Abstract
Objective
Osteoarthritis (OA) is an inflammatory and degenerative disease, and the numerous treatments currently used are not fully effective. Mesenchymal stem cells (MSCs) and platelet-rich plasma (PRP) are proposed for OA treatment as biologic therapies. The aim of the study was to observe the role of autologous protein solution (APS), a type of PRP, on chondrogenic differentiation of 2 types of MSCs, from bone marrow (BMSCs) and adipose tissue (ADSCs), in an in vitro osteoarthritic microenvironment.
Design
Inflammatory culture conditions, mimicking OA, were obtained by adding interleukin-1β (IL-1β) and tumor necrosis factor α (TNFα), or synovial fluid from patient osteoarthritic knees (OSF), to the culture medium. MSCs were then treated with APS.
Results
After 1 month of culture, both cell types formed mature micromasses, partially altered in the presence of IL-1β and TNFα but quite preserved with OSF. Inflammatory conditions hindered differentiation in terms of gene expression, not counterbalanced by APS. APS triggered type I collagen deposition and above all contributed to decrease the expression of metalloproteinases in the most aggressive conditions (IL-1β and TNFα in the culture medium). ADSCs originated micromasses more mature and less prone toward osteogenic lineage than BMSCs, thus showing to better adapt in an aggressive environment than BMSC.
Conclusions
APS seems to act better on inflammation front and, between cell types, ADSCs respond better to the inflammatory microenvironment of OA and to the treatment with APS than BMSCs.
Introduction
Osteoarthritis (OA) is a chronic disease affecting the articular cartilage. Bruyère et al. 1 estimated that 250 million of the world’s population, aged over 50 years, suffer from symptomatic knee OA and its incidence is expected to increase, with huge socioeconomic impact. 2
The dual nature of this pathology, degenerative and inflammatory, is now explained by the sequence of events characterizing it. OA starts with an early extracellular matrix (ECM) degradation, that triggers the release of inflammatory cytokines and catabolic mediators, and proceeds with the increase of the cartilaginous tissue erosion until the uncovering of the subchondral bone. 3
Different pro-inflammatory cytokines, such as interleukin (IL)-1β, IL-6, tumor necrosis factor α (TNFα) and several chemokines are recognized and well described as great mediators of the complex cross-talk between chondrocytes and synoviocytes, while the metalloproteinases (MMPs) are the critical effectors of cartilage degradation.4-6
Multiple causes of OA, including age, prolonged physical effort, traumatic events, correspond a common clinical picture: pain, deformity and loss of function until joint replacement with artroprosthesis.7,8
Despite the widespread diffusion of OA, to date a therapy able to really counteract the progression of this clinical condition is lacking. 9 The most common approaches include nonsteroidal anti-inflammatory drugs (NSAIDs), analgesics and opioids, chondroprotecting agents, and intra-articular infiltrative treatment with steroids and hyaluronates. 10 Physical therapies, such as pulsed electromagnetic fields (PEMFs), are also being studied for their anti-inflammatory and regenerative effects, and already adopted in the clinic. 11 These treatment options are widely used but, while they are able to mitigate symptoms, ameliorating pain and functionality, they are unable to prevent or delay OA progression. 9 Furthermore, side effects are also reported in relationship to some treatments, and conflicting opinions remain about the true effectiveness of these approaches. 12 To date, the challenge of identifying a new, resolutive tool in order to address OA is open.
The therapeutic potential of mesenchymal stem cell (MSC) injections has been known for several decades from preclinical and clinical research on animals or patients affected by OA.13-15 Among the MSC sources, the most used in OA are bone marrow (BMSCs) and adipose tissue (ADSCs),16,17 but there is still the need of knowledge on the behavior of these MSCs.
In a previous study of our group, an in vitro model was set up with ADSCs or BMSC micromasses cultured in a chondrogenic medium containing IL1β and/or TNFα or human osteoarthritic synovial fluid (OSF) to simulate, as much as possible, the inflammatory microenvironment of a joint affected by OA. After 4 weeks of culture, a better response toward inflammation was observed by ADSCs compared with BMSCs. 3
A biological treatment proposed to address OA, not only widely studied but also highly debated, is platelet-rich plasma (PRP). 18 Despite a biological variability in the concentration of platelets (PLTs), PRP generally contains around 5-fold the PLT amount present in the peripheral blood, providing a high amount of growth factors (GFs) content in PLT granules. 19 Thanks to its characteristics, PRP might counteract the degrading effects of inflammatory mediators and improve the regenerative ability of cartilage, a tissue characterized by poor healing capacity. This could restore a favorable joint environment, reducing the need and therefore the risks of intolerance and side effects ascribed to conventional drugs.20,21 Conflicting results emerge from different studies, due to the different preparation protocols used to concentrate whole blood, the activators used, and PLTs and white blood cell (WBC) concentrations, that still make clinical evaluation of the true potential of PRP, as a biologic therapy in OA, difficult. 22
With the aim to improve the potential of blood derivatives for OA treatment, recently another product has been developed: autologous protein solution (APS), a blood derivative obtained after 2 consecutive centrifugations performed with a polyacrylamide beads concentration device. The resulting APS is characterized by a high concentration of PLTs, WBCs, and plasma containing both anabolic and anti-inflammatory molecules. WBCs are the main source of IL1 receptor antagonist (IL-1ra), inhibiting catabolic effect of IL-1β. Alpha PLT granules contain anabolic GFs, such as epidermal growth factor (EGF), insulin-like growth factor (IGF-1), platelet derived growth factor (PDGF)-AB and -BB, vascular endothelial growth factor (VEGF) and transforming growth factor β1 (TGF-β1), that induce cartilage regeneration. Plasma contains anti-inflammatory cytokines, such as soluble interleukin-1 receptor antagonist-type II (sIL-1RII) and soluble tumor necrosis factor receptor-type I and -type II (sTNF-RI and sTNF-RII). On the other hand, APS contains low levels of proinflammatory molecules (eg, IL1β and TNFα). 23
A number of experimental studies have evaluated the features, effectiveness and side effects of APS treatment. These observations focused on in vitro cultures of chondrocytes and synoviocytes,24-27 in vivo28-30 and clinical studies.23,31-34 The ability of APS to block inflammation, regenerate cartilage and improve the clinical scenario of subjects affected by OA has been shown.
Starting from these premises, the present study aimed at evaluating the biological effects of APS on the chondrogenic ability of BMSCs and ADSCs cultured in micromasses in an inflammatory environment. In continuity with our previous research, 3 in vitro alternative conditions were set up: IL-1β and TNFα, or OSF were added to the cell culture medium, with or without APS. The differentiation toward chondrocytes, the synthesis of ECM and the possible tendency toward osteogenesis have been evaluated through histology, histomorphometry, immunohistochemistry, and gene expression analysis after 4 weeks of culture.
This study hypothesis was that APS improves chondrogenesis and reduces the production of pro-inflammatory mediators in an in vitro model, trying to mimic the clinical conditions in which MSCs are injected into OA joints.
Methods
Experimental Setup
Human BMSCs and ADSCs (ATCC, Virginia, USA), certified for stemness and their trilineage differentiation ability, were expanded in Dulbecco’s modified Eagle Medium (DMEM) (MesenCult MSC Basal Medium, Stem Cell Technologies, Canada) added with 10% fetal bovine serum (FBS; EUROCLONE, Pero, Milan, Italy) until the second passage and then cultured into a 3-dimesional system, as previously reported. 3 Briefly, 2.5 × 105 cells were pelleted (10 minutes, 160g) per tube (Sarstedt, Nümbrecht, Germany) and kept in 1 mL of chondrocyte differentiation medium (Stem Pro, GIBCO, Grand Island, NY, USA) to induce the micromass growth and organization.
Three different culture conditions were set up in tubes (n = 8 for each culture condition):
Control (CTR): Cells were cultured with chondrocyte differentiation medium
IL-1β + TNFα: Cells were cultured in chondrocyte differentiation medium supplemented with 10 ng/mL IL-1β and 50 ng/mL TNFα (Roche Diagnostics GmbH, Mannheim, Germany)
OSF: Cells were cultured in chondrocyte differentiation medium supplemented with 20% of human synovial fluid harvested from patients affected by OA.
For each culture condition, APS conditioned medium (CM) was added to half of the tubes (CTR + CM, IL-1β + TNFα + CM, and OSF + CM groups), while the other half of tubes did not receive CM (CTR, IL-1β + TNFα, and OSF groups).
The micromasses (n = 4 for each group) were cultured for 4 weeks at 37°C in a 5% CO2/air humidified atmosphere to reach a mature state. The medium was changed twice a week, one of which employing CM in the groups with APS. Every time the APS was freshly obtained from donor blood.
APS Conditioned Medium
APS was obtained starting from 55 mL blood sample of a healthy male volunteer, after signing of a research donation consent and after local ethical committee approval (IOR Ethical Committee, prot. 0003409). APS was prepared by using the NStride APS kit (Zimmer Biomet, Zimmer GmbH, Sulzerallee 8, 8404 Winterthur, Switzerland) according to the manufacturer’s instructions. Briefly, peripheral blood was collected in a 60-mL syringe with 5 mL anticoagulant citrate dextrose solution–formula A (ACD-A) and slowly transferred into the APS separator. The device was processed using a centrifuge (BIOMET) at 1800g for 15 minutes. The resulting cell suspension was then collected, shaken, and loaded in the APS concentrator, which uses filtration through polyacrylamide beads to concentrate the cytokines in the final output. 31 After brief centrifugation (1800g, 2 minutes), the final product of the APS kit (approximately 2-3 mL) contained high concentrations of PLTs, WBCs, and plasma proteins.25,26,31 No PLT activation agents were used in this study following the kit instructions.
APS was also characterized for its content of inflammatory cytokines and GFs by immunoenzymatic assay: IL-1β, IL-1RA (Boster Biological Technology, Pleasanton, CA, USA). In the 3 experimental groups that required APS, it was added to the chondrogenic culture medium immediately after each preparation in a ratio of 1:10 to prepare CM. After 4 hours of incubation at 37°C, the resulting mixture was deprived of the unavoidable formed clot and defined CM to be added to the cultures.
Synovial Fluid
The OSF was obtained as waste material from a pool of 17 donor specimens collected from patients suffering from knee OA (Kellgren-Lawrence grade III-IV), who had given written consent (IOR Ethical Committee, No. 0003409), and scheduled for treatment in our institution. Before being added to the micromasses, the OSF pool was characterized regarding some pro-inflammatory factors, such as human IL6, IL8, and degradative enzymes, including MMP1 and MMP13, by using a Multi-Plex system (PROCARTA PLEX, eBioscience, ThermoFisher Scientific) that exploits a bead-based sandwich immunoassay technology with high sensitivity and dynamic range. The evaluation was performed by following the manufacturer’s instructions, and the characterization is reported in the “Results” section.
Gene Expression Analysis
Before proceeding to RNA extraction, the micromasses were chopped with a pestle (Argos Technologies, Vernon Hills, IL, USA) in 1 ml of Trizol reagent (Life Technologies, Carlsbad, CA, USA). The procedure was continued by using the commercial Purelink RNeasy Mini Kit (AMBION, Life Technologies,), after that the extracted RNA was quantified by NANODROP spectrophotometer (NANODROP 2720, Thermal Cycler, Applied Biosystem) and reverse transcribed with Superscript VILO cDNA Synthesis kit (Life Technologies) following the manufacturer’s instructions. cDNA of each sample was diluted to the final concentration of 5 ng/μL. Gene expression was evaluated by quantitative polymerase chain reaction (qPCR), using the SYBR green PCR kit (Qiagen, Hilden, Germany) in a Light Cycler 2.0 Instrument (Roche Diagnostics, Monza, Italy). Five nanograms of each sample were tested in duplicate. The protocol included a denaturation cycle at 95°C for 15 minutes, 25 to 40 cycles of amplification (95°C for 15 seconds, appropriate annealing temperature for each target, as detailed in Table 1 , for 20 seconds, and 72°C for 20 seconds) and a melting curve analysis to check for amplicon specificity.
Details of Primers Used for Gene Expression Analysis.

The mean threshold cycle was used for the calculation of relative expression using the 2−∆∆Ct method (Schmittgen and Livak, 2008), with GAPDH as reference gene.
Histology and Immunohistochemistry
After 4 weeks, micromasses were fixed in 10% buffered formalin for 30 minutes, washed in distilled water, dehydrated in graded ethanol series, and finally embedded in paraffin (ThermoFisher Scientific, Waltham, MA, USA). The blocks were sectioned along the frontal plane of the micromasses, obtaining histological sections of 5-µm thick with a Microm HM355S rotatory microtome (ThermoScientific, Walldorf, Germany). Three sections were then staining with hematoxylin-eosin (HE, Bio Optica, Milano, Italy) and other 3 with Alcian blue (AB, Sigma-Aldrich, Saint Louis, MO, USA) to qualitatively and quantitatively evaluate cellularity, ECM organization, glycosaminoglycans (GAG) content, and micromass area.
Three histological sections were immunostained for each micromass.
Other sections were treated with 0.2% pronase enzyme (Sigma-Aldrich, Saint Louis, MO, USA) in phosphate buffered saline (PBS) at 37°C for 30 minutes for epitope unmasking. After washing, the slides were incubated at room temperature for 1 hour with Blocking Serum (Vectastain Universal Quick Kit, Vectors Laboratories, Burlingame, CA, USA) to prevent nonspecific bindings, then incubated with specific rabbit polyclonal antibodies against human Type II collagen (COLL II) (ThermoFisher Scientific, Waltham, MA, USA) followed by incubation with an anti-rabbit HRP-conjugated secondary antibody (Bethyl Laboratories, Montgomery, TX, USA). The reactions were finally developed using the Vector NovaRed Substrate Kit for Peroxidase (Vectors Laboratories, Burlingame, CA, USA) and negative controls, by omitting the primary antibody, were included to check proper specificity and performance of the applied reagents.
Histomorphometric Measurements
Stained and immunostained sections were acquired with the digital scanner Aperio ScanScope (Aperio ScanScope CS, Aperio Technologies, Leica Biosystems, USA) to obtain histological images to perform histomorphometric evaluations:
Micromass area and the number of cells were quantified on slides stained with Alcian blue. The number of cells were then normalized for area of the corresponding micromass to obtain the cell density.
The quantification of type II collagen was performed by dividing the immunostained area of each micromass for the total area of micromass and the results were expressed in percentage (%).
Statistical Analysis
The statistical analysis was conducted using the software R v. 4.0.2, 35 and packages ARTool v.0.10.736,37 and emmeans v. 1.4.8. 38 After having assessed normality (Shapiro-Wilk test) and homogeneity of variance (Levene test) of data, they were analyzed with a linear model considering the effects of the factors (‘culture condition’, 3 levels: CTR, IL-1β + TNFα and OSF; and “APS conditioned medium,” 2 levels: “CM” and “no CM”) or their interaction on data. Data are reported as mean ± SD or ± SE at a significant level of P < 0.05. The reference control group was CTR.
Pairwise comparisons were conducted with estimated marginal means (also known as least squares means) and adjusting P value according to Tukey. If the data were not normally distributed or presented heteroskedasticity of the variance, then they were analyzed using an aligned-rank transform (ART) allowing for nonparametric analyses of variance. Since with the aligned rank tests it is not possible to correctly calculate the differences in the pairwise combinations of levels between the factors in the interactions by using the interaction contrasts, these differences were calculated by the software as differences in the rank differences between the factors in the interaction.39,40 Cohen’s d effect size of standardized mean differences was calculated.
Results
Synovial Fluid Characterization
The following values were obtained by the pool of patient synovial fluids: IL6 (20.63 ± 1.17), IL8 (22.70 ± 10.11), MMP1 (20244 ± 1733) and MMP13 (123.63 ± 48).
Morphology, Gene Expression, and Protein Expression
Before analyzing the results in the different experimental conditions, the results of gene and protein expression of BMSC and ADSC CTR cultures were compared at 4 weeks ( Table 2 ). In consideration of the fact that significant differences were found between the 2 cell populations in this experimental time with regard to the expression of SOX9 (P < 0.0005) and ACAN (P = 0.0007), and Alcian blue staining (P = 0.0066), it was preferred to proceed in the statistical analysis considering the experimental setting adopted separately for each type of cells.
Gene Expression (2−ΔΔCt), Coll-II, and Alcian Blue Percentage Area (%) of ADSCs and BMSCs Cultured with Chondrogenic Differentiating Medium at 4 Weeks (Mean ± SD, n = 3 Replicates).
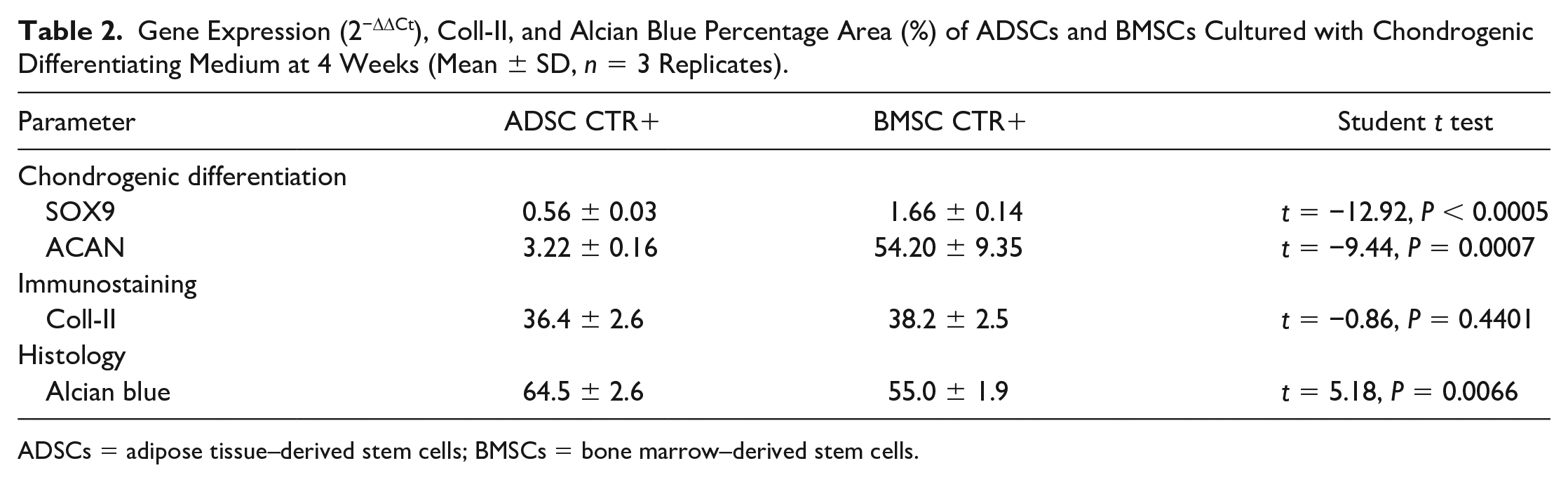
ADSCs = adipose tissue–derived stem cells; BMSCs = bone marrow–derived stem cells.
Table 3 shows the Cohen’s d effect size and 95% confidence interval of the following assessed parameters for contrast: IL-1β + TNFα + CM versus IL-1β/TNFα, and OSF + CM versus OSF in ADSC and BMSC micromasses.
Cohen’s d Effect Size and 95% Confidence Interval (in Brackets) of Assessed Parameters for Contrast: IL-1β + TNFα + CM versus IL1β/TNFα and OSF + CM versus OSF in ADSC and BMSC micromasses.
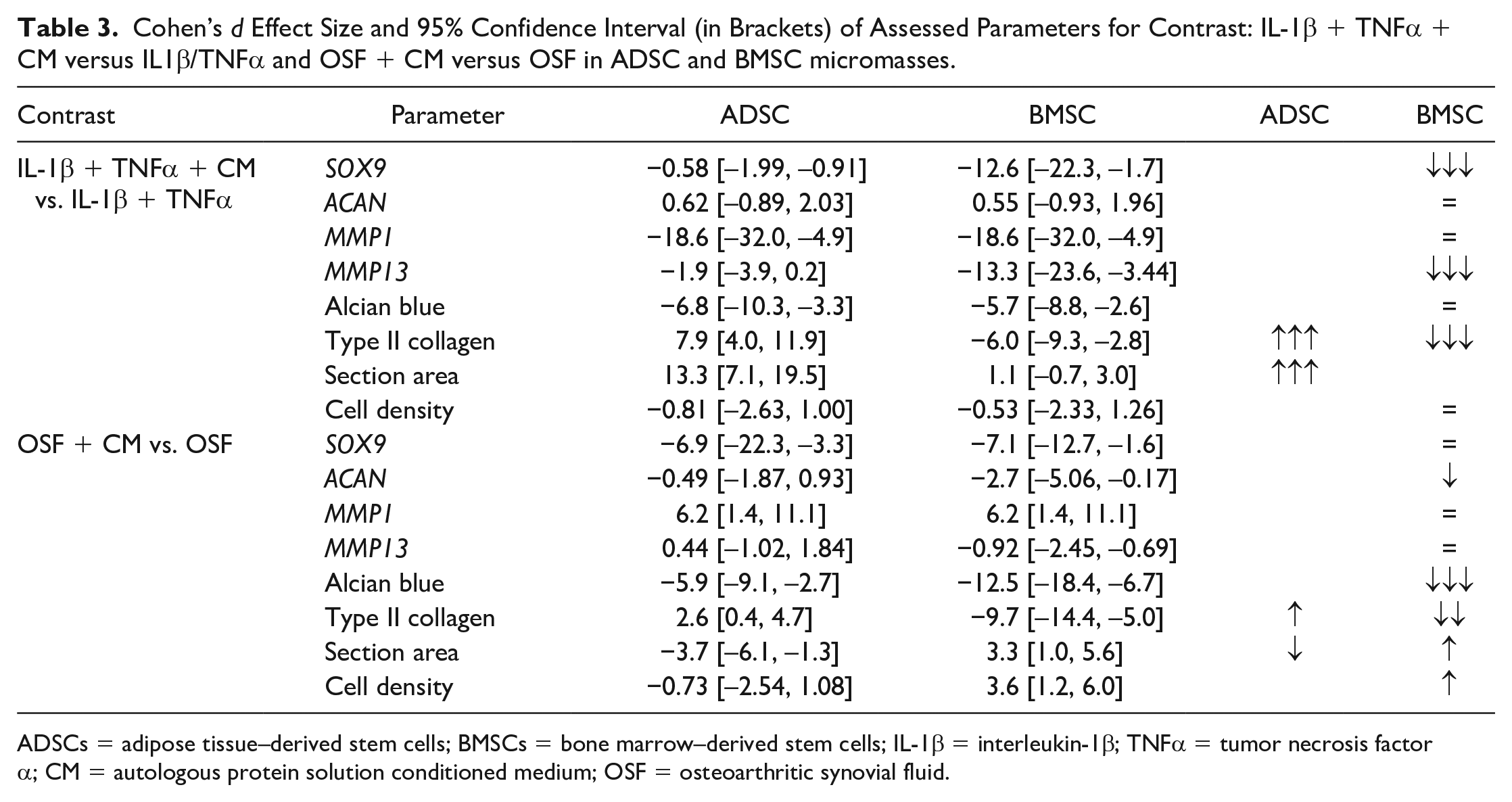
ADSCs = adipose tissue–derived stem cells; BMSCs = bone marrow–derived stem cells; IL-1β = interleukin-1β; TNFα = tumor necrosis factor α; CM = autologous protein solution conditioned medium; OSF = osteoarthritic synovial fluid.
hBMSCs
Comparison among Groups without APS
Before the analysis of the APS role, a preliminary comparison among CTR, IL-1β + TNFα and OSF groups was performed.
In CTR and OSF groups the consistency of micromasses was strong and the shape regularly round.
The micromasses in the IL-1β + TNFα group were still compact, presented a marked Alcian blue staining, and some signs of irregularity ( Fig. 1A , C , and E ). Data comparisons of IL1β+TNFα and OSF groups with CTR group confirmed no significant differences in Alcian blue staining, section area and cellular density ( Fig. 2A-C ).
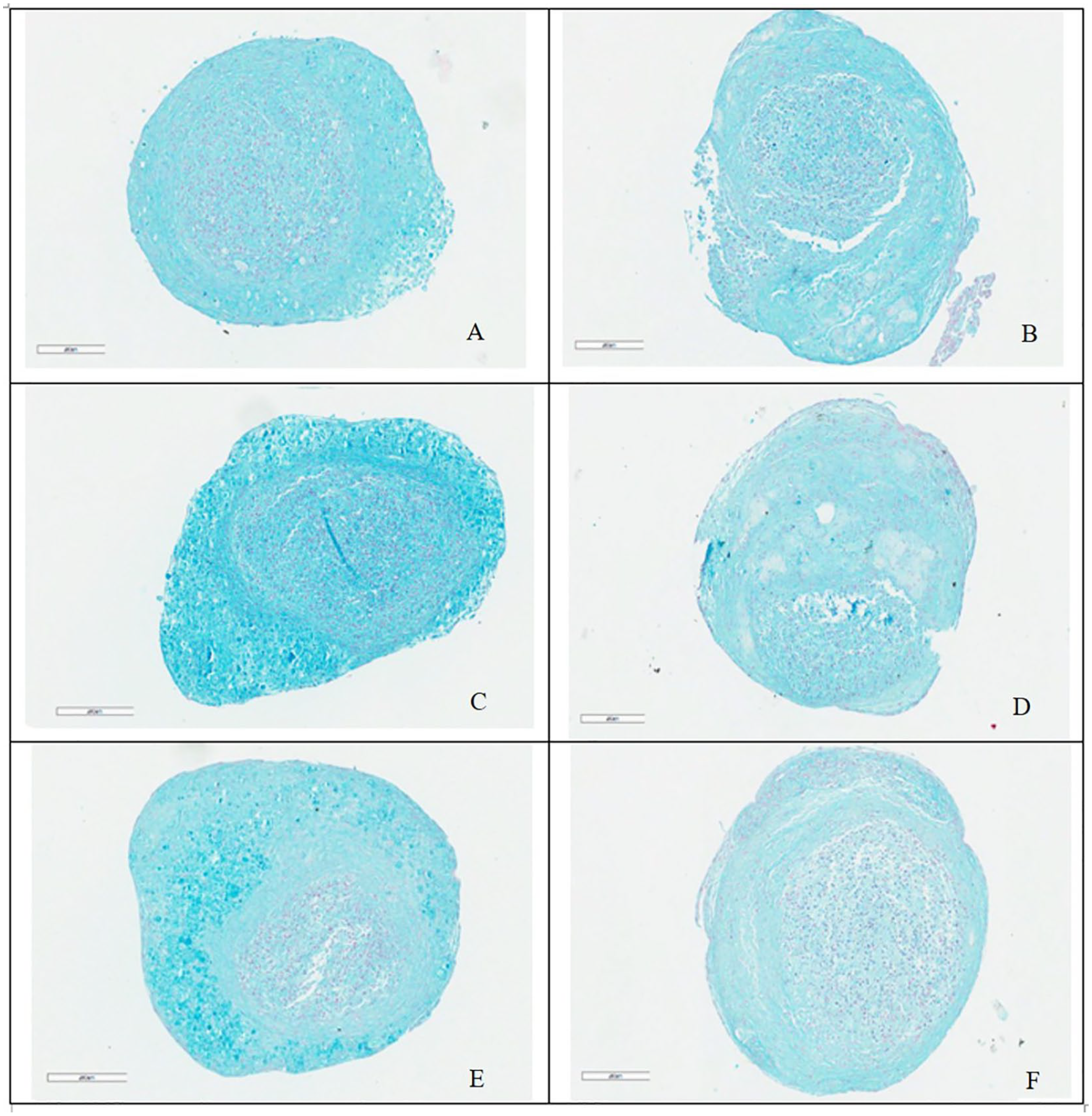
Histological images of BMSCs in CTR (
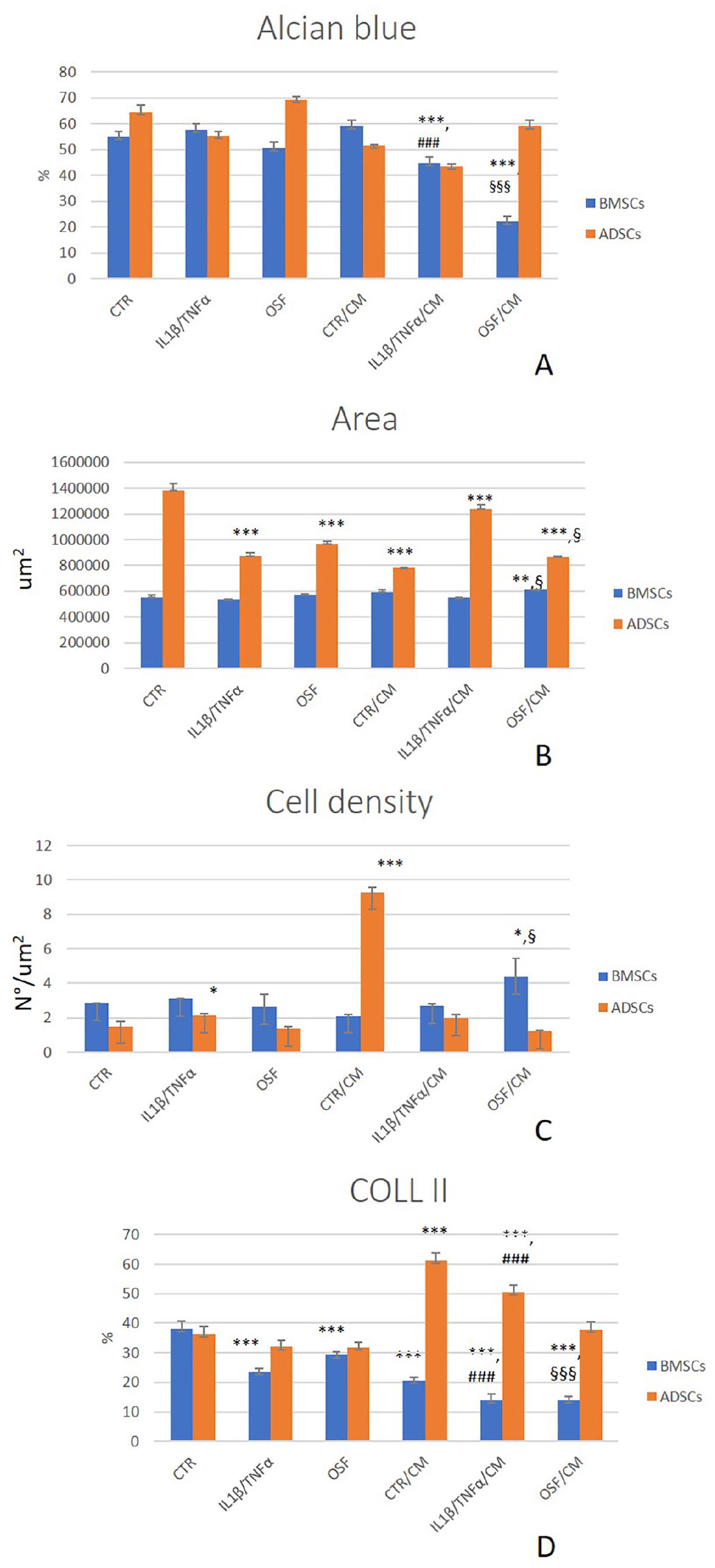
Histograms of Alcian blue staining (
SOX9 gene expression decreased in inflammatory conditions with regard to the CTR (IL-1β + TNFα: d = −2.4, P = 0.0153; OSF: d = −4.7, P < 0.0005) ( Fig. 3A ). A similar trend was observed for ACAN gene that strongly decreased in presence of IL-1β + TNFα or OSF (d = −4.8, P < 0.0005) until values close to zero ( Fig. 3B ). COL2A1 expression resulted undetectable by molecular biology, but protein type II collagen was observed in micromass sections of all groups: IL-1β + TNFα (d = −9.2, P < 0.0005) and OSF (d = −5.6, P < 0.0005) results were significantly lower than CTR ( Fig. 2D ). As confirmation of the inflammatory mediator role, a strong increases in MMP1 and, even if less evident, of MMP13 gene expression were observed for IL-1β + TNFα compared with CTR (MMP1: d = 8.6, P < 0.0005; MMP13: d = 2.8, P = 0.004) ( Fig. 3c and d ). In the OSF group, MMP1 (d = −2.2, P = 0.0289) and MMP13 (d = −8.4, P < 0.0005) expression decreased compared with CTR ( Fig. 3c and d ).
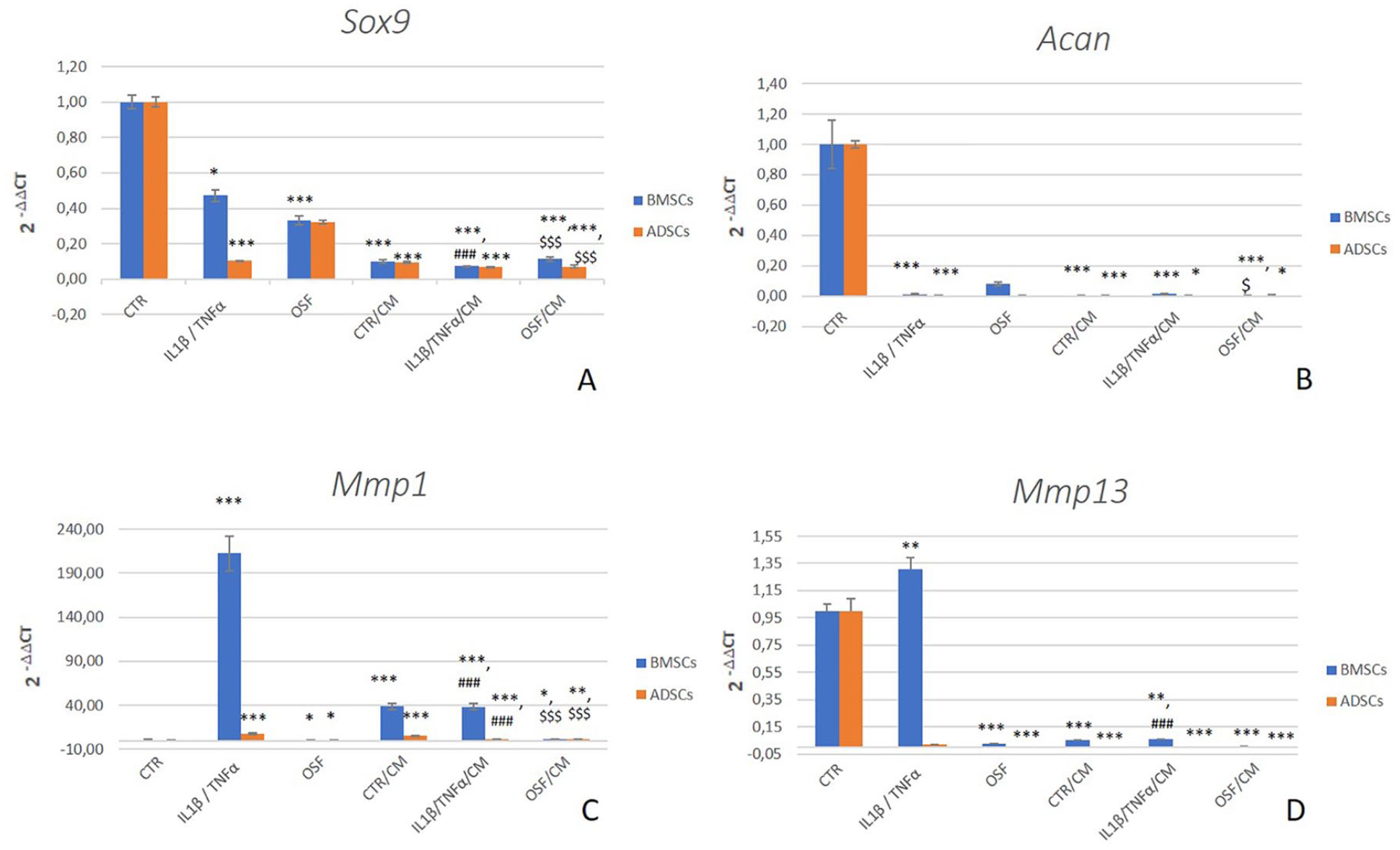
Histograms of gene expression results for SOX9 (
Comparison among Groups with and without APS
To investigate the role of APS, a comparison between each of CM treated groups and the related untreated one was performed, beside the comparison with CTR.
The regular shape and the consistency of the CTR micromasses seemed slightly altered in the presence of CM with little peripheral zone broken (Fig. 1 B, D, and F). Regarding Alcian blue staining, the association of APS CM and inflammatory culture conditions made this staining weaker than CTR (IL-1β + TNFα + CM: d = −4.5, P < 0.0005; OSF + CM: d = −14.5, P < 0.0005) ( Fig. 2A ). In addition, micromasses cultured in IL-1β + TNFα + CM showed lower Alcian staining than IL-1β + TNFα (d = −5.7, P < 0.0005), as well as OSF + CM ones in comparison with OSF (d = −12.5, P < 0.0005). The area of sections resulted wider in OSF + CM than CTR (d = 4.9, P < 0.005) and no significant difference was reported for IL-1β + TNFα + CM versus CTR. The comparison between the section area of CM treated and untreated ones highlighted a significant higher value for OSF + CM (d = 3.3, P = 0.0159) ( Fig. 2B ). Regarding cell density, the highest value was observed in the OSF + CM group that was significantly different from CTR (d = 3.4, P = 0.0120) and OSF groups (d = 3.6, P = 0.0078) ( Fig. 2C ).
SOX9 and ACAN gene showed an expression approaching zero, therefore a net decrease with respect to CTR in all groups (P < 0.0005) ( Fig. 3a and b ). The addition of APS to inflammatory conditions determined a further significant decrease in SOX2 (IL-1β + TNFα + CM: d = −12.6, P < 0.0005; OSF + CM: d = −7.1, P < 0.0005) and ACAN (OSF + CM: d = −2.7, P = 0.0159) expression versus APS untreated micromasses. As observed for the groups grown without CM, COL2A1 resulted undetectable by molecular biology, but present as protein in the observed micromasses, even if at lower level than CTR (P < 0.0005) ( Fig. 2D ). In addition, the presence of CM to inflammatory standard conditions determined a significant lower synthesis of type II collagen than in the absence of CM (IL-1β + TNFα + CM: d = −6.0, P < 0.0005; OSF + CM: d = −9.7, P < 0.0005). MMP1 was always more expressed than in CTR (CTR + CM: d = 5.4, P < 0.0005; IL-1β + TNFα + CM: d = 5.4, P < 0.0005; OSF + CM: d = 2.2, P < 0.05); the presence of CM decreased or increased MMP1 expression in IL-1β + TNFα + CM (d = −18.6, P < 0.0005) or OSF + CM (d = 6.2, P < 0.0005) groups, respectively, when compared with the same inflammatory conditions without CM ( Fig. 3C ). On the contrary, MMP13 gene expression was always lower than CTR in treated groups, and the addition of APS significantly decreased the gene expression of IL-1β + TNFα (d = −13.3, P < 0.0005) ( Fig. 3d ).
hADSCs
Comparison among Groups without APS
Morphology of hADSC micromasses revealed compact structure, large dimensions, and almost oval shape for CTR and OSF (CTR section area: 1.4 × 106 µm2), and a little irregular aspect for IL-1β + TNFα. ( Fig. 4a , c , and E ). Alcian blue staining quantification revealed similar content of cartilage matrix components among the 3 groups ( Fig. 2A ). Area of micromass section was smaller than CTR when grown in inflammatory environments (IL-1β + TNFα: d = −18.5, P < 0.0005; OSF: d = −15.0, P < 0.0005), while the cellular density resulted in general low, but higher in IL-1β + TNFα group than CTR (IL-1β + TNFα: d = 3.4, p = 0.0123) ( Fig. 2B and C ).
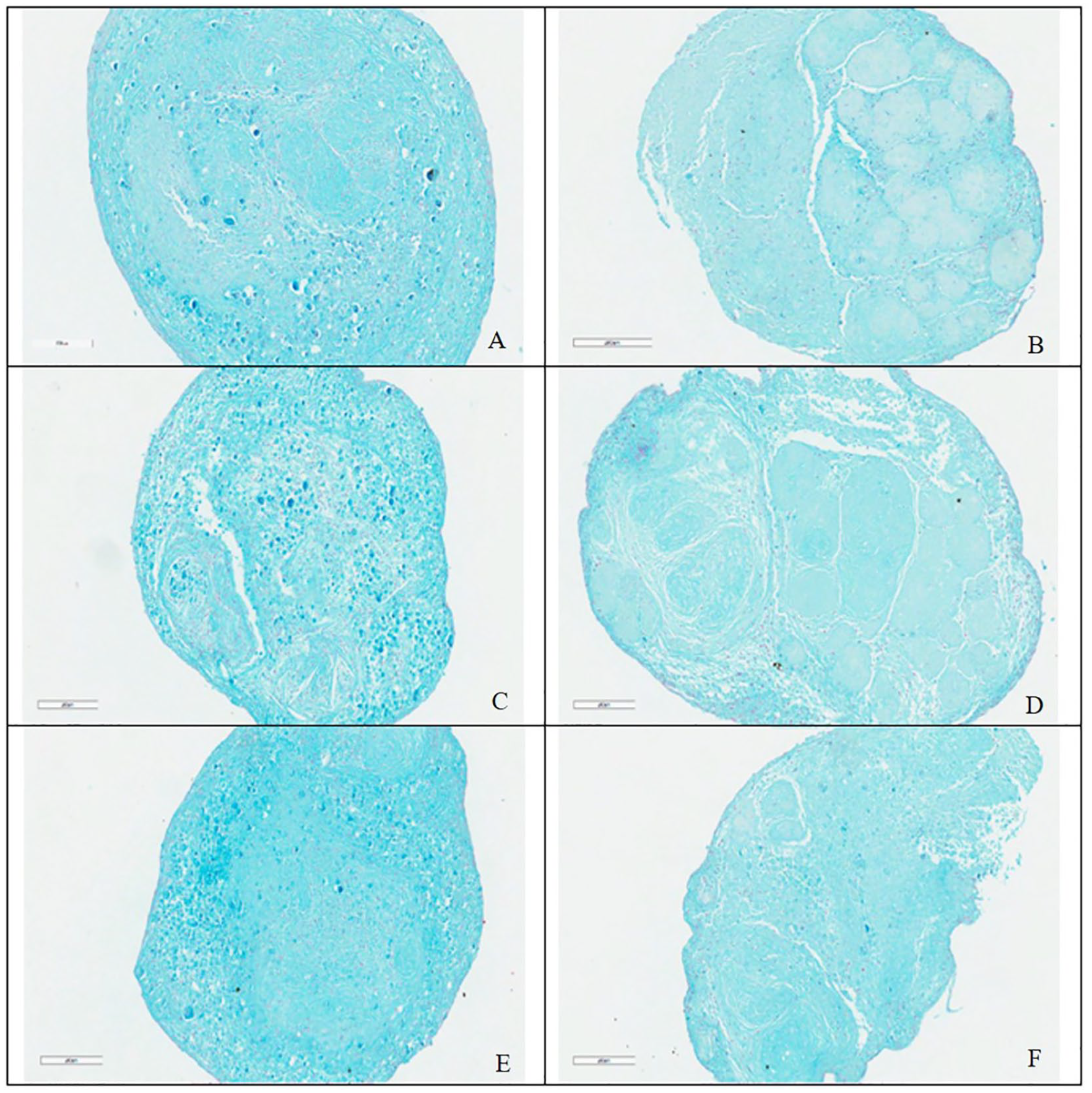
Histological images of ADSCs in CTR (
The trend of SOX9 and ACAN gene expression was similar to what observed for BMSCs, with a more pronounced decrease in SOX9 (d = −4.1) and ACAN (d = −3.6) for IL-1β + TNFα group compared with CTR (P < 0.0005) ( Fig. 3A and B ). Type II collagen protein showed similar amount in the 3 groups ( Fig. 2D ), and no expression by molecular biology. Also, MMP1 gene expression can be compared with what is observed for hBMSCs: the IL-1β + TNFα group was still the group that more expressed this effector of matrix degradation respect to CTR (d = 10.2, P < 0.0005) ( Fig. 3C ). OSF significantly reduced MMP1 expression (d = −2.6, P < 0.05). A significant decrease in MMP13 expression was found for OSF (d = −3.6, P < 0.0005) compared with CTR ( Fig. 3D ).
Comparison among Groups with and without APS
The histology of hADSC micromasses cultured with CM showed lower Alcian blue staining than those with no CM (d = −6.7, P = 0.0018), but no significant differences were observed among CM groups ( Fig. 2A ). Human ADSC micromasses cultured with inflammatory cytokines and CM showed an increase in irregularity of the section shape ( Fig. 4B , D , and F ); section areas of all CM-treated groups were lower that CTR (IL-1β + TNFα + CM: d = −5.2, P < 0.0005; OSF + CM: d = −18.7, P < 0.0005; and CTR + CM: d = −21.8, P < 0.0005) ( Fig. 2B ). When cultured with CM, IL-1β + TNFα, and OSF groups presented higher and lower section area results, respectively, in comparison with no CM ones (IL-1β + TNFα + CM: d = 13.3, P < 0.0005; OSF + CM: d = −3.7, P = 0.0067). Conversely, cellularity was higher in CTR + CM (d = −40.5, P < 0.0005) than CTR ( Fig. 2C ).
The expressions of tested genes in micromasses cultured with CM showed the same trend observed for hBMSCs ( Fig. 3 ). The addition of APS to inflammatory conditions produced significant decreases in SOX9 expression for OSF + CM (d = −6.9, P < 0.0005) and MMP1 expression for IL-1β + TNFα + CM (d = −18.6, P < 0.0005), and an increase in MMP1 expression for OSF + CM (d = 6.2, P < 0.0005) in comparison with the same no CM groups. Regarding type II collagen, CTR + CM and IL-1β + TNFα + CM were significantly higher than CTR (CTR + CM: d = 10.6, P < 0.0005; IL-1β + TNFα + CM: d = 6.1, P < 0.0005) and IL-1β + TNFα + CM versus IL-1β + TNFα (d = 7.9, P < 0.0005) ( Fig. 2D ).
Discussion
Clinicians use MSCs as a therapy for OA because of their potential to regenerate damaged cartilage and also to relieve joint inflammation and to modulate the immunologic response. In this regard, there are still many opinions. Some authors have proposed the idea that MSCs could differentiate to chondrocytes. Currently, several authors have expressed the opinion that MSCs, more important treatment role in OA, may be as a source of GFs having a trophic role on the surrounding tissue.41-43
To continue what was previously observed on the chondrogenic ability of BMSCs and ADSCs in an aggressive environment typical of OA, 3 in the present study, we cultured these cells in the same culture condition of the previous study, but in presence of APS. In this way, we assessed the behavior of both MSCs and the role of this blood derivative, a solution rich with PLTs, WBCs, and plasma.
Among the numerous alternative formulations of blood derivatives,20,21,44 APS is characterized by a high content of anti-inflammatory cytokines and IL1ra. O’Shaughnessey et al. 26 observed that OA patients, similarly to healthy donors, presented higher quantity of anti-inflammatory than inflammatory cytokines in APS with regard to peripheral blood. Of note, even if also the inflammatory molecules increase in APS (with regard to whole blood), this increase is at a lower fold if compared with the anti-inflammatory ones, thus keeping a favorable ratio.26,34
APS contained IL1ra (563.90 ± 203.33 pg/mL) and IL-1β (53.02 ± 19.15 pg/mL) with an IL1ra/ IL1β ratio of 10.6. IL1ra (1518.73 ± 213.63 pg/mL) and of IL-1β (27.90 ± 12.31 pg/mL) were detected in APS CM. IL1ra (252.65 ± 80.34 pg/mL) and IL-1β (15.36 ± 1.5 pg/mL) were in CM added with the inflammatory cytokines, thus confirming what was observed by O’Shaughnessey et al. 26 : a great increase of IL1ra after incubation with APS, which maintains high levels also after addition of IL1β.
All micromasses obtained with ADSCs and BMSCs in standard conditions were characterized by good shape and organization, partially modified in presence of inflammatory stimuli, as already noted in our previous study. 3 One month of culture confirmed to be a suitable experimental time to obtain solid micromasses, with a matrix rich in GAG. In this regard, a very low expression of SOX9 and ACAN genes, in particular in inflammatory conditions, was already observed by other authors,43,45,46 but it did not prevent us from observing an abundant matrix, sign of the timing of these genes, most likely turned off after 4 weeks.
An interesting review on the MSC potential for cartilage regeneration well described as an environment with the presence of IL-1β and TNFα, which inhibits SOX9, is detrimental for the complete chondrocyte differentiation. 46 Our observations agree with this picture, confirming also how the synovial pool may be less aggressive than the experimental condition in which IL-1β and TNFα are present at standard concentrations and in the absence of other physiological factors.
The presence of APS as CM did not increase the level of expression of SOX9 and ACAN, and the matrix was scarcer in all groups, and the micromass shape less regular and organized. Conversely, COLL II revealed a positive influence of APS on ADSCs, with a significant increase of this matrix component, in particular in CTR and IL-1β + TNFα conditions. The same effect was not evidenced in BMSCs micromasses.
The analysis of cell density clearly revealed a higher cell number, but a correspondingly slightly lower amount of GAG (Alcian staining) in BMSCs groups than ADSCs, without APS. This observation, together with the higher values of ADSC area measurement, leads to suppose a stronger activity of ADSCs and faster maturation of these micromasses. The presence of APS increased the cell number compared with CTR in OSF group of BMSCs, and in CTR and IL-1β + TNFα of ADSCs, perhaps taking the cells back to an earlier stage. Matuska et al. 25 referred to the anabolic GFs, such as EGF, IGF-I, PDGF-AB and -BB, VEGF, and TGF-β1, contained in APS the proliferative effects on bovine cartilage explants, not observed by adding rhIL1ra alone. The decrease of Alcian quantification in almost all groups of both cell types in presence of APS could confirm a global low influence of APS on chondrogenic differentiation. Exception was the most abundant COLL II observed in ADSCs micromasses. Liou et al. 47 set up a study that compared ADSCs and BMSCs in micromasses, treated with PRP at different concentrations and doses. The obtained findings were very similar to what is reported above: The number of doses of PRP was inversely proportional to the maturation of mesenchymal cells into chondrocytes.
Another interesting aspect was the different expression of MMP1 gene among the groups and between cell types: the stimulation with IL-1β + TNFα triggered a significant increase of MMP1 expression with regard to the standard condition in ADSCs micromasses, much more evident in BMSCs. Only a weak increase was observed in OSF condition of ADSCs and BMSCs, according with the results already reported in our previous research. 3 In ADSCs, the addition of APS induced an increase of MMP1 at gene level in CTR and OSF groups, but a sharp decrease in the most aggressive environment (IL-1β + TNFα) for both cell types. MMP13 seemed more stable and even less expressed than CTR, maybe being not involved in the remodeling process. Only in the IL-1β + TNFα group of BMSCs the expression of this gene resulted increased, as further demonstration of a more marked tendency of these cells to respond to inflammatory stimuli respect ADSCs. Typically, MMP13 is evaluated as a chondrocyte response to inflammatory cytokines and APS is considered an indirect inhibitor of this metalloprotease as well as of MMP1. 48
In the present study, we did not investigate chondrocytes but MSCs. However, we could still appreciate a modulation of the most aggressive environment carried out by the APS administration. In order to extend our observations on micromasses set up with both MSC types, we evaluated also some elements of a possible undesirable osteogenic differentiation, with particular attention on the role of APS. The trend of alkaline phosphatase (ALPL) gene expression showed to be very interesting: in BMSCs, under inflammatory conditions, ALPL strongly increased its expression, modulated by the presence of APS. ADSCs showed to be much less responsive to the ALPL expression in these aggressive environments (data not shown). Instead, vascular endothelial growth factor (VEGF) was more stimulated by APS in BMSC micromasses (data not shown). Finally, collagen I (COLL1a1) seemed to decrease in presence of APS. Considering that ALP, VEGF, and COLL I could be three of signal markers of incorrect differentiation, it is important to note that also for this aspect the ADSCs seemed more suitable to be used in chondrogenic regeneration and APS a partial positive support.
In conclusion, this experimental model confirmed to be useful in observing the chondrogenic differentiation of MSCs, especially in an environment mimicking, as much as possible, the osteoarthritic one. By analyzing results and images it was possible to infer that the pooled synovial fluid provided a milder condition than IL-1β and TNFα. The variability of the synovial fluid cytokine content in knee joints of 17 OA patients, and in particular the absence of IL-1β probably made this biological sample only partially aggressive. APS, in this condition, seemed to be less effective than in a strong inflammatory environment. As also observed in the previous study, 3 ADSCs seem to adapt and respond better to both inflammatory stimuli and APS than BMSCs.
Footnotes
Acknowledgments and Funding
We thank Zimmer Biomet, Zimmer GmbH (Sulzerallee 8, 8404 Winterthur, Switzerland) for providing NStride APS Kit and the Ministry of Health-Ricerca Corrente to the Rizzoli Orthopedic Institute. The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Blood samples were obtained from volunteers after local ethical committee approval (IOR Ethical Committee, prot. 0003409). Osteoarthritic synovial fluid was obtained from patients after local ethical committee approval (IOR Ethical Committee, No. 0003409).
Informed Consent
Blood samples were obtained from volunteers after signing of a research donation consent. Osteoarthritic synovial fluid was obtained from patients after written consent.
Trial Registration
Not applicable.
