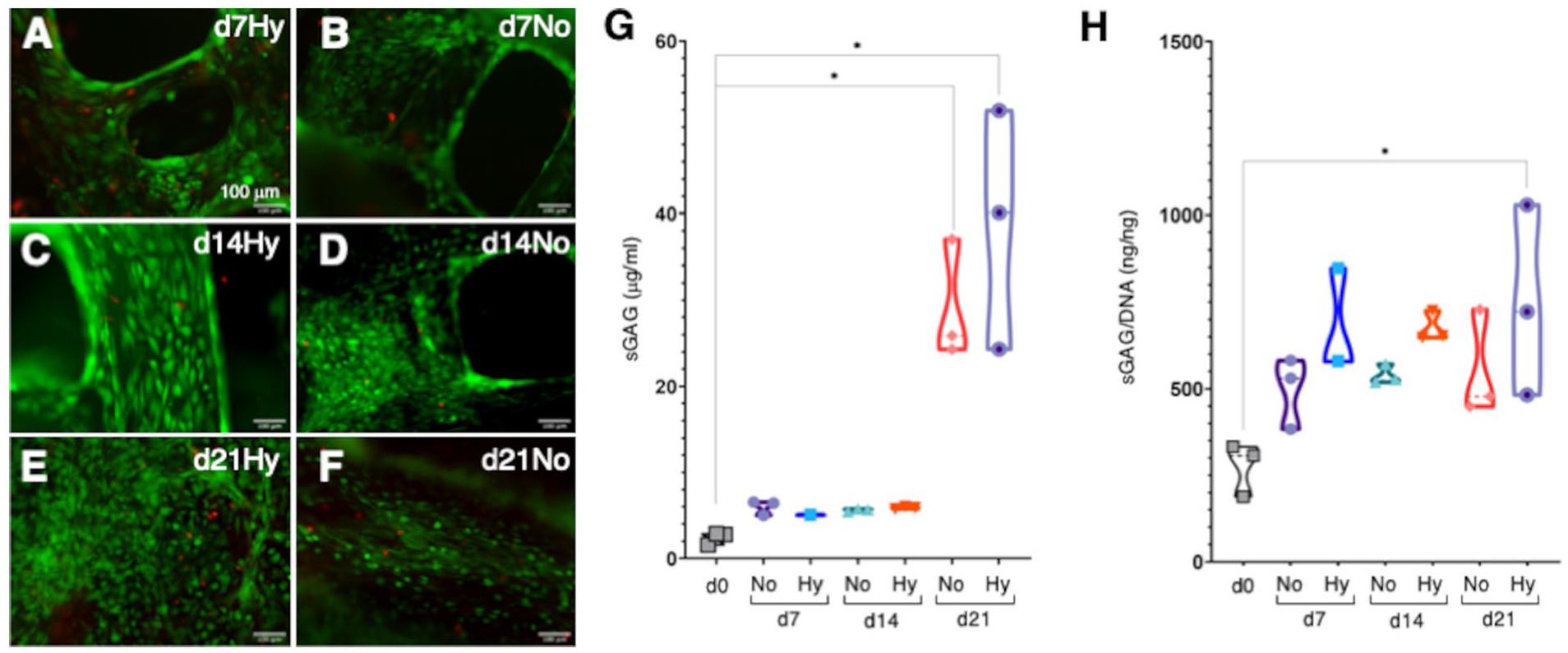
Cell viability and biochemical characterization of in vitro matured constructs. (
Quantification of total sGAG demonstrated increased extracellular matrix (ECM) deposition by day 21, with no significant differences between study groups. Maximum sGAG values deposited on the scaffolds were 38.78 ± 7.99 µg/ml in hypoxic conditions compared with 29.07 ± 4.00 µg/ml in normoxia, perhaps indicating a tendency of prochondrogenic media and hypoxia to induce increased ECM deposition (
As varying cell numbers may affect the amount of sGAG deposited in the scaffolds, sGAG values were normalized to DNA content. In this analysis, scaffolds matured in hypoxia showed again a tendency to increase sGAG secretion compared with normoxia (
Chondrogenic gene expression in 3D scaffolds in vitro (RT- qPCR)
Chondrogenic differentiation may be quantitatively determined by measuring the expression of chondrogenic genes such as COL1A1, COL2A1, SOX9, ACAN, and COL10A1 by RT-qPCR. Expression analysis at day 7, 14, and 21 of in vitro maturation culture revealed interesting differences between the normoxia (No) and hypoxia (Hy) culture groups (
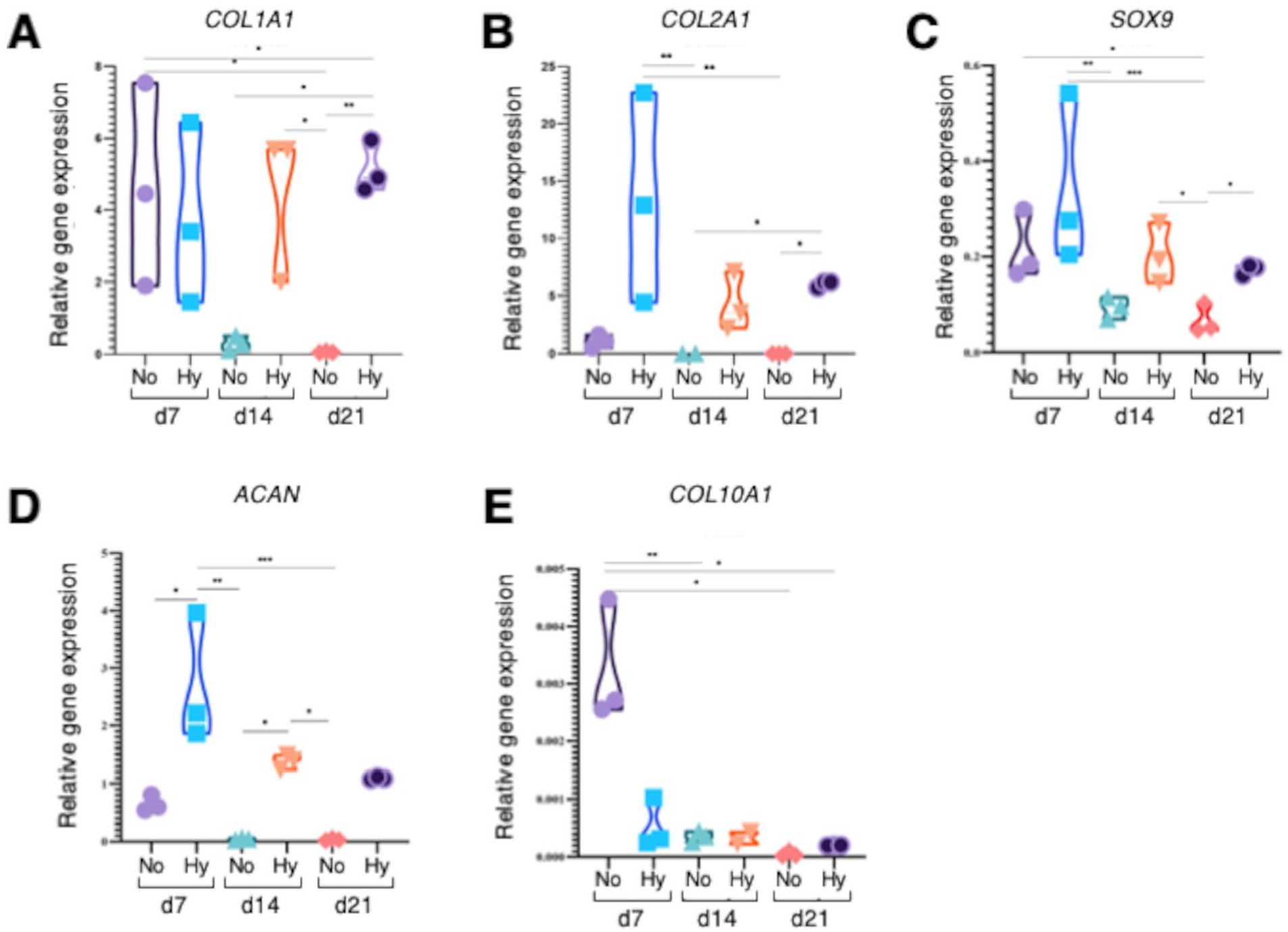
Chondrogenic gene expression profile by in vitro matured constructs. RT-qPCR analysis was done for chondrogenic markers such as collagen type I (COL1A1) (
Mechanical properties of neocartilage
To determine the mechanical properties of the matured scaffolds, constructs cultivated in normoxia (No) and hypoxia (Hy) were compared in their performance in uniaxial compression tests to acellular scaffolds (Ac) and native rabbit ear cartilage (
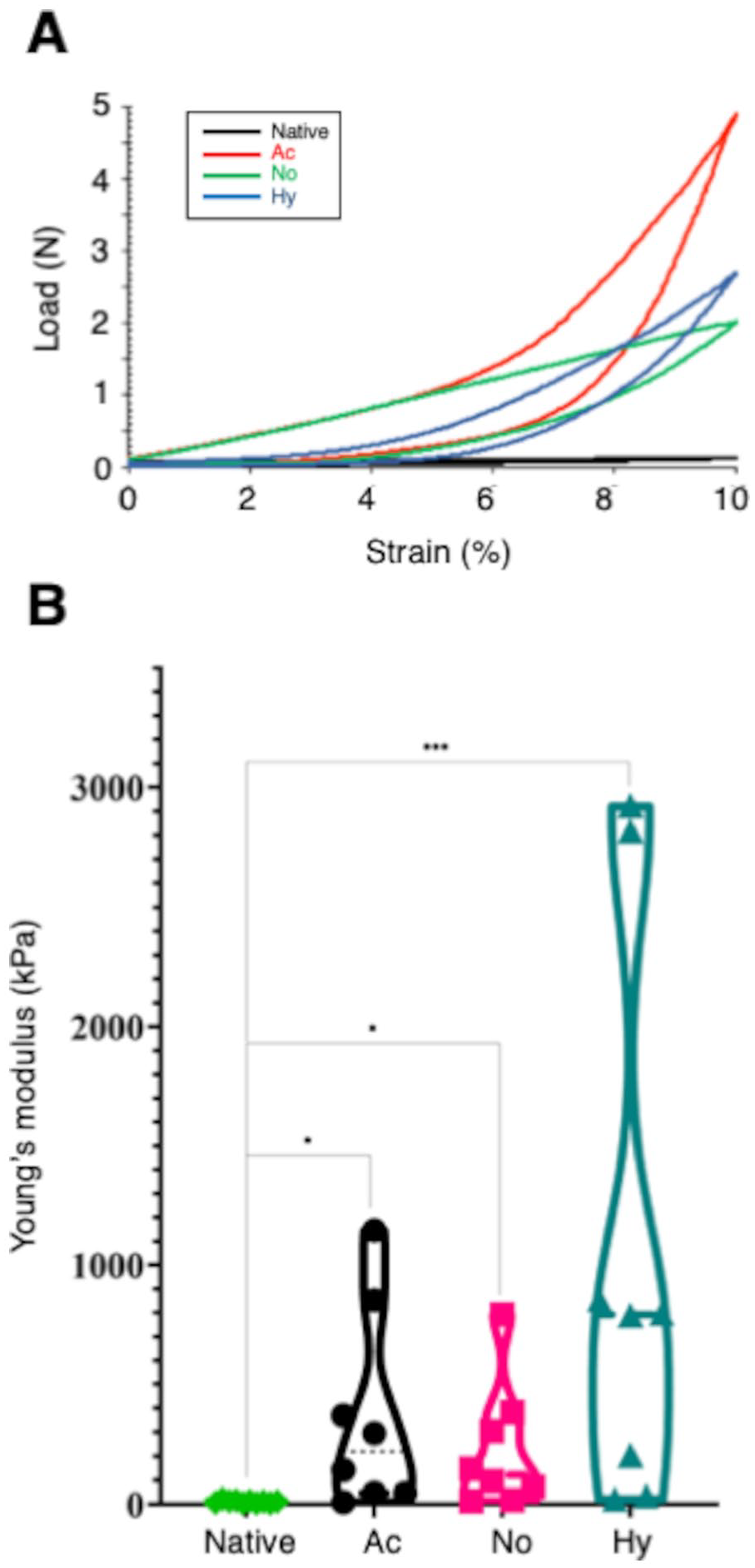
Biomechanical analysis of in vitro matured constructs. (
SEM analysis and immunofluorescence of the scaffolds
SEM was performed to observe the scaffolds’ microstructure and chondrocyte distribution (
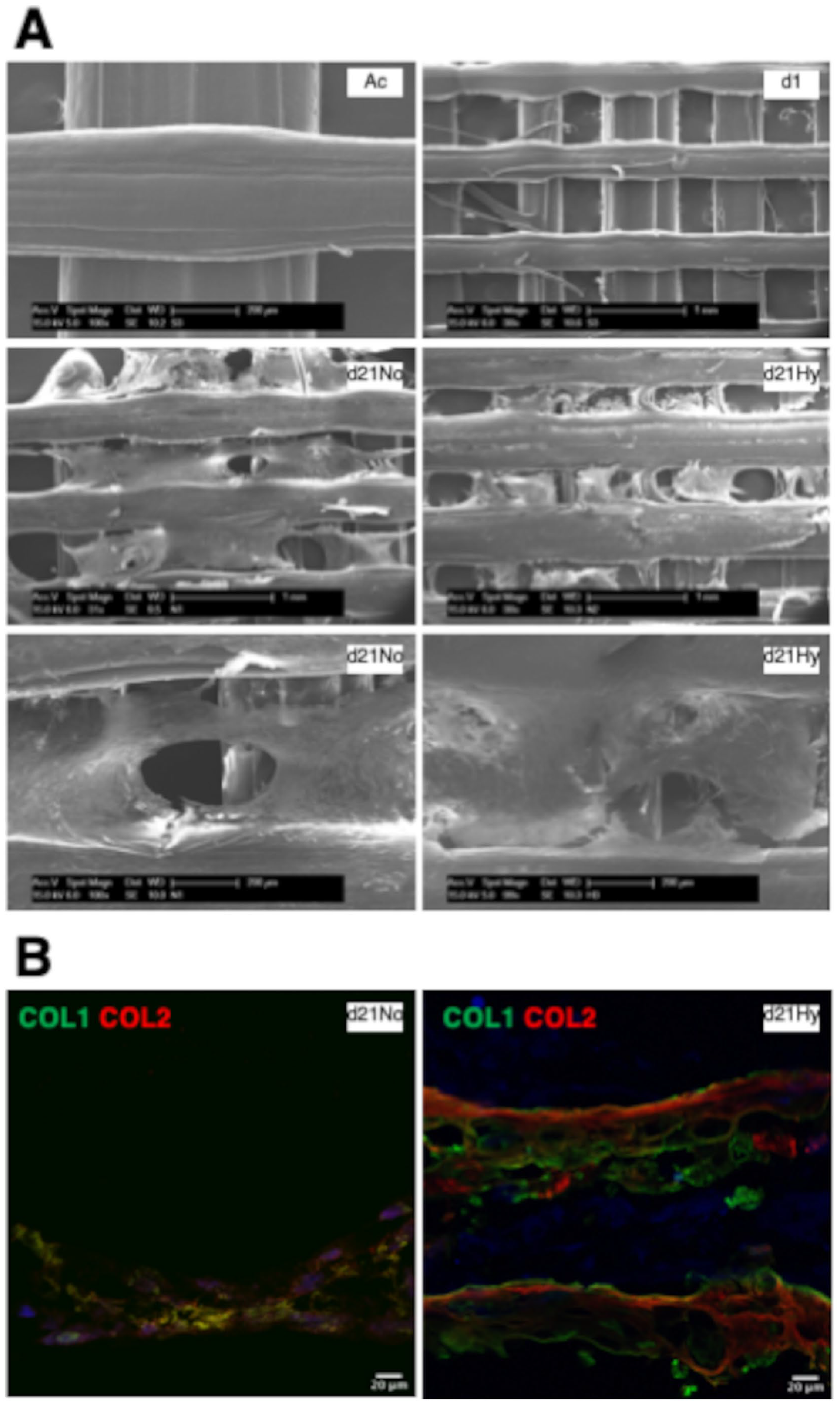
Ultrastructural characterization of in vitro matured constructs. (
In Vivo Transplantation Assays on Rabbit Ears
Perichondrium pockets were generated in rabbits’ ears, and in the case of the control group (N = 5 ears), the wound was closed untreated. In experimental groups, tissue void was refilled with acellular scaffolds (N = 5), scaffolds with chondrocytes cultured in non-prochondrogenic conditions (N = 5), and scaffolds with chondrocytes cultured in prochondrogenic conditions (N = 5;
Chondrogenic gene expression (RT-qPCR) in 3D scaffolds after in vivo implantation
Chondrogenic gene expression (COL1A1, COL2A1, ACAN, SOX9, and COL10A1) was evaluated in extracted constructs after 12 weeks of transplantation, in the following groups: acellular scaffolds (Ac), scaffolds with cells grown in basal conditions in normoxia (No), and scaffolds with cells grown in prochondrogenic hypoxic conditions (Hy) (
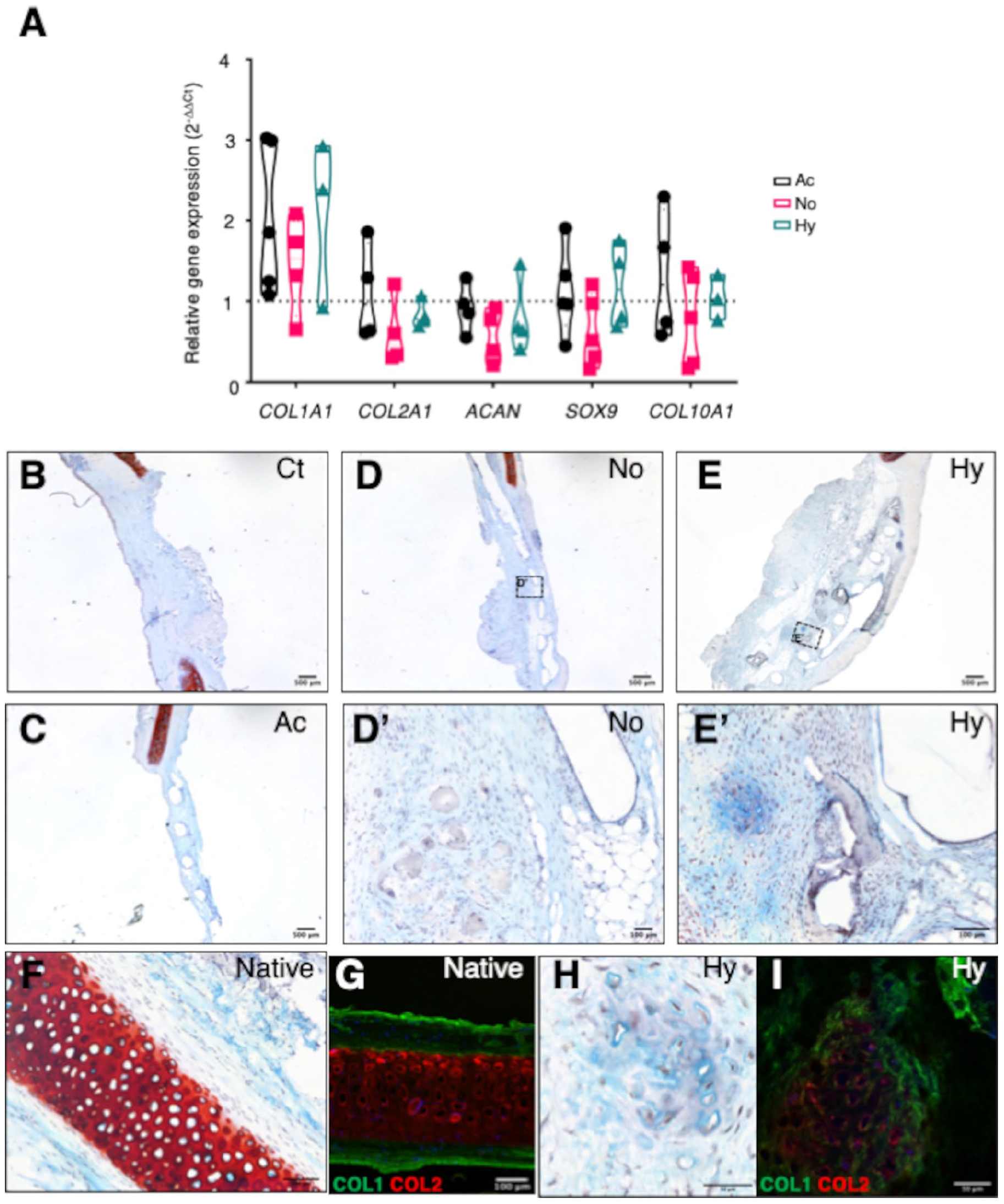
Gene expression and histological analyses of grafted constructs at the end point of in vivo study. (
Histological evaluation and immunofluorescence analysis of the neocartilage formation in vivo
At the 12-week end point of study, Alcian Blue-Safranine O staining was performed on extracted tissue sections (
Discussion
Due to its relative low cost and easiness of use, fused filament fabrication method represents nowadays one of the most used methods for 3D-printed scaffold fabrication, presenting a wide variety of available biomaterials.38,39 Here, we confirmed the feasibility of transplanting a chondrocyte-laden, 2-layered PCL scaffold to an acute cartilage defect on rabbit ears. In this study, our main goal was to improve maturation of the constructs prior to transplantation and ideally emulate hyaline cartilage structure. Of note, as recently demonstrated by Hou et al., 40 at least in the case of MSC-laden scaffolds, the native cartilage niche at the implantation site has a dominant role in cartilage type specification, that is, in the long-term, it does specify which specific types of cartilage will be generated from the implanted cell-laden constructs. In our study, we chose the perichondrium pocket in rabbit ear because it is structurally similar to nasoseptal cartilage, whose defects are clinically relevant. We also used auricular chondrocytes because, in our opinion, it is a more suitable source for clinical use than chondrocytes originating from nasoseptal biopsies. Thus, with regard to potential future use of these scaffolds in nasal septal cartilage reconstruction, we believe that other elastic or hyaline-like cartilage (e.g. auricle, meniscus) could be convenient sources of cells for autologous use in the clinic.
Primary chondrocytes cultured both under standard normoxic expansion and prochondrogenic hypoxic conditions showed detectable deposition of sGAG and type I and II collagens. In vitro, cartilage-like ECM deposition was most clear in the hypoxia prochondrogenic group. SEM images confirmed the secretion of a compact proteoglycan-based matrix in matured scaffolds, composed of collagen types I and II as revealed by immunofluorescence. RT-qPCR experiments revealed lower expression of chondrogenic markers in standard expansion conditions compared with the hypoxia group. Wongin et al. 41 have argued that 2D cell growth induces stress fiber formation, lowering Sox9 production by blocking RhoA signaling. In contrast, prochondrogenic hypoxia conditions in our study may have provided a thick and dense ECM layer in which chondrocytes mimicked their round shape at the native niche, which perhaps promoted an improved retention of the expression of cartilage markers.
Histological analyses performed on scaffolds extracted of the in vivo experiments demonstrated again the presence of collagen types I and II as well as cartilage regeneration. These are the major collagens in cartilage. Type II collagen is primarily found in hyaline cartilage (such as articular and nasoseptal cartilage) and is considered the first choice as a cartilage substitute in many surgical procedures. 42 Unfortunately, type II collagen shows unwanted arthritogenic activities.43,44 By contrast, type I collagen does not elicit adverse immune reaction particularly in the absence of its telopeptides, and lacks arthritogenic effects.45,46 In addition, due to its higher biocompatibility, type I collagen is used commonly in cartilage tissue engineering. 47
Collagen type I is the main fibrillar type of fibrocartilage and elastic cartilage. The highest presence of COL1A1 mRNA in vivo might indicate the presence of elastic cartilage, by dominance of the receptor site, or dedifferentiation of the implanted chondrocyte pool. From RT-qPCR data obtained in the in vitro assays, the expression of collagen I was maintained in the case of scaffolds subjected to prochondrogenic differentiation in hypoxia, suggesting partial dedifferentiation as argued by Rampichová et al. 48 Histologically, we observed clear neocartilage formation after 12 weeks in vivo. In addition, a high percentage of the cross-sectional area was occupied by GAG surrounding cells, and immunostaining demonstrated type II collagen deposition, with cluster formation of collagen II positive cells surrounded by a collagen I matrix. These results can be considered as a qualitative evidence of cartilage-like tissue formation over the 3D-printed scaffolds.
With regard to mechanical properties of the matured scaffolds, Young’s modulus of printed scaffolds was higher than the rabbits’ ear native cartilage. According to bibliography, the elastic modulus of human nasal septal cartilage presents values of 2.72 ± 0.82 MPa 49 that could be in line with the values obtained for hypoxia group in our study (1.054 ± 0.41 MPa). The absence of complications like extrusions or skin erosions in this subset of animals may be related to the relatively short follow-up. We are currently running long-term follow-up studies to evaluate the behavior of scaffolds in 6- and 12-month periods. In a second study currently in progress, we are now cultivating human nasal septal chondrocytes to create a human 3D-printed nasal septal cartilage substitute that will be tested in preclinical studies.
In addition to the aforementioned, a number of strategies could be used to further improve in vitro chondrogenic differentiation and possibly shorten the time required for scaffold maturation. On the cellular side, chondrocyte-derived extracellular vesicles immobilized onto electrospun PCL nanofibers have been shown to promote chondrogenic cell fate. 50 Co-cultures of MSCs and primary chondrocytes seem to enhance differentiation properties in a synergistic manner. 51 Similarly, the use of coatings based on decellularized hyaline cartilage ECM promotes chondrogenic differentiation of periosteal cells. 52 On the materials side, pore geometry and size could be tailored to improve chondrogenic differentiation properties.53,54 Cartilage-derived ECM could also be included on composite material formulations. 55 Finally, use of more biomimetic microenvironments such as application of dynamic compression forces may further improve functional graft characteristics. 56
Conclusions
In vitro maturation of engineered cartilage scaffolds under prochondrogenic conditions that better mimic the in vivo environment may be beneficial to improve functional properties of the engineered grafts. The proposed maturation strategy may also be of use for other tissue-engineered constructs and may ultimately impact survival and integration of the grafts 57 in the damaged tissue microenvironment. 58
Footnotes
Acknowledgments and Funding
We acknowledge the excellent technical assistance of Edurne Bereciartua from the Biochemistry Service of OSI Donostialdea-Donostia University Hospital. The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: National grants from Instituto de Salud Carlos III (ISCIII) through the projects PI16/01430, PI19/01621, and PT20/00030 and, co-funded by the European Union (EU); grants from the Department of Health (17BU207, 19BU203, 20BU206, 20BU210, 2020111004, 21BU203) and the Department of Economy and Competitiveness of the Basque Government (KK-2020/00010, KK-2019/00006, KK-2019/00093); and Diputación Foral de Gipuzkoa. R.H.-M. was supported by the ISCIII Platform Biobanks and Biomodels, PT20/00030, co-funded by the EU
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The Biodonostia Animal Care Committee, Authorized Body, and Competent Authority approved the animal experimentation reported in this study with reference numbers CEEA 19/001, OH19/001, and PRO-AE-SS-131, respectively, in accordance with Spanish Royal Decree 1386/2018, European Directive 2010/63/EU, and other relevant guidelines.
