Abstract
Objective
The study was performed to evaluate cartilage within the knee following a first-time patellar dislocation, using elevated MRI-based T1ρ relaxation times as an indicator of low proteoglycan concentration. The hypothesis is that MRI-based T1ρ relaxation times for patellofemoral and tibiofemoral cartilage are significantly longer for knees being treated for patellar dislocation than for healthy control knees.
Design
Twenty-one subjects being treated for a first-time, unilateral dislocation of the patella and 16 healthy controls participated in MRI-based T1ρ relaxation time mapping. Mean relaxation times were quantified for patellofemoral and tibiofemoral regions for injured knees, the contralateral knees, and healthy controls. T1ρ values for each region were compared between the 3 groups with generalized estimating equations. Linear regressions were also performed to correlate T1ρ relaxation times with time from injury.
Results
The knees with a disloction had longer T1ρ relaxation times than the contralateral knees and control group at the medial patella and longer relaxation times than the control group at the lateral tibia (P < 0.05). T1ρ relaxation times at the medial patella also decreased with time from injury (r2 = 0.21, P = 0.037).
Conclusions
Compositional changes to cartilage on the medial patella are related to traumatic impact during a dislocation. Potential exists for cartilage properties at the medial patella to improve with time. Cartilage degradation at the lateral tibia is not directly related to traumatic impact. The current baseline data are a starting point to characterize the pathway from a first-time dislocation to progressive cartilage degradation and osteoarthritis.
Keywords
Introduction
A lateral patellar dislocation is a traumatic event with annual incidence rates of approximately 150 and 50 per 100,000 person years for age ranges of 14 to 18 and 19 to 24 years, respectively. 1 In the acute phase following a first-time dislocation, more than 50% of subjects show signs of cartilage lesions on the medial or central patella, likely related to impact of the medial patella against the lateral femoral condyle during a dislocation.2-4 Cartilage lesions are also noted at the lateral femoral condyle and lateral trochlear groove for approximately one-third of patients.3-6 Follow-up evaluations show cartilage lesions on the patella for more than 95% of patients,2,7,8 with an increased frequency of lesions within the trochlear groove and tibiofemoral joint. 2 Cartilage lesions following patellar dislocation can progress to patellofemoral post-traumatic osteoarthritis (PTOA). Patellofemoral OA is identified for approximately 50% of patients by 25 years following a patellar dislocation, 9 accounting for more than 25% of all cases of patellofemoral OA. 10 Progression to tibiofemoral OA following patellar dislocation has not been characterized to date.
A more quantitative assessment of cartilage properties is needed to characterize the pathway from patellar dislocation to OA. Quantitative MRI is increasingly being used to identify cartilage matrix degeneration11-14 that could progress to OA 13 based on elevated T1ρ and T2 relaxation times. T1ρ relaxation times are more sensitive to concentration of proteoglycans, and T2 relaxation times are more sensitive to collagen structure. 15 Previous studies focused on general patellar instability, 8 multiple dislocations,16,17 and surgical treatment for patellar dislocation18 have shown elevated T2 relaxation times for cartilage on the patella. 18 A specific focus on the acute phase following a first-time patellar dislocation is lacking, but needed for longitudinal characterization of cartilage degradation. Quantitative MRI-based evaluation of cartilage within the trochlear groove and tibiofemoral joint is also lacking. Only one of the previous studies 17 evaluated contralateral knees to specifically relate cartilage degradation to traumatic impact. Characterization of the influence of demographic parameters on T1ρ and T2 relaxation times following patellar dislocation is also lacking. Studies that have related T1ρ and T2 relaxation times for patellofemoral and tibiofemoral cartilage to demographic parameters have shown that the relationships vary with the type of knee disorder.19,20 Relationships between patellofemoral T1ρ/T2 relaxation times and demographic parameters are also lacking for healthy knees.
The current study was performed to characterize cartilage properties throughout injured and contralateral knees following a first-time patellar dislocation to address cartilage degradation related to the initial traumatic injury and set the baseline for characterization of longitudinal cartilage degradation. The hypothesis of the study is that T1ρ relaxation times for patellofemoral and tibiofemoral cartilage are significantly longer for knees being treated for a first-time patellar dislocation than for healthy knees. The study also includes characterization of the influence of demographic parameters on T1ρ relaxation times for healthy knees and following patellar dislocation.
Methods
Subjects
Twenty-one injured subjects between the ages of 14 and 36 years were prospectively recruited from January 2019 to December 2021. The study was approved by the Cleveland Clinic Institutional Review Board (ID # 18-1388), and all subjects (plus a guardian for minors) signed a consent or assent form prior to participating. Patients were recruited based on treatment for a first-time, complete, traumatic dislocation of the patella from the trochlear groove for one knee. All dislocations were confirmed by identification of a bone bruise on the medial patella and lateral femoral condyle on MRI. Exclusion criteria were: (1) age less than 13 years, (2) prior patellar dislocation for either knee, (3) prior surgery for either knee, (4) more than 6 months since the initial dislocation, (5) existing OA, and (6) soft tissue injuries or fractures that required surgical treatment. All patients were initially treated conservatively with activity modification, physical therapy, and bracing. Most patients were no longer wearing a brace by the time of evaluation. An additional 16 subjects between the ages of 13 and 33 years with no history of prior surgeries, injuries, or OA for either knee were recruited to participate in the control group.
Demographic data collected from the injured subjects and controls included age, sex, and body mass index (BMI). Time from dislocation to imaging was also acquired for the injured subjects. Skeletal maturity was assessed for each subject, based on the MRI data. 21 Subjects were considered skeletally mature if all growth plates on the femur, tibia, and fibula were fully closed.
Magnetic Resonance Imaging
MRI scans were performed on a 3T scanner (Prisma, Siemens) using a 1Tx/15Rx knee coil (Quality Electrodynamics). Both knees were scanned for the injured subjects, and one knee for the controls. Anatomical parameters were measured from a 3D non-fat saturated Sampling Perfection with Application optimized Contrasts using different flip angle Evolution (SPACE) scan. Cartilage was segmented from a 3D fat saturated Dual Echo Steady State (DESS) scan. Relaxation times were quantified using a fat saturated T1ρ Magnetization-prepared Angle-modulated Partitioned-k-space Spoiled gradient-echo Snapshots (MAPSS) imaging sequence.22,23 The major parameters of the MRI protocol are shown in Table 1 .
Parameters for 3D MRI Scans.
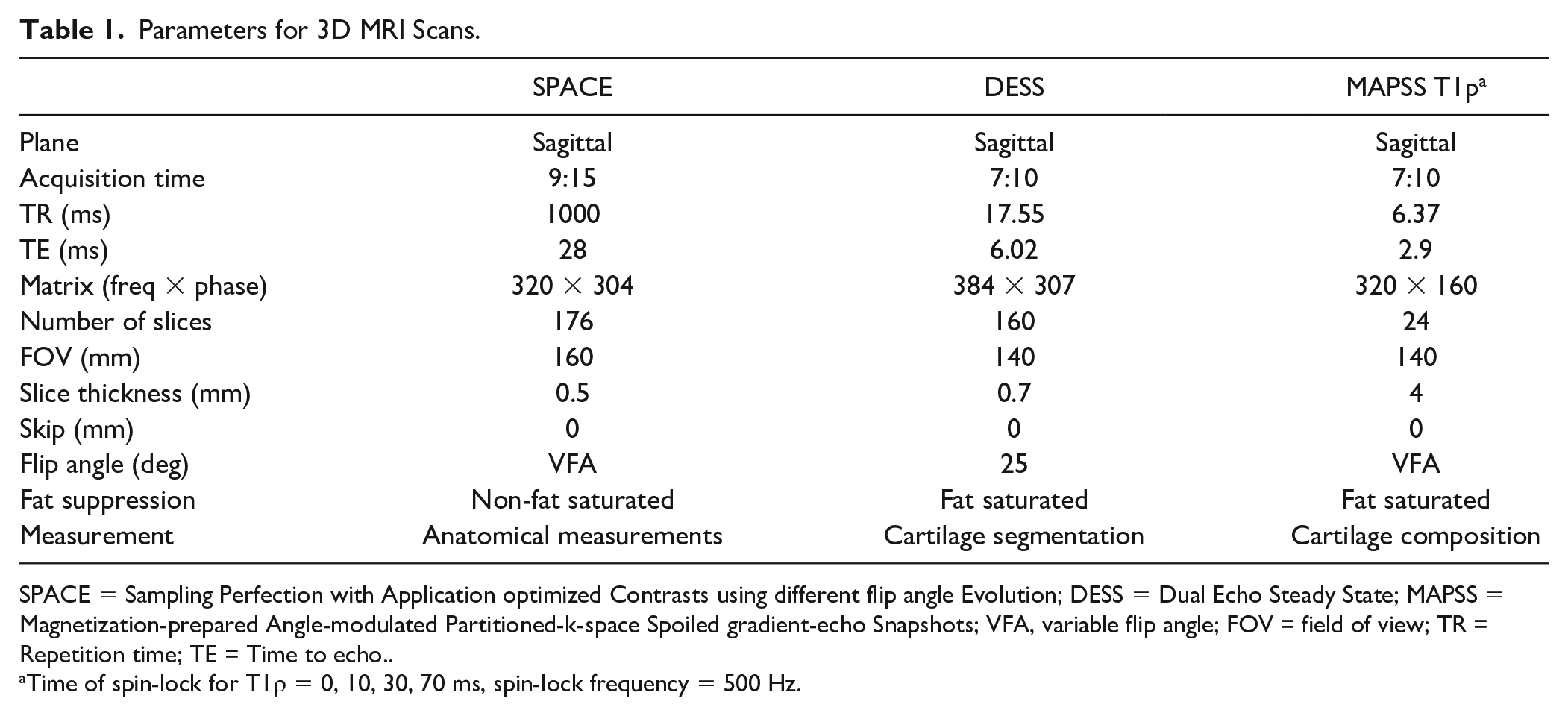
SPACE = Sampling Perfection with Application optimized Contrasts using different flip angle Evolution; DESS = Dual Echo Steady State; MAPSS = Magnetization-prepared Angle-modulated Partitioned-k-space Spoiled gradient-echo Snapshots; VFA, variable flip angle; FOV = field of view; TR = Repetition time; TE = Time to echo..
Time of spin-lock for T1ρ = 0, 10, 30, 70 ms, spin-lock frequency = 500 Hz.
Anatomical measurements characterized trochlear depth, patellar height, and position of the tibial tuberosity for each knee. Trochlear depth was based on lateral trochlear inclination, as the angle between a line along the lateral ridge of the trochlear groove and a line tangent to the posterior condylar axis of the femur ( Fig. 1 ). 24 The measurement was made in an axial plane, using the most proximal slice showing cartilage spanning the trochlear groove to represent the lateral ridge and a slice with the largest anterior-posterior dimension of the femoral condyles to represent the posterior condylar axis. A lateral trochlear inclination less than 11° was considered trochlear dysplasia. 25 Position of the tibial tuberosity was based on the tibial tuberosity to trochlear groove (TT-TG) distance, measured as the lateral distance from the deepest point on the trochlear groove to the most prominent point of the tibial tuberosity. 24 TT-TG distance was measured in an axial plane, using the same slices for the posterior condylar axis and deepest point of the trochlear groove, with a separate slice at the patellar tendon attachment to identify the tibial tuberosity point. A TT-TG distance exceeding 20 mm was considered lateral malalignment. 26 Patellar height was based on the Caton-Deschamps index, calculated by dividing the distance from the most distal point of the articular surface of the patella to the most superior-anterior point on the tibia by the superior-inferior length of the cartilage on the patella. Measurements were made from a sagittal slice with the largest superior-inferior dimension of the patella. 24 A Caton-Deschamps index ≥ 1.2 was considered patella alta. 24
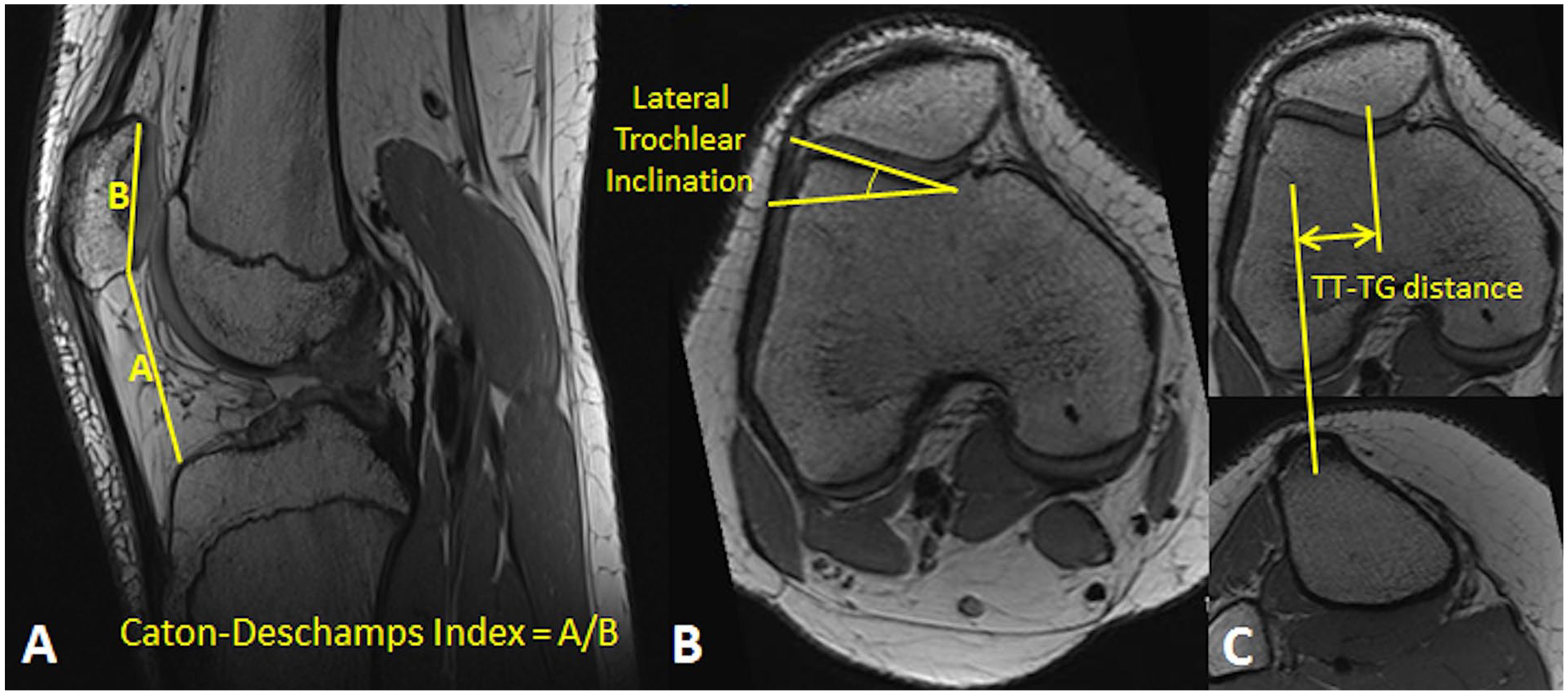
Measurements used to characterize knee anatomy. The lines drawn to calculate the Caton-Deschamps index (
Cartilage Mapping
Regions of patellofemoral and tibiofemoral cartilage were segmented from the DESS scan for T1ρ cartilage mapping. Cartilage compartments representing the medial and lateral femoral condyle, medial and lateral tibial plateau, trochlear groove, and patella were automatically segmented with a custom, deep learning algorithm that combines conditional generative adversarial networks and convolutional neural networks to identify cartilage borders at the articular surface and interface with underlying bone. 27 Manual corrections were applied to the segmentation if automated segmentation failed.
Cartilage on the patella and within the trochlear groove were further divided into medial, lateral, and central regions ( Fig. 2 ). Landmarks were identified on the surfaces of the bones with an automated closed-contour mapping algorithm. For the patella, the most medial and lateral points and the most prominent point on the patellar ridge were identified within axial slices through the patella. The central region extended from the patellar ridge, one-third of the distance to the medial and lateral edges of the patella. The remaining cartilage on the facets were identified as the medial and lateral regions. A similar approach was applied to the trochlear groove, with points identified on axial slices at the medial and lateral edges of the cartilage and the deepest point of the trochlear groove. The landmarks were individually examined for each knee and manually corrected if necessary. A division between cartilage in the trochlear groove and on the femoral condyles was based on a projection of Blumensaat’s line, which traces the roof of the intercondylar notch on a sagittal view, through the cartilage on the femur.
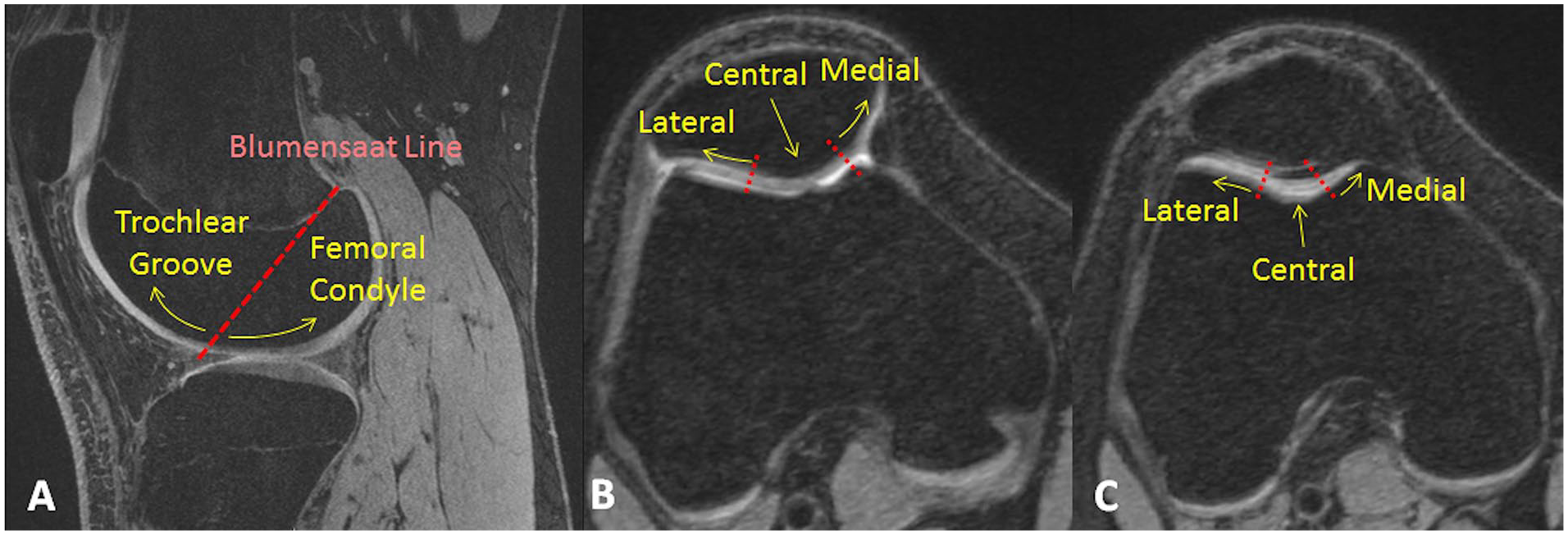
A sagittal view showing separation of cartilage on the femur into trochlear groove and femoral condyle regions based on a projection of Blumensaat’s line (
T1ρ relaxation times were mapped to the reconstructed cartilage surfaces ( Fig. 3 ).28,29 Images from the DESS scan were rigidly registered with the first echo of the T1ρ images, using piecewise rigid registration to account for variations in the positions of the bones. An intensity-based, multi-resolution pyramidal approach was used for non-rigid registration between the first echo and a template mask covering areas surrounding the patella, tibia, fibula, and femur to obtain a predefined mask for precision. The resulting transformation was applied to a rigid registration of the later echoes of the T1ρ images to the first echo. The T1ρ images were fit pixel by pixel based on a 2 parameter, monoexponential Levenberg-Marquardt algorithm relating image signal to exponential decay based on time of spin lock to T1ρ relaxation time. The average T1ρ relaxation time was calculated based on all voxels within each sub-region. Using the same techniques, the scan-rescan root mean square coefficient of variation for T1ρ values has been reported to be less than 5%.23,30
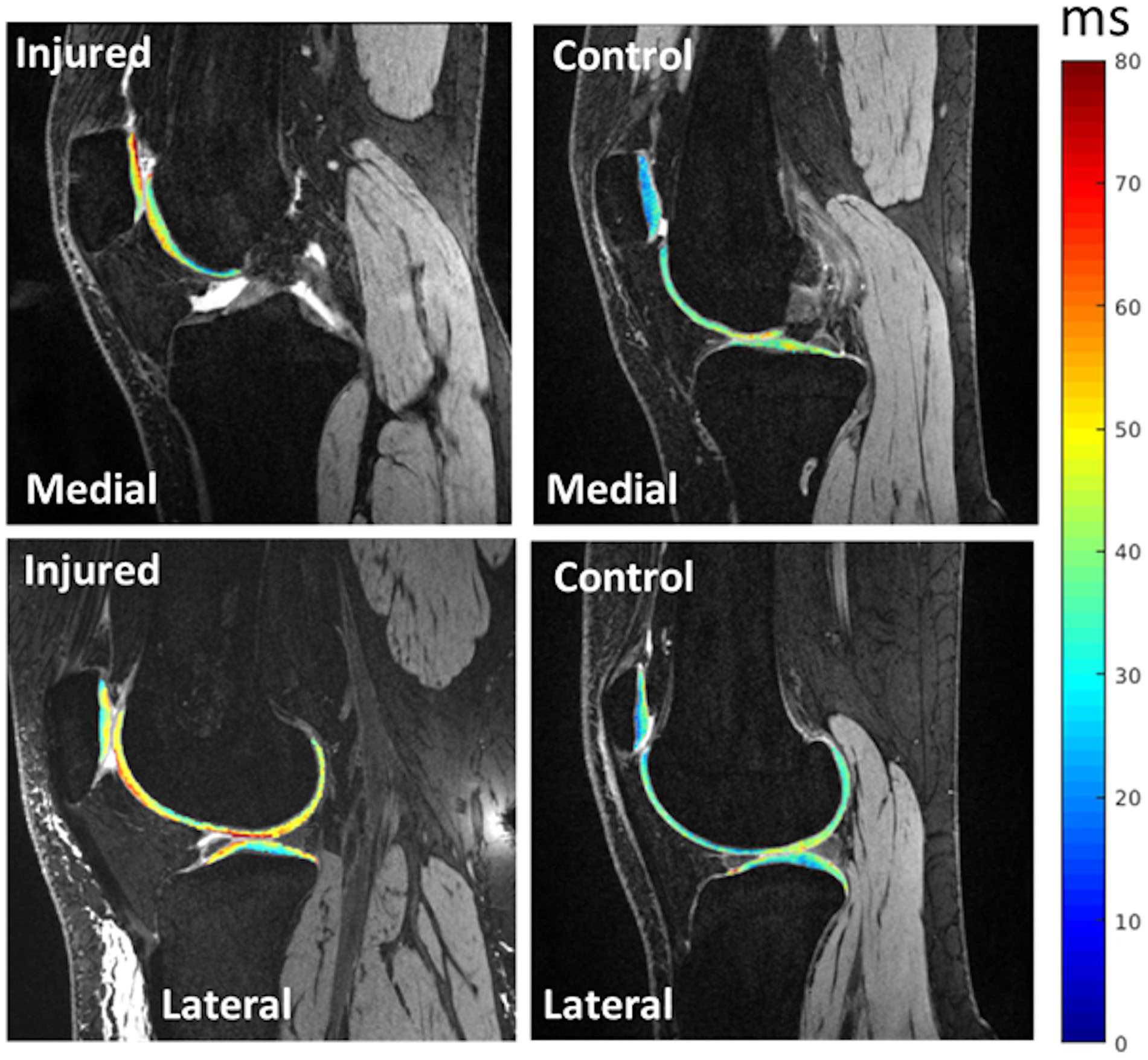
T1ρ relaxation times mapped to patellofemoral and tibiofemoral cartilage from a Dual Echo Steady State scan for injured and uninjured knees. The images give examples of elevated relaxation times for cartilage at the medial patella and lateral tibia for an injured knee.
Statistical Analysis
Statistical analysis focused on comparisons between injured and control knees for each region to characterize cartilage degradation that could lead to PTOA. Comparisons between injured and contralateral knees were also performed to focus on the influence of traumatic impact on cartilage degradation. Age and body mass index were compared between injured and control subjects with a Mann-Whitney U test, based on assessment of normality from Shapiro-Wilk tests (SPSS version 24, IBM). Distribution of sex and skeletal maturity was compared between injured and control subjects with a chi-square test. Anatomical parameters and T1ρ relaxation times were compared between the groups with generalized estimating equations accounting for unpaired comparisons between injured and control knees and paired comparisons between injured and contralateral knees. Due to a weak trend for higher BMI for injured subjects than controls, BMI was treated as a covariate in the generalized estimating equations for T1ρ relaxation times. The sample size was designed to achieve a power of 0.95 for an effect size of 1.3 and statistical significance set at P < 0.05 based on previous studies that compared injured knees to controls, but did not focus on the acute phase of injury.8,16
To characterize the influence of demographic parameters on T1ρ relaxation times, linear regressions were performed to relate age and BMI to T1ρ relaxation times in each region for the dislocation and control groups. Data were analyzed to confirm assumptions of normally distributed residuals based on a PP-plot and homoscedasticity based on a scatterplot of the residuals. Standardized β coefficients were quantified to determine the strength of correlations. Time from dislocation to imaging was included in the regressions for the dislocation group. T1ρ relaxation times were also compared between males and females and between skeletally mature and immature subjects for injured and control knees with t tests. The groups were checked for normality and for equality of variances with Levene tests.
Results
Anatomical parameters differed between injured knees and healthy controls, but demographic parameters were similar ( Table 2 ). The median time from initial injury to imaging was 48 days (range: 3-151). Lateral trochlear inclination was significantly smaller for the injured knees than the control and contralateral knees (P ≤ 0.01). Lateral trochlear inclination was less than 11° for 5 injured knees. TT-TG distance was significantly larger for the injured knees than the control and contralateral knees (P < 0.01). TT-TG distance exceeded 20 mm for 5 injured knees. Caton-Deschamps index did not vary significantly between the 3 groups (P = 0.17). Caton-Deschamps index was ≥ 1.2 for 11 injured knees. Distribution of sex, skeletal maturity, and age were similar for injured subjects and controls. The injured subjects also tended to have a higher BMI, although the difference did not reach statistical significance (P = 0.34). Due to the mean difference of 3.7 kg/m2, the tendency for higher BMI for the injured subjects was accounted for as a covariate in the generalized estimating equations comparing T1ρ relaxation times between injured knees and controls.
Demographic and Anatomical Characteristics for the Control, Dislocation, and Contralateral (Anatomical Only) Groups.
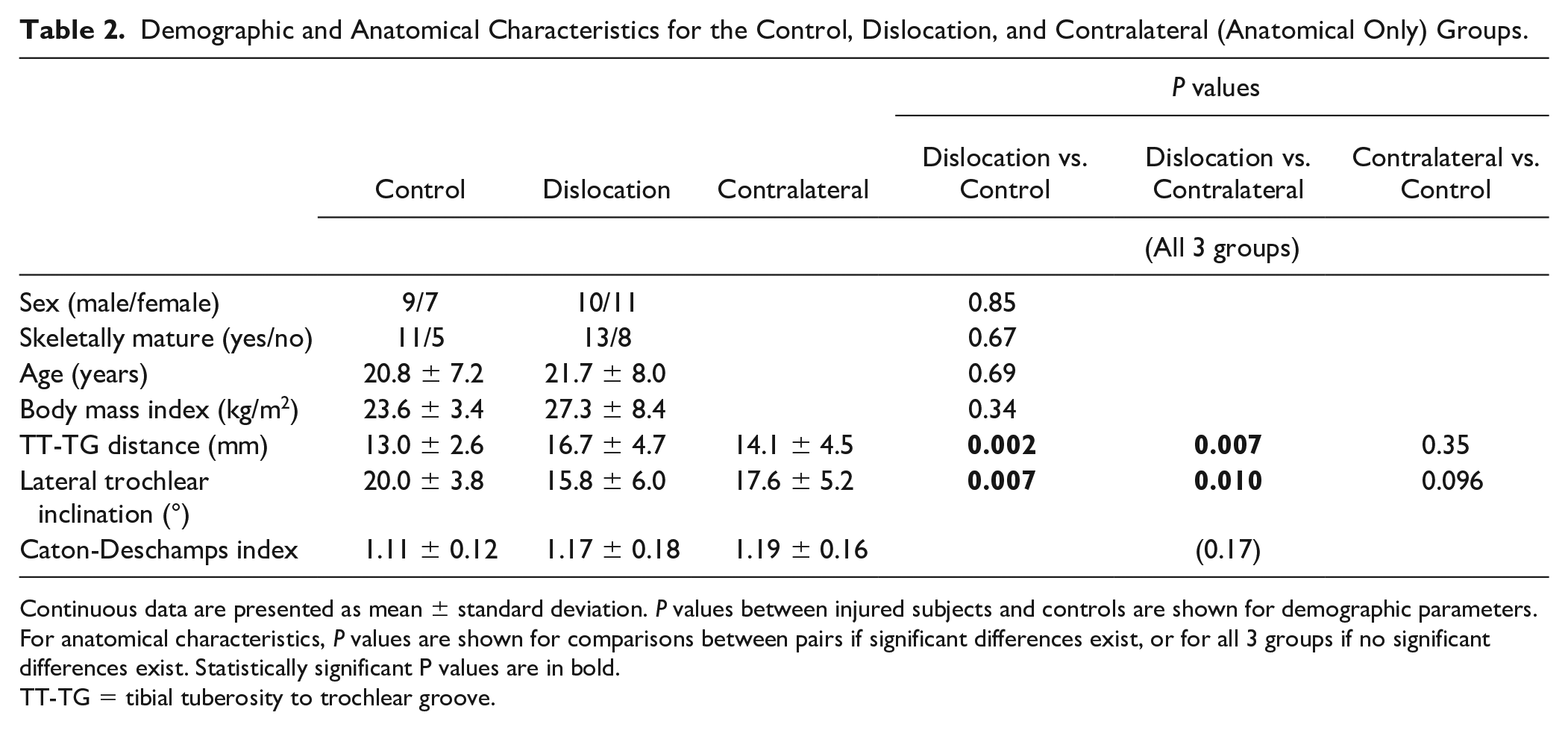
Continuous data are presented as mean ± standard deviation. P values between injured subjects and controls are shown for demographic parameters. For anatomical characteristics, P values are shown for comparisons between pairs if significant differences exist, or for all 3 groups if no significant differences exist. Statistically significant P values are in bold.
TT-TG = tibial tuberosity to trochlear groove.
T1ρ relaxation times were longer for the dislocation group than the control group at the medial patella and lateral tibia ( Table 3 ). At the medial patella, the mean relaxation time was 6.0 ms longer for the dislocation group than the control group (P = 0.012). The mean relaxation time was also longer for the dislocation group than the contralateral knees (difference = 4.5 ms, P = 0.006). At the lateral tibia, the difference between injured and control knees was 3.1 ms (P = 0.006). Relaxation times were not significantly longer for the injured knees than the contralateral knees at the lateral tibia (P = 0.78). T1ρ relaxation times tended to be longer for the dislocation group than control knees for other patellofemoral regions, but no other statistically significant differences were identified.
Mean (± Standard Deviation) T1ρ Relaxation Times (ms) for the 3 Groups.
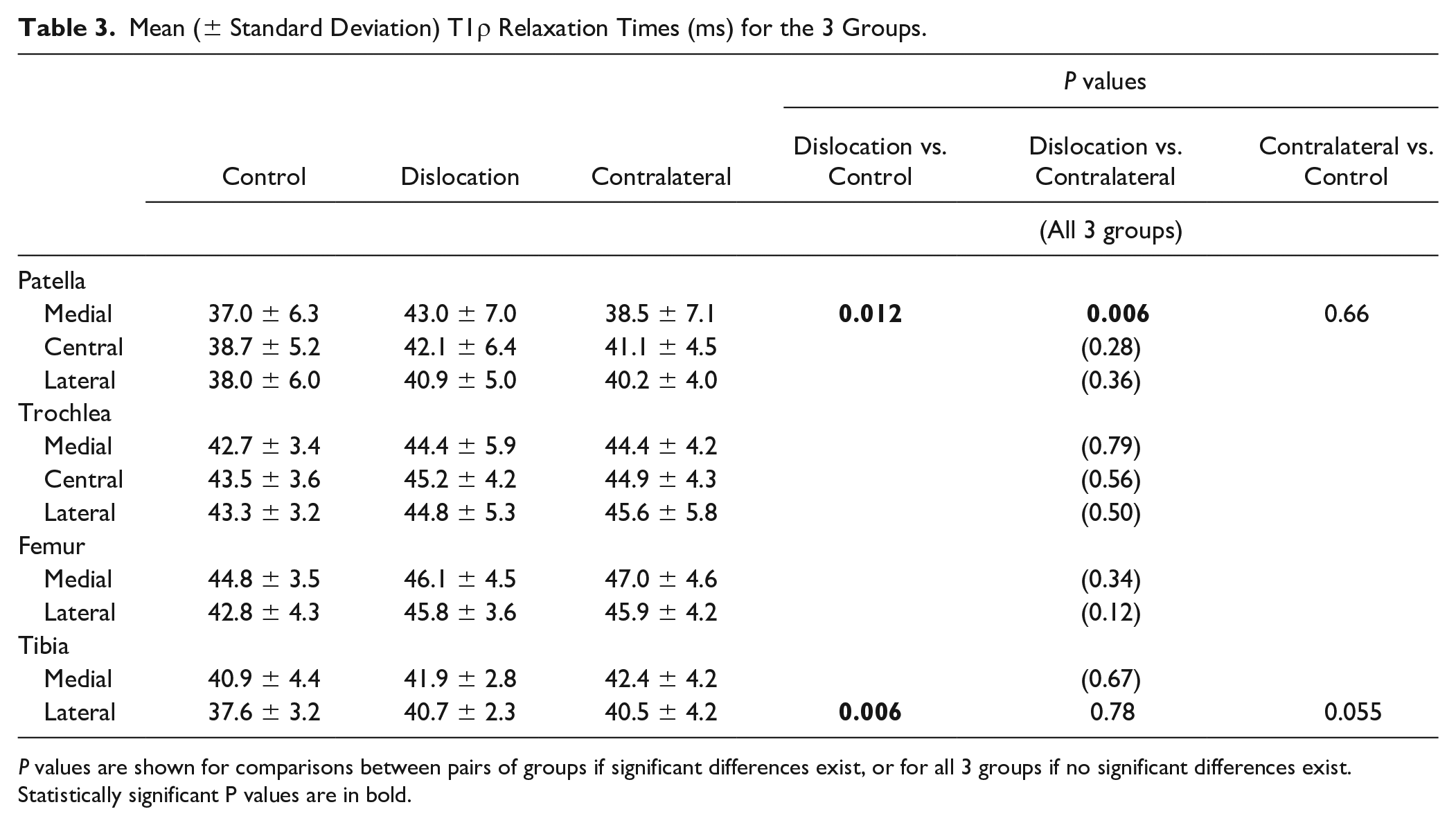
P values are shown for comparisons between pairs of groups if significant differences exist, or for all 3 groups if no significant differences exist. Statistically significant P values are in bold.
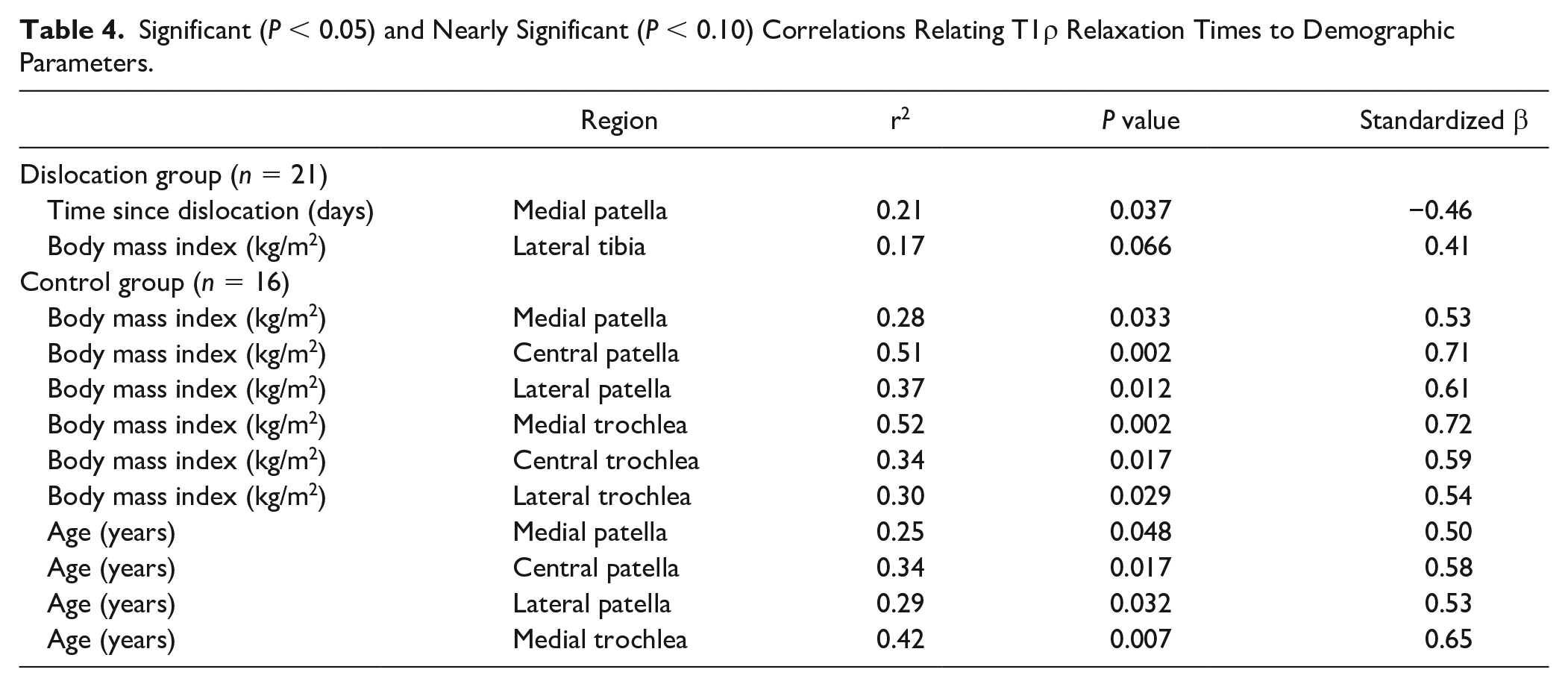
For the dislocation group, the only parameter correlated with T1ρ relaxation time was time from dislocation, while age and BMI were correlated with T1ρ relaxation time for the control group ( Table 4 ). For the dislocation group, T1ρ relaxation time at the medial patella decreased with time from injury (r2 = 0.21, P = 0.037). The trend was primarily driven by patients evaluated longer than 2 months after dislocation ( Fig. 4 ). No other statistically significant correlations were identified between T1ρ relaxation time and time from injury, age, or BMI for any region. For the control group, T1ρ relaxation time increased with BMI for all 6 patellofemoral regions (P < 0.04). T1ρ relaxation time also increased with age for all 3 regions on the patella and the medial trochlear groove (P < 0.05). The standardized β coefficients were larger for BMI than age for every region in which both were significantly correlated with T1ρ relaxation time. For the control group, no correlations between T1ρ relaxation time and demographic parameters were identified for any region of the tibiofemoral joint.
Significant (P < 0.05) and Nearly Significant (P < 0.10) Correlations Relating T1ρ Relaxation Times to Demographic Parameters.
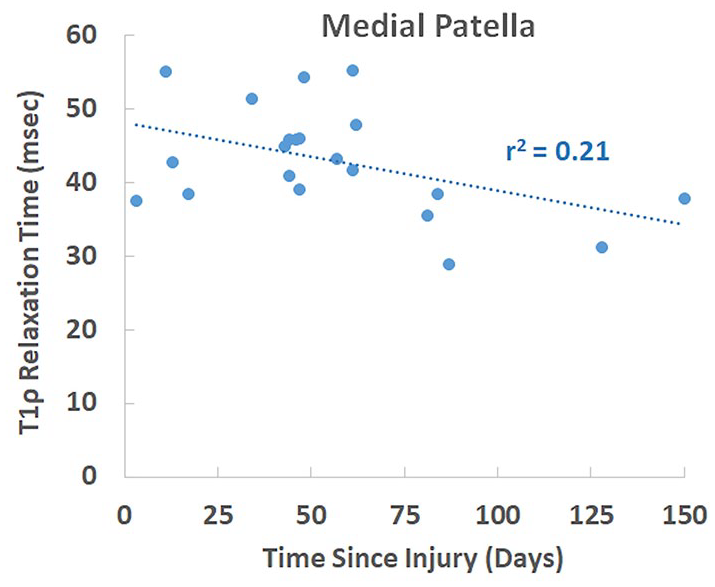
T1ρ relaxation time at the medial patella versus the number of days since dislocation. The decrease in T1ρ relaxation time with days since injury was statistically significant (P = 0.037).
Significant differences between injured and control knees related to sex and skeletal maturity were limited. The only significant difference between males and females was longer relaxation times for females than males at the lateral patella for the dislocation group (43.0 ± 4.2 vs. 38.7 ± 5.0 ms, P = 0.045). The only significant difference related to skeletal maturity was longer relaxation times for skeletally mature than immature subjects at the lateral patella for the control group (40.2 ± 4.9 vs. 33.3 ± 5.8 ms, P = 0.026).
Discussion
The primary result is that a first-time patellar dislocation is associated with elevated T1ρ relaxation times for cartilage on the medial patella and lateral tibia. The longer T1ρ relaxation times indicate loss of proteoglycan concentration within the cartilage that reduces cartilage integrity and could be an initiation point for progression to OA. Elevated T1ρ relaxation times at the medial patella are consistent with previous studies focused on T2 relaxation times for patients being treated for patellar instability.8,16,31 The previous studies did not focus on acute first-time patellar dislocations. The current results are also consistent with cartilage lesions on the medial facet of the patella for approximately 60% of patients following a first-time patellar dislocation.2-4 In addition to the patellofemoral joint, the current study provides the first analysis of cartilage composition in the tibiofemoral joint after patellar dislocation, to the best of the authors’ knowledge. Relaxation times at the lateral tibia were elevated following patellar dislocation. Previous studies have identified cartilage lesions on the lateral femoral condyle for approximately one-third of patients following patellar dislocation.3,5,6
Traumatic injury during a dislocation episode contributes to longer T1ρ relaxation times at the medial patella following a lateral patellar dislocation. The medial patella impacts against the lateral femoral condyle during a dislocation event, with bruises forming on each bone at the location of impact.2,4 Relaxation times at the medial patella were longer for injured knees than controls, indicating early cartilage degradation, and were also longer for injured than contralateral knees, indicating the traumatic impact was a primary contributor to the cartilage degradation. In contrast, one study evaluated patients with multiple dislocations at least 5 years after the initial dislocation and identified longer T2 relaxation times within superficial cartilage on the medial patella for uninjured contralateral knees than injured knees. 17 The decrease in T1ρ relaxation times at the medial patella beyond 2 months after dislocation indicates potential for cartilage to recover from the initial injury. Any recovery may be partial and short term. A study based on delayed gadolinium-enhanced MRI of cartilage (dGEMRIC) indicated proteoglycan concentration at the medial patella decreases with time for patients evaluated at least 6 months following a recurrent dislocation. 32 Furthermore, a previous study indicated early cartilage lesions are still evident an average of 8 years following patellar dislocation. 2
The lateral tibia is not considered a site of direct trauma during a patellar dislocation. At the lateral tibia, T1ρ relaxation times were longer for injured knees than healthy controls, but not longer than for injured knees than contralateral knees. Elevated T1ρ values at the lateral tibia did not decrease with time from injury. Direct trauma does not seem to be the primary factor influencing cartilage degradation at the lateral tibia.
Multiple factors other than direct trauma can contribute to cartilage degradation observed following a patellar dislocation. Pathologic anatomy contributes to patellar dislocations. Injured knees demonstrated a shallow trochlear groove and a lateral position of the tibial tuberosity in comparison to contralateral knees and the control group, based on TT-TG distance and lateral trochlear inclination, respectively. Both conditions are commonly associated with patellar dislocations24,33 and could contribute to re-dislocations 34 that are associated with an increased risk of patellofemoral OA. 9 Without weightbearing full limb anterior-posterior radiographs, valgus alignment that could contribute to both patellar dislocations 35 and degradation of lateral tibiofemoral cartilage 36 was not characterized. Pathologic anatomy and alignment could contribute to longer T1ρ relaxation times at the medial patella and lateral tibia. Inflammatory effects that contribute to cartilage degradation following anterior cruciate ligament (ACL) injury37,38 could also contribute to elevated T1ρ relaxation times. The current study lacks characterization of T1ρ relaxation times for knees with high levels of pathologic anatomy but without the direct impact and inflammatory effects associated with a dislocation episode to distinguish between these 3 factors. Further studies including a more comprehensive characterization of anatomy, analysis of inflammatory biomarkers, and evaluation of knees with pathologic anatomy prior to dislocation are warranted.
The current data provide a baseline measure of cartilage properties following patellar dislocation for comparison to longitudinal measures mapping the pathway to PTOA. Approximately 50% of patients progress to patellofemoral OA by 25 years after a first-time patellar dislocation. 9 Progressive cartilage degradation and OA are not isolated to the medial patella. Studies focused on patients being treated for multiple dislocations and general instability identified significant differences between injured knees and controls at the central8,16 and lateral 8 patella. Increased prevalence of cartilage lesions at the central and lateral patella, within the trochlear groove, and within the tibiofemoral joint have been noted at approximately 8 years following a first-time dislocation. 2 Progressive cartilage degradation is likely related to several factors in addition to the initial injury. Abnormal function related to pathologic anatomy and injury to the medial patellofemoral ligament during the initial dislocation could contribute to progressive cartilage degradation. Trochlear dysplasia, in particular, has been related to an elevated risk of OA. 9 Inflammatory effects could also contribute to progressive cartilage degradation.
The results suggest patellar dislocation alters the relationship between T1ρ values and demographic factors. For control subjects, T1ρ values for patellofemoral cartilage increased with age and BMI. The correlations were stronger for BMI than age (suggested by the larger standardized β coefficients, Table 4 ). These relationships were not identified for tibiofemoral cartilage. A previous study showed T1ρ relaxation times increase with BMI for cartilage on the tibia but not the femur. 39 The minimum age for the previous study was 22 years, as opposed to 13 for the current study. The authors are not aware of other previous publications correlating age and BMI with patellofemoral and tibiofemoral cartilage T1ρ relaxation times for healthy knees. For injured knees, neither age nor BMI was correlated with patellofemoral T1ρ relaxation times, likely due to the influence of traumatic injury on T1ρ values. Longer T1ρ relaxation times for females than males were identified at the lateral patella following dislocation. This difference was not observed for control subjects, suggesting potential for differing responses to injury between sexes. Trends between demographic factors and relaxation times seem to be injury specific. For patients treated surgically for ACL injury, one study reported T1ρ relaxation times for patellofemoral and tibiofemoral cartilage increased with age, but decreased with BMI 19 ; females also had higher T1ρ relaxation times than males. 19 For patients treated for other forms of knee pain or injury, not including patellar dislocation, patellar cartilage T2 relaxation times decreased with age and were greater in males than females. 20 Follow-up studies are warranted to longitudinally evaluate correlations between cartilage health and demographics, including potential differences based on sex, following patellar dislocation.
The increase in patellofemoral relaxation times with age for the control group but not for the injured subjects gives an indication that relatively short T1ρ relaxation times for the young controls contribute to the difference noted at the medial patella and trends noted for the other patellofemoral regions. The T1ρ relaxation times were also longer at the lateral patella for skeletally mature than immature controls. Without a significant correlation relating age to T1ρ relaxation times for the control subjects for the tibiofemoral regions, the difference at the lateral tibia does not seem to be driven by young subjects. Further studies with a larger sample size seem warranted to divide the subjects into young and old sub-groups.
The primary limitation of the study is the relatively small sample size for identifying elevated T1ρ relaxation times for regions other than the medial patella and lateral tibia. Effect sizes for comparisons between injured knees and controls at the central and lateral regions of patellar cartilage exceeded 0.5. Approximately 50 subjects would be needed per group to reach a power of 0.8 for comparisons between dislocation subjects and controls. Analysis of cartilage degradation is based only on T1ρ relaxation times, which are related to proteoglycan concentration. Additional analysis based on T2 relaxation times could also assess cartilage properties based on water and collagen content, along with organization of collagen fibrils. The current data provide a baseline assessment of post-injury cartilage degradation. Longitudinal evaluation is needed to characterize progression of cartilage degradation to PTOA and identify patients at greatest risk.
In conclusion, the current study indicates a first-time patellar dislocation is associated with early cartilage degradation within multiple regions of the knee when compared with asymptomatic healthy controls. Compositional changes to cartilage on the medial patella are related to the traumatic impact during a dislocation event. In the acute phase of the injury, potential exists for cartilage properties at the medial patella to improve with time. Traumatic impact has less influence on cartilage degradation at the lateral tibia. Additional studies including a more comprehensive characterization of anatomy, analysis of inflammatory biomarkers, and evaluation of knees with pathologic anatomy prior to dislocation are warranted to identify factors that contribute to cartilage degradation. The current baseline data can be considered a starting point to characterize the pathway from a first-time dislocation to progressive cartilage degradation and OA.
Footnotes
Authors’ Note
The study was performed at the Cleveland Clinic, Cleveland, OH.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Funding was provided by the Office of the Assistant Secretary of Defense for Health Affairs through the Peer Reviewed Medical Research Program Discovery Award under Award No. W81XWH-20-1-0040 and a grant from the Orthopaedic Research and Education Foundation (OREF) with funding made possible by the American Arthritis Society.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Carl S. Winalski has acted as a consultant for CartiHeal, Parexel, Janssen Research and Development, and Siemens Medical Solutions.
Ethical Approval
The study was approved by the Cleveland Clinic Institutional Review Board.
