Abstract
Objective. To compare demographics and cartilage lesion characteristics of patients enrolled in clinical trials investigating autologous chondrocyte implantation (ACI) in the ankle joint with those actually scheduled for matrix-assisted chondrocyte implantation (MACI) using database records. Design. Anonymized data from patients scheduled for MACI treatment in the ankle in Australia/Asia and Europe were obtained from the Genzyme/Sanofi database. Average age, defect size, and male-female ratio were analyzed and compared by country. A literature search was performed on PubMed and Google Scholar and clinical cohort studies and prospective comparative trials using ACI and related treatments in the ankle joint were identified. Weighted average age, weighted defect size, and male-female ratio were analyzed and compared with database data. Results. The 167 patients included from the databases from Europe and Australia had a mean age of 33.4 years (range 14-64 years) and a mean defect size of 2.27 cm2 (range 0.25-16 cm2). Male-female ratio was 4:3. Patients from European countries were significantly younger and had significantly larger defects compared with patients from Australia. From the literature search a total of 472 patients were included from 28 studies. The mean age was 32.2 years (range 15-62 years). Male-female ratio was 3:2. Weighted mean size was 1.94cm2 (range 0.3-16). There were no significant differences between previous studies and databases. Conclusion. No differences in sizes and age were found between patients enrolled in clinical trials and patients scheduled for MACI outside clinical trials. The sizes of treated defects followed the general recommendations. There were, however, significant differences between countries.
Introduction
Chondral and osteochondral lesions in the ankle joint represent a significant challenge for orthopedic surgeons. MACI (matrix-assisted chondrocyte implantation) is a commercially available 2-stage surgical treatment for chondral injuries with implantation of in vitro cultured chondrocytes on a 3-dimensional scaffold into the cartilage defect. The treatment represents the third generation of autologous chondrocyte implantation (ACI), which was first introduced for the treatment of cartilage injuries in the knee. 1 Since its introduction it has become increasingly popular with positive short- and long-term results.2-5 In the knee, ACI-based treatments are generally limited to larger chondral defects >3 to 4 cm2 due to the high expense and availability of less costly alternatives, although long-term outcomes tend to favor the cell-based treatment. In the ankle, defects larger than 1 cm2 are considered appropriate for treatment with ACI as these defects generally have a poor repair response with other treatment modalities such as marrow stimulation techniques. 6
The literature on ACI for treatment of cartilage defects in the ankle is sparse compared with the knee joint and no randomized controlled trials evaluating ACI in the ankle joint are available. 7 While prospective case series have been undertaken, there is a lack of reports on patient and lesion characteristics across large patient populations. In a previous study, we reported that patients enrolled in clinical trials with ACI of the knee had smaller defects compared with patients undergoing MACI in clinical practice outside the trial environment, 8 with potential implications for the generalizability of data from clinical trials.
The aim of this study was to compare demographics and lesion characteristics of patients enrolled in clinical trials on ACI of the ankle joint with the characteristics of patients receiving MACI in clinical practice. We hypothesized that patient and defect characteristics were similar between patients scheduled for MACI of the ankle compared to patients enrolled in clinical trials.
Methods
Database Review
The Genzyme/Sanofi clinical databases from Europe and Australia were used to obtain data on patients scheduled for MACI in the ankle between 2009 and 2013. The database included anonymized data from all patients scheduled for MACI treatment in that period. Demographic data and surgeon-assessed defect size at time of biopsy were available. The database was discontinued in the present form when the treatment and cell culture facilities were acquired by Aastrom Biosciences (now Vericel Corp.). Patient demographics (age, gender) and characteristics of the cartilage defects (size, number of defects) were evaluated. Age and defect size were reported at the time of biopsy, rather than at implantation. The database is used as an indicator of the characteristics of patients actually receiving ACI treatment as it includes a large number of patients.
Literature Review
A literature search on PubMed and Google Scholar was performed to identify studies on ACI in the ankle joint (on December 10, 2018). Patient demographics and lesion sizes were retrieved from each study to allow for comparison with the database.
Search words were “ACI,” “ACT,” “MACI,” autologous chondrocyte implantation,” “autologous chondrocyte transplantation,” “matrix-assisted chondrocyte implantation” combined with “ankle” or “talus.” Studies in English and German were included. Both studies on chondral and osteochondral lesions were included. The database did not distinguish between open and arthroscopic procedures and both studies on open and arthroscopic procedures were included.
Study design, mean patient age, mean defect size, number of defects, and male-female ratio in the included studies were noted and compared with the database. Studies with no information on average age and average defect size were excluded if it was not possible to extrapolate this information from the provided data. Weighted average age and lesion sizes were calculated using the number of enrolled patients in each study. Studies presenting follow-up data on previously published patient data were excluded. Novel applications, such as gel-based administration, were also excluded. Studies where it was not possible to determine if the patients had been enrolled in previous trials were included. The included studies were grouped by country and compared with the databases from the respective countries whenever possible.
Statistical Analysis
Bartlett’s test revealed equal variance between the European and Australian databases in terms of age and defect size. An unpaired t test was used to test the hypothesis of no difference between patients from Europe and Australia. Register data and data in studies (age and defect size) was investigated using t test with weighting of studies based on enrolled patients. 9 A significance level of P < 0.05 was used.
Results
Database Characteristics
A total of 167 patients were included in the Genzyme/Sanofi databases from Europe and Australia ( Table 1 ). Male-female ratio was 4:3. The mean age of patients was 33.4 years (range 14-64 years) and the mean size of the defects was 2.27 cm2 (range 0.25-16 cm2). Four patients had multifocal lesions and the remainder had single lesions.
Number of Patients, Average Age, and Defect Size Divided by Country.
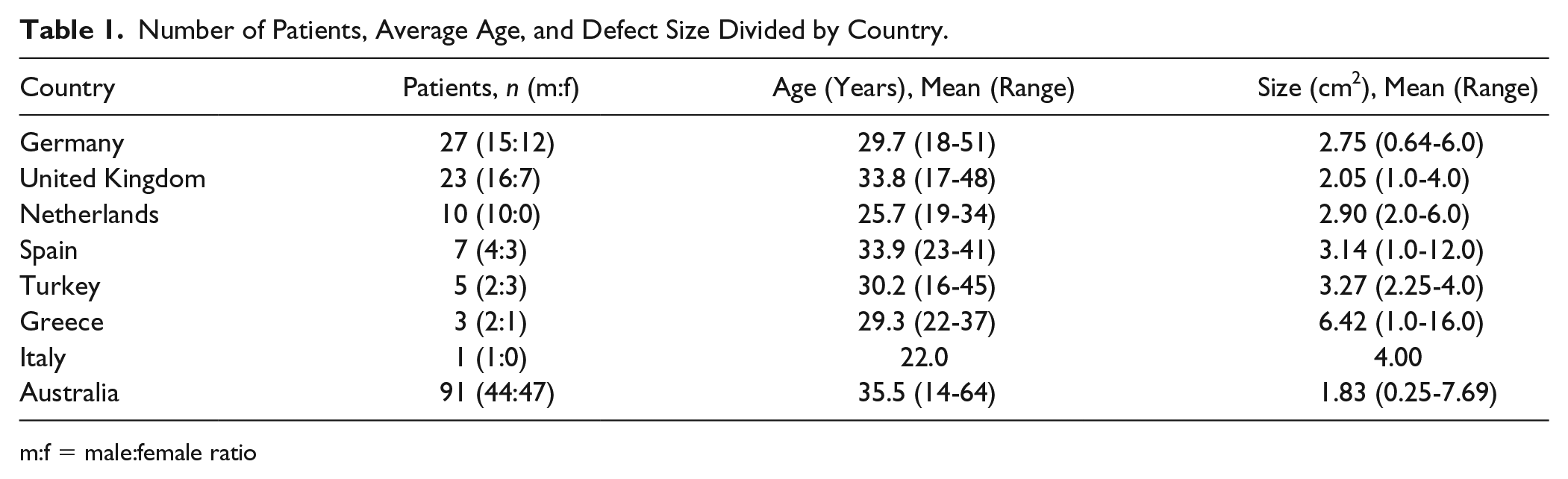
m:f = male:female ratio
Of the 167 patients, 76 were from the European database and originated from 7 different European countries; Germany, the United Kingdom, Netherlands, Spain, Turkey, Greece, and Italy. The mean age was 30.9 years (range 16-51 years). Male-female ratio was 3:2. Mean defect size was 2.79 cm2 (range 0.25-16.0 cm2).
Ninety-one patients were from the Australian database. The mean age of these patients was 35.5 years (range 14-64 years). Male-female ratio was 1:1. Mean defect size was 1.83 cm2 (range 0.25-7.69 cm2).
Literature Demographics Review
A total of 472 patients were included from 28 studies ( Table 2 ). The mean age was 32.2 years (range 15-62 years). Male-female ratio was 3:2. Weighted mean size was 1.94 cm2 (range 0.3-16 cm2).
Demographics of Patients Receiving Chondrocyte Implantation Enrolled in Clinical Trials.
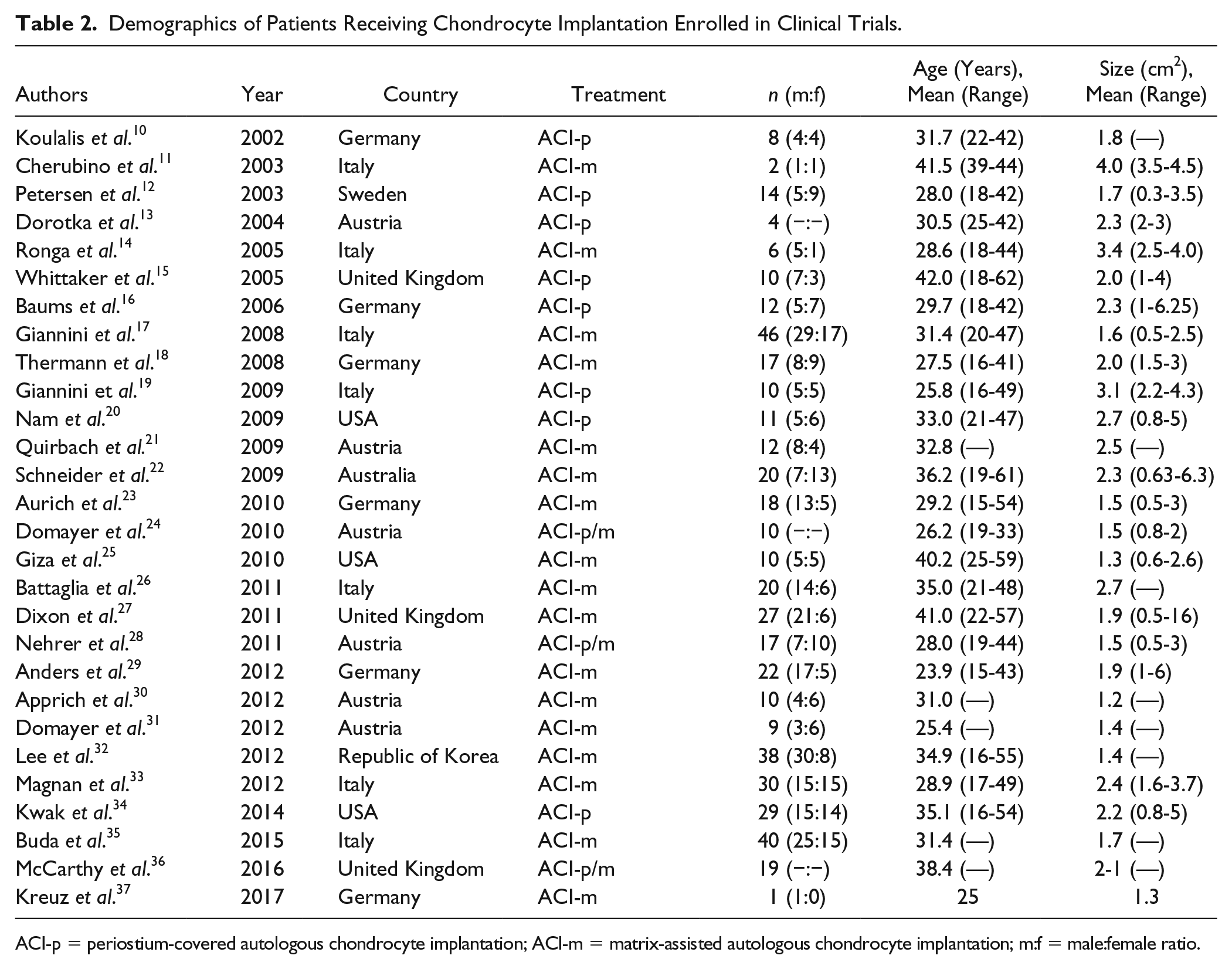
ACI-p = periostium-covered autologous chondrocyte implantation; ACI-m = matrix-assisted autologous chondrocyte implantation; m:f = male:female ratio.
One case report, 2 retrospective case series, 2 retrospective comparative studies, and 23 prospective case series were included.
Comparison of Databases and Studies
There were no significant differences between previous studies and databases in age or size of defect, although there was a trend toward larger defects in the European database compared with studies (P = 0.07). The databases and most studies did not allow for discrimination between chondral or osteochondral defects.
International Comparisons
Patients from European countries were significantly younger (P = 0.005) and had significantly larger defects compared with patients from Australia (P = 0.003).
Patients from Greece had the largest average defects, but this was affected by 1 of only 3 patients with a 16 cm2 defect. No significant differences were found when comparing with defect sizes of other countries. Patients from Turkey, although only 5 patients, had significantly larger defects compared with patients from the United Kingdom (3.3 vs. 2.1 cm2, P = 0.01). Patients from the Netherlands were significantly younger than patients from the United Kingdom and Spain (25.7 vs. 33.8 and 33.9 years, P = 0.003 and P = 0.02, respectively).
Average defect sizes in the database were compared with average defect sizes in studies for Germany and the United Kingdom, since these countries were the only countries with enough patients in both the database and studies to allow for a meaningful comparison. No differences were found.
Discussion
Our results demonstrate that patient and defects characteristics reported in clinical trials of ACI/MACI of the ankle are comparable to those treated clinically, outside the trial environment. Overall, most trials have inclusion/exclusion criteria stricter than those of common clinical indications, and there has been considerable debate regarding the generalizability of trial data to a “normal” clinical population. Highlighting this point, one study on ACI of the knee demonstrated that only 4.4% of patients seen at a cartilage center would have been candidates for inclusion in a typical clinical trial. 38
Our study compared 472 patients that were reported on in several publications with 167 patients from databases maintained by the manufacturer of the ACI/MACI implant. Surgeons report patient and defect characteristics to the manufacturer at the time of cartilage biopsy. While there were size and age differences between countries, these were not significantly different from trial data and fell within the general recommendations for clinical use of chondrocyte implantation in the ankle. Importantly, given that the trial and clinical use populations were not significantly different it can be assumed that outcomes of patients treated in clinical practice will match those reported in the literature. This is different from the situation in the knee, where larger defects were seen in clinical practice patients than in trial patients, raising concerns over the usefulness of trial data to discuss expectations and outcomes with patients in clinical practice. 8
The database demonstrated regional differences, with larger defects seen in Europe than in Australia. These differences are likely based on different indications and treatment alternatives established in the different countries or mandated by the respective insurance systems.
Our study has limitations since the databases do not contain information regarding the surgical approach (open or arthroscopic); however, since our study focuses on patient demographics rather than outcomes, this would not be expected to affect our results. Similarly, no information was provided on whether procedures were performed in isolation or in conjunction with bone graft for the treatment of osteochondral defects. The estimation of defect size noted in the database is surgeon-assessed with no standardization and the estimation was performed at the time of biopsy. This, however, is similar to the situation in many clinical studies. Standardization of defect size estimation is needed as the choice of treatment is based on this estimation and there may be huge variability between calculation methods and whether estimation is done based on magentic resonance imaging or arthroscopy. 39 This may influence the comparability among studies and also the application of study results in clinical practice.
In conclusion, our study utilized data from a database reflecting clinical use of ACI/MACI in the ankle, demonstrating no significant differences in comparison to data previously published on this topic. These findings support the generalizability of these published results to a wider clinical population.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
No ethical approval was required for the completion of this work.
