Abstract
Objective
We evaluated the ability of miR-379-5p to influence the proliferation of osteoarthritis chondrocytes and elucidated the regulatory mechanism of miR-379-5p in osteoarthritis.
Methods
Real time polymerase chain reaction (RT- PCR) was used to detect the expression of miR-379-5p and YBX1 in knee articular cartilages of human. Cell proliferation, inflammatory factors, extracellular matrix (ECM) degradation-associated proteins and proteins in PI3K/Akt pathway were assessed in rat primary chondrocytes treated with interleukin (IL)-1β or/and miR-379-5p mimics or miR-379-5p inhibitor via cell counting assay kit-8 (CCK-8), enzyme-linked immunosorbent assay (ELISA), immunofluorescence and Western blotting (WB). The target of miR-379-5p predicted by TargetScan and miRwalk software was verified by luciferase reporter assay. Safranin O-fast green staining, immunohistochemistry, and WB were performed to observe the effect of miR-379-5p agomir on development of osteoarthritis in rats.
Results
MiR-379-5p was down-regulated in human osteoarthritic tissues and negatively correlated with YBX1 expression. High level of miR-379-5p in chondrocytes with IL-1β stimulated increased cell viability, the expression of proliferation-related protein and extracellular matrix (ECM)-related proteins collagen II and aggrecan. However, the expression of inflammatory factors and ECM-related proteins matrix metalloproteinases (MMP-1) and MMP-13 was decreased. Luciferase reporting assay verified the targeting relationship between miR-379-5p and YBX1. This function of miR-379-5p was exerted through PI3K/Akt pathway and could be blocked by the PI3K/Akt pathway inhibitor LY294002. MiR-379-5p agomir promoted the articular chondrocytes proliferation and alleviated cartilage degradation in vivo.
Conclusion
Our findings reveal that miR-379-5p can promote the articular chondrocytes proliferation in osteoarthritis (OA) by interacting with YBX1 and regulating PI3K/Akt pathway. Restoring miR-379-5p might be a future therapeutic strategy for OA.
Introduction
Osteoarthritis (OA) is one of the most common joint diseases with degenerative changes in articular cartilage that involve the entire joint tissue. It also involves focal cartilage loss, bone hyperplasia (osteophytes and subchondral sclerosis), thickening of the synovial capsule, and structural changes in the ligaments and surrounding muscles around the joint. 1 OA increases with longevity and has become one of the global clinical problems. 2 The pathophysiology of OA remains unclear. However, the current view is that mechanical, inflammatory, and metabolic factors are involved in the complex pathogenesis leading to joint destruction and failure. 3
MicroRNAs (miRNAs) are a class of non-coding single-stranded RNA molecules encoded by endogenous genes with a length of about 22 nucleotides. They can regulate gene expression at the transcriptional and post-transcriptional levels by directly binding to specific sequences in target mRNAs. 4 MiRs have been shown to be regulators of hundreds of genes involved in chondrogenesis, internal environmental stability, and OA pathology. 5 Mechanical loading is very important for the regulation of cartilage metabolism and affects miRNAs expression. 6 Some miRNAs have been shown to regulate chondrocyte hypertrophy, proteolytic enzyme synthesis, and OA-related signaling pathways. 7 In addition, pathological phenomena generally associated with cartilage degeneration and OA, including inflammation, obesity, apoptosis, and autophagy defects, are also associated with miRNAs specific activity. 8
Recent research has found that miR-379-5p was lowly expressed in cartilage tissues of patients with knee OA through deep sequencing of microRNAs. 9 MiR-379-5p is located on chromosome 14q32.31, and it has been found to be lowly expressed in breast cancer and hepatocellular carcinoma.10,11 High level of miR-379-5p can inhibit the migration, invasion, and epithelial-mesenchymal transition of hepatocellular carcinoma cells. 12 Another new study showed that A-to-I–edited miR-379-5p inhibited cell proliferation and promoted apoptosis of breast cancer, ovarian cancer, renal cancer, and lung cancer, compared to wild-type miRNA. 13 In human hepatic cells, miR-379-5p regulation of gasdermin D suppressed arsenite-induced activation of hepatic stellate cells and fibrosis. 14 Although the high level of miR-379-5p has a role in promoting the proliferation of chondrocytes, 15 the possible pathological mechanism between miR-379-5p and OA is still unclear.
We screened out a total of 10 target genes (UBE2E3, EIF4G2, ZBTB26, SLC20AL, LRRC8C, ELMOD2, YBX1, ADAMTS15, HSPA5, TNRC6B) for miR-379-5p through TargetScan 7.2 (http://www.targetscan.org/vert_72/) and miRDB (http://mirdb.org/) online tools and all 10 target genes were conserved among human beings, mice and rats. And there was an evidence showed that YBX1 was indeed identified and validated as a direct target of miR-379-5p. 16 Then, we found that Y-box binding protein 1 (YBX1) is a DNA- and RNA-binding protein that contains a highly conserved cold shock domain and is involved in many different cellular processes, such as cell proliferation and development. 17 The expression of YBX1 also can affect the downstream phosphatidlinositol-3-kinase-protein kinase B (PI3K/Akt) signaling pathway and activate it. 18 PI3K/Akt mediated signaling pathway is one of the most critical pathways in the regulation of cell survival, proliferation, differentiation and apoptosis. 19 PI3K/Akt was closely related to the occurrence and development of inflammation. 20 It was a signaling pathway that was significantly activated during the pathogenesis of OA.21,22 Inhibition of this pathway can significantly alleviate bone damage caused by OA. 23 Therefore, we hypothesize that YBX1 may be involved in the proliferation process of chondrocytes in OA. The purpose of this study was to investigate whether miR-379-5p affects the occurrence and development of OA by targeting YBX1 and thereby inhibiting the PI3K/Akt signaling pathway and promoting chondrocyte proliferation.
Materials and Methods
Cartilage Samples
OA cartilage samples were collected from 32 OA patients (age 64.3 [6.2] years) underwent total knee replacement surgery in the Department of Orthopedics, Henan Provincial People’s Hospital, and another 14 normal cartilage samples (age 56.8 [4.4] years) were collected from healthy patient underwent amputation in the Department of Orthopedics, Henan Provincial People’s Hospital. These samples were used to detect the level of miR-379-5p and YBX1, and isolate the chondrocytes. The study has been approved by the ethical committee of the Henan Provincial People’s Hospital, People’s Hospital of Zhengzhou University, School of Clinical Medicine, Henan University. All participants provided their written informed consents.
Cell Culture
We took cartilage from the knee joint of the sacrificed rats under aseptic conditions and placed it in a sterile plate. The cartilage was cut into 1 mm3 pieces and placed in a conical flask. We added 0.2% type II collagenase and shake for 30 min. Then we added 0.25% trypsin and digest for 20 min. The supernatant was filtered through a 200-mesh screen and centrifuged. After supernatant was discarded, the cells were re-suspended in Dulbecco’s modified Eagle medium (DMEM) containing 100 U/mL penicillin, 100 mg/L streptomycin, and 10% fetal bovine serum. The cells were collected by centrifugation and cultured in DMEM medium. The cells were subcultured when the bottom of the flask was filled with cells. When the cells were passed on to the third generation, they were used for subsequent experiments.
Cell Treatment
We divided the isolated and cultured rat chondrocytes into the following groups: NC (negative control) group, BC (blank control, without any transfection) group, interleukin (IL)-1β (adding IL-1β and adjusting the final concentration to 10 ng/ mL) group, IL-1β+NC mimics (adding IL-1β and transfecting NC mimics) group, IL-1β+NC inhibitor (adding IL-1β and transfecting NC inhibitor) group, IL-1β+miR-379-5p mimics (adding IL-1β and transfecting miR-379-5P mimics) group, IL-1β+miR-379-5p inhibitor (adding IL-1β and transfecting miR-379-5P inhibitor) group, YBX1 (4 µg) group, sh-YBX1 (4 µg) group, IL-1β+miR-379-5p mimics+LY294002 (with IL-1β, miR-379-5p mimics and LY294002) group, IL-1β+miR-379-5p inhibitor+LY294002 (with IL-1β, miR-379-5p inhibitor and LY294002) group. Before transfection, rat chondrocytes were seeded into 12-well plates with 1 × 105 per well to culture 24 to 48 h. Chondrocytes of 3 to 5 generations were selected and transfected into chondrocytes with LipofectamineTM 2000 (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions, and then the corresponding indexes were detected.
Luciferase Reporter Assay
The target of miR-379-5p was predicted using TargetScan and miRwalk software, and the 3’UTR of YBX1 mRNA may be complementarily paired with the seed sequences of miR-379-5p. The luciferase reporter assay was used to determine that miR-379-5p directly targets to the 3’-UTR of YBX1 mRNA. The YBX1 3’UTR including miR-379-5p binding sites (YBX1 3’-UTR-WT) and according mutations (YBX1 3’-UTR-MT) were cloned into a pmirGLO vector (Promega, Madison, WI, USA), respectively, between the PmeI and XbaI restriction sites. Then NC mimics (20 nM) or miR-379-5p mimics (20 nM) or NC inhibitor (20 nM) or miR-379-5p inhibitor (20 nM), and 0.5 μg YBX1 3’-UTR-WT (20 nM) or 0.5 μg YBX1 3’-UTR-MT (20 nM) were cotransfected into chondrocytes using lipofectamine 3000 (Invitrogen, Carlsbad, CA, USA). The luciferase activity was measured using the Dual-Glo® Luciferase Assay System (promega, Madison, WI, USA) and a MicroLumatPlus LB96V luminometer (Berthold, Hamburg, Germany) at 24 h after cotransfection. The miR-379-5p mimics sequence (5’-3’) was GCGCUGGUAGACUAUGGAAUU; The NC mimics sequence (5’-3’) was UUGUACUACACAAAAGUACUG.
The miR-379-5p inhibitor sequence (5’-3’) was UUCCAUAGUCUACCAGCGCUU. The NC inhibitor sequence (5’-3’) was CAGUACUUUUGUGUAGUACAA.
OA Modeling
Twelve-week-old Sprague-Dawley rats were purchased from Shanghai SLAC Laboratory Animal CO. LTD. All rats were divided into 4 groups: sham, OA, OA+NC and OA+ agomir, 6 rats in each group. All rats were anesthetized by intraperitoneal injection of 3% pentobarbital sodium (30 mg/kg) (Tocris, Minneapolis, MN, USA) before surgery. After anesthesia, the articular cavity was exposed through an anterior incision on the medial side of the left knee. The anterior and posterior cruciate ligaments were transected and the medial meniscus was resected. The bleeding was stopped and the surgical wound was sealed layer by layer. Rats in sham group underwent sham surgery only to expose the surface of the right knee cartilage following by suture. Rats in the other 3 groups underwent anterior cruciate ligament transection (ACLT) to induce arthritis before suture. Rats in OA+NC group were injected miR-379-5p NC with a dose of 50 nmol (100 μL saline) into the joint cavity 6 h before ACLT and per week in 12 weeks after surgery. In the OA+agomir group, miR-379-5p agomir was injected in the same way as OA+NC group. We give it by local intra-articular injection after mixed agomir with LipofectamineTM2000 (Invitrogen, Carlsbad, CA, USA). Six rats in each group were sacrificed every 4 weeks after surgery, and knee articular cartilage were isolated to detect the levels of miR-379-5p and YBX1 mRNA using real time polymerase chain reaction (PCR) (RT-PCR). Knee articular cartilage samples (with bone) from rats 12 weeks after surgery were used for safranin O-fast green staining. Then, cartilage sample was carefully separated from the bone with a No.10 anatomical blade under aseptic conditions. The whole piece of tissue was fixed with large tweezers during separation. Sterilized phosphate buffered saline (PBS) was added to the surface to keep the cartilage moist. Finally, the isolated cartilage was placed in a culture dish containing PBS for subsequent experiments including RT-PCR, immunohistochemistry (IHC), and western blotting. The severity of articular cartilage degeneration was graded using the Osteoarthritis Research Society International (OARSI) scoring system by blinded observers. 24 All animal studies were approved by the Ethics Committee of Zhengzhou University.
Real Time PCR
Once the samples were isolated, they were stored in liquid nitrogen tanks unless we used them right away. Before adding Trizol, we take tissue samples from liquid nitrogen tanks and grind them with liquid nitrogen. Then, total RNAs were isolated from cartilage samples and chondrocytes using the TRIzol reagent (Invitrogen, Carlsbad, CA, USA) and were reversed transcribed using PrimeScript™ RT reagent Kit with gDNA Eraser (Perfect Real Time) (Takara, Kyoto, Japan). cDNA synthesis was performed with 1 μg of total RNA utilizing oligo dt-primer with a cDNA Synthesis Kit (Thermo Scientific, Waltham, MA, USA). Real time PCR was performed using TB Green™ Premix Ex Taq™ (Tli RNaseH Plus) (Takara, Kyoto, Japan) on the qTOWER3G real time PCR system (analytikjena, Jena, Germany). Relative gene expression was calculated using ΔΔCt method and was normalized to U6 or GAPDH. 25 The specific primer sequences (5’→3’) were as follows: miR-379-5p-F: GCGCTGGTAGACTATGGAA; miR-379-5p-R: GTGCAGGGTCCGAGGT, U6-F: CTCGCTTCGGCAGCACATATACT; U6-R: ACGCTTCACGAATTTGCGTGTCYBX1, YBX1-F: GGGTGCAGGAGAACAAGGTA; YBX1-R: TCTTCATTGCCGTCCTCTCT, PI3K-F: AGCATGGTCAGCTTTCTTCTT; PI3K-R: GAATGGAAGACGGGAGATTCA, AKT-F: CCAGGATCCATGGGTAGGA; AKT-R: GCAGCCCCTTTGACTTCTT, GAPDH-F: CCATGTTCGTCATGGGTGTGAACCA; GAPDH-R: GCCAGTAGAGGCAGGGATGATGTTCPI3K.
Western Blotting
Total protein was isolated from chondrocytes and rat cartilage samples using Radio-Immunoprecipitation Assay (RIPA) lysis (Beyotime, Shanghai, China) buffer and was qualitied with bicinchoninic acid (BCA) Assay Kit (Thermo Scientific, Waltham, MA, USA). Protein (30 μg per sample) was separated by SDS-PAGE on a 10% gel and transferred to a polyvinylidene difluoride membrane (Millipore, Boston, MA, USA). The protein-carrying membranes were blocked in 5% nonfat dry milk, and then incubated overnight at 4°C with primary antibodies against ki-67 (ab16667, 1:100), proliferating cell nuclear antigen (PCNA) (ab92552, 1:2,000), IL-6 (ab9324, 1:3000), tumor necrosis factor-α (TNF-α) (ab66579, 1:1,000), IL-8 (ab154390, 1:1000), IL-17 (ab150719, 1:1,000), MMP1 (10371-2-AP, 1:1,000), MMP13 (ab39012, 1:30,00), YBX1 (ab50575, 1:2,000), PI3K (ab86714, 1:10,000), p-PI3K (ab86714, 1:10,000), AKT (ab8805, 1:10,000), p-AKT (ab8805, 1:1,000), collagen II (ab34712, 1:2000), aggrecan (sc-166951, 1: 500) and GAPDH (ab181602, 1:10,000). The membranes were incubated with Goat Anti-Rabbit IgG H&L (HRP) preadsorbed (ab97080, 1:5,000) or Rabbit Anti-Mouse IgG H&L (HRP) preadsorbed (ab6728, 1:2,000) for 1 h at room temperature and protein was detected using an enhanced chemiluminescence kit (Thermo Scientific, Waltham, MA, USA). Antibodies against MMP1 and aggrecan were purchased from Applygen Technologies (Beijing, China) and Santa Cruz (CA, USA), respectively, and the other antibodies were purchased from Abcam (Cambridge, UK).
Cell Counting Assay Kit-8 (CCK-8) Assay
Cultured chondrocytes were seeded into a 96-well plate with 1 × 104 cells in each well (150 μL). The corresponding IL-1 β, NC mimics, miR-mimics, NC inhibitor and miR-inhibitor were added to the well according to the previous group. Then, 20 μL of 5% CCK-8 solution (PA137267, Pierce, Rockford, IL, USA) was added to each well. The plate was incubated for additional 2.5 h before measuring the absorbance at 450 nm wavelength using a microplate reader (SPECTROstar Nano; BMG Labtech, Ortenberg, Germany).
Immunohistochemistry (IHC) and Safranin O-Fast Green Staining
Cartilage samples were fixed 4 h with formalin and decalcified with formic acid solution followed by embedding with parrffin and cut into 4 μm slices. For IHC, samples were undergoing a heat mediated antigen retrieval step using citrate buffer and then blocked with 1% bovine serum albumin (BSA) for 10 min at 21°C followed by incubation with the primary antibody, anti-ki-67 antibody (ab16667, 1:200) and anti-PCNA antibody (ab92552, 1:300), for 30 min. An anti-Rabbit IgG H&L (HRP) preadsorbed (ab97080) was used as secondary antibody at a 1/1000 dilution, and subsequently the sample was counter-stained with hematoxylin. For safranin O-fast green staining, samples were stained with weigert’s iron hematoxylin and safranin O solution according to the instruction of Safranine O Cartilage Stain solution (Solarbio, Beijing, China).
Enzyme Linked Immunosorbent Assay (ELISA)
Centrifuge cell culture media at 2,000× g for 10 min to remove debris. Collect supernatants to detect the levels of IL-6, IL-8, IL-17, TNF-α, MMP-1, MMP-13, and aggrecan according to the instructions of Human IL-6 SimpleStep ELISA® Kit (No: ab178013, Sensitivity: 1.6 pg/ml, Range: 7.8 pg/ml-500 pg/ml), Human IL-8 SimpleStep ELISA® Kit (No: ab214030, Sensitivity: 1.8 pg/ml, Range: 3.91 pg/ml-250 pg/ml), IL-17 Human ELISA Kit (No: ab119535, Sensitivity: 0.5 pg/ml, Range: 1.6 pg/ml-100 pg/ml), Human MMP1 ELISA Kit (No: ab100603, Sensitivity: < 8 pg/ml, Range: 24.69 pg/ml-18000 pg/ml), Human MMP13 ELISA Kit (No: ab221839, Sensitivity: 5.7 pg/ml, Range: 39 pg/ml-2500 pg/ml), and Human Aggrecan ELISA Kit (No: ab213754, Sensitivity: 50 pg/ml, Range: 1.56 ng/ml-100 ng/ml). The level of collagen II in supernatant was detected using sandwich ELISA method with Anti-Collagen II antibody (ab34712, at a dilution of 1: 10000).
Immunofluorescence (IF)
Chondrocytes were fixed with 100% methanol for 5 min, if necessary, permeabilized with 0.1% Triton X-100 for 5 min, and then blocked with 1% BSA in 0.1% PBS-Tween for 1 h. Then cells were incubated with primary antibodies against ki-67 (ab16667, 1:250), PCNA (ab92552, 1:200), IL-6 (ab9324, 1:1,000), TNF-α (ab66579, 1:1,000), MMP13 (ab39012, 1:150) and collagen II (ab34712, 1:200) overnight at 4°C. Finally, cells were incubated with Goat Anti-Rabbit IgG H&L (Alexa Fluor® 488) preadsorbed (ab150081, 1:1,000) or Alexa Fluor® 488 goat anti-rabbit (ab150077, 1:1,000) 1 h at room temperature followed by counter-staining with 4′,6-diamidino-2-phenylindole (DAPI).
Statistical Analysis
All data were expressed as the mean ± standard deviation (SD). The relationship between the levels of miR-379-5p and YBX1 mRNA was explored with Pearson correlation analysis. Comparison between different groups was performed using student’s t test or 2-way analysis of variance (ANOVA) with Bonferroni posttests, and difference with P < 0.05 was considered statistically significant. All analyses were performed using GraphPad Prism 6 (GraphPad Software Inc., La Jolla, CA, USA).
Results
The Expression Levels of miR-379-5p and YBX1 in OA Cartilage Samples
The results of real time PCR showed that miR-379-5p was down-regulated in OA cartilage samples (P < 0.001) ( Fig. 1A ) while the level of YBX1 mRNA was up-regulated in OA cartilage samples (P < 0.001) ( Fig. 1B ) compared with those in normal cartilage samples. There was a negative relationship between miR-379-5p and YBX1 mRNA in OA cartilage samples (R2 = 0.1974, P < 0.05) ( Fig. 1C ). Further IHC analysis also showed that YBX1 protein level was significantly increased in OA cartilage samples (P < 0.01) ( Fig. 1D ).
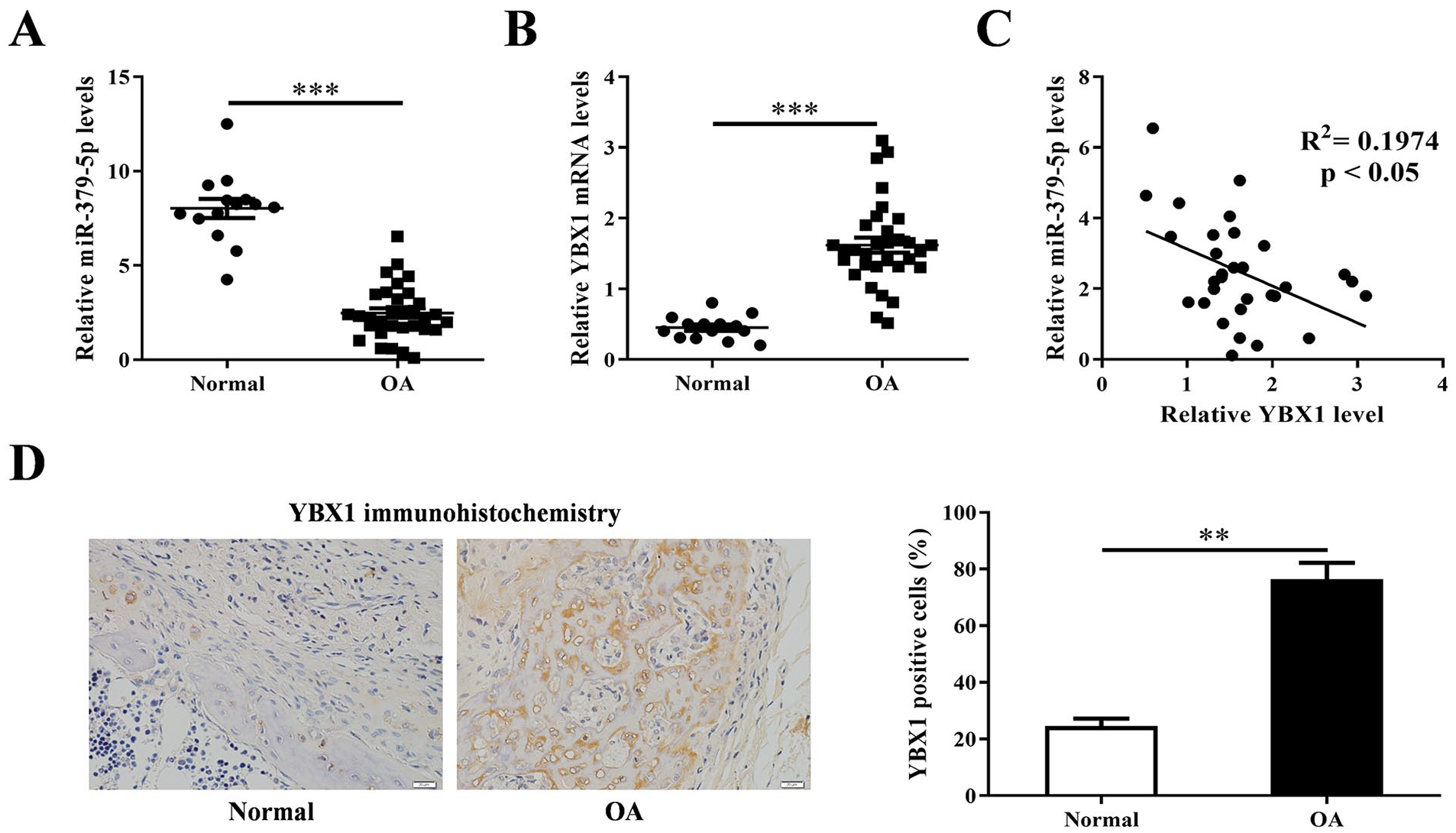
Expression levels of miR-379-5p and YBX1 in cartilage tissues from OA and normal patients. (
The Effect of miR-379-5p on IL-1β-Mediated Inhibition of Chondrocytes Proliferation
Real time PCR and CCK-8 methods were used to detect the effects of miR-379-5p mimics/inhibitor and IL-1β on intracellular miR-379-5p expression level and cell proliferation. The results showed that IL-1β significantly decreased miR-379-5p level in chondrocytes (P < 0.001), and this decrease would be rescued by miR-379-5p mimics (P < 0.001) rather aggravated by miR-379-5p inhibitor (P < 0.05) ( Fig. 2A ). At the same time, IL-1β weakened the cell viability of chondrocytes, which would be rescued by miR-379-5p mimics (P < 0.01) ( Fig. 2B ). The results of IF and western blotting showed that IL-1β down-regulated the level of ki-67 and PCNA proteins in chondrocytes, which were increased in miR-379-5p mimics + IL-1β group rather decreased in miR-379-5p inhibitor + IL-1β group ( Figs. 2C and 3A and B ).
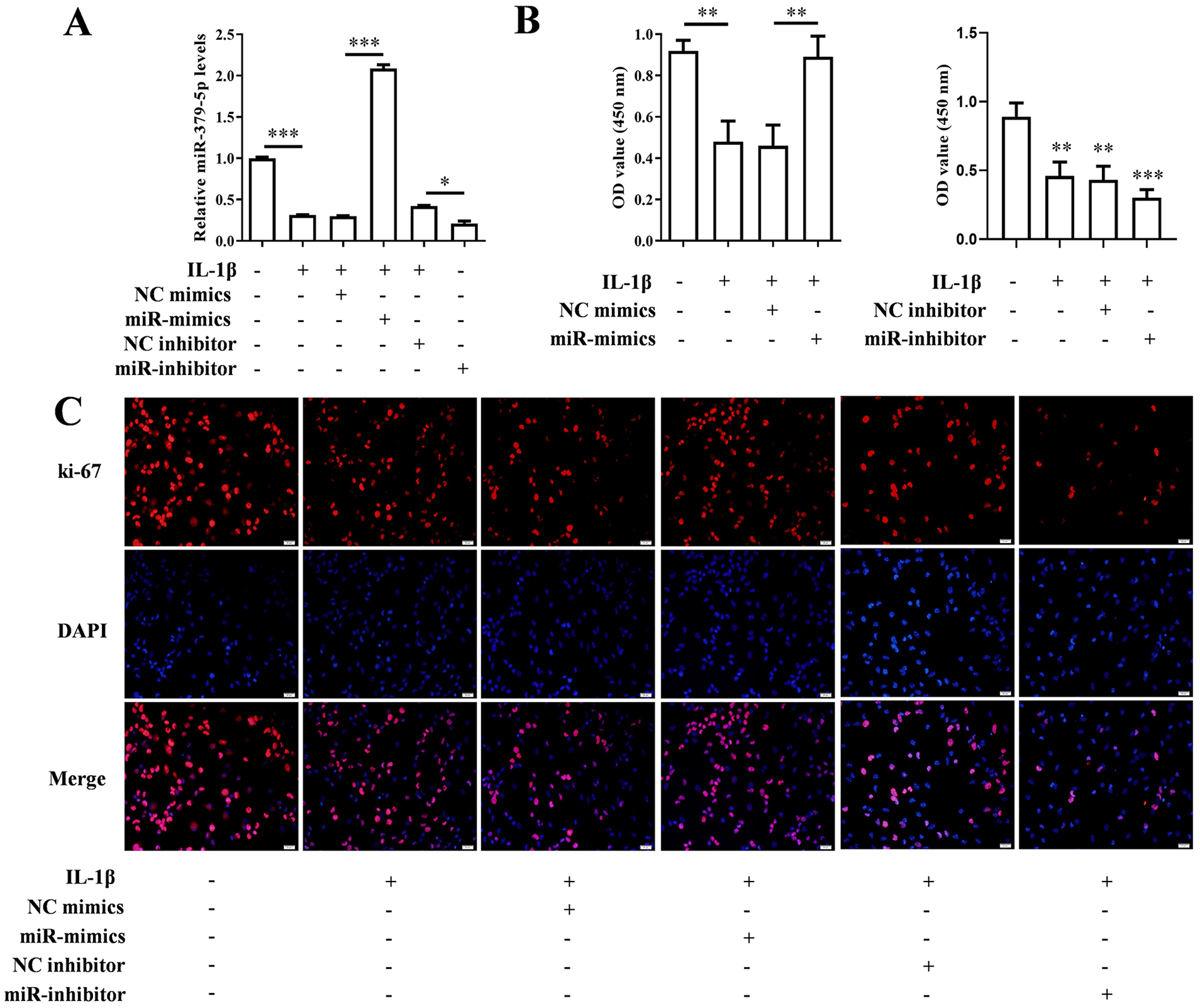
The expression of miR-379-5p increased or decreased, and the changes of cell activity and Ki-67. (
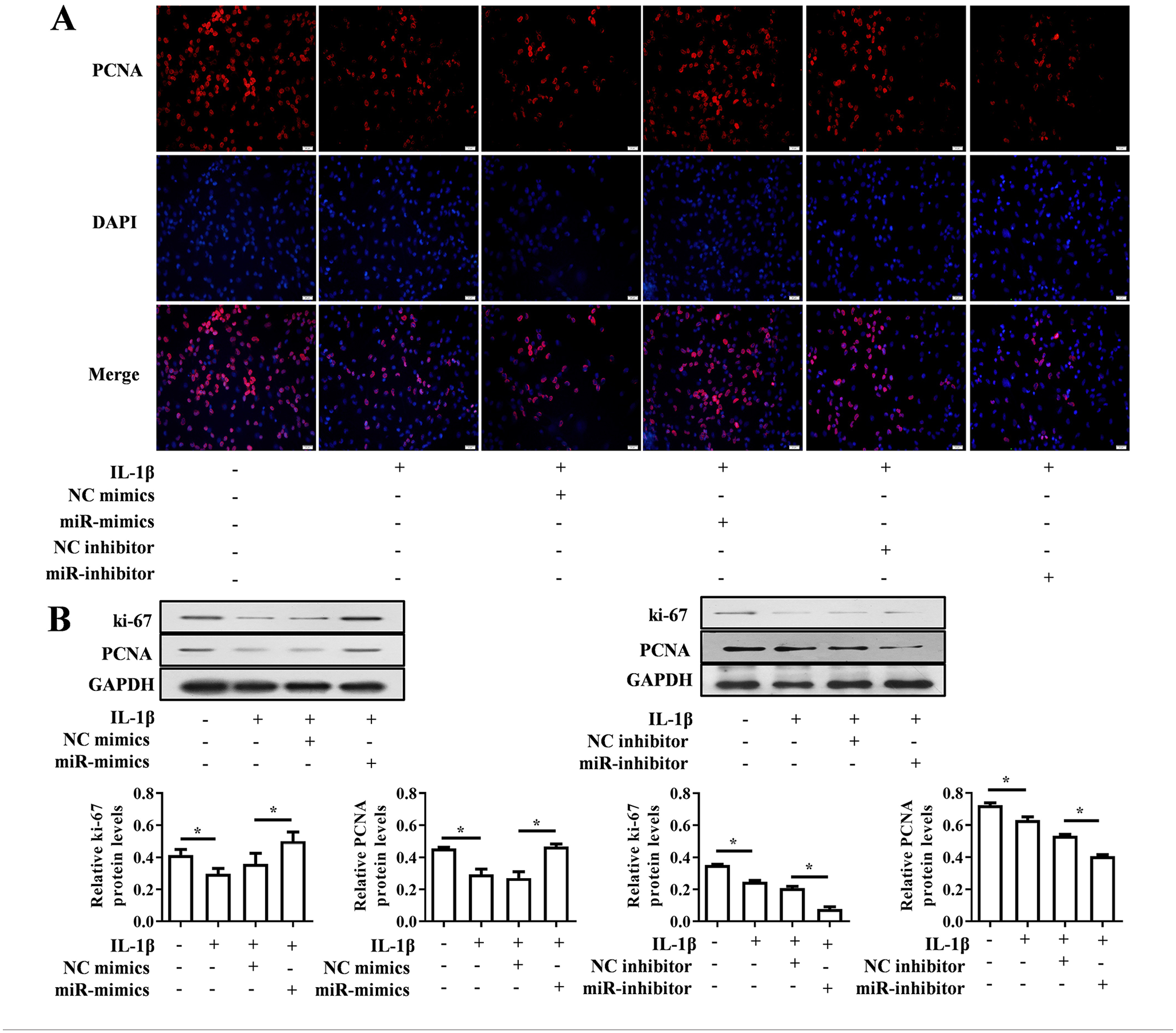
Expression levels of value-added related proteins PCNA and Ki-67 when miR-379-5p up-regulated or down-regulate. (
The Effects of miR-379-5p on IL-1β-Mediated Inflammatory Response and ECM Degradation of Chondrocytes
The results of ELISA assay showed that the levels of inflammatory factors IL-6, IL-8, IL-17, and TNF-α in the cell culture supernatant were increased by IL-1β, and this would be pulled down by miR-379-5p mimics (P < 0.001) rather aggravated by miR-379-5p inhibitor (P < 0.001) ( Fig. 4A ). IF again confirmed the effect of IL-1β and miR-379-5p on the expression of IL-6 and TNF-α in chondrocytes, which was consistent with ELISA results ( Fig. 4B and C ). The levels of ECM degradation-related proteins (MMP-1, MMP-13, collagen II, and aggrecan) in cell culture supernatant were also assessed using ELISA and IF. And the ELISA results showed that IL-1β promoted the secretion of MMP-1 and MMP-13 (P < 0.05), but prevented the secretion of collagen II and aggrecan (P < 0.001). However, this change was prevented by miR-379-5p mimics and promoted by miR-379-5p inhibitor (P < 0.01) ( Fig. 5A ). This was also verified by IF assay on MMP-13 and collagen II proteins performed in chondrocytes ( Fig. 5B and C ).
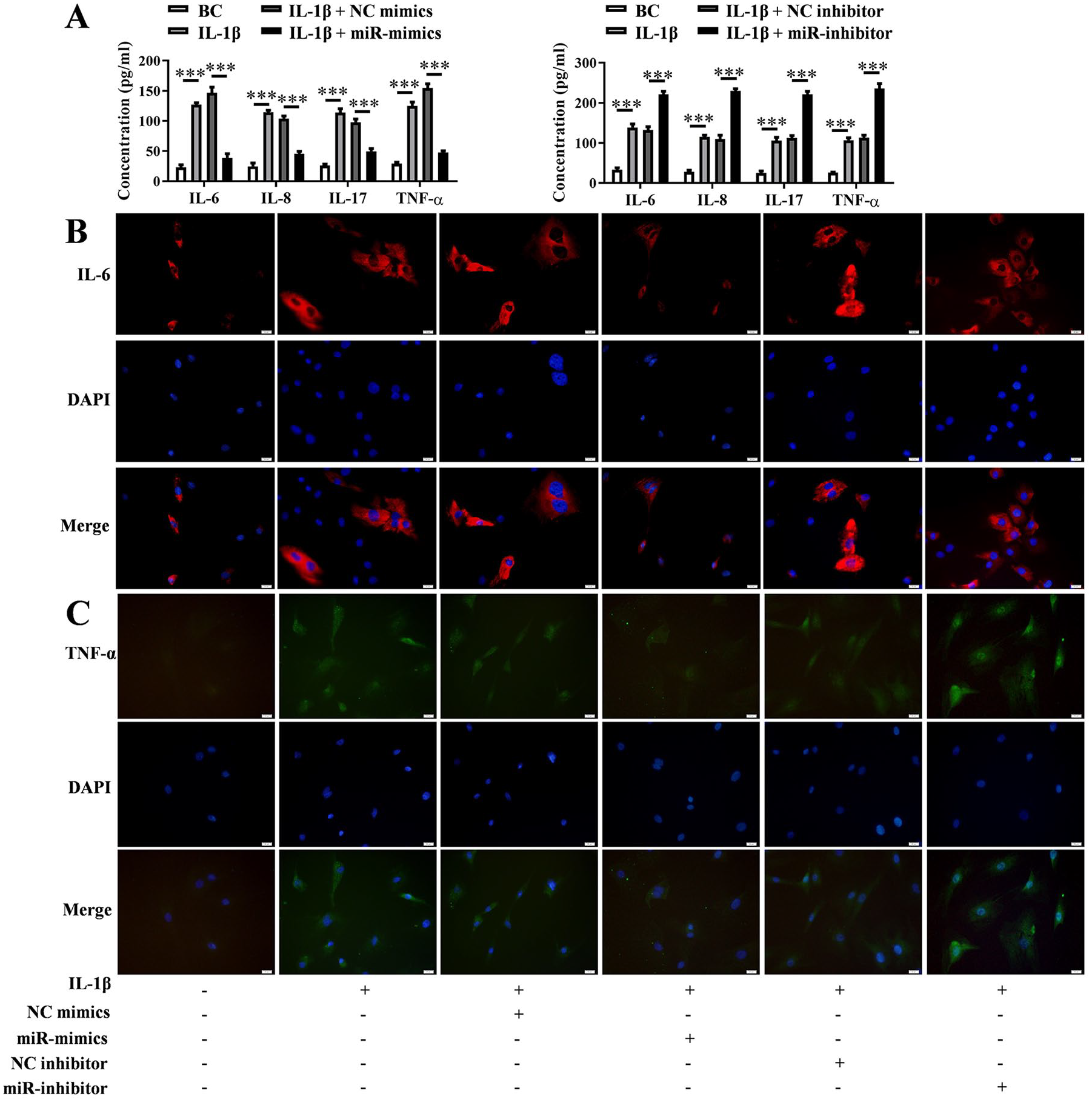
Expression levels of inflammatory cytokines when miR-379-5p up-regulated or down-regulate. (
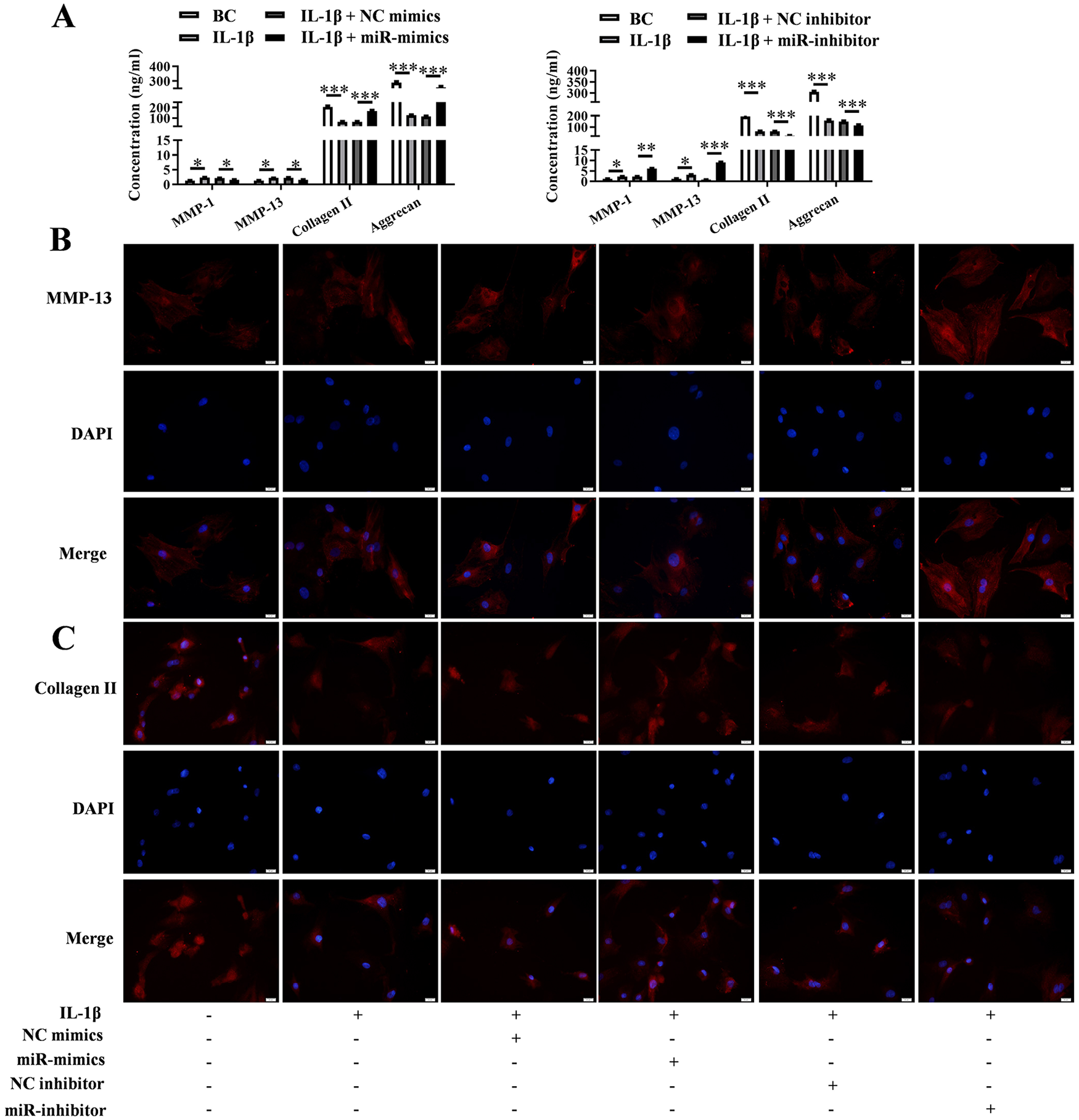
Expression levels of ECM-related proteins when miR-379-5p up-regulated or down-regulate. (
MiR-379-5p Regulated YBX1 and the Influence on PI3K/Akt Pathway
The results of real-time PCR showed that IL-1β up-regulated the expression level of YBX1, which was contrary to the effect of miR-379-5p on YBX1 expression (P < 0.01) ( Fig. 6A ). TargetScan and miRwalk software predicted that 3’UTR of YBX1 mRNA was completely paired with the seed sequence of miR-379-5p ( Fig. 6B ), and luciferase reporter assay verified that miR-379-5p inhibited firefly luciferase by targeting to the 3’UTR of YBX1 mRNA ( Fig. 6C ). Furthermore, YBX1 protein and PI3K/Akt pathway related proteins (PI3K, p-PI3K, AKT, p-AKT) were also regulated by miR-379-5p via targeting YBX1 mRNA ( Fig. 6D and E ).
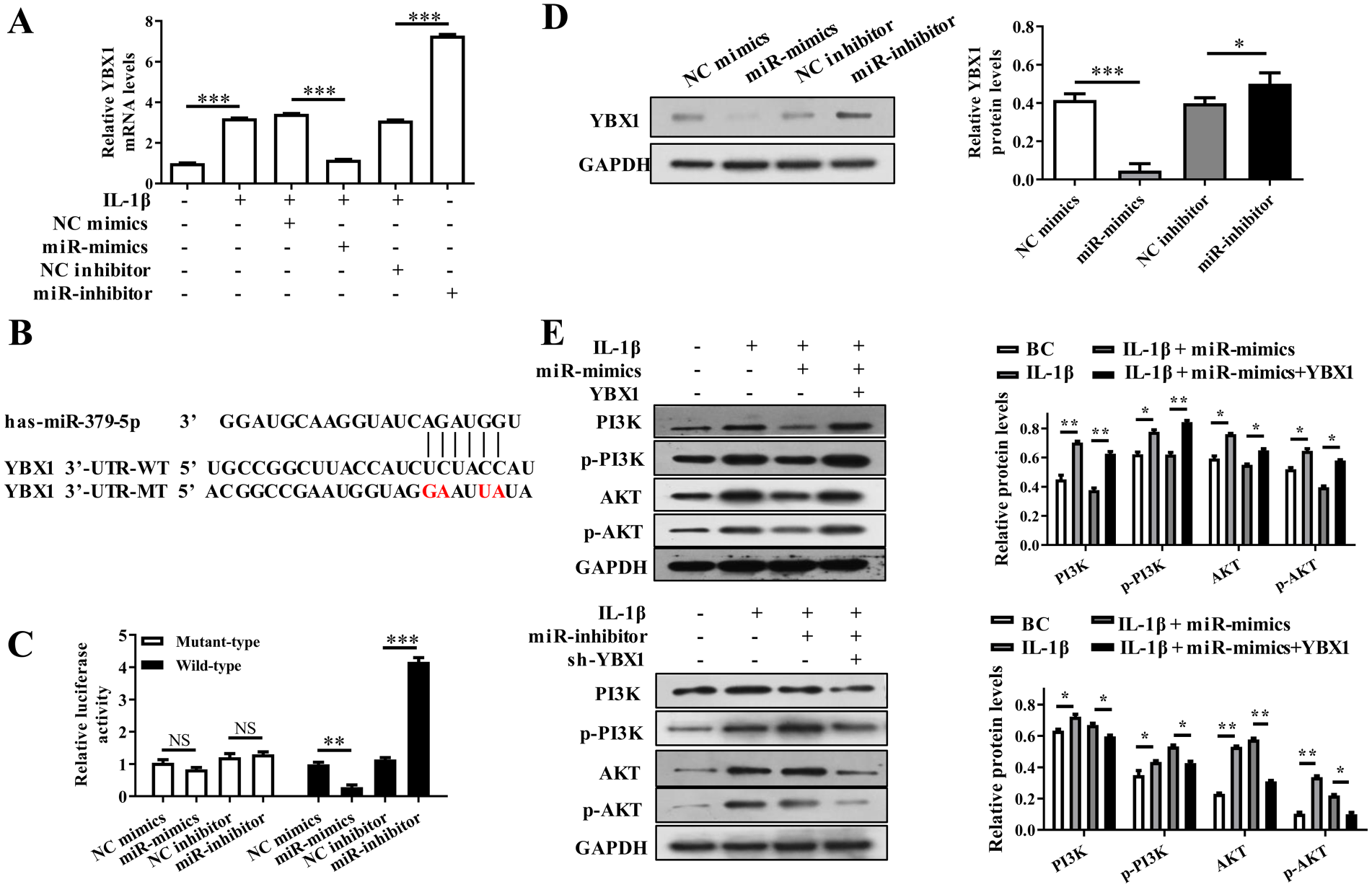
MiR-379-5p inhibited the activation of PI3K/Akt signaling by targeting YBX1 in IL-1β stimulated chondrocytes. (
The Effects of Inhibited PI3K/Akt Pathway on miR-379-5p-Mediated Cell Proliferation and Inflammatory Response
To confirm the effects of inhibited PI3K/Akt pathway on miR-379-5p-mediated cell proliferation and inflammatory response, the PI3K/Akt pathway inhibitor LY294002 was added in miR-379-5p inhibitor + IL-1β treated chondrocytes. The mRNA levels of PI3K/Akt pathway related proteins (PI3K, p-PI3K, AKT, p-AKT) were up-regulated by IL-1β and miR-379-5p inhibitor (P < 0.001), which was decreased by LY294002 (P < 0.001) ( Fig. 7A ). The cell viability decreased by IL-1β and miR-379-5p inhibitor was also rescued by LY294002 (P < 0.001) ( Fig. 7B ), as well as the expression of ki-67 and PCNA protein (P < 0.05) ( Fig. 7C and D ). The results of ELISA showed that secretion of inflammatory factors (IL-6, IL-8, IL-17, and TNF-α) and ECM degradation-related proteins (MMP-1 and MMP-13) were increased by IL-1β and miR-379-5p inhibitor but decreased by LY294002 (P < 0.01) ( Fig. 7E ). ECM degradation-related proteins (collagen II and aggrecan) were decreased by IL-1β and miR-379-5p inhibitor but increased by LY294002 (P < 0.01) ( Fig. 7F ).
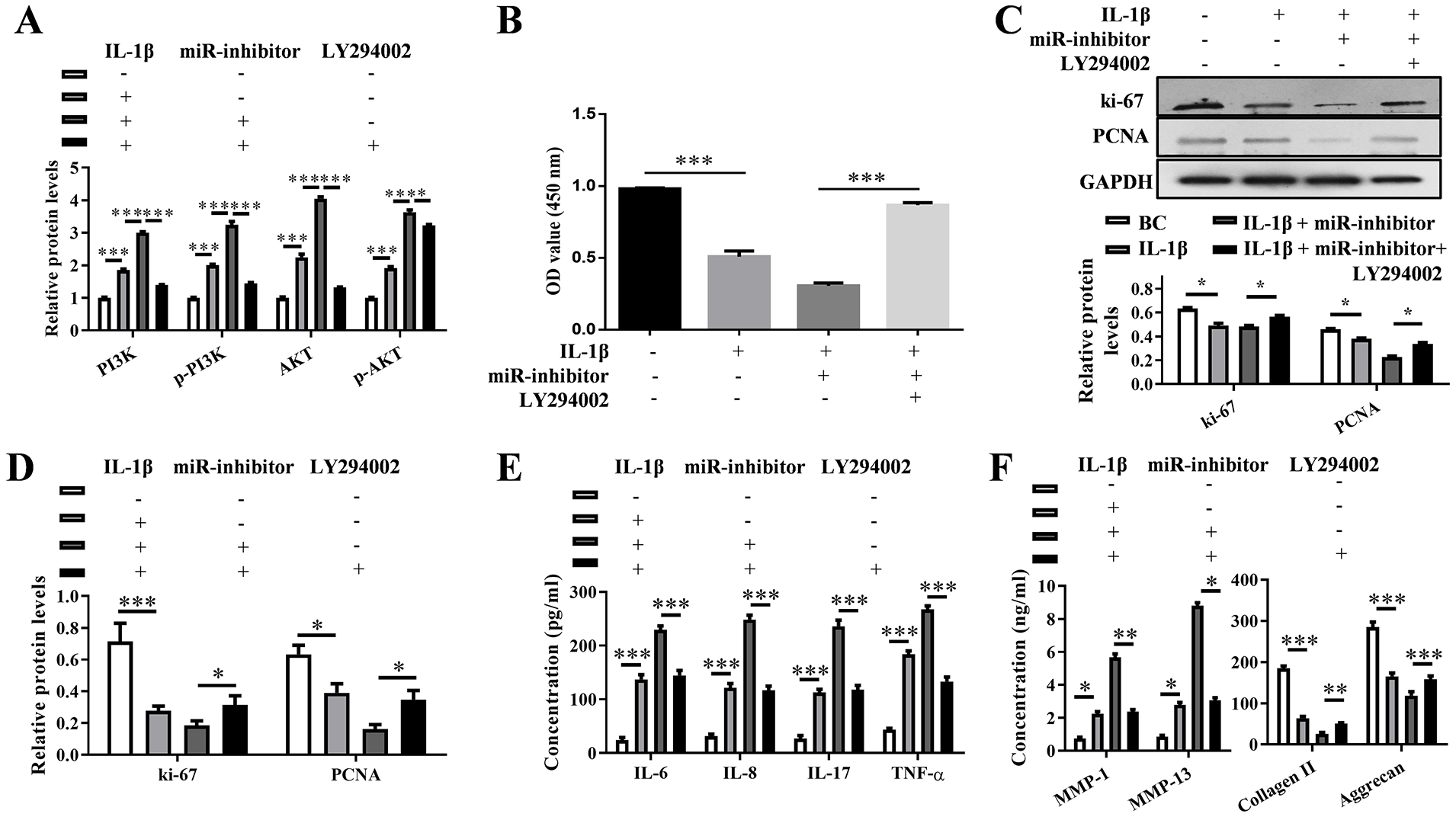
Blocking PI3K/Akt pathway attenuates the effect of miR-379-5p inhibitor on OA progression. (
The Effect of miR-379-5p on Chondrocyte Proliferation and Cartilage Degradation
The level of miR-379-5p in OA samples of rats were down-regulated and reached the extreme at 4 weeks, while miR-379-5p agomir increased the miR-379-5p level in OA tissues (P < 0.001) ( Fig. 8A ). The YBX1 mRNA was up-regulated reached the extreme at 4 weeks in OA tissues, which was decreased by miR-379-5p agomir (P < 0.001) ( Fig. 8B ). Safranin O-fast green staining showed that there was a severe erosion of cartilage surface and decreased safranin O staining in superficial layer in OA samples, while the erosion of cartilage surface was alleviated and safranin O staining was increased in OA samples with miR-379-5p agomir injection ( Fig. 8C ). The severity of articular cartilage degeneration in each rat was graded according to OARSI scoring system ( Fig. 8D ). Furthermore, miR-379-5p agomir increased the ki-67 and PCNA protein levels in OA tissues ( Fig. 8E ). And up-regulated IL-6, IL-8, IL-17, TNF-α, MMP-1, and MMP-13 in OA tissues were decreased by miR-379-5p agomir while down-regulated collagen II and aggrecan were increased ( Fig. 8F and G ).
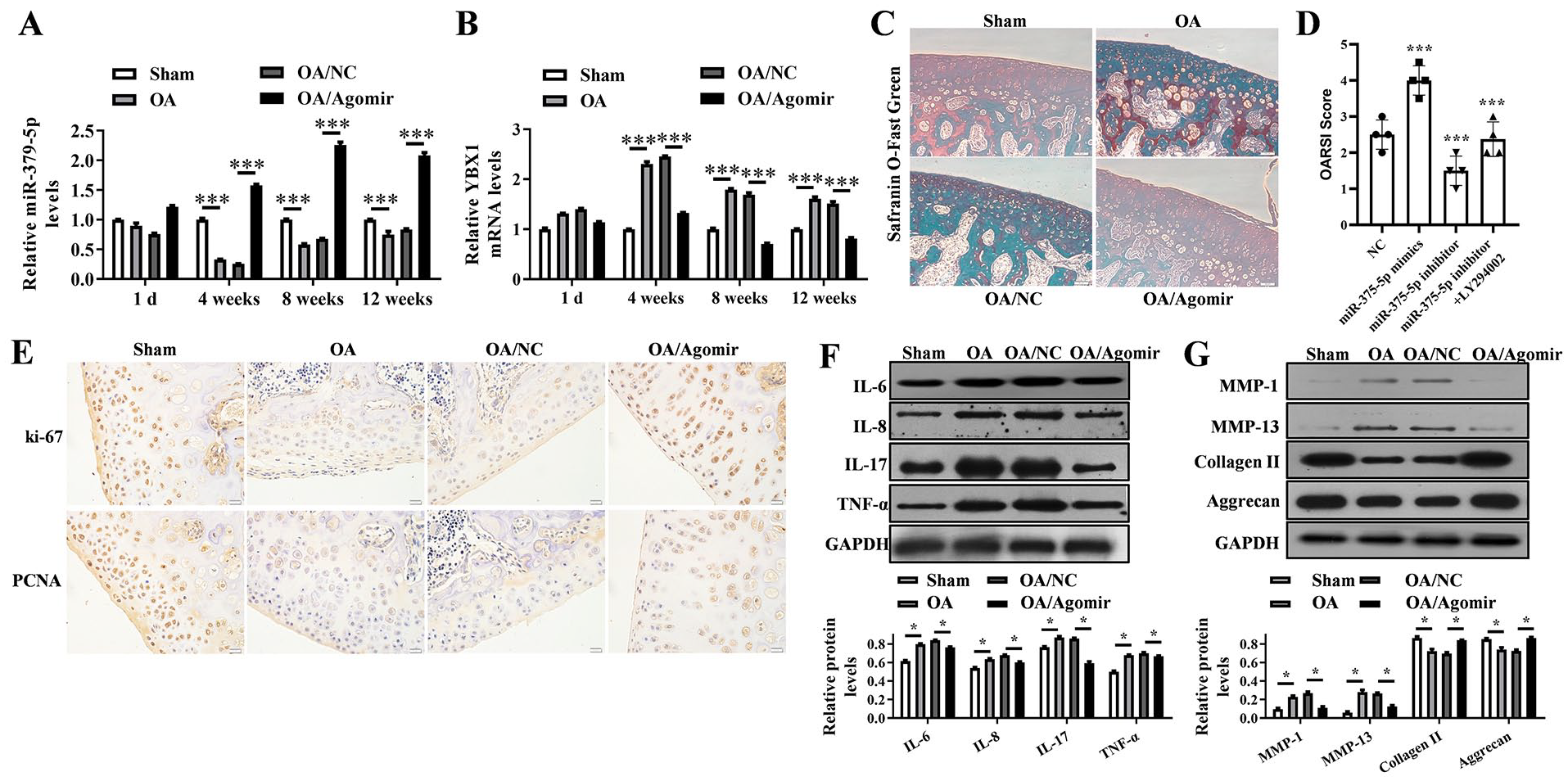
MiR-379-5p agomir promotes the articular chondrocytes proliferation and alleviates cartilage degradation in vivo. (
Discussion
Cartilage damage and inflammatory response are important features of OA. 26 IL-1β signaling pathway is involved in the progression of OA. 27 Therefore, IL-1β-treated cartilage cells were used as in vitro OA model in this study. Inflammation of articular tissues, degradation of ECM and apoptosis of chondrocytes were important factors in the pathogenesis of OA. 28 Chondrocytes in articular cartilage were cells that produce ECM components 29 and chondrocytes, stimulated by the pro-inflammatory cytokine IL-1β, produce matrix metalloproteinases and other inflammatory cytokines, 30 causing cellular inflammation and ECM degradation. In this study, we found that elevated level of miR-379-5p promoted the proliferation of IL-1β stimulated chondrocytes, reduced the production of inflammatory factors and matrix metalloproteinases, and promoted extracellular ECM formation. These effects might be exerted by targeting YBX1 and inhibiting the activation of PI3K/Akt pathway in IL-1β stimulated chondrocytes. In addition, intra-articular injection of miR-379-5p agomir was an effective means of administering miRNA-379-5p in OA rats, which promoted the proliferation of chondrocytes in the knee articulation of OA rats and alleviated the inflammation and ECM degradation in OA tissue.
High level of miR-379-5p could promote chondrocyte proliferation. 15 While in this study, miR-379-5p was down-regulated in human knee OA tissues, which was consistent with the result of miRNA sequencing. 9 This indicated that inhibition of miR-379-5p was associated with the progression of OA. Ki-67 protein was associated with cell cycle and PCNA protein participant in the process of DNA replication. Ki-67 and PCNA were standard markers of cell proliferation.31,32 Up-regulated miR-379-5p promoted both cell viability and levels of ki-67 and PCNA in IL-1β stimulated chondrocytes, indicating miR-379-5p promotion of cell proliferation was associated with up-regulation of ki-67 and PCNA. Inflammatory cytokines IL-6, IL-8 and IL-17 were implicated in OA. 33 TNF-α was related to the produce of matrix metalloproteinase (MMPs) degrading all components of the extracellular matrix, MMP-13 was also degrading aggrecan, which aggravated the matrix destruction. 34 The present results showed that up-regulated miR-379-5p inhibited the expression and secretion of inflammatory cytokines IL-6, IL-8, IL-17, and TNF-α and matrix metalloproteinase MMP-1 and MMP-13, and the inhibition of collagen II and aggrecan was relieved in IL-1β stimulated chondrocytes. This indicated that miR-379-5p, in addition of promoting cell proliferation, protected cells against IL-1β stimulated inflammation and ECM degradation in chondrocytes. The rescue by miRNA mimics after IL-1β treatment was sometimes clearly exceeding the control level, and this was because there were other pathways effects as well. The regulatory amplitude of miRNA mimics to different downstream molecules was different.
YBX1 is a modulator of MIA/CD-RAP-dependent chondrogenesis. 35 YBX1 also plays an important role in signaling DNA damage and cell proliferation. 36 YBX1 expression can affect the downstream PI3K/Akt signaling pathway and activate it. 18 Therefore, YBX1 plays an important role in PI3K/Akt. Akt also plays an important role in preventing cell proliferation, migration, and invasion. 37 In this study, YBX1 was targeted by miR-379-5p in chondrocytes. PI3K, p-PI3K, AKT, p-AKT, key proteins in PI3K/Akt signaling, were elevated in IL-1β- or YBX1-induced chondrocytes, while this effort was reversed by miR-379-5p. Furthermore, we demonstrated that inhibition of PI3K/Akt signaling in miR-379-5p and IL-1β treated chondrocytes elevated ki-67, PCNA, collagen II and aggrecan, and decreased IL-6, IL-8, IL-17, TNF-α, MMP-1, and MMP-13. These suggested that miR-379-5p regulated the cell proliferation and ECM homeostasis in chondrocytes by targeting YBX1 and PI3K/Akt signaling. The PI3K/Akt pathway has also been shown to be an important regulator of bone formation and remodeling, and its reduced expression can induce delayed ossification, short-bone and the formation of secondary ossification centers. 38 In addition, we performed the intra-articular injection of miR-379-5p agomir in OA rats, and demonstrated that miR-379-5p agomir elevated the miR-379-5p level in OA tissue, promoted cell proliferation and inhibited inflammation and ECM degradation, this alleviated the erosion of cartilage surface caused by MMPs and inflammatory cytokines.34,39
In addition to PI3K/Akt, AMP-activated protein kinase (AMPK) activation is also associated with YBX1. 40 It is a critical cellular energy sensor, which could take part in the tumorigenesis and OA development.41,42 At transcriptional level, AMPK has been reported to be regulated by miR-379-5p. 43 However, due to limited funds and time, we only confirmed that miR-379-5p can inhibit PI3K/Akt pathway and promote chondrocyte proliferation by targeting YBX1, but did not pay much attention to AMPK signaling in chondrocytes, which may also serve as a key regulator in OA development.
Taken together, our results suggested that miR-379-5p was lowly expressed in human knee OA tissues. The up-regulation of miR-379-5p in chondrocytes promoted cell proliferation, decreased the expression and secretion of inflammatory cytokines, and inhibited the degradation of ECM by inhibiting the PI3K/Akt signaling pathway. Intra-articular injection of miR-379-5p agomir also alleviated the progression of OA in rats with OA. Thus, miR-379-5p may be used for the treatment of human OA in clinical. Moreover, there are few studies on miR-379-5p and OA. Our study provides a theoretical basis for subsequent studies on the role of miR-379-5p in OA. MiR-375-5p via inhibition of PI3K/Akt pathway by targeting YBX1 may represent a novel and promising target for the treatment of OA.
Footnotes
Author Contributions
Jia Zheng made substantial contributions to research design; Hongjun Zhang and Wendi Zheng were responsible for the acquisition and interpretation of data; Wendi Zheng and Du Li performed statistical analysis and data visualization; Hongjun Zhang drafted the paper; and Jia Zheng revised it critically for important intellectual content. All authors have read and approved the final manuscript.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Ministry of Science and Technology National Key Research and Development Program (Grant No.: 122300411149), Natural Science Foundation of Henan Province (Grant No.: 182300410349), and the Joint Construction Project of Henan Provincial Health Committee and Ministry of Health (Grant Nos.: 201701017 and B20190316).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethics Committee of the Henan Provincial People’s Hospital, People’s Hospital of Zhengzhou University, School of Clinical Medicine, Henan University. All animal studies were approved by the Ethics Committee of Zhengzhou University.
