Abstract
Objective:
To explore the utilization of platelet-rich plasma (PRP) for degenerative cartilage processes and evaluate whether there is sufficient evidence to better define its potential effects.
Design:
Systematic literature reviews were conducted in PubMed/MEDLINE and Cochrane electronic databases till May 2015, using the keywords “platelet-rich plasma OR PRP OR autologous conditioned plasma OR ACP AND cartilage OR chondrocyte OR chondrogenesis OR osteoarthritis (OA) OR arthritis.”
Results:
The final result yielded 29 articles. Twenty-six studies examined PRP administration for knee OA and 3 involved PRP administration for hip OA. The results included 9 prospective randomized controlled trials (RCTs) (8 knee and 1 hip), 4 prospective comparative studies, 14 case series, and 2 retrospective comparative studies. Hyaluronic acid (HA) was used as a control in 11 studies (7 RCTs, 2 prospective comparative studies, and 2 retrospective cohort). Overall, all RCTs reported on improved symptoms compared to baseline scores. Only 2 RCTs—one for knee and one for hip—did not report significant superiority of PRP compared to the control group (HA). Nine out of 11 HA controlled studies showed significant better results in the PRP groups. A trend toward better results for PRP injections in patients with early knee OA and young age was observed; however, lack of uniformity was evident in terms of indications, inclusion criteria, and pathology definitions in the different studies.
Conclusion:
Current clinical evidence supports the benefit in PRP treatment for knee and hip OA, proven to temporarily relieve pain and improve function of the involved joint with superior results compared with several alternative treatments. Further research to establish the optimal preparation protocol and characteristics of PRP injections for OA is needed.
Introduction
Background
Degenerative osteoarthritis (OA) is the most common form of arthritis and a major public health problem worldwide. It affects nearly 27 million adults in the United States alone. 1 Often causing pain, loss of function, and disability, it has a substantial social impact due to the rising mean age of the population, increasing rates of obesity, and a growing emphasis on physical activity in all age groups. 2 Attempts to analyze and understand the biology of this disease have not yet led to satisfactory results. OA is regarded as the result of a long chain of events; however, some parts in that chain are still a mystery and uncertainty exists regarding which part of this process should be targeted in order to inhibit and prevent disease progression. Cartilage possesses limited regeneration properties. Joint tissue damage and OA may result from loss of proper tissue homeostasis due to trauma or chronic repetitive overload, but could also be the result of metabolic and biological predispositions. 3 Numerous approaches have been proposed as noninvasive treatment options 4 ; however, none has shown any distinct ability to change the natural history of the disease. The fact that current conservative OA therapies are unable to supply consistent satisfying results is attributed mainly to insufficient understanding of the molecular basis of disease development and progression, as well as the lack of dynamic biomarkers that might reflect specific biological or pathological processes. There are currently no disease modifying medical interventions for OA, and treatments are primarily aimed at symptom relief. 5 In recent years, research efforts have shifted toward identifying key biochemical pathways that can be targeted therapeutically through biological intervention. One of the major foci of research in the past decade has been platelet-rich plasma (PRP), drawing much attention as an innovative and promising therapeutic modality. PRP therapies are considered a major potential breakthrough in the treatment of many medical conditions and are currently one of the hottest topics in regenerative medicine. Among these, PRP has emerged as a biological therapy for the treatment of cartilage injuries and for intraarticular application to address knee pain. The purpose of this study was to systematically review the scientific evidence for utilization of PRP in pathologic processes of cartilage and as a potential addition to the management strategy algorithm for OA. Using the existing data, our aim was to explore the rationale behind the utilization of PRP in pathologic degenerative cartilage processes and evaluate whether there is sufficient evidence to better define its potential effects.
Rationale behind the Use of PRP for Degenerative Cartilage Disease
PRP is defined as a biological therapeutic modality consisting of preparations containing a supraphysiological concentration of platelet and plasma proteins that accelerate the repair process by direct and indirect mechanisms. The aim of this treatment is to improve the reparability of endogenous cells. It is based on the intraarticular delivery of autologous platelet-rich preparations containing a large pool of growth factors and proteins stored in the alpha-granules of platelets. These growth factors and proteins, which have been implicated in tissue repairing mechanisms and have been found to take part in the regulation of articular cartilage, 6 are aimed at stimulating repair and replacement of damaged cartilage. Numerous growth factors in PRP stimulate cartilage matrix synthesis and counteract the effects of catabolic cytokines such as interleukin-1 and tumor necrosis factor-α. Growth factors, as a pool, have been found to possess synergistic effects on cartilage matrix synthesis7,8 and are known to induce further growth factor protein production by neighboring articular chondrocytes. 9 Basic science evidence supports the therapeutic potential of PRP. Chondrocytes treated in vitro with releasate from thrombin-clotted leukocyte-PRP (L-PRP) 10 resulted in significantly increased cell proliferation, synthesis rate, and accumulation of glycosaminoglycans and collagen type II (COL2) compared with controls. 11 In another study, human OA chondrocytes were removed from patients undergoing total hip arthroplasty, and PRP application induced the expression of proteins involved in chondrocytic differentiation compared with platelet poor plasma (PPP) and fetal bovine serum. 12 In this study, PRP was shown to be more effective than PPP or fetal bovine serum at increasing cell proliferation and inducing expression of genes associated with normal chondrocyte phenotype, including aggrecan and Sox-9 with sustained but not increased levels of COL2. Other basic science research demonstrated that PRP causes inhibition of the transactivating activity of nuclear factor-κB (NF-κB) and decreased expression of cyclooxygenase-2, which are important inflammatory regulators. 13 These findings suggest that PRP has the ability to stimulate local host cartilage and improve repair. Anitua et al. examined the effect of a platelet-derived preparation rich in growth factors (PRGFs) in OA synovial cell biology and revealed the ability of PRGF to stimulate HA synthesis, driving the secretion of HA by the synovial fibroblasts. 14 Synovial cells were isolated from 10 osteoarthritic patients and cultured in serum-free media (basal conditions) and exposed to either a platelet-poor preparation or PRGF (no or very low leukocyte penetration) for 72 hours. Cells activated with interleukin-1 for 48 hours were also exposed to PRGF. They found that PRGF significantly enhanced HA secretion compared with platelet-poor preparations. PRGF also significantly enhanced the secretion of HA induced by interleukin-1 activation, suggesting that pure platelet rich plasma could serve to induce chondroprotection and joint lubrication after intraarticular application even in the face of inflammation. These data support the therapeutic potential for PRP in the articular environment with anabolic effects on cartilage, mesenchymal stem cells, and synovial coverage.
Methods
Two independent reviewers performed a search using the PubMed/MEDLINE and Cochrane electronic databases till May 2015. Combinations of the terms “platelet-rich plasma OR PRP OR autologous conditioned plasma OR ACP AND cartilage OR chondrocyte OR chondrogenesis OR osteoarthritis OR arthritis” were used. Our inclusion criteria were studies evaluating the effect of PRP or any blood product with platelet concentration higher than baseline values published in peer-reviewed journals in the English language. We included different variations of PRP preparations in this study: activated and nonactivated PRP, PRP releasate, and PRP gels. Of all the articles, we selected only articles reporting on clinical studies which had utilized PRP, PRGF, or autologous conditioned plasma (ACP) for the treatment of degenerative cartilage pathology or OA of the knee or hip joint in humans and those articles that were published in the English language. Exclusion criteria were (1) animal studies; (2) patients with previous surgical intervention (i.e., total knee arthroplasty or anterior cruciate ligaments reconstruction); (3) articles involving nonosteoarthritic indications (osteonecrosis, osteochondral lesions, etc.); and (3) articles not published in English. In addition, articles that used PRP as a supplemental treatment to alternative modalities like bone marrow aspirate concentrate, scaffold-based cartilage repair, and mesenchymal stem cells were also excluded, as well as articles that included PRP as augmentation for different cartilage-related procedures (microfractures, cartilage implantation). Reference lists of included studies were also reviewed to ensure that no relevant articles were overlooked/missed. Review articles, expert opinions, case reports, and letters to editors were excluded from this review. Two authors performed the literature search, and 4 authors independently reviewed the search results. For the search results, articles titles and abstracts were reviewed, and potentially eligible studies received full-text review. Final agreement on inclusion was discussed among all authors, and the main author had the definitive decision vote in controversial cases. The initial search included 288 articles, from which 259 were excluded after screening for duplicates, title, and/or abstract. The final result yielded 29 articles ( Fig. 1 ) in which PRP was a prime treatment and are described in brief in Tables 1 and 2 .
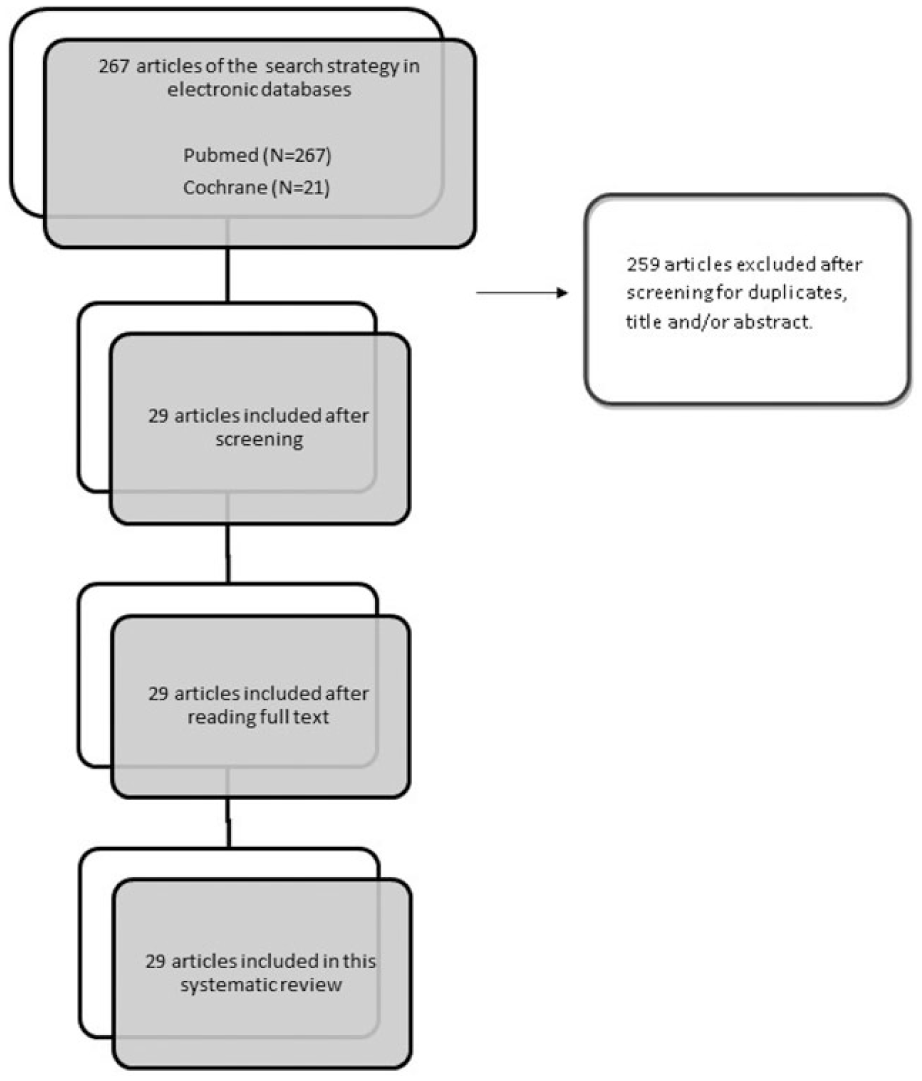
Study flow diagram.
Clinical Studies.
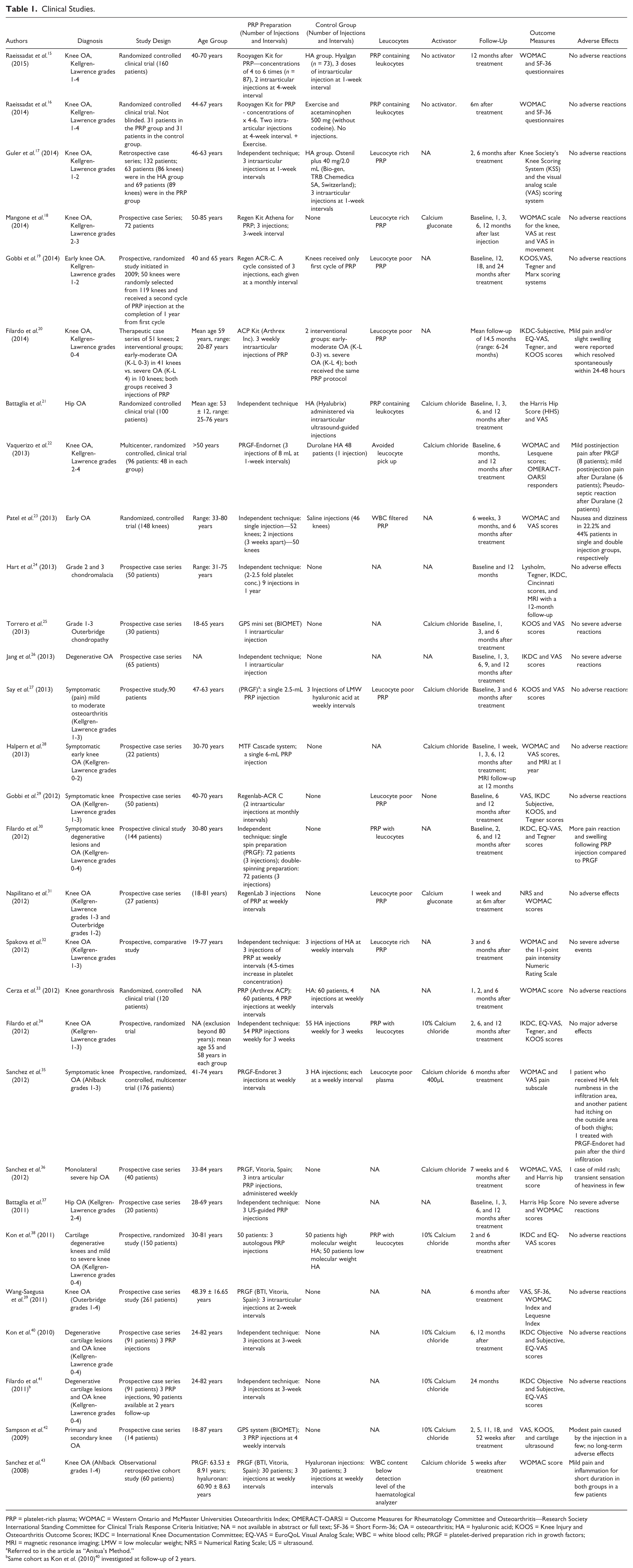
PRP = platelet-rich plasma; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index; OMERACT-OARSI = Outcome Measures for Rheumatology Committee and Osteoarthritis—Research Society International Standing Committee for Clinical Trials Response Criteria Initiative; NA = not available in abstract or full text; SF-36 = Short Form-36; OA = osteoarthritis; HA = hyaluronic acid; KOOS = Knee Injury and Osteoarthritis Outcome Scores; IKDC = International Knee Documentation Committee; EQ-VAS = EuroQoL Visual Analog Scale; WBC = white blood cells; PRGF = platelet-derived preparation rich in growth factors; MRI = magnetic resonance imaging; LMW = low molecular weight; NRS = Numerical Rating Scale; US = ultrasound.
Referred to in the article as “Anitua’s Method.”
Same cohort as Kon et al. (2010) 40 investigated at follow-up of 2 years.
Randomized Controlled Trials.
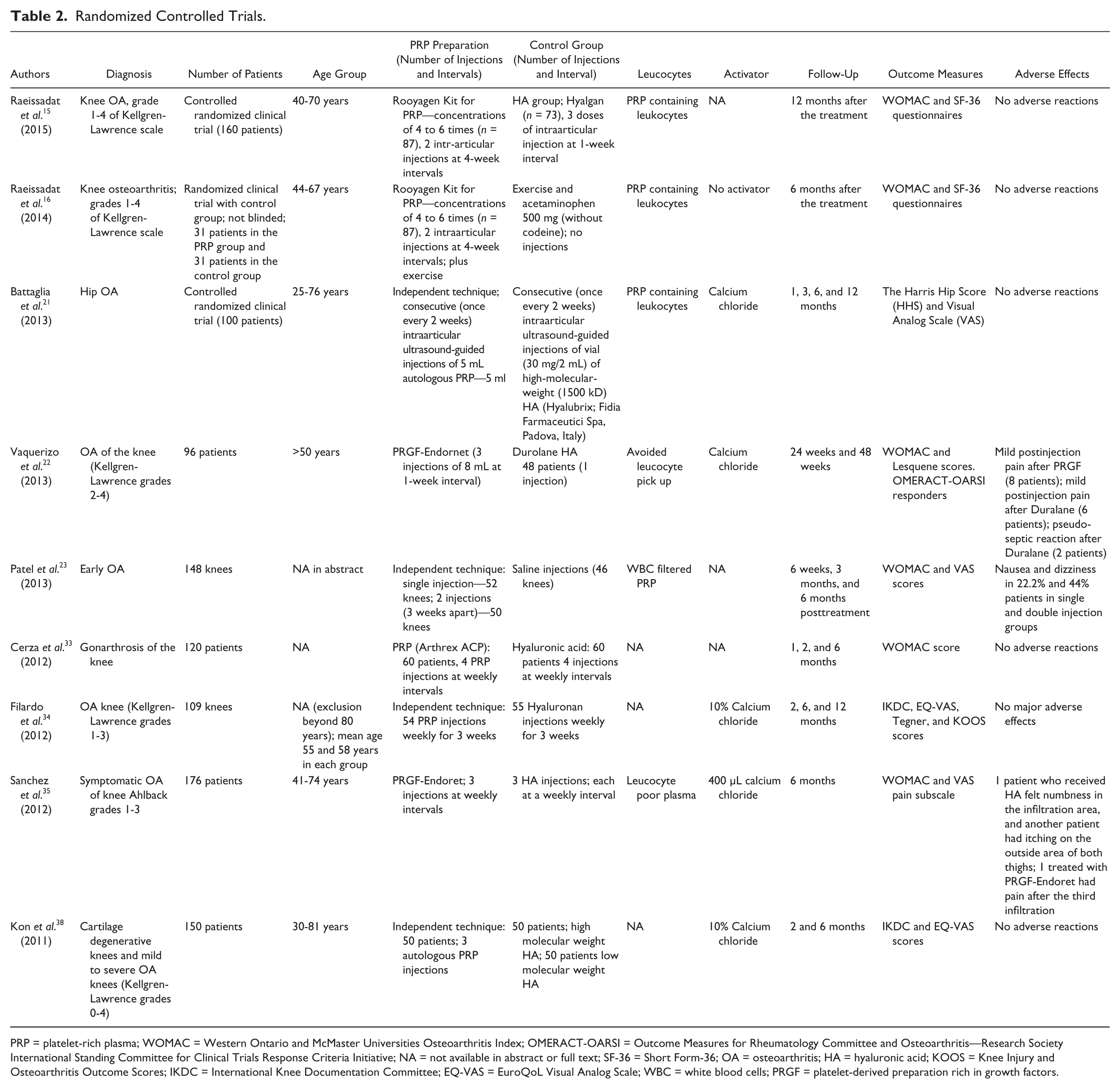
PRP = platelet-rich plasma; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index; OMERACT-OARSI = Outcome Measures for Rheumatology Committee and Osteoarthritis—Research Society International Standing Committee for Clinical Trials Response Criteria Initiative; NA = not available in abstract or full text; SF-36 = Short Form-36; OA = osteoarthritis; HA = hyaluronic acid; KOOS = Knee Injury and Osteoarthritis Outcome Scores; IKDC = International Knee Documentation Committee; EQ-VAS = EuroQoL Visual Analog Scale; WBC = white blood cells; PRGF = platelet-derived preparation rich in growth factors.
Results
The results of our systematic review yielded 29 clinical studies ( Tables 1 and 2 ). The majority of these studies (26) examined PRP administration for knee OA, while only 3 of the studies involved PRP administration for hip OA. The results included 9 prospective randomized controlled trials (RCTs), 4 prospective comparative studies, 14 case series, and 2 retrospective comparative studies. One of the studies, by Hart et al., was not clear with regard to whether a randomization process was involved and was therefore regarded as a prospective comparative study. Eight of the RCTs were conducted for knee OA and 1 for hip OA. Hyaluronic acid was used for the control groups in 7 RCTs, saline was used in 1 study, and an exercise program was used in 1 study. In the prospective comparative studies, HA was used for the control group in 2 studies, mesocaine was used in 1 study, 24 and in another study 19 the control group consisted of knees not receiving a second cycle of PRP injection at the completion of 1 year from first cycle. In the retrospective comparative studies group HA was used for the control group.
Age range in the studies was wide and not uniform. Various studies included patients as young as 18 years and as old as 85 years, with different age ranges. Four studies did not report age range.
Nineteen studies used the Kellgren Lawrence scale for the quantification of OA changes of which 7 studies included patients with Kellgren Lawrence grades 0 to 4, and 6 studies included patients with Kellgren Lawrence grades 0 to 3. Three studies used the Outerbridge condromalacia scale, while 1 study used the Ahlback scale, and 5 studies did not report the OA scale used. Within the RCTs group, 4 used the Kellgren Lawrence scale, 1 used the Ahlback scale, and the rest did not report the usage of any scale.
The follow-up period in the different studies ranged from 5 weeks to 24 months, with the majority of studies reporting outcomes with a follow-up period of 12 months (13 studies) and 6 months (12 studies). Only 2 studies reported a follow-up of 24 months.
The studies included in this review vary in the PRP preparation method and different production techniques yielding different resulting preparations. Twelve different preparation techniques—commercial and independent noncommercial methods—were used in the different studies. Table 3 summarizes the different types of solutions used in the studies included in this systematic review according to currently used classification systems.
PRP Preparation, Concentration, Number of Injections, Interval, and Classifications.
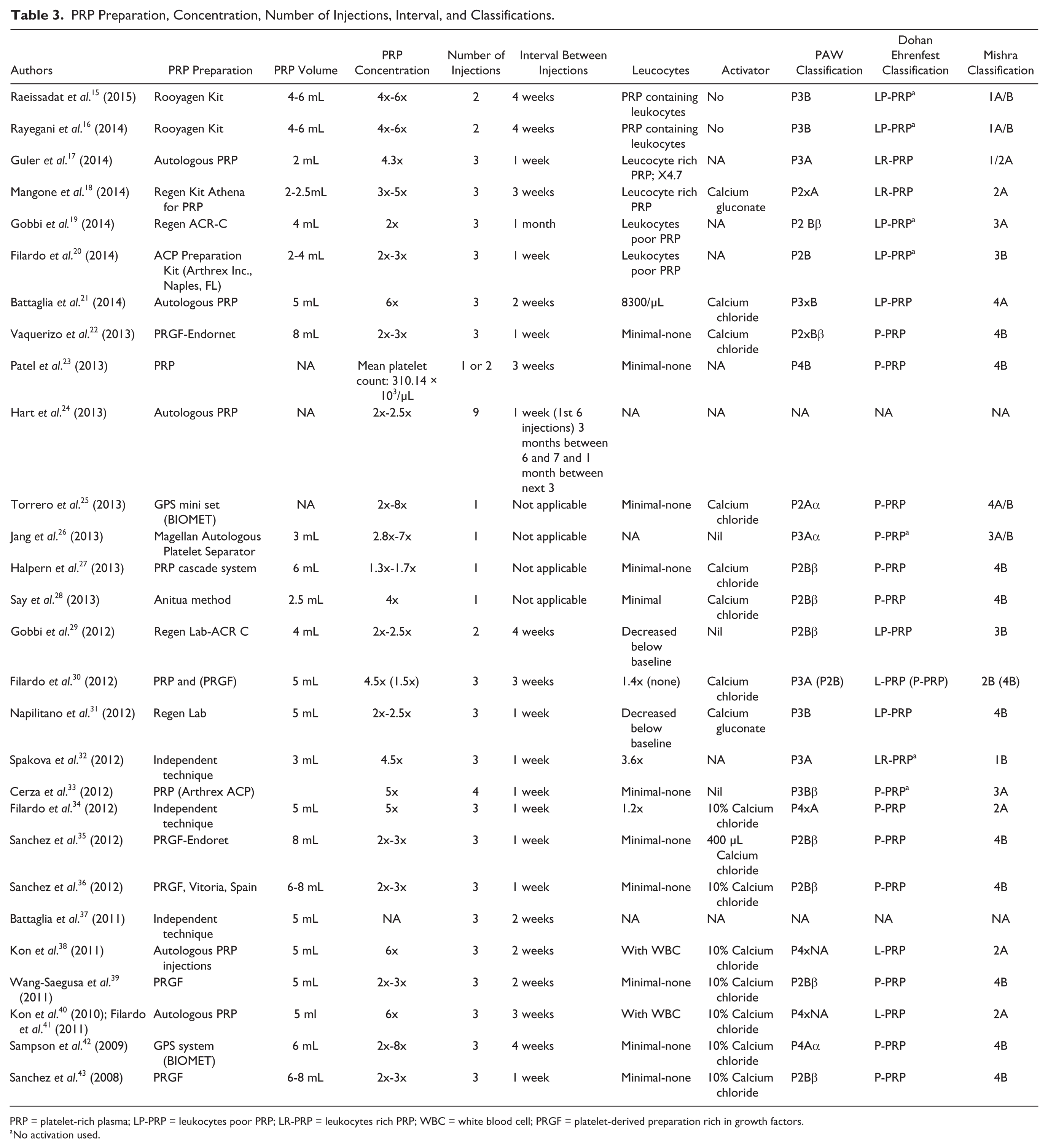
PRP = platelet-rich plasma; LP-PRP = leukocytes poor PRP; LR-PRP = leukocytes rich PRP; WBC = white blood cell; PRGF = platelet-derived preparation rich in growth factors.
No activation used.
Platelet concentration in these studies ranged between 1.3 times to 8 times the blood concentration, with the majority ranging between 2 and 4 times the blood concentration. Sixteen studies reported use of an activator prior to injection, and 14 of these studies used calcium chloride 10%. Fourteen studies used white blood cell (WBC)–containing preparations, 5 of which studies reported the use of PRP preparations with WBC content above blood concentration (1.2 to 4.7 times the blood concentration). Thirteen studies used WBC-free or minimal WBC preparations. Two studies did not report on WBC content.
As for adverse effects, no severe adverse effects of intraarticular PRP injections were reported in any of the studies. Most studies reported no adverse effects at all. Mild and self-limiting adverse effects were reported in a small number of patients in 7 studies (see Tables 4 and 5 ).
PRP Preparation Systems.
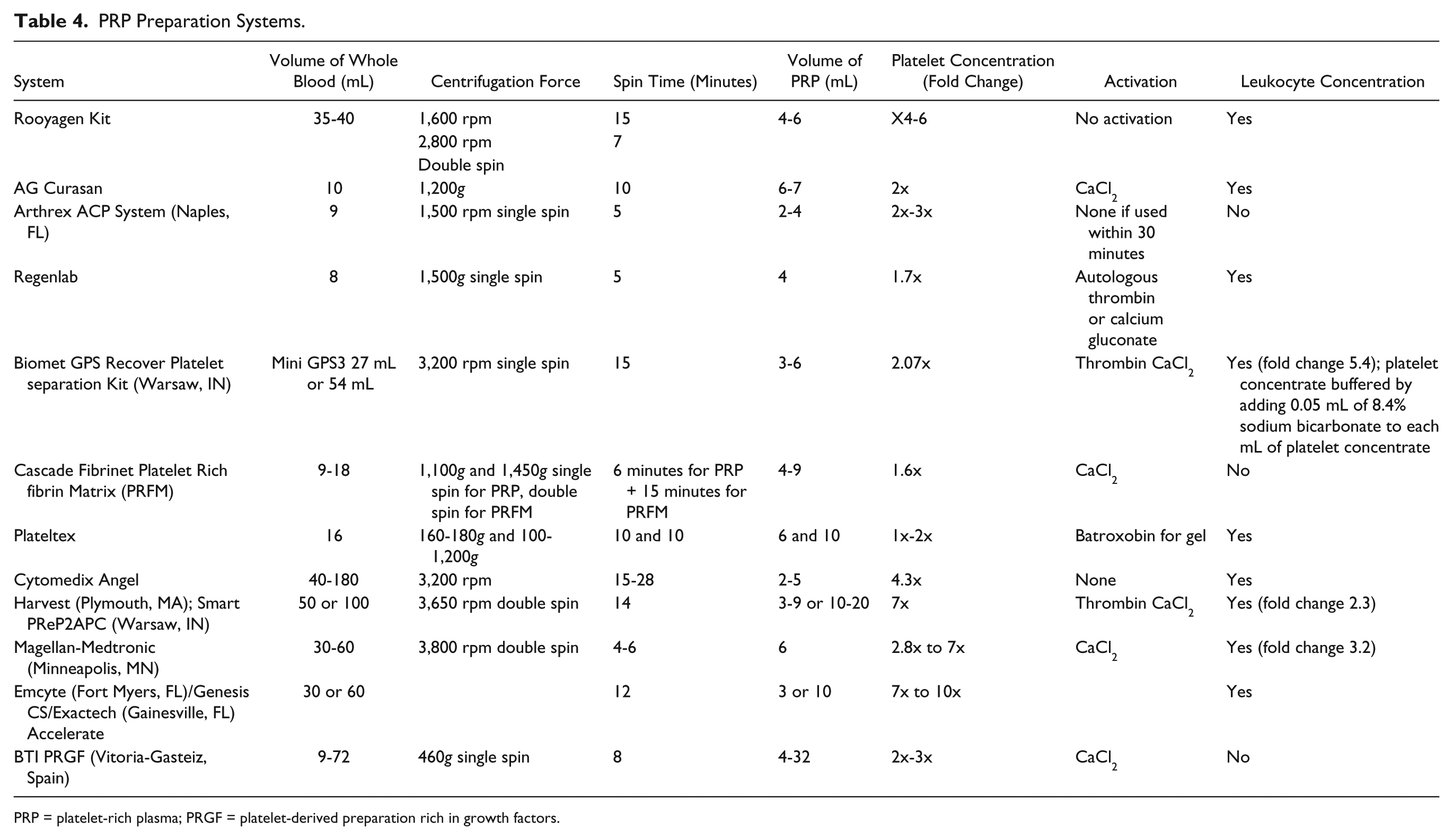
PRP = platelet-rich plasma; PRGF = platelet-derived preparation rich in growth factors.
Studies Included in This Systematic Review: Description and Results.
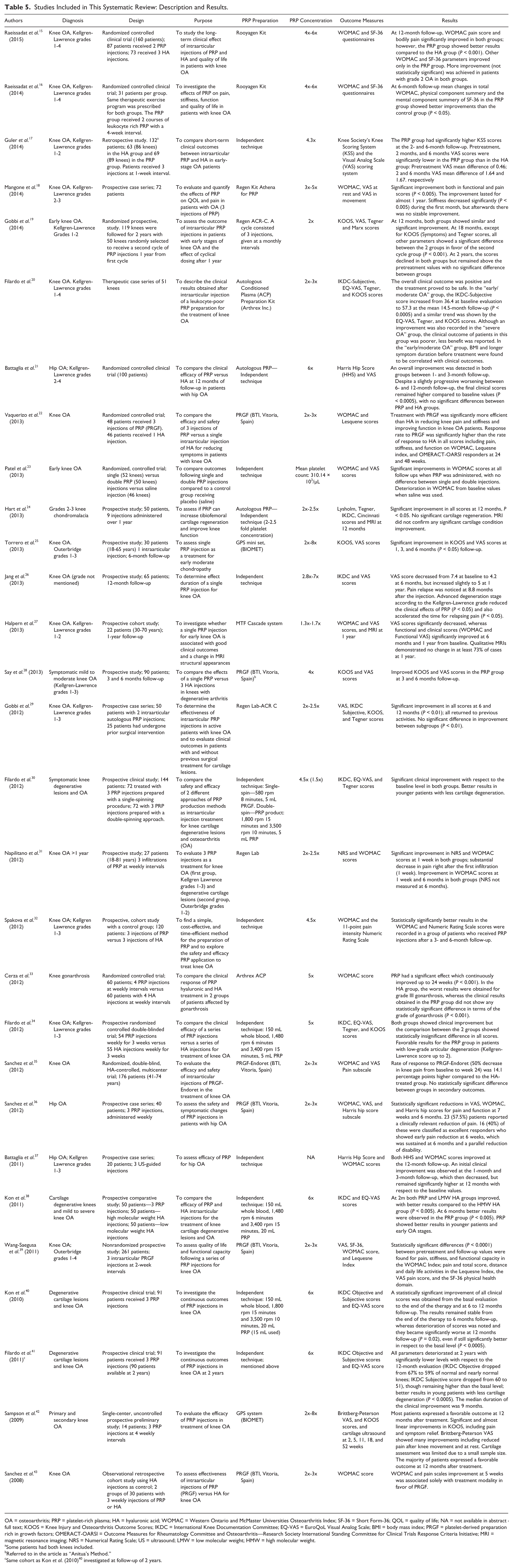
OA = osteoarthritis; PRP = platelet-rich plasma; HA = hyaluronic acid; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index; SF-36 = Short Form-36; QOL = quality of life; NA = not available in abstract or full text; KOOS = Knee Injury and Osteoarthritis Outcome Scores; IKDC = International Knee Documentation Committee; EQ-VAS = EuroQoL Visual Analog Scale; BMI = body mass index; PRGF = platelet-derived preparation rich in growth factors; OMERACT-OARSI = Outcome Measures for Rheumatology Committee and Osteoarthritis—Research Society International Standing Committee for Clinical Trials Response Criteria Initiative; MRI = magnetic resonance imaging; NRS = Numerical Rating Scale; US = ultrasound; LMW = low molecular weight; HMW = high molecular weight.
Some patients had both knees included.
Referred to in the article as “Anitua’s Method.”
Same cohort as Kon et al. (2010) 40 investigated at follow-up of 2 years.
Discussion
When analyzing the results of our search, what stood out most was the lack of uniformity in treatment protocol with respect to PRP preparation, administration, and dosing. While some articles have mentioned the specific company product that was used for the preparation and reported the platelets concentrations, others have not, presenting an obstacle in the attempts to understand and interpret the results. The large spectrum of variations in treatment protocols included more than 12 different preparation techniques (commercially available and independent noncommercially available methods), different administration protocols varying in number of total injections (from 1 injections up to 9), and intervals between injections (1 week to 1 month). A total of 3 injections was the common protocol used in 18 studies. A 1-week interval was the common protocol used in 10 studies. Additional major differences were noticed in the volumes of PRP injections, which varied from 2 mL to 8 mL; platelet concentrations, which varied from 1.3 times to 8 times the blood concentration; WBC concentration, which varied from none to 4.7 times the blood concentration; and the use of an activator, which varied from none, calcium gluconate, and calcium chloride (the most commonly used, 14 studies). In addition, it is important to mention that some studies did not report all of the above-mentioned parameters (PRP kit used, platelets and WBC concentration, PRP volume, activator used, etc.) as illustrated in Tables 1 and 3 . One study included in our results differed significantly in the administration protocol. 24 The authors administered 9 injections of PRP over a 12-month period (6 injections in 6 weeks, a 3-month interruption, followed by 3 additional injections at 1-month intervals). This raises questions regarding the number and frequency of injections, considering there have been reports of improved knee function following protocols that involved less injections and even after only a single injection. 25 The injections were initiated 6 weeks after a diagnostic arthroscopy, in which the grade of chondromalacia was determined. The study included a control group, in which mesocaine was injected at the same time points. The PRP preparation method, although described, was independent and data were lacking regarding the leukocytes profile in the preparation and activation. As portrayed in Table 1 , there was lack of consistency with regard to the PRP preparation techniques in the studies in our review. Eleven different types of PRP preparation techniques were used, with the PRGF technique (PRGF-Endoret, BTI, Vitoria, Spain) being the most common (6 studies). Filardo and Kon used a reproducible independent technique in 3 interventions, leading to 4 studies. The rest of the studies separately used 9 different preparation methods, 4 of which were independent and 5 were commercially available systems. There was also variability in terms of the resulting volumes in each injection in different studies, ranging from 3 mL up to 8 mL per injection. Two studies did not report the injected volume. Platelet concentration or level in relation to the basal platelet count were reported in only 12 studies, while information was lacking in 9 studies. The significant differences mentioned above emphasize the great difficulty of analyzing the results of the studies included in this review, as all the parameters mentioned have a direct biological effect on the activity of the administrated PRP preparation. When trying to understand the biological activity of PRP, it should be taken into consideration that PRP contains many prochondrogenic growth factors, including TGF-β1, IGF-1, bFGF, and bone morphogenetic protein-2; however, most preparations also contain high levels of antichondrogenic growth factors, such as VEGF, IGF binding proteins, PDGFs, and EGF. Investigations have shown that although chondrogenic growth factors such as TGF-β1 directly stimulate COL1 production in skin, synovium, and tendons, PRP preparations containing the same amount of TGF-β1 actually inhibit collagen production. 43 An insight to this paradoxical action suggests that the platelet concentration of PRP preparations is crucial to its potency and its effect on different conditions and injuries. Preparations containing only moderately elevated platelet concentrations have been suggested to induce optimal biological benefit, whereas lower platelet concentrations produce suboptimal effects and higher concentrations produce inhibitory effects. The “therapeutic dose” of PRP is considered at a range of at least 2 to 6 times higher than the normal platelet count. These observations reflect the complex molecular pool contained in PRP. It is therefore believed that maintaining what can be a delicate balance between pro-chondrogenic and anti-chondrogenic effects is crucial and the volumes of the PRP preparations and platelet concentrations play an important role, emphasizing the importance of optimal characterization of the studied PRP preparations. An additional factor contributing to the biologic characteristics of the PRP preparation is the activation method. Platelet activation triggers degranulation and release of the growth factors. The activation method (endogenous or exogenous with CaCl or CaG) determines the timing and cumulative release of the growth factors. However, this release may continue throughout the platelets’ 7- to 10-day life span, 1 thus offering the benefit of maintaining the normal physiologic ratios of these molecules.
Proper terminology for classifying and describing the many different variations of platelet concentrates are essential, especially when comparing results of several studies and analyzing the benefits of these treatments. The need for clarification, terminology, categorization, or classification was highlighted in recent years and yielded several classification systems. The first described and most comprehensive classification system, also known as the Dohan Ehrenfest classification, 44 is based on the presence of cell content (mostly leukocytes) and the fibrin architecture. Four main families were defined: Pure Platelet-Rich Plasma (P-PRP)—or Leukocyte-Poor Platelet-Rich Plasma—products are preparations without leukocytes and with a low-density fibrin network after activation; Leukocyte- and Platelet-Rich Plasma (L-PRP) products are preparations with leukocytes and with a low-density fibrin network after activation; Pure Platelet-Rich Fibrin (P-PRF)—or Leukocyte-Poor Platelet-Rich Fibrin—are preparations without leukocytes and with a high-density fibrin network; and Leukocyte- and Platelet-Rich Fibrin (L-PRF) products are preparations with leukocytes and with a high-density fibrin network. This terminology and classification are now considered as a basis of consensus in many fields. Another 2 classification systems were proposed in recent years, but are considered limited and applicable for sports medicine applications only. These are the Mishra classification and the PAW classification. Mishra et al. 45 proposed a classification that takes into consideration the presence of leukocytes, activation of platelets, and platelets concentration. This classification established 4 types of PRP: type 1 PRP is an L-PRP solution, type 2 PRP is an L-PRP gel (with activation), type 3 PRP is P-PRP solution, type 4 PRP is a P-PRP gel (with activation). Each type can be described as an A or B subtype. A subtype is 5 times or more the blood concentration of platelets, and B subtype is less than 5 times the blood concentration of platelets. The PAW classification 46 is similar to the Mishra classification and is based on the absolute number of platelets, the manner in which platelet activation occurs, and the presence or absence of white cells. Table 3 illustrates the different PRP preparations used in the studies included in this review according to currently used classifications.
Only 2 studies were PRP controlled, one comparing different PRP administration protocol: 1 versus 2 PRP injections 23 and the other comparing different preparation method: single spinning versus double spinning procedures. 30 Both studies showed no significant changes between groups. These studies emphasize the fact that current literature is lacking randomized controlled studies aimed to introduce the optimal PRP treatment (preparation characteristics, administration protocol, etc.) for knee or hip OA.
Randomized Controlled Trials
Nine RCTs were included in the results,15,16,21 -23,33 -35,38 3 of which were double blinded ( Table 2 ). Hyaluronic acid was used for the control groups in 7 RCTs, saline was used in 1 study, and an exercise program was used in another.
Eight studies targeted knee OA whereas 1 study targeted hip OA. 21 The follow-up period in these studies ranged from 6 to 12 months.
Overall, all RCTs reported on improved symptoms at the last follow-up when compared to the baseline scores; however, 2 RCTs—one for knee OA 34 and one for hip OA 21 —did not report significantly superior results for the PRP group compared to the control group (HA). All other studies showed significantly better results for the PRP group.
In a multicenter study, 35 the efficacy and safety of intraarticular injections of PRGF were evaluated in 176 patients with knee OA (aged 41-74 years). At 6-month follow-up, the PRGF group had significantly more patients who achieved greater than 50% decrease in the WOMAC (Western Ontario and McMaster Universities Osteoarthritis Index) pain score, compared to the HA group; however, although absolute WOMAC and VAS (Visual Analog Scale) scores were better in the PRGF group, there was almost no statistical significance compared to the HA hyaluronan group. In a double-blinded RCT, 34 Filardo et al. found no statistically significant difference between the PRP and the HA treated groups. A trend favorable of the PRP group was only found in patients with low-grade articular degeneration (Kellgren-Lawrence score up to 2) at 6 and 12 months of follow-up. In another RCT, Cerza et al. 33 compared treatment with ACP in 60 patients with knee OA to 60 patients treated with HA. The PRP showed a significant effect using the WOMAC score, which continuously improved up to 24 weeks (P < 0.001). In the HA group, the worst results were obtained for grade III gonarthrosis, whereas the clinical results obtained in the PRP group did not show any statistically significant difference in terms of the grade of gonarthrosis (P < 0.001). In their RCT, Patel et al. 23 used saline injections for the control group. The study included 78 patients with bilateral knee OA, divided into 3 groups: single PRP injection (n = 52 knees); 2 PRP injections (n = 50 knees); single saline injection (n = 46 knees). Significant improvement in WOMAC score was recorded at all follow-ups (1.5, 3, and 6 months) when PRP was administered, compared to the saline group, with no difference between the single and double injections groups. An interesting finding in this study was that the improvement was maximal at first follow-up with deterioration in the second and third follow-ups, suggesting a temporary nature of the benefits of PRP injections. In their multicenter RCT, Vaquerizo et al. 22 compared 3 injections of PRGF to one single intraarticular injection of HA. Each group included 48 patients. They reported that PRGF was significantly more efficient than treatment with Durolane HA in reducing knee pain and stiffness and improving function in patients with knee OA. The rate of response to PRGF was significantly higher than the rate of response to HA for all scores including pain, stiffness, and function on the WOMAC, Lequesne index, and OMERACT-OARSI responders at 24 and 48 weeks. In the most recent RCT, 15 Raeissadat et al. compared 2 intraarticular injections of PRP (Rooyagen Kit) to 3 injections of Hyalgan HA for patients with knee OA. The PRP group included 87 patients and the HA group 73 patients. At the 12-month follow-up, WOMAC pain score and bodily pain significantly improved in both groups; however, better results were determined in the PRP group compared to the HA group (P < 0.001). Other WOMAC and SF-36 (Short Form-36) parameters improved only in the PRP group. Another interesting RCT by Raeissadat et al. 16 compared intraarticular PRP injections and a prescribed exercise program with a prescribed exercise program alone. After 6 months, the mean changes of total WOMAC, physical component summery, and mental component summery of SF-36 in the PRP group showed better improvement than the control group (P < 0.05). Only 1 RCT by Battaglia et al. 21 investigated intraarticular injections of PRP and HA in hip OA. One hundred patients with symptomatic hip OA were randomly assigned to 1 of 2 groups: group A received PRP and group B received HA intraarticular injections. Patients were evaluated at baseline and after 1, 3, 6, and 12 months using the Harris Hip Score (HHS) and visual analog scale (VAS). An overall improvement was detected in both groups between 1- and 3-month follow-up. Despite a slightly progressive worsening between 6- and 12-month follow-up, the final clinical scores remained higher compared with baseline (P < 0.0005), with no significant differences between PRP and HA.
HA Controlled Studies
Nine out of 11 HA controlled studies (7 RCTs, 2 prospective comparative studies, and 2 retrospective cohorts) showed significantly better results in the PRP groups. In most of the studies, improvement in pain and functional scales was noted in both the HA and PRP groups, with significantly better results in the PRP groups. Most studies in this subgroup followed the participants up to 6 months (6 studies), while 4 studies had a follow-up of up to 12 months. One study followed the patients only for 5 weeks. 43 In a prospective double-blinded RCT 34 both groups showed clinical improvement, and the comparison between the 2 groups showed statistically insignificant difference in all scores. In a recently published randomized controlled clinical trial (160 patients), 15 87 patients received 2 PRP injections and 73 received 3 HA injections. At the 12-month follow-up, WOMAC pain score and bodily pain significantly improved in both groups; however, better results were determined in the PRP group compared to the HA group (P < 0.001). Other WOMAC and SF-36 parameters improved only in the PRP group. More improvement (not statistically significant) was achieved in patients with grade 2 OA in both the groups.
Prospective Case Series
Positive results for PRP treatment in knee and hip OA was evident in all 14 prospective case series studies included. These studies showed improvement in pain and functional performance compared to pretreatment measurements for follow-ups of up to 24 months. The majority of studies followed the patients up to 12 months. However, a deterioration in the positive effect of PRP was also noted during follow-up time. Kon et al. 40 showed a 9-month median duration of clinical improvement for knee OA patients treated with PRP. Despite this deterioration, functional and pain scores remained higher than the basal level of pretreatment in all studies.
Follow-Up Period
The majority of studies reported a follow-up period of 12 months (13 studies) or 6 months (12 studies). The shortest follow-up was 5 weeks in one study. 43 In a prospective study including 91 patients, a follow-up of 24 months has been reported.40,41 In this study group, patients received 3 intraarticular PRP injections at monthly intervals showed symptomatic improvement at 12 months but significant worsening of symptoms at 2 years with respect to the 12-month evaluation (IKDC [International Knee Documentation Committee] objective decreased from 67% to 59% of normal and nearly normal knees; IKDC subjective score decreased from 60 to 51), although they remained higher than the basal level. This is an interesting finding and reiterates the need for studies with longer follow-ups. A closer analysis of the data in this study revealed that older patients had a weaker response compared to younger patients (P = 0.049) and that only 30% of patients over 65 years of age with advanced OA showed significant improvement. In another study with a follow-up of 24 months, 50 knees were randomly selected from 119 knees and received a second cycle of PRP injection at the completion of 1 year from first cycle. At 12 months, both groups showed similar and significant improvement. At 18 months, except for KOOS (Knee Injury and Osteoarthritis Outcome Score–Symptoms) and Tegner score, all other parameters showed a significant difference between the 2 groups in favour of the patients who had received the second cycle (P < 0.001). At 2 years, the scores declined in both groups but remained above the pretreatment value with no significant difference between the groups. This study raises many questions with regard to the temporary effect of PRP treatment and the proper ways of prolonging its positive effect.
Level of OA
A trend toward better results for PRP injections in patients with early knee OA changes and young age was observed in several studies.15,20,26,30,34,38 This might suggest that PRP treatment should be considered more seriously in this subgroup. It is important to mention that improved outcomes have been shown in up to grade 3 OA (according to the Kellgren-Lawrence classification).25,26
Lack of uniformity was evident in terms of indications, inclusion criteria, pathology definitions, and classifications used in the different studies. In the knee studies, not all authors reported the degree of OA or degeneration, using a general description such as “degenerative joint disease,” or “primary and secondary OA,” or even “osteoarthritis,” whereas others varied in the classifications used to quantify the level of OA. Another variable posing difficulty in uniformly assessing the data was regarding the methods of reporting the level of OA treated. The Outerbridge classification 47 was used to describe the lesions in 2 studies,4,25 and another study 35 used the Ahlback classification, 48 whereas the Kellgren-Lawrence classification 49 was used in most others (18 studies). Some studies have simply used the broad term “early to severe OA.”23,33,26 Although most authors used the Kellgren-Lawrence classification, there was variability in the grades included in the studies, with some focusing only on grades 0 to 2 and others including also grade 3 and even grade 4. Six studies included patients with grade 0 Kellgren-Lawrence,20,27,30,38,40,41 meaning symptomatic knees with no radiographic signs of OA, which could potentially add additional confounding factors to the results. Not all studies describe the distribution of the study population according to the OA grades. Although these studies have targeted degenerative cartilage pathology, the range of the age group included has been as wide as from 18 years to 81 years,25,31 which could possibly generate a bias in the results.
Activator Use
Fourteen studies reported use of an activator right before PRP injection, with the majority 14 using calcium chloride 10%. Platelet activation triggers degranulation and release of the growth factors. The activation method determines the timing and cumulative release of the growth factors. However, this release may continue throughout the platelets’ 7- to 10-day life span, 1 thus offering the benefit of maintaining the normal physiologic ratios of these molecules.
Imaging Evaluation
Two studies were using imaging modalities to evaluate cartilage changes following PRP administration. In an uncontrolled prospective study, in which 14 patients with knee OA were treated with PRP, the authors reported improvement using ultrasonographic measurement of femoral articular cartilage thickness in 6 out of 13 patients at 6-month follow-up. 42 In a prospective study by Halpern et al. 27 involving 22 patients, qualitative magnetic resonance imaging was performed at baseline and at 1 year of follow-up, demonstrated no change in at least 73% of cases at 1 year. Although these studies involved a small sample size and a short-term follow-up period, they point out the need for a more objective measure to evaluate the effects of PRP on cartilage, as none of the other studies used an arthroscopic or consistent radiological evaluation after treatment administration to assess cartilage regeneration and tissue histopathology for regenerative features.
Adverse Effects
Only 7 studies reported on mild and self-limiting adverse effects in a small number of patients.
Filardo et al. 20 reported mild pain and/or slight swelling, which resolved spontaneously within 24 to 48 hours, without reporting number of patients. Vaquerizo et al. 22 reported mild postinjection pain after PRGF injection in 8 patients (out of 48). Patel et al. 23 reported nonspecific 19 adverse effects during the course of PRP treatment (148 knees). These included local pain, stiffness, syncope, dizziness, headache, nausea, sweating, and tachycardia, all self-limited. Filardo et al. 30 reported temporary pain and swelling reaction for the PRP treatment (number of patients not mentioned). Spakova et al. 32 documented temporary mild worsening of pain in the knee joint after application of PRP in 6 cases (out of 60), which was spontaneously resolved after 2 days. Sporadic adverse effects of temporary mild rash, local numbness, and itching sensation were also reported.35,36
PRP for Hip OA
Only 3 studies21,36,37 have been conducted to assess the efficacy of PRP for OA of the hip joint. While the first, published in 2011, showed clinical improvement in the short term, follow-up at 1 year was not as good. In a larger group of 40 patients, Sanchez et al. 36 reported 57.5% patients had clinically relevant relief of pain, assessed by the WOMAC subscale. Of these, 40% were classified as excellent responders who showed early pain reduction at 6 weeks, which was sustained at 6 months, with a parallel reduction of disability. A notable observation in this study was that 10 of the 11 patients in which treatment was not effective had a higher grade of hip OA (Tonnis grade 3). The authors concluded that PRP was effective for the management of pain from OA of the hip. However, the criteria for significance defined in the study was a 30% reduction in pain and disability, which, as shown by Richette et al., 50 can also be achieved with a placebo. In a recent randomized controlled clinical trial (100 patients), 21 Battaglia et al. compared the clinical efficacy of PRP versus HA at 12 months of follow-up in patients with hip OA. They showed that an overall improvement was detected in both groups between 1- and 3-month follow-up. Despite a slightly progressive worsening between 6- and 12-month follow-up, the final clinical scores remained higher compared with baseline (P < 0.0005), with no significant differences between PRP and HA.
Leukocyte Concentration
Leukocyte concentration in PRP preparations is another major confounding factor when analyzing the results included in this study. There is no doubt that leukocytes have a biological effect that must be understood. The difficulty of reaching an acceptable conclusion or trend when considering the optimal leukocyte concentration is based on the large spectrum of leukocytes content in PRP preparations injected in the studies reviewed and the lack of an acceptable and clear definition of what can be considered leukocyte rich or poor concentration. As reported in the studies included in this review, 5 studies used PRP preparations with leukocytes content above blood concentration (1.2 to 4.7 times the blood concentration), 9 used leukocytes containing preparations, 13 used minimal or no leukocytes preparations, and 2 studies did not report on leukocyte content. No trend related to leukocyte content and better or worse results was identifiable. This issue is of major relevance in future studies to better understand the role of leukocytes content in PRP preparations. It is essential that the terms “leukocyte rich” and “leukocyte poor” be defined more precisely and perhaps a third group of leukocyte blood concentration should be introduced for that matter.
Despite the obvious methodological variability, all studies seem to show overall positive results and a clinical benefit for the use of PRP. These benefits are emphasized in RCTs when compared to HA as a control15,22,33 -35 and even saline. 23 The duration of these benefits is still very controversial, as long-term follow-up studies are lacking, and many of the existing studies report only up to 6 months of follow-up. Studies with 1 year of follow-up are inconsistent in terms of the outcomes at this time point, with some reporting an improvement trend and others already showing deterioration at this time point. One study 26 observed relapsed pain 8.8 months after PRP treatment. This is an important aspect when assessing the benefits of PRP treatment, suggesting it has a limited or short-term effect that might necessitate further treatment cycles to maintain and prolong these effects.
It should be emphasized that no correlation was found between better results and any specific type of PRP characteristics and/or administration protocol. Any discussion involving PRP therapies should carefully acknowledge the differences between the preparations and the administration procedures used. Regardless of the fact that pure PRP and leukocyte PRP formulations are not comparable in terms of leukocyte content, platelet count, or plasma volume, the resulting improvements in pain and function were not exclusive to any type of formulation. This finding leaves the mystery of the optimal PRP treatment (protocol and formulation) as an exciting challenge for future research.
As described earlier, intraarticular PRP injections are as safe as other intraarticular injection treatments and present no significant risk for serious adverse effect. In a minority of patients, adverse effects were minor and temporary, with no long-term implications.
Conclusion
Current clinical studies evidence support the concept that PRP treatment can be beneficial for degenerative joint pathology in the knee and hip. It has been proven to temporarily relieve pain and function of the involved joint with superior results compared with several alternative treatments. Current variability in methodologies used in different studies and the numerous PRP preparation methods, in addition to the highly inconsistent systems to classify PRP formulations, make it difficult to establish firm and uniform recommendations and guidelines regarding which type of PRP to use and for which indications. A broad overview of the results clearly implies a positive influence of PRP on knee OA with encouraging clinical results in almost all the studies included in this review. The simplicity and safety of PRP use makes it an interesting and promising option for both researchers and clinicians. Further research with RCTs that will address the issues of the optimal preparation protocol and formulation of intraarticular PRP injections for OA of the knee, hip, and other joints, as well as longer follow-ups and larger population sizes is of major need.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
