Abstract
Degenerative diseases associated with articular cartilage pose a huge burden on health care economics. The nature of the tissue involved and the changes therein do not allow self-healing; and most of these problems are progressive. Tissue engineering offers some solutions provided we focus on the right kind of cells and the appropriate surrounding niches created for a particular tissue. The present study deals with the formation of polysaccharide rich stable scaffold of collagen after cross-linking with oxidized gum arabic. The scaffold was tested for its biocompatibility and ability to support cells. The in vitro cytotoxicity of the scaffolds toward induced pluripotent stem cells and chondrocytes was evaluated. Evaluation of expression of lineage specific markers indicates differentiation of induced pluripotent stem cells to chondrogenic lineage and maintenance of chondrocytes per se when grown in the scaffold. Animal studies were carried out to study the efficacy of the scaffold to repair the knee injuries. Cells along with the scaffold appeared to be the best filling, in repair of injured cartilage. These studies show that these scaffolds are potential candidates in applications such as tissue engineering of cartilage.
Introduction
Cells reside in very specific niches; cells and extracellular matrix (ECM) have a reciprocal influence over each other resulting in homeostasis in the tissue. ECM is a product of locally secreted proteins and polysaccharides arranged in a cell-/tissue-specific combination, pattern/topology, and mechanical properties.1,2 How ECM guides the cell response would offer clues to design specific scaffolds for tissue engineering. Scaffolds are created using a wide variety of materials and equally large variations in the fabrication strategies. 3 Natural components of the ECM are a preferred material while designing scaffolds as they would be nonreactive and hence biocompatible.
Collagen, the most predominant protein found ubiquitously in all tissue types, is one of the proteins extensively used in scaffold design. Collagen is nonimmunogenic and biocompatible, supports cell adherence and growth. The scaffolds prepared with collagen lack mechanical strength and are easily degradable4,5 unless crosslinked. Collagen has been used as gels, sheets, and so on after necessary cross-linking procedures using chemicals with known toxicity limiting their application. In an earlier study, 6 we have cross-linked collagen using gum arabic (GA) and made stable biocompatible scaffolds that could be custom modulated to suit the need of appropriate cell type.
GA, a variable mixture of arabinogalactan, oligosaccharides, polysaccharides, and glycoproteins, is obtained from the acacia trees as an exudate. The chemical composition of GA is complex; it contains about 97% carbohydrates consisting of variety of sugars like arabinose, rhamnose, and so on. GA can be oxidized due to the presence of sugars, creating reactive groups for cross-linking with other polymers/proteins, and so on.6 -9
Cartilage has been the major focus for tissue engineering but we still lack dependable ideal procedures to generate an articular cartilage that resembles the native tissue. In spite of the resilience to tolerate repetitive stress, the articular cartilage cannot repair itself; mainly due to the low mitotic potential of the chondrocytes, their immobility and avascularity of the tissue. The matrix of the cartilage is rich in proteoglycans along with collagen and elastin. It is a good strategy to focus on creating a matrix mimicking the natural one for an avascular tissue like cartilage with low cell number and activity. 10 In this study, we composed a matrix of proteins cross-linked with polysaccharides to create a prestressed matrix resembling a native matrix in composition. For the present work, natural polymers, namely, collagen and GA were selected and a biocompatible scaffold was developed by cross-linking collagen with GA, a complex polysaccharide. Based on our earlier study, 6 where we showed that a variety of scaffolds can be designed by changing the ratio of collagen and the oxidation level of GA and it can be suited to match the tissue requirement. For this study we used 50% oxidized GA with 1% collagen to make scaffolds for cartilage repair. These blocks were inoculated with cells and monitored for growth and differentiation in culture conditions. These constructs were tested in animal models involving knee-cartilage injury as a proof of concept. Our results find them suitable for chondrogenic engineering.
Materials and Methods
Preparation of Gum Arabic Aldehyde
Gum arabic aldehyde (GAA) was prepared from GA (Sigma Aldrich) using periodic acid. Ten grams of GA were dissolved in 70 mL of water and 4.3 g of sodium periodate (Sigma Aldrich) in 30 mL of water was added making up to 100 ml. The reaction mixture was rotated in dark at room temperature for 6 hours and precipitated with acetone. The precipitate was frozen and lyophilized. The lyophilized powder was dissolved in 0.1 M borax solution to make 10% solution of GAA. Extent of aldehyde formation was estimated using 2,4-dinitrophenylhydrazine (Merck) in the oxidized GA. 11
Preparation of Gum Arabic Aldehyde–Collagen (GACO) Scaffolds
Collagen isolated from rat tails and checked for purity by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) was made into 1% solution in 0.15 M acetic acid. 6 Five milliliters of solution of GAA was mixed with 5 mL of 1% collagen solution and vortexed for 30 seconds till the solution turned into a thick homogenous emulsion. Cells were seeded at this stage and the mixture was allowed to solidify.
Characterization of GACO Scaffolds
Scanning electron microscopy (SEM) was used to visualize the topology and morphology of the GACO scaffolds. Scaffolds seeded with cells were also processed for SEM imaging. Scaffolds were fixed in 2% glutaraldehyde, followed by dehydration, critical point drying. Dried samples were mounted and gold coated before examining in SEM.
The amount of free amino acids was estimated in the scaffolds, 1% collagen gels by Ninhydrin method using glycine as standard. Stability of the formed scaffolds was checked by the degradation profile of the scaffold blocks left in sterile phosphate buffered saline (PBS) for up to 12 weeks. Weighed wet samples about 1 g (average 1.18 + 0.07 g) were immersed in 10 mL PBS in a tight container and left at room temperature; the samples were blotted and weighed after 4 and 12 weeks. Weight ratio was calculated for each set of block (N = 4). 6
Cell Culture Studies
Induced pluripotent stem cells (iPSCs) derived from human fibroblasts in the laboratory were used after obtaining clearance from Institutional Ethical Committee and Institutional Committee for Stem Cell Research of our institute. 12 iPSCs were induced to chondrocytic lineage using small molecules. Cells were incubated in basal medium (DMEM-F12 medium with 1% ITS, 1% NEAA, and 50 µg/mL ascorbic acid) containing 100 nM TTNPB for 10 days. Ten micrograms of CHIR99021 was added for initial 2 days. Cells were characterized using immunohistochemistry to assess the differentiation status. 13 . iPS/induced chondrocytic cells were seeded into the scaffolds before it solidifies and cultured in DMEM with 10% serum. GAA and sterile collagen solution were mixed in a biosafety hood and vortexed for 30 seconds, cells were seeded and thoroughly mixed with the gel mixture and poured into wells of a culture dish. The dish was kept in CO2 incubator at 37°C with 5% CO2. Cells prestained with tracker dyes (CFSE and Dil [Invitrogen]) were grown in scaffolds and examined in Leica confocal microscope on day 30 in XZ and YZ orientations. Some of the tissue constructs were used for histology and immunohistology after 30 and 90 days in culture. Immunofluorescence staining was performed using primary antibodies for aggrecan, PAX1, Coll 2a, and FOXF1; and counterstained with appropriate fluorescent tagged second antibody ( Table 1 ). The slides were examined in Leica TCS SP5 with appropriate filters after counter staining with DAPI for nuclear staining. Analysis was performed using Leica Application suite X version 2.0. 2.15022.
Details of Antibodies Used for Immunocharacterization.
Cytotoxicity Studies
Biocompatibility of scaffolds toward iPSCs was examined by MTT assay. The cells were seeded on a scaffold at 30,000 cells/cm3 in DMEM containing 10% fetal bovine serum (FBS) and kept at 37°C in a 5% CO2 atmosphere for 24 hours, 7 days, and 30 days. Cells cultured without any scaffold were used as control. After 24 hours, 800 µL of MTT solution (5 mg/mL) was added to each sample and incubated for 4 hours. After 4 hours, 0.01 N HCl dissolved in isopropanol was added and incubated for 40 minutes. To prevent any interference from the remaining scaffold, the plate was centrifuged and supernatant was transferred to fresh dish. The color product developed was quantified by measuring absorbance at 555 nm using Multimode spectrum (Thermo Electron Corporation) plate reader.
Gene Expression of Specific Markers
Total RNA was isolated from the cells grown on scaffolds for 30 days, directly using Trizol reagent and converted to cDNA using Superscript II. Glucose 6 phosphatase dehydrogenase and β-2 microglobin served as internal controls. Real-time polymerase chain reaction (PCR) was carried out for chondrogenic marker genes ( Table 2 ). The primers were custom synthesized by Bioserve India Pvt. Ltd. and optimized using a crosswise combination matrix. A total of 25 ng DNA was used for real-time PCR with SyBR Green as indicator using Applied Biosystem (7900) HT fast real-time PCR system. Fold changes in gene expression were calculated by 2∆CT method. Analyses were done for fold increase in the expression of genes in comparison with control iPSCs grown without scaffolds. Statistical analysis was carried out using GraphPad Prism.
Primers Used for Real-Time Polymerase Chain Reaction Analysis.

Western Analysis
iPSC cells were grown on scaffolds for 30 days were analyzed by Western analysis. Samples were homogenized-lysed for 10 minutes in Tissue Homozilyser II (Qiagen). The proteins were analyzed on SDS-PAGE and transferred by electrotransfer on a Millipore IVD HYBOND–C Extra membrane. The membrane was incubated in buffer containing milk proteins to block nonspecific binding. Membranes were incubated with primary antibody against aggrecan, collagen 2a, OVOS (ovostatin 2), FOXF1, and β actin as internal control. After washing, the membranes were treated with HRP (horseradish peroxidase) conjugated secondary antibodies and visualized by enhanced chemiluminescence.
Animal Studies
Skeletally mature 4+-month-old female Wistar rats of body mass 250 to 300 g from the animal house were used for the experiments. The animals were maintained at standard environmental conditions (temperature 22°C to 25°C, humidity 40% to 70% with 12:12 dark/light photoperiod) approved by the Committee for the Purpose of Control and Supervision of Experiment on Animals, and all the experimental protocols were approved by Institutional Animal Ethics Committee of the Centre for Cellular and Molecular Biology.
Experimental animals were divided into 5 groups. During surgery group 1 received only scaffold; group 2—scaffolds with 5 × 105 chondrocytes/100 µL was injected; group 3—scaffolds with 5 × 105 iPSCs/100 µl; group 4 received only 5 × 105 chondrocytes; and in group 5—only 5 × 105 iPSC were injected. Contralateral knee of each animal was either sham operated and served as control or was used for injury without any intervention.
Animals received anesthesia via intraperitoneal root with 10% ketamine HCl 2% xylazine HCl, acepromazine 0.3 % in the saline buffer (0.5 mL/kg IP). Body hairs from knee were removed using surgical blades. After general disinfection, an incision was made to open the joint capsule, and the patella was dislocated to expose the femoral condyles, using a motorized drill (diameter 5 mm) we removed cartilage from the frontal femoral condyles. The surgical wound was closed with suture materials. Povidone-iodine ointment was applied over the wound externally for 3 days.
All the animals were left to heal for the next twelve weeks and were observed daily for any signs of abnormality or inflammation. Twelve weeks later all the animals were culled, and knees were harvested. The resected samples were fixed in 10% neutral buffered formalin, decalcified using 10% EDTA solution for 20 days and processed for histopathology. Five-micrometer sections were stained with hemotoxylin-eosin and Safranin-FCF green. The stained slides were examined under a light microscope Axioplan II.
Results and Discussion
Preparation of Scaffold
Tissue engineering of cartilage has been at the forefront but we still lack reliable methods to mimic the native tissue. Cartilage lacks vascular tissue and has only a single type of cells, that is, the chondrocytes and yet it has been difficult to construct. 10 In this study, we have designed the construct to match its native matrix in terms of protein to carbohydrate content and encapsulate iPSCs or chondrocytic progenitor cells in a matrix. The matrix produced has cross-linked collagen without any chemical cross-linkers. The cross-linking has been carried out using glycans that has also been a strategy to increase the glycan component in the matrix. 6 We envisaged this matrix would influence the phenotype of the cells and would be suitable for in situ repair.
GA (40 mmol) was mixed with periodate (20 mmol) corresponding to 50% oxidation of the gum. The yield of the product was about 122%. GA oxidized in presence of periodate showed the presence of aldehyde groups to the extent of 62.97 ± 23.5 mmol (n = 8). Ten percent oxidized GAA solution in borax buffer was mixed with 1% collagen solution in the ratio of 1:1 and vortexed until the frothy mixture turned viscous. Cells were seeded into the GACO mixture and were poured into multiwell dishes before they set. GA is a glycan-rich plant exudates containing polysaccharides and proteins. The sugar moieties get oxidized with periodate to generate aldehyde groups. 6 These can be conjugated to imine/amine groups of other biopolymers by Schiff’s reaction ( Fig. 1 ). The amount of crosslinking was confirmed both by estimation of free amino acids and the amount of glucose present in the scaffolds. The amount of free amino acids in the crosslinked scaffold was 1.8 µg/100 µg ± 0.022, about one-tenth of that estimated for collagen gels 19.8 µg/100 µg ± 4.328 ( Fig. 1 ). These gels were subjected to glucose estimation and the amount of polysaccharide assessed was 19.139 ± 0.45 µg/mg of scaffold (n = 4). The polysaccharide to protein (hydroxyproline) ratio was found to be 2.9 ± 0.24:1. The natural scaffold of a cartilage is generally having a polysaccharide:protein molar ratio of 3:1; the hyaline cartilage is almost like proteoglycan and matrix proteins trapped in collagen fibril network.14,15
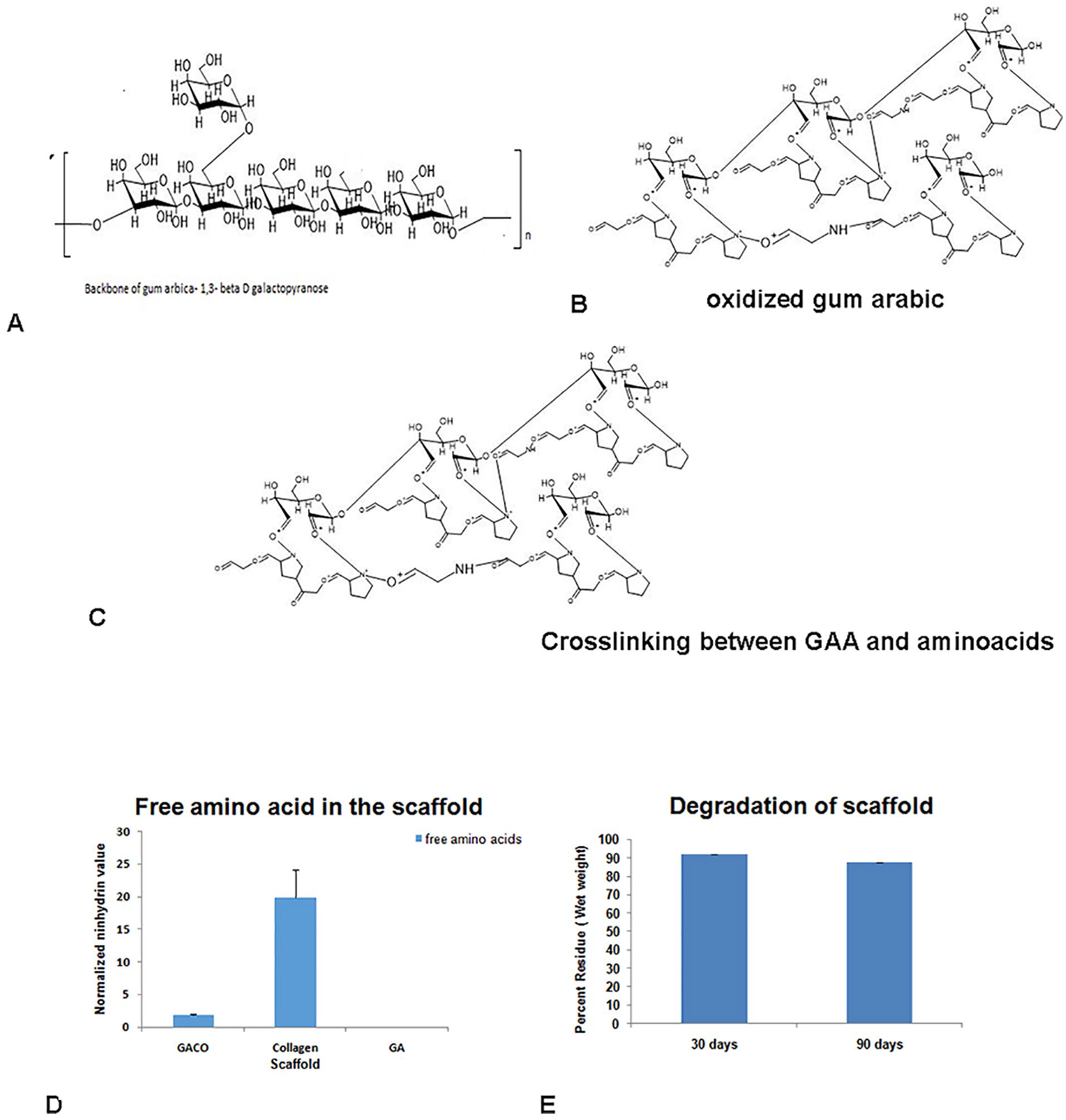
Schematic diagram of the structure of gum arabic (GA) (
The stability of gels was analyzed by leaving about 100 mg (wet weight) crosslinked gel in sterile PBS for about 3 months and estimated for weight at 30 and 90 days. The scaffold remained stable up to 90 days without much loss in weight whereas the collagen gel had loosened and was not amenable to weighing ( Fig. 1 ).
Cell Culture Studies
The GACO blocks seeded with cells were examined using SEM. The surface of the gels containing cells mostly appeared smooth without revealing any porosity ( Fig. 2A ) and occasional cells adhering to the surface ( Fig. 2B ).
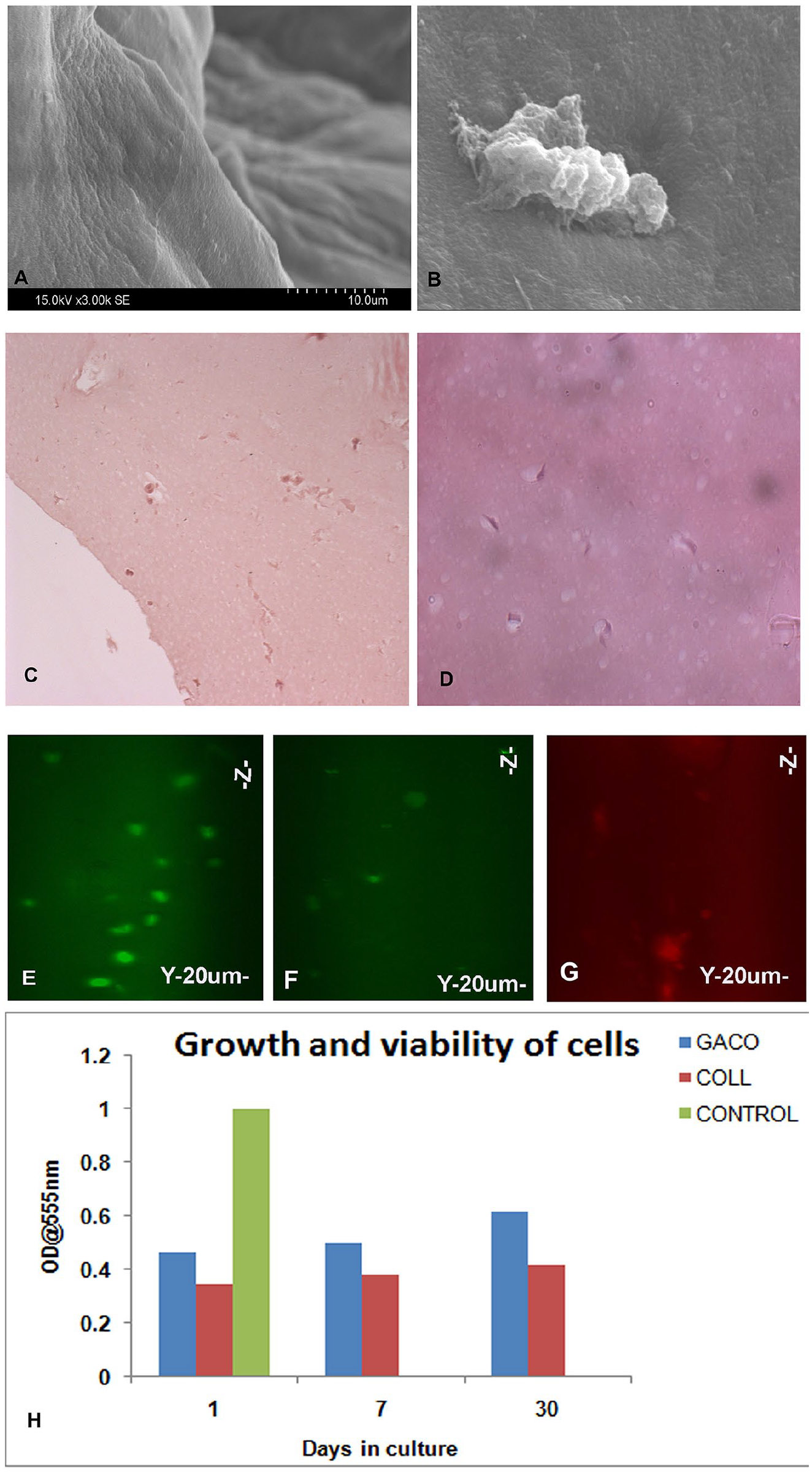
Scanning electron micrograph of the scaffold (
Study of the constructs in light microscopy showed that the cells remained viable and one could see the cells deep into the scaffolds. The histopathology studies showed presence of cells at different heights suggesting good distribution of cells in the scaffold ( Fig. 2C and D ). Cells preloaded with tracking dyes (CFSE and Dil tracker) were also used to locate the cells deep in the scaffolds with the confocal imaging; cells could be seen across the depth of scaffolds at various levels up to day 30 ( Fig. 2E-G ). Detection of stained cells in the scaffolds revealed their viable status. Besides, viability of cells in the scaffolds was also assessed based on the metabolic activity of the cells as measured by MTT assay. The scaffold construct was biocompatible as the cells remained viable on longer incubations of up to 30 days. MTT assay revealed the presence of viable cells and growth ( Fig. 2H ). Moreover, staining of these cells with specific markers also confirmed the presence of viable cells.
iPSCs were induced to chondrogenic lineage. After characterization of chondrocytic cells by immunohistochemistry ( Fig. 3A-C ), these cells were also seeded into the GACO scaffold and incubated for 30 days. Chondrocytic cells remained viable and retained their phenotype ( Fig. 3D-G ). The chondrocytic cells embedded in the scaffold continued to show positive staining for chondrogenic markers like Coll2a, SOX9, aggrecan up to 30 days suggesting that the scaffolds support the viability and maintenance of the chondrocytic cells. The glycan to protein ratio of the scaffold after being cultured with the cells for 30 days remained more or less the same around 2.8 to 3.0. This homeostasis may be maintained by some amount of degradation of the scaffold, whereas at the same time with secretion of new ECM by the cells.
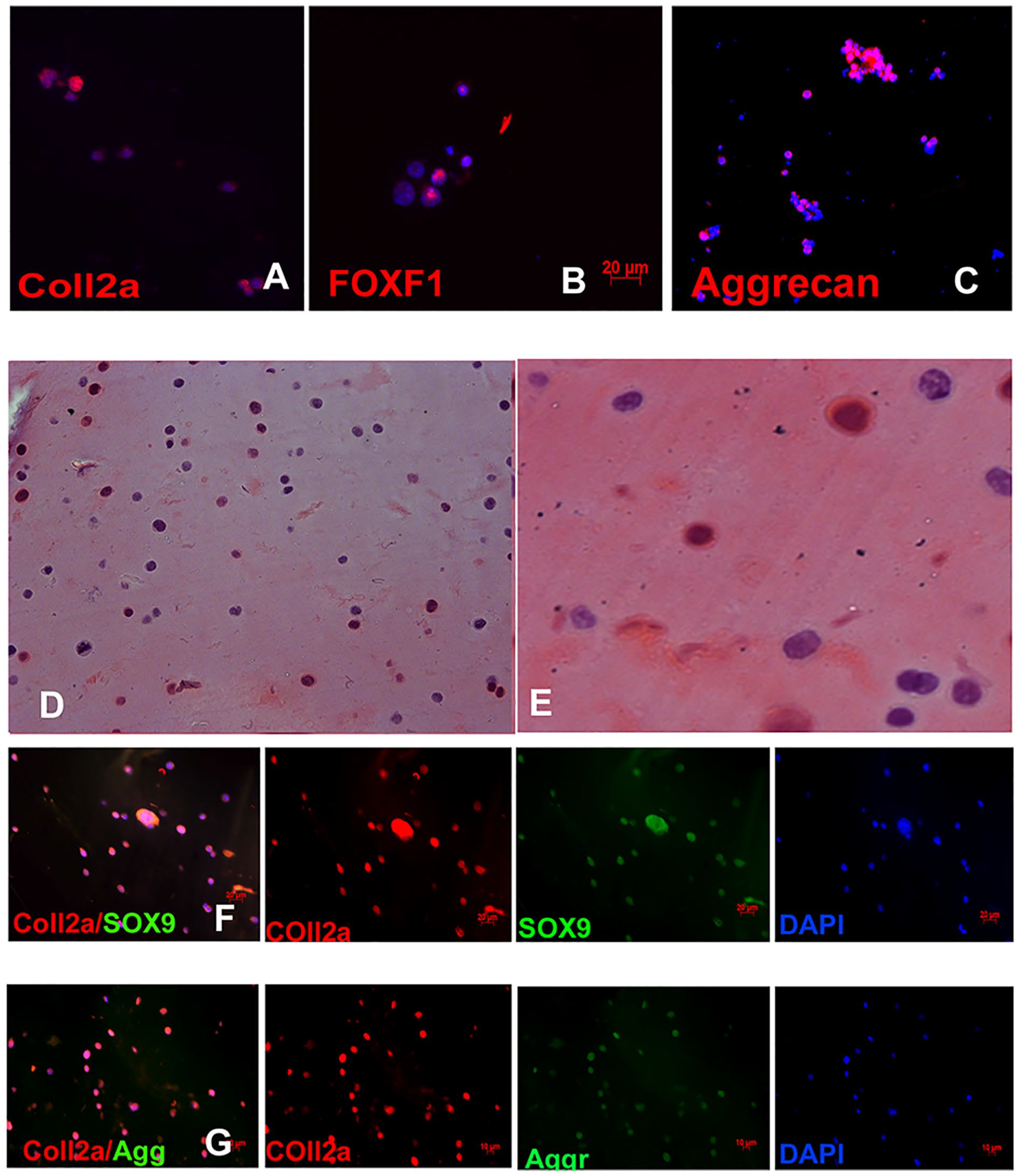
Induced pluripotent stem cells (iPSCs) were differentiated into chondrocytes and the cells expressed markers like Coll2a (
Differentiation of Cells
ECM promotes cell signaling and is able to regulate the cell differentiation by virtue of its ability to bind growth factors, chemokines, matrix constituents itself, topology generated, and other molecules.2,16,17 Changing the composition of the matrix by varying ratios of glycans may change the fate of cells.18-20 Cells transduced with promoter reporters for beta cells, osteogenesis and chondrogenesis were added to the scaffolds. The daily screening of the dishes showed appearance of markers by day 7 as shown in Table 3 .
Appearance of Lineage Specific Markers. a

GACO, gum arabic–collagen; Coll, collagen; Neg. negative; Pos. positive; Sl. Pos. slightly positive.
Mesenchymal stem cells transduced with reporter vectors for beta cells, osteogenesis, and chondrogenesis were grown with the scaffolds and screened for about 10 days. Appearance of fluorescence indicated activation of lineage specific gene expression. We start seeing expression of aggrecan and RUNX2 by day 7.
iPSCs grown in scaffolds were characterized by immuno-fluorescence staining using antibodies against chondrocyte and iPSC markers. Cells grown in GACO scaffolds for 30 days showed positive staining for Coll 2, SOX9, aggrecan, and ovostatin2 ( Fig. 4 ). GACO scaffold showed chondroinductivity, this was further analyzed and established by quantitative PCR studies and Western analysis. Presence of polysaccharides in the biocomposites was shown to improve cartilage repair.20-22
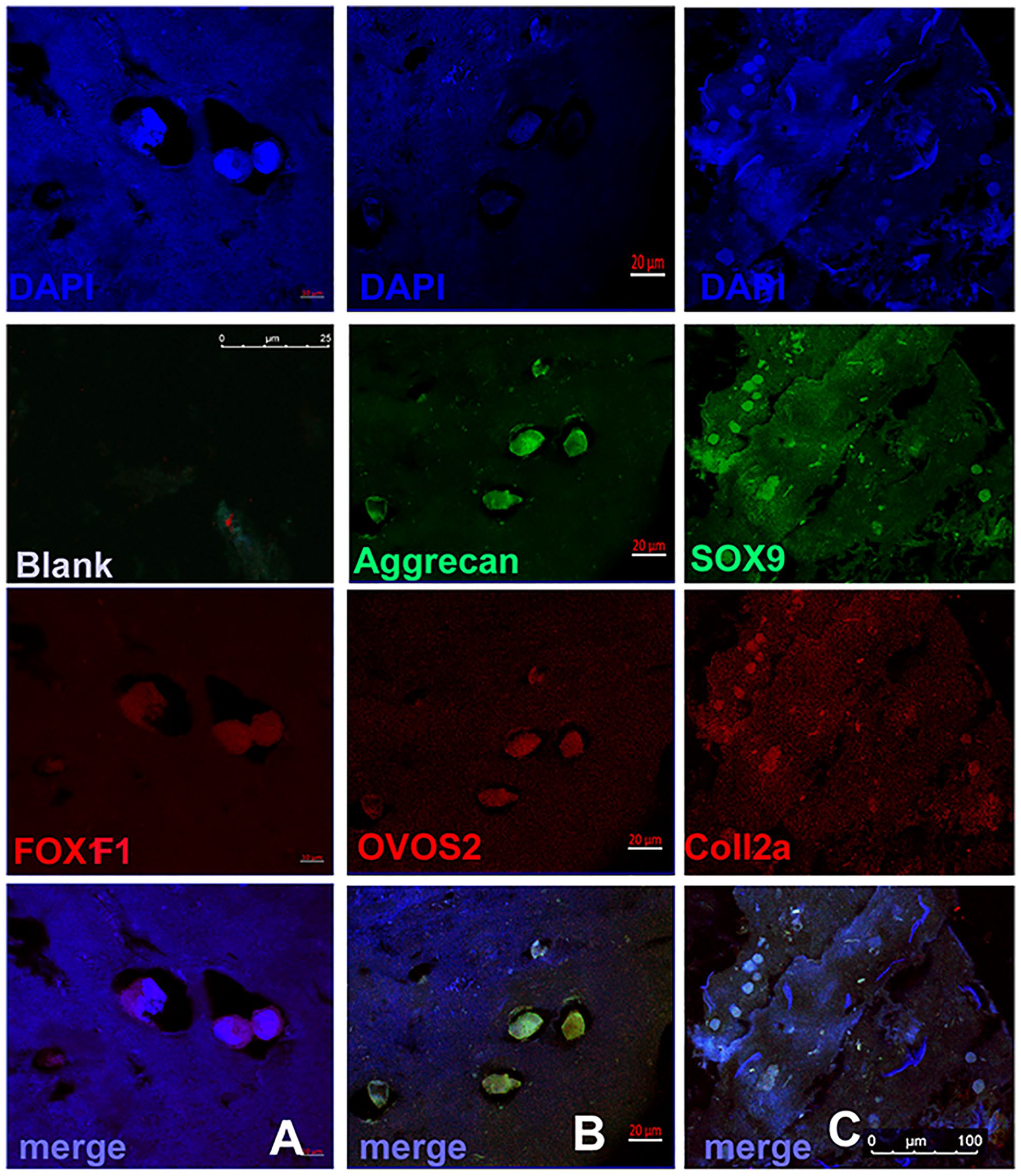
Induced pluripotent stem cells (iPSCs) grown in scaffold for 30 to 45 days also showed expression of chondrogenic markers (
Gene Expression Analysis and Western Blot
To evaluate the status of our cells grown in the GACO scaffold, aggrecan, FOXF1, Collagen 2a, CA, CYTL1, PAX1, fibulin, OVOS2, and SOX9 were analyzed for relative gene expression. In relation to control iPSCs, there was manifold enhancement in gene expression of FOXF1, OVOS2, PAX1, aggrecan (ACAN) and fibulin in cells grown in scaffolds ( Fig. 5A ). The fold increase was significantly (P < 0.05) more in iPSCs grown with the scaffold indicating a change in lineage. iPSCs induced into chondrogenic cells do show an enhanced expression of chondrogenic markers like Coll2a, SOX9, ACAN in 3-dimensional cultures23,24 and our studies also correspond to the cells in 3-dimensional cultures entrapped in gels.
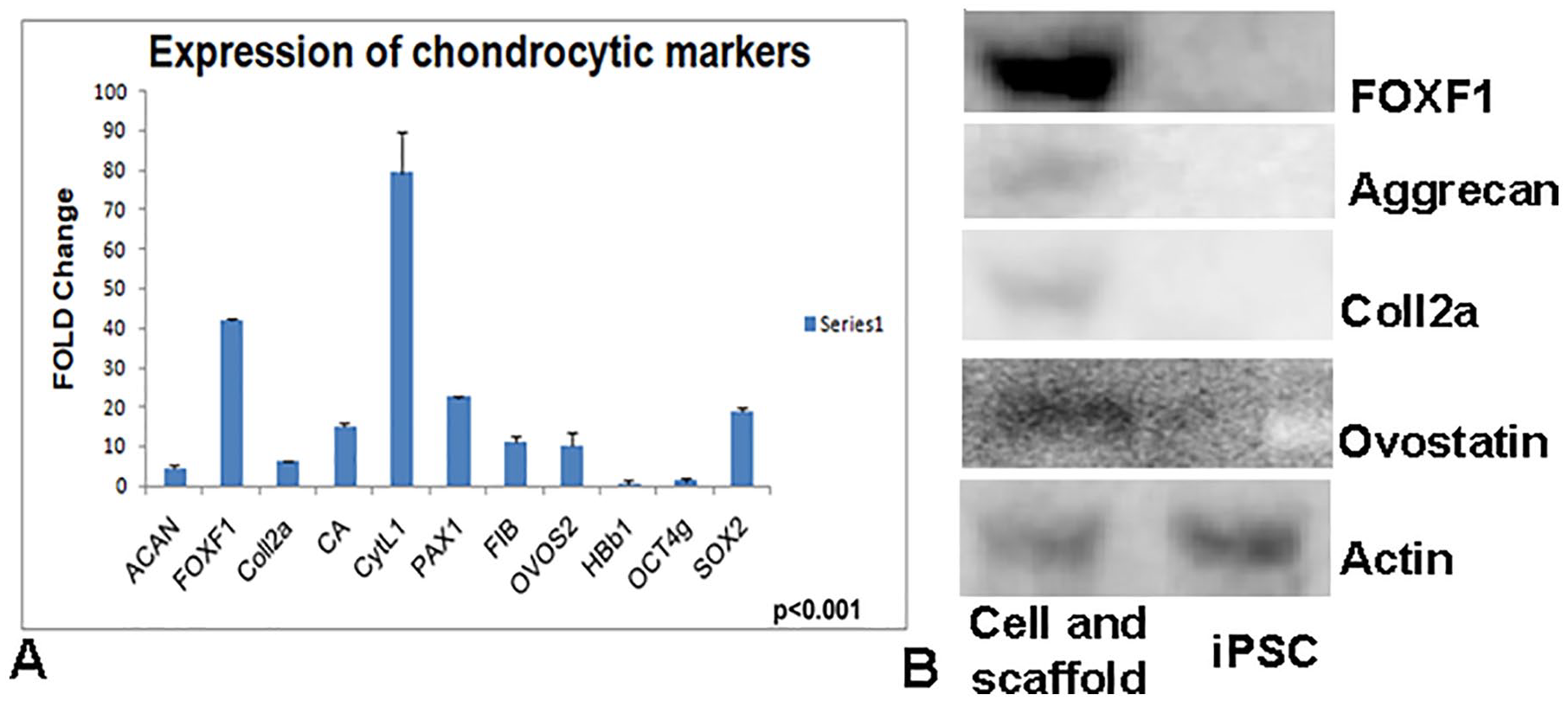
(
In this study, we used FOX F1, collagen type 2a, aggrecan, and ovostatin2 expression as key markers for chondrogenesis. Cells grown with the scaffolds for 30 days were analyzed for Western analysis. Using antibodies against FOX F1, Coll2a, aggrecan and ovostatin2 we found their expression in iPSC grown in the scaffolds and none in iPSC grown without the scaffold. β-Actin used as internal control expressed equally in both iPSCs and iPSCs grown in the scaffolds ( Fig. 5B ). Gene expression, immunostaining and Western analysis indicate that the iPSCs grown in the GACO scaffold differentiate into chondrogenic/nucleus pulposus cells.
The influence of the scaffold is established here as we did not use any growth factors to induce chondrocytic differentiation. Addition of growth factors could enhance the performance as the growth factors can be bound to the matrix as well. 25
Animal Studies
Proof of Principle, Clearance, Toxicology, and Safety
The material, that is, GACO scaffolds clearly showed differentiation of iPSCs into chondrocytes as studied by expression of marker genes and immunohistochemistry. The material was also injectable and can be injected at the injury site before it sets. The scaffolds take about 3 ± 0.05 minutes to set once injected. Animals were divided into 5 groups with 3 animals each and were inflicted with knee cap injury in open surgery. The animals in all the groups remained healthy and did not show any immunological or infectious reaction to the material injected. None of the animals was seen dragging their feet due to injury. Assessments such as macroscopic, histological, and biocompatibility were used as endpoints in proof of efficacy and safety. After the end of the recovery period of 12 weeks, the knee portion was excised and subjected to histopathology studies.
For tissue processing, the femoral axis was adjusted to be upright against the embedding surface so that each sample was in the same orientation and while sectioning we get a similar profile for all the sets.
The outcome of the cartilage repair was determined at 12 weeks, and there was a difference in the repair profile of the cartilage depending on the choice of filling. The medial meniscus remained normal in all cases. In the control animal with cartilage injury and no intervention, defect remained evident as an indentation at surface and fibrous tissue with thinning of cartilage compared to control animals ( Fig. 6A-D ). The scaffold material injected at the injury site had filled in the injury gap, the material integrated well with the surrounding tissue and cells had infiltrated the injury area into the scaffold ( Fig. 6E and F ). Only cells whether iPSC or chondrocytes at the injury site did not help the healing process the injury site had thin layer of dense fibrous tissue ( Fig. 6G , H , K , and L ). Cells alone were not sufficient to heal the injury. There was a thinning and indentation at the injury site. Cells and the scaffold material injected at the injury site had the best healing effect on the injury. New cartilage formation was evident in the injury site as opposed to the fibrous growth in other sets. In the group of animals that received chondrocytes and scaffold ( Fig. 6I and J ), the wound area covered without any indentations; there was chondrocytic matrix flow-through near the edges of the injury site (asterisk). At 12 weeks, the cartilage did not appear like hyaline but was progressing ( Fig. 6I and J ). Animals that received iPSC with scaffolds also showed progressive healing ( Fig. 6M and N ). The incorporation of cells into biomaterial has always been significantly helpful in regeneration of any tissue.10,26 Earlier studies have shown that the scaffolds support the development of cartilage injury.10,27-31
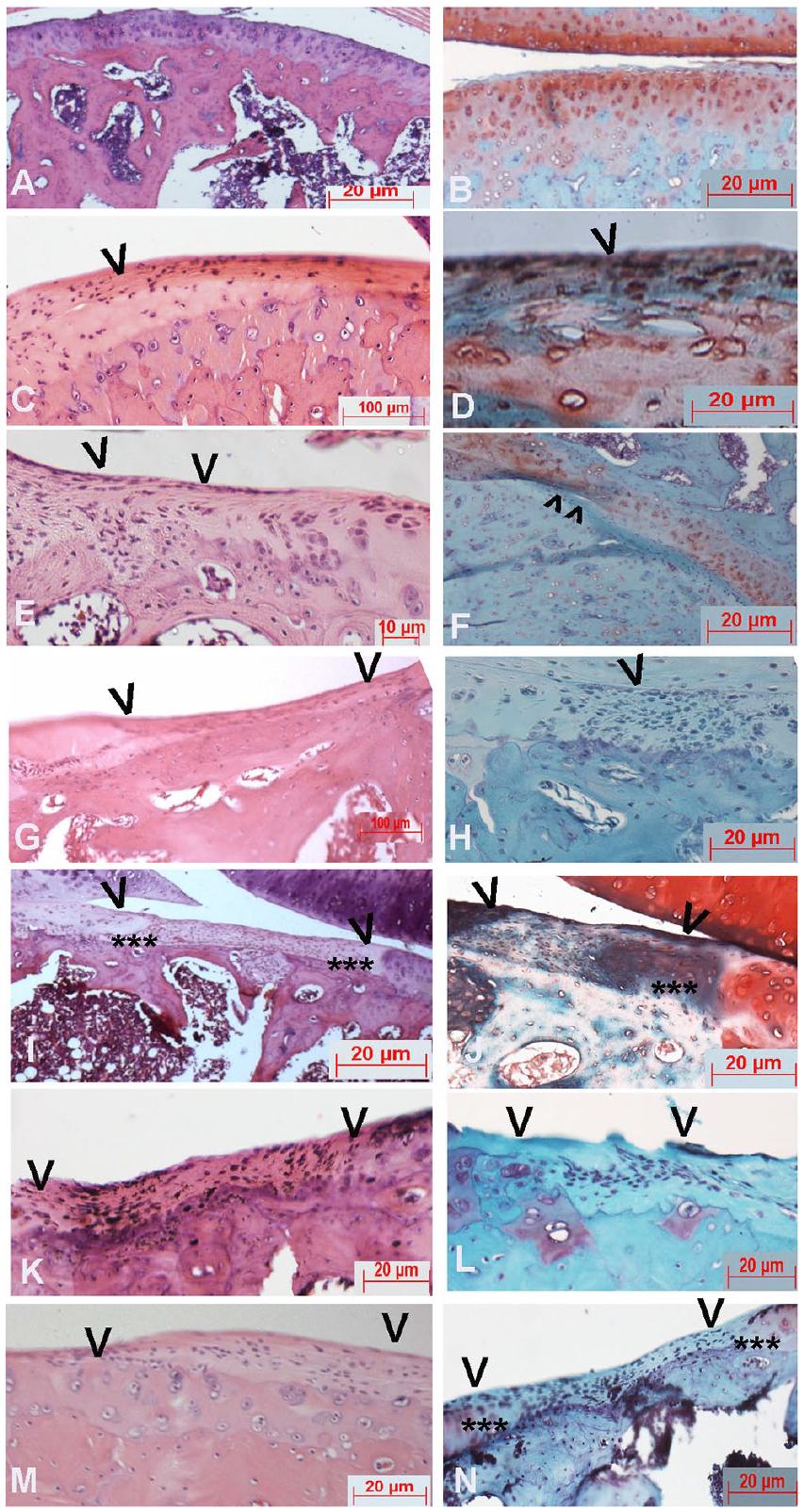
Histopathology studies of the animal experiments. (
Staining with safranin/fast green revealed that the scaffold material is not degraded even after 3 months also as the staining for collagen and glycan is still high at the spot of injury in the group where chondrocytes with scaffolds were injected ( Fig. 6F and J ). Safranin stained the cells indicating the presence of proteoglycans at the cartilage. In all the other groups, safranin only stained the cells and the matrix area usually remained devoid of the glycans, whereas the cartilage surrounding the injury had copious staining for safranin ( Figs. 6 and 7 ).
We also studied the type II collagen synthesis at the site of injury with the scaffold material and cells as healing was best in these cases. Collagen 2a expression was evident in the areas surrounding cells and so was aggrecan. In the case of rats with scaffold and iPSCs, the aggrecan expression was not very high ( Fig. 7 ). The best healing was observed in presence of chondrocytes and scaffold injected together at the injury site in terms of filling, histology, and marker expression.
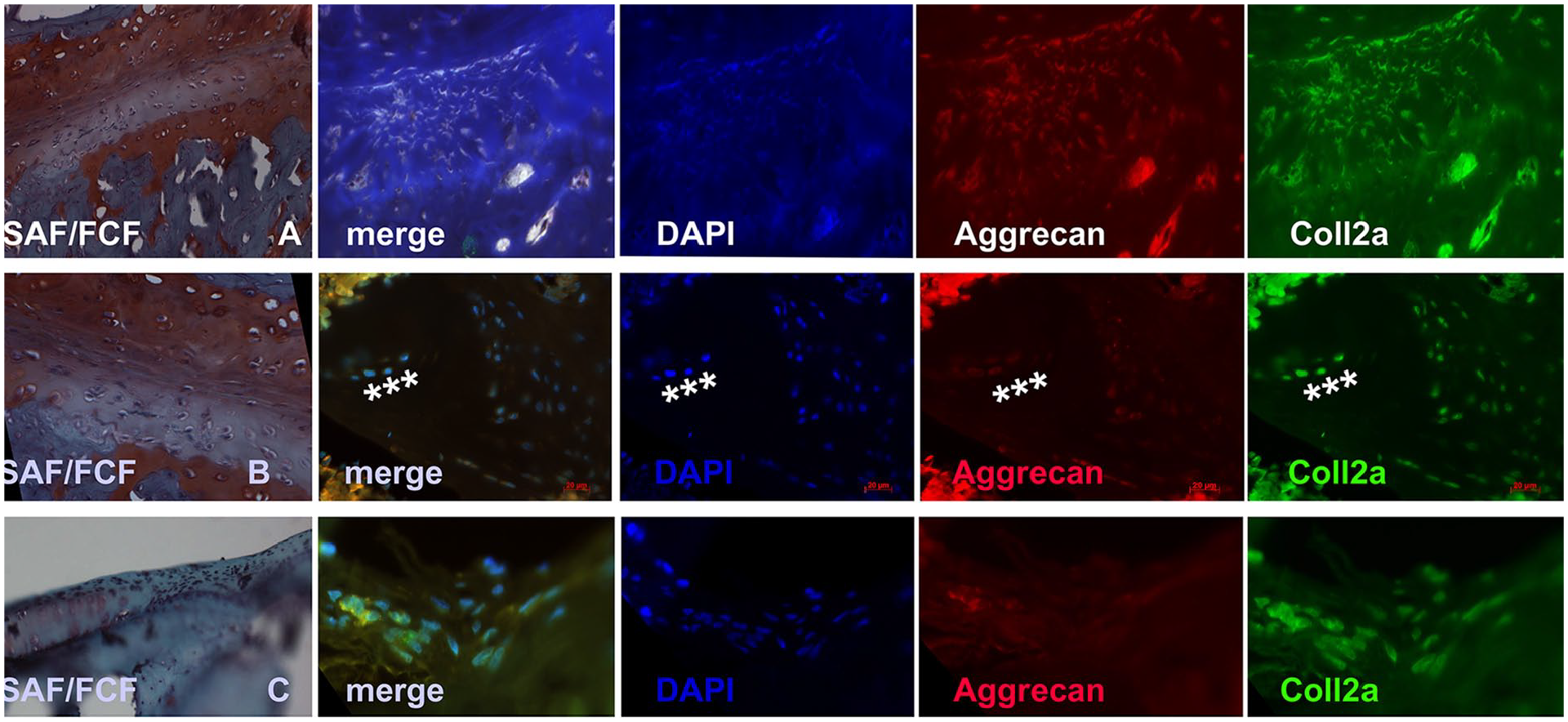
Immunohistochemistry of the site of injury after 12 weeks of recovery shows that the scaffold and chondrocytes (
The results of animal experiments suggested that the defects of the chondrocytic scaffold group had been adequately filled in the cases where scaffold has been included with or without cells, the boundary between the injured area and surrounding tissues were merged and the morphology of cells was also changing and the cells in the injury site had the chondrocytic markers ( Figs. 6 and 7 ). The results suggest that the cartilage scaffold is able to guide the recovery of the cartilage.
Cartilage cells are slow growing and the repair of these organs is painfully slow. The primary intervention is by seeding cells or scaffolds. Various cell types tried for treatment are chondrogenic or mesenchymal stem cells.32-34 Among the scaffolds for cartilage repair, a number of natural and synthetic materials have been tried and tested.28-31 Natural polymers explored include collagen, hyaluronic acid, gelatin, alginate, chondroitin sulfate, fibrin glue and cellulose but lose out on synthetic material for want of mechanical strength, stability, and host enzyme degradation. Glycans or polysaccharides in the composites of scaffolds have always shown to improve cartilage repair. 35 Our scaffold using collagen and polysaccharide-rich GA also appears to support the differentiation of iPSCs as well as growth of the chondrocytes. ECM of the cartilage is rich in proteoglycans and loss of PGs initiates a degenerative process35; addition of glycans to enhance cross-linking and stability was also a step to increase the carbohydrate content of the scaffold. The scaffold was able to support cell growth in cultures and in vivo conditions.
Conclusion
GACO scaffolds prepared by cross-linking collagen with oxidized GA, result in formation of stable blocks rich in polysaccharides; appear to be closely mimicking the native ECM and helps in repair of the cartilage injury in rat model. These blocks support the growth and sustenance of chondrocytes and human iPSCs. These scaffolds are chondroinductive in nature as iPSC plated on these scaffolds display chondrogenic/nucleus pulposus cell markers. The animal studies showed that the scaffolds appear to be promising for cartilage repair because of its biointegrity and chondroinduction.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Induced pluripotent stem cells (iPSCs) derived from human fibroblasts in the laboratory were used after obtaining clearance from Institutional Ethical Committee and Institutional Committee for Stem Cell Research of our institute. The animals were maintained at standard environmental conditions approved by the Committee for the Purpose of Control and Supervision of Experiment on Animals, and all the experimental protocols were approved by Institutional Animal Ethics Committee of the Centre for Cellular and Molecular Biology.
