Abstract
Objective
Rebamipide has antioxidant effects and is a drug with a local rather than systemic mechanism of action. Oxidative stress and inflammation in chondrocytes are the major factors contributing to the development and progression of osteoarthritis (OA). Since OA is mainly developed in weight bearing or overused joints, the locally sustained therapy is effective for targeting inflammatory component of OA. We investigated the effects of intra-articular injection of rebamipide loaded nanoparticles (NPs) in OA rat model.
Design
We fabricated rebamipide-loaded methoxy poly(ethylene glycol)-b-poly(D,L-lactide) (mPEG-PDLLA) and poly(D, L-lactide-
Results
Conclusions
Using a chemically induced rat model of OA, intra-articular delivery of rebamipide was associated with decreased local and systemic inflammatory response decreased joint degradation and arthritic progression.
Keywords
Introduction
Osteoarthritis (OA) is the most common chronic degenerative disease in the elderly and various factors affect the development and progression of OA. Continuous oxidative stress in chondrocytes is an important factor contributing to the development of OA. 1 Oxidative stress is produced by the imbalance between the production and clearance of reactive oxygen species (ROS), which is high in OA cartilage, causing chronic inflammation. 2 One of the oxidants, nitric oxide (NO), and ROS, plays a role in chondrocyte insensitivity to the anabolic actions of insulin-like growth factor-1, which leads to the pathological degradation of the cartilage extracellular matrix. ROS cause cellular damage, and excessive production of oxidants is associated with the apoptosis of cartilage chondrocytes. 3 Apoptotic cell death is associated with senescence, and oxidative stress-induced cell death is observed in OA chondrocytes. 4 In addition to aging, various inflammatory cytokines affect the development of OA, and one of the key cytokines, interleukin-1 beta (IL-1β), also stimulates the production of ROS. 5 Inflammatory cytokines, such as IL-1β, IL-6, and tumor necrosis factor-α (TNF-α) are highly upregulated in OA joints and expression of matrix degrading proteases leads to cartilage extracellular matrix degradation. 2 ROS and inflammation are considered as possible targets for the treatment of OA.
Oxidative stress is caused by an imbalance of ROS and antioxidant defense, and attempts have been made to suppress ROS production in OA. 6 Tenoxicam has an antioxidant effect and improves OA, 7 and celecoxib or hyaluronan can reduce nitrite and NO.7,8 In an animal study, statin were shown to be effective in improving OA through antioxidant action. 9 Rebamipide protects the gastrointestinal mucosa by inhibiting inflammation through an antioxidant mechanism that scavenges hydroxyl radicals and suppresses superoxide production. 10 In addition, rebamipide inhibits the signal transducer and activator of transcription factor 3, suppresses the nuclear factor (NF) kappa-B transcription factor, and inhibits the expression of the receptor activator of nuclear factor kappa-B ligand, preventing the progression of arthritis.10 -12 NF-kappa B activation is inhibited in the presence of antioxidants. 13 The antioxidant and anti-inflammatory effects of rebamipide have been applied to various diseases, such as liver injury, dry eye, and Behçet’s disease.3,11 Rebamipide also attenuates subchondral trabecular bone resorption and produces proteoglycans.14,15 In previous animal studies, systemic administration of rebamipide was found to be effective in improving arthritis.3,16 However, rebamipide is considered to have a local rather than systemic effect with respect to the mechanism of action. 17 Thus, topical rebamipide is used as a topical agent for the treatment of ocular dryness. 18
Currently, intra-articular injections are mainly administered with steroids and hyaluronic acid. Rebamipide may directly affect directly joints presenting OA
Methods
Materials
The mPEG-PDLLA (molecular weight [MW]: 5,000–20,000 Da) was purchased from Akina Inc. (West Lafayette, IN, USA). PLGA (50:50; Resomer® RG505) was supplied by Boehringer Ingelheim (Ingelheim, Germany). Rebamipide was purchased from the Tokyo Chemical Industry Co., Ltd. (Tokyo, Japan). Dimethyl sulfoxide (DMSO) and lipopolysaccharide (LPS) were obtained from Sigma-Aldrich (St. Louis, MO, USA). A cellulose-ester dialysis membrane (MW cut-off [MWCO]: 6–8 kDa) was obtained from Spectrum Laboratories (CA, USA). Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum, phosphate-buffered saline (PBS), IL-1β and penicillin-streptomycin were obtained from Gibco BRL (Rockville, MD, USA). Chondrocytes were purchased from Lonza Ltd. (Walkersville, MD, USA).
Fabrication of mPEG-PDLLA and PLGA NPs and Rebamipide-Loaded NPs
To fabricate the rebamipide-loaded NPs, mPEG-PDLLA (40 mg mL-1) and PLGA (160 mg mL-1) were dissolved in DMSO solution and slowly stirred for 30 min at room temperature, followed by the addition of rebamipide (0.2 or 2 mg for 1 and 10 mg [w/w] of the total polymer) and stirred for 24 h. The resulting solution was dialyzed against distilled water (DW) and lyophilized. The mPEG-PDLLA and PLGA NPs without rebamipide were similarly fabricated as the mPEG-PDLLA and PLGA NPs with rebamipide. The mPEG-PDLLA and PLGA NPs loaded with rebamipide (1 or 10 mg) are hereafter referred to as rebamipide (1 mg)/NPs and rebamipide (10 mg)/NPs. In addition, mPEG-PDLLA and PLGA NPs without rebamipide are hereafter referred to as NPs.
Characterization of Bare NPs and Modified NPs
We confirmed the shapes of NPs with or without rebamipide using a JEM-F2100 (JEOL Ltd., Japan) field emission-transmission electron microscope (FE-TEM). Before the FE-TEM exhibition, each specimen (100 µg) was placed in a tube with EtOH (1 mL) and scattered by a bath-type sonicator (Powersonic 405; 40 kHz, power: 350 W, Hwashin Tech Co., Ltd. Korea) for 1 h at 4°C. Each NP (10 µg) was carefully moved on to a TEM grid (CF200-Cu, Electron Microscopy Sciences, Hatfield, PA, USA), and the shape of each NP was monitored using FE-TEM at 200 kV. To perform particle range tests using dynamic light scattering (DLS), NPs with or without rebamipide were dispersed using a bath-type sonicator containing DW for 1 h at 4°C. The size distribution and zeta potential values were determined using a DLS instrument (Malvern Zetasizer 3000, Malvern, UK) equipped with a helium-neon laser at a wavelength of 633 nm. The zeta potential, the electrostatic potential at the slipping plane, is used to exam colloid-electrolyte interactions, and the basic principle is that particles with opposite charge surfaces are attracted, while particles of similar charge are repelled.22,23 Zeta potential is one of the main properties that can affect the stability and cell immersion of NPs by measuring the surface charge in suspension through electrophoretic light scattering. The higher zeta potential, the higher is the repulsion force between the particles and the lower the probability for a collision with subsequent aggregating. 24 To investigate the amount of rebamipide loaded on NPs, 10 mg of rebamipde (1 mg)/NPs and 10 mg of rebamipde (10 mg)/NPs were dissolved in 2 mL of dichloromethane. Then, PBS (2 mL) was added to the rebamipide/NPs solution. The rebamipide concentration was determined from the standard curve of various rebamipide solution using the following standard curve equation:
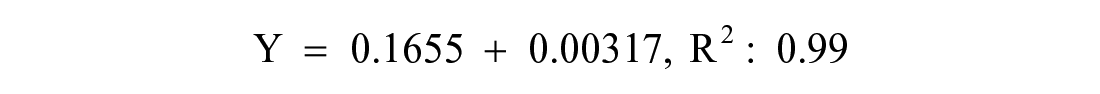
The drug concentration in each group was measured using a Flash Multimode Reader (VarioskanTM, Thermo Scientific, USA) at 315 nm.
Rebamipide Release from Rebamipide/NPs
To investigate the release of rebamipide from rebamipide (1 mg)/NPs and rebamipide (10 mg)/NPs, each concentration of rebamipide/NPs, distributed in 1 mL of PBS solution (pH 7.4), was added to a dialysis bag (MWCO 6–8 kDa). Each bag containing the substrate was carefully transferred into a 50 mL tube with 5 mL of PBS solution (pH 7.4), followed by gentle shaking (100 rpm) in a water bath at 37°C. At predetermined time intervals points (1, 5, 9, and 12 h, and 1, 3, 5, 7, 14, 21, and 28 days), the supernatant of each rebamipide/NPs was harvested and replaced with fresh PBS. The amount of rebamipide from the NPs was investigated by measuring the absorbance at 315 nm using a multimode reader.
Cytotoxicity Test
The cytotoxicity of NPs, rebamipide (1 mg)/NPs, rebamipide (10 mg)/NPs, and rebamipide (10 mg) solution was tested against chondrocyte on days 1 and 3. Cells (5 × 104 cells/well) were seeded into 96-well culture plates and allowed to adhere for 24 h, followed by the addition of DMEM in the presence or absence of 100 µg/mL NPs with or without rebamipide. The cell were treated with rebamipide solution as positive control. On the first and third day of incubation, the cells were washed with PBS and cell counting kit-8 (CCK-8) reagents (Dojindo, Tokyo, Japan) were added, followed by incubation for 1 h. The optical density was monitored at 450 nm using a Flash Multimode Reader (VarioskanTM, Thermo Scientific).
In Vitro Anti-Inflammatory Effects on Inflamed Chondrocytes
To examine the effect of rebamipide/NPs on the mRNA levels of pro-inflammatory cytokines in LPS + IL-1β-promoted chondrocytes, cells (1 × 105 cells/well) were seeded in 24-well plates and treated with LPS (10 ng/mL) and IL-1β (10 ng/mL) solutions and with NPs, rebamipide (1 mg)/NPs, and rebamipide (10 mg)/NPs (at a concentration of 100 µg/mL) or rebamipide (100 µg/mL) and incubated at 37°C in a humidified 5% CO2 atmosphere. LPS- and IL-1β-stimulated cells were used as positive controls, and non-stimulated cells were used as negative controls. The expression of IL-1β, IL-6, TNF-α, matrix metalloproteinase-3 (MMP-3), MMP-13, and cyclo-oxygenase-2 (COX-2) was measured. At 1- and 3-day time-points, cells were harvested for total RNA isolation. Total RNA was isolated using the RNeasy Plus Mini Kit (Qiagen, Valencia, CA, USA). For the reverse transcription of the total RNA (1 μg) into complementary deoxyribonucleic acid (cDNA), AccuPower RT PreMix (Bioneer, Daejeon, Korea) was used. All PCR amplifications were performed using AccuPower PCR PreMix (Bioneer, Daejeon, Korea). The primer sequences of the pro-inflammatory genes are described in the Supplementary Materials (Table 1). PCR amplification and detection were conducted using an ABI7300 Real-Time Thermal Cycler (Applied Biosystems, Foster City, CA, USA). The relative mRNA levels of inflammatory markers were normalized to those of glyceraldehyde 3-phosphate dehydrogenase (GAPDH).
All Primers of the Pro- and Anti-Inflammatory Component Target Genes Subjected to Real-Time PCR
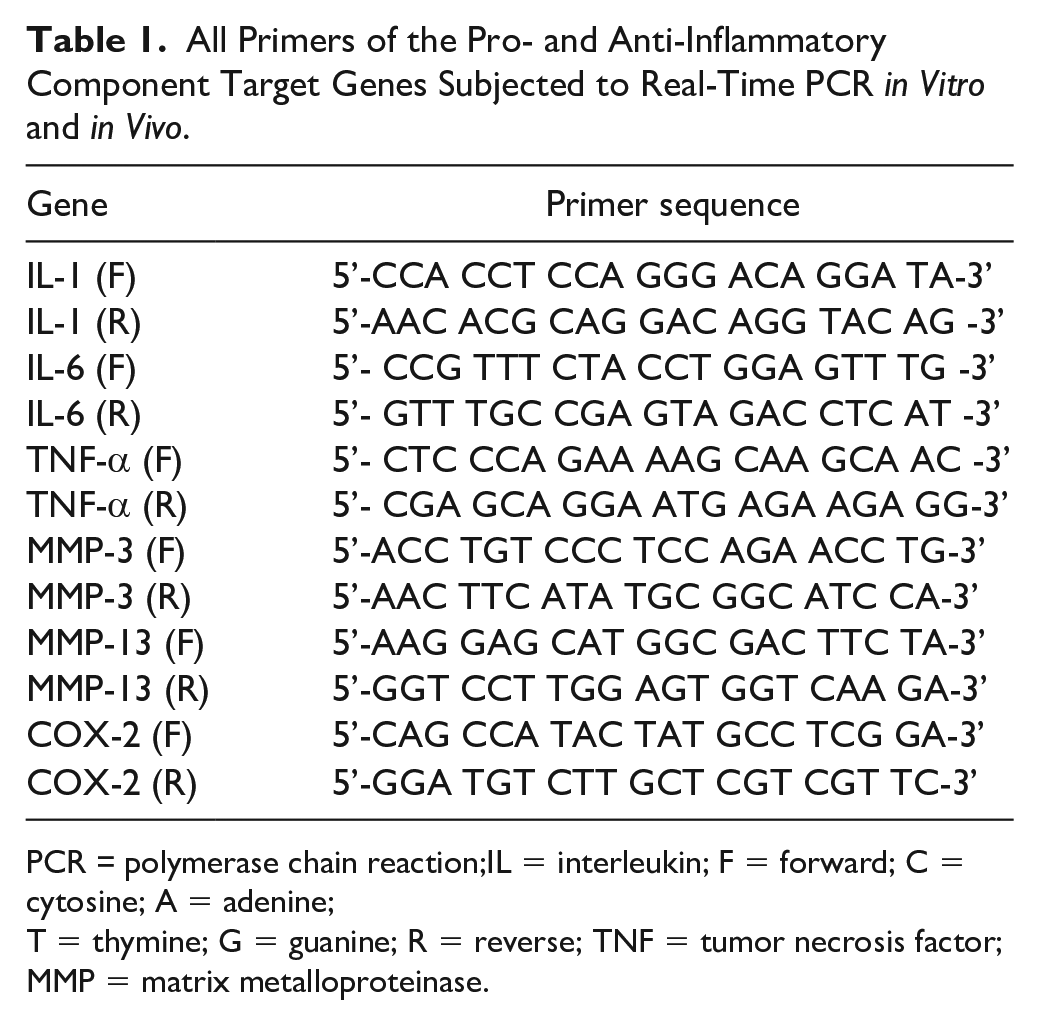
PCR = polymerase chain reaction;IL = interleukin; F = forward; C = cytosine; A = adenine;
T = thymine; G = guanine; R = reverse; TNF = tumor necrosis factor; MMP = matrix metalloproteinase.
OA-Induced Animal Model and Experimental Plan
For the
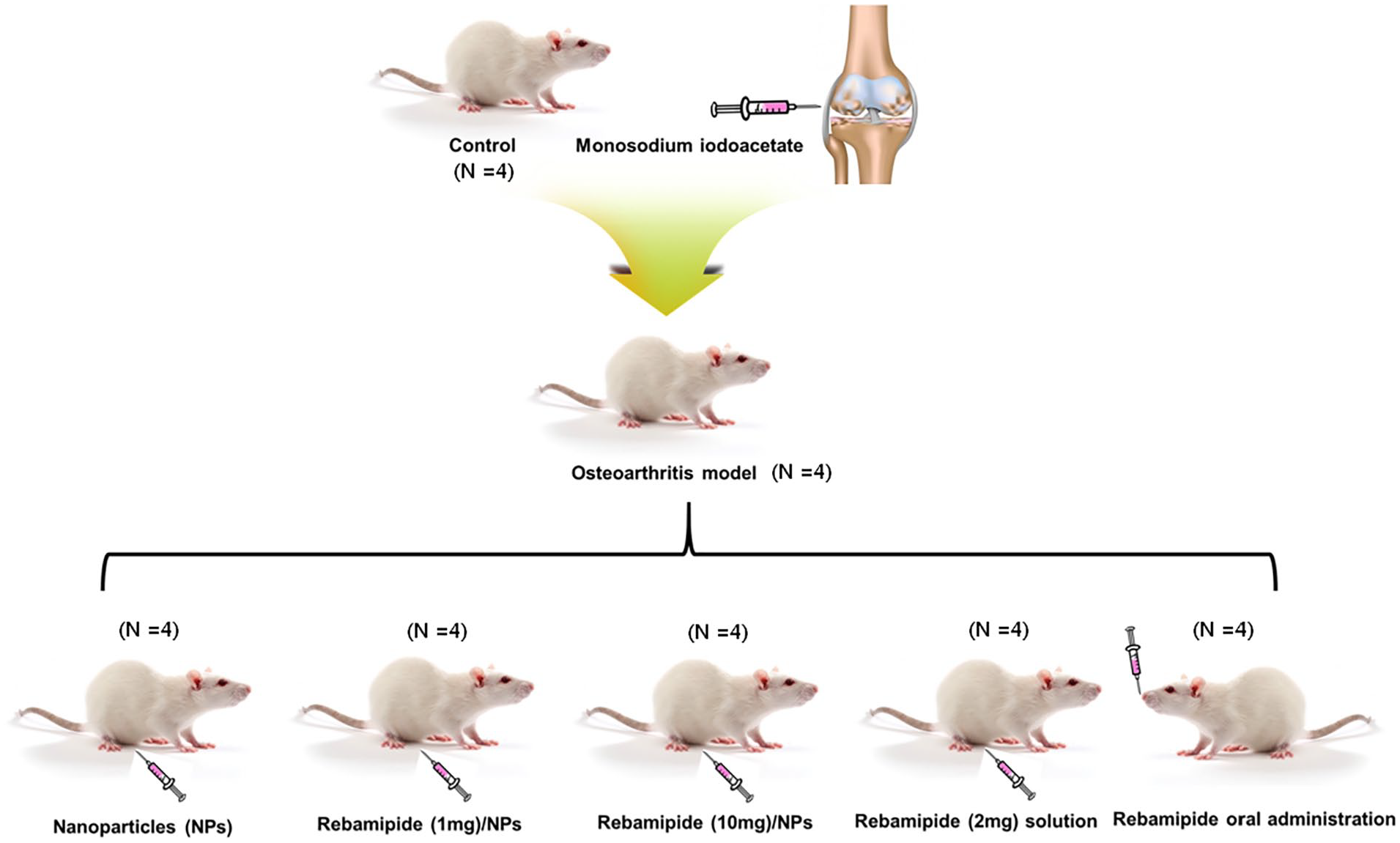
The experimental overview of the study. NPs = nanoparticles.
Histological Examination and Micro-CT
For
In Vivo Anti-Inflammatory Effects
Total RNA was extracted from the whole blood of rats using the QIAamp RNA Blood Mini Kit (Qiagen, Valencia, CA, USA) according to the manufacturer’s instructions at 4 and 8 weeks after the administration of NPs with or without rebamipide, rebamipide solution, and oral rebamipide. Then, 1 μg of total RNA was used for cDNA synthesis
Statistical Analysis
All tests were repeated 3 times. Data are expressed as mean ± standard deviation, and statistical significance was determined using one-way ANOVA in SigmaPlot (Systat Software, Inc., IL, USA)
Results
Characteristics of NPs with or without Rebamipide
The morphology of the NPs, rebamipide (1 mg)/NPs, and rebamipide (10 mg)/NPs were confirmed by FE-TEM. Each NP group exhibited by TEM had a spherical form and was nanosized. The average NP sizes and polydispersity indices were 241.70 ± 38.29 nm and 0.312 for NPs, 231.00 ± 40.80 nm and 0.294 for rebamipide (1 mg)/NPs, and 238.00 ± 34.07 nm and 0.307 for rebamipide (10 mg)/NPs (Fig. 2), respectively. The demonstrated zeta potential values were -14.20 ± 2.17 mV for NPs, -19.73 ± 1.11 mV for rebamipide (1 mg)/NPs and -33.97 ± 2.61 mV for rebamipide (10 mg)/NPs (Fig. 3). The loading amount (percentage) of rebamipide in the rebamipide (1 mg)/NPs and rebamipide (10 mg)/NPs was 7.73 ± 0.54 µg (77.33 ± 5.40%) and 69.08 ± 3.34 µg (69.08 ± 3.34%), respectively.
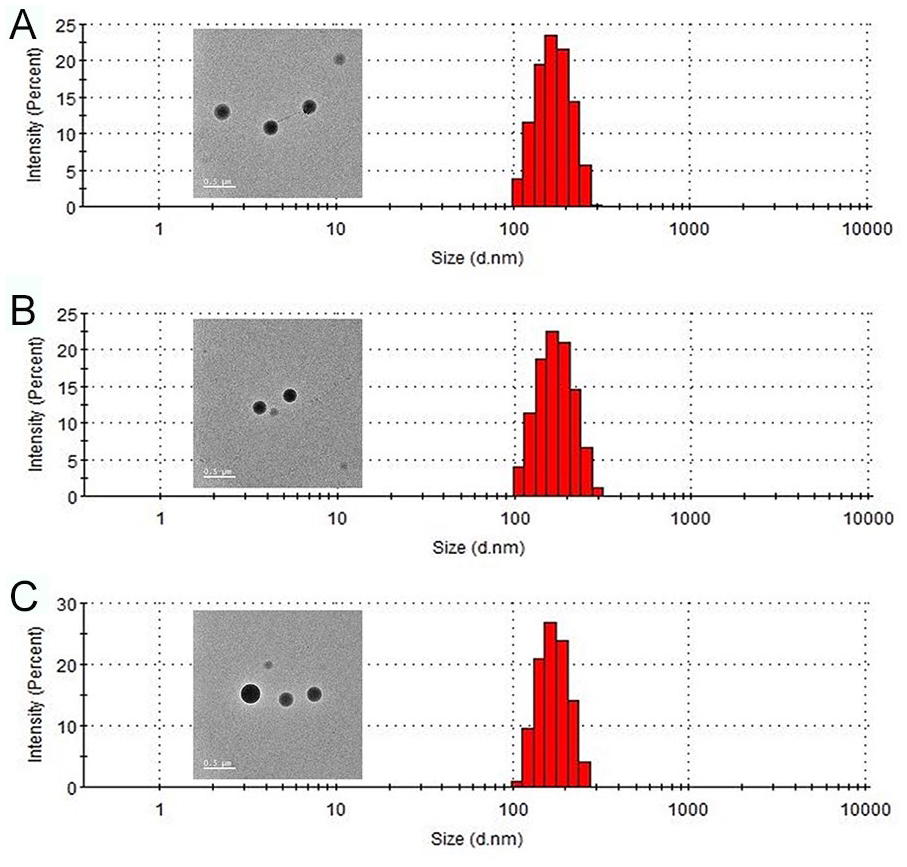
Particle size and distribution of (
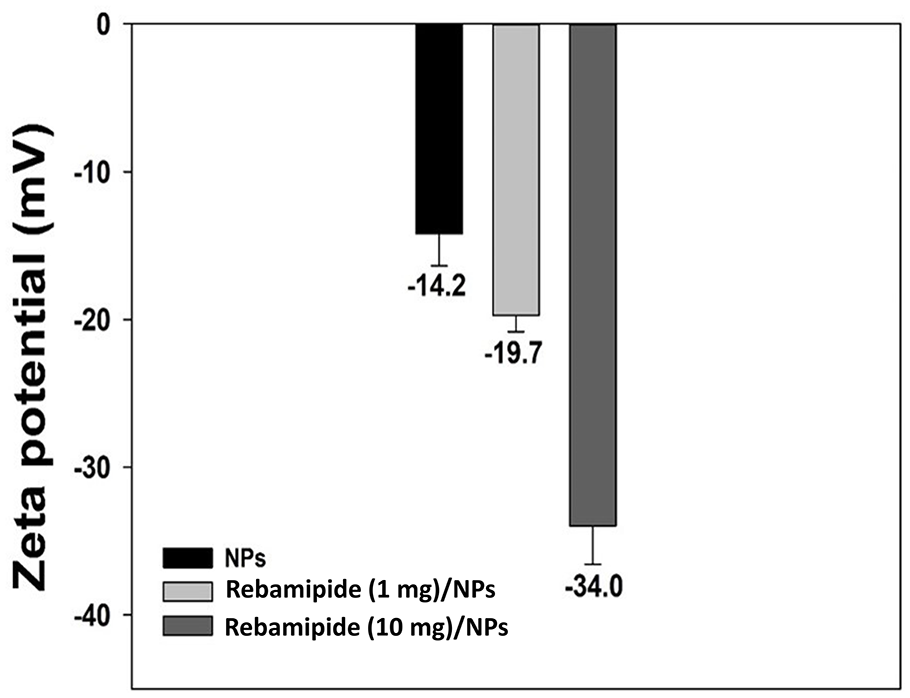
Zeta potential of NPs, rebamipide (1 mg)/NPs, and rebamipide (10 mg)/NPs. NPs = nanoparticles.
Dialysis membrane method was used to confirm the release kinetics of rebampide from NPs. A dialysis membrane method set-up is divided into two compartments, 1 consisting of a dialysis device surrounded by a NP formulation or free drug solution, and the other consisting of a sink transmitter.26,27 The release kinetics of NPs was measured by combining results of drug diffuse in a sink transmitter and the drug releasing from NPs the medium inside the dialysis device. The release kinetics of rebamipide from rebamipide (1 mg)/NPs and rebamipide (10 mg)/NPs for up to 28 days are shown in Figure 4A. On day 1, the amounts of the released rebamipide (percentage) were 4.49 ± 0.03 µg (58.16 ± 0.39%) for rebamipide (1 mg)/NPs and 41.18 ± 0.36 μg (60.27 ± 0.52%) for rebamipide (10 mg)/NPs. After 28 days, rebamipide (1 mg)/NPs and rebamipide (10 mg)/NPs released 7.22 ± 0.06 μg (93.45 ± 0.82%) and 62.17 ± 0.75 μg (90.65 ± 1.09%) of rebamipide, respectively. Figure 4B shows the cytotoxicity results for each group at 1 and 3 days of incubation with the chondrocytes. Cell viability after treatment with each NP substance was over 95% for 3 days compared with that of the control.
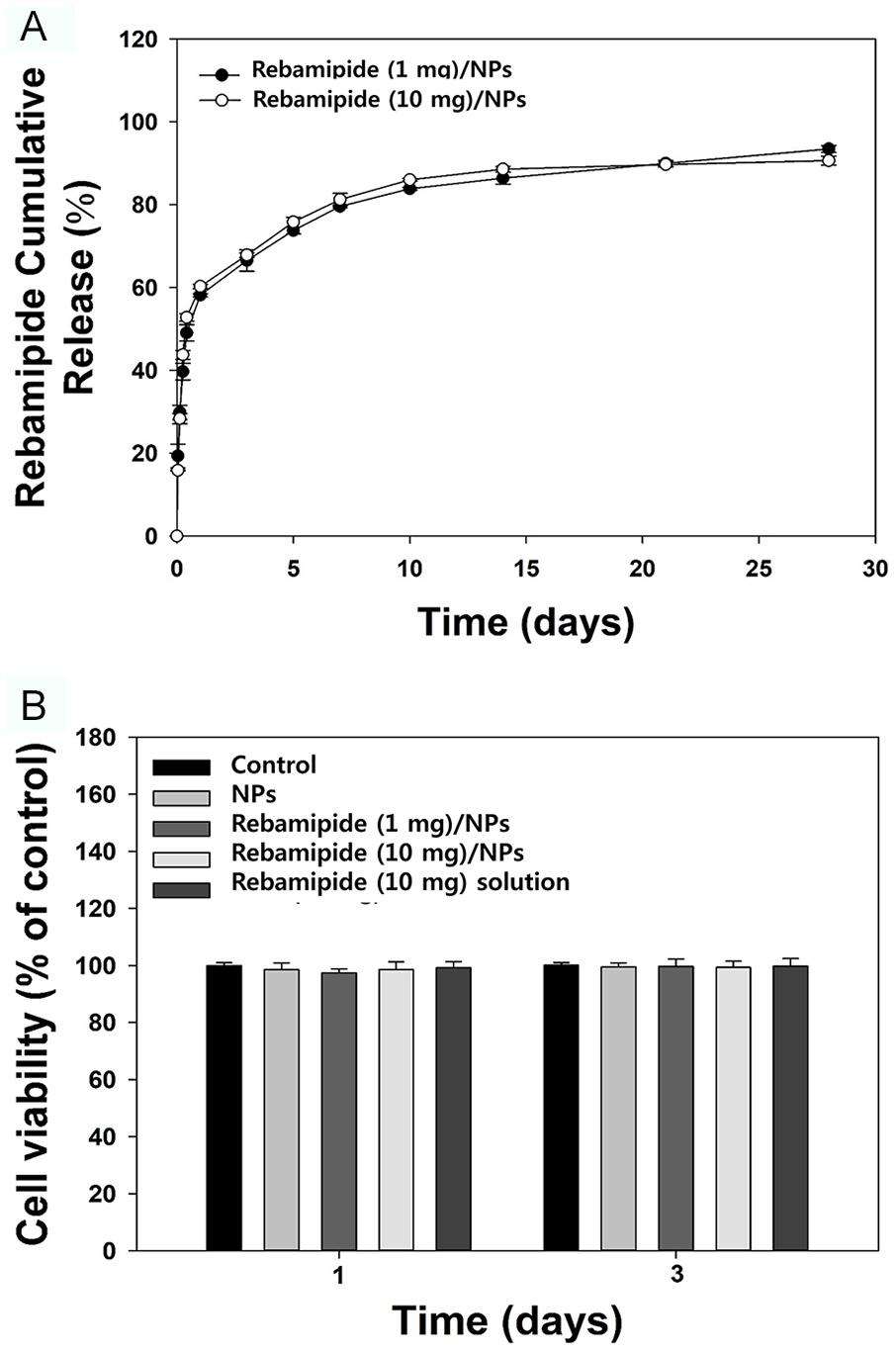
(
Gene Levels of Cytokines in IL-1 & LPS-Promoted Chondrocytes after Treatment with Rebamipide/NPs
To determine whether rebamipide/NPs remarkably suppressed the IL-1- and LPS-promoted inflammatory response in chondrocytes, we conducted real-time PCR to estimate the mRNA levels of pro-inflammatory elements (i.e., IL-1β, IL-6, TNF-α, MMP-3, MMP-13, and COX-2) in chondrocytes treated with NPs, rebamipide (1 mg)/NPs, rebamipide (10 mg)/NPs or rebamipide for 1 or 3 days (Fig. 5 and Suppl. Table S1). Compared with cells without IL-1 and LPS induction, IL-1- and LPS-induced cells exhibited an upregulation of pro-inflammatory elements for 3 days. The mRNA levels of IL-1β, IL-6, TNF-α, MMP-3, MMP-13, and COX-2 in cells treated with NPs were similar to those in cells treated with IL-1 and LPS, whereas the expression levels of pro-inflammatory elements in IL-1- and LPS-induced chondrocytes cultured with rebamipide/NPs decreased in a time- and concentration-dependent manner. Moreover, IL-1β, IL-6, TNF-α, MMP-3, MMP-13, and COX-2 gene levels in IL-1- and LPS-promoted cells treated with rebamipide solution were prominently suppressed compared with those in all other groups.
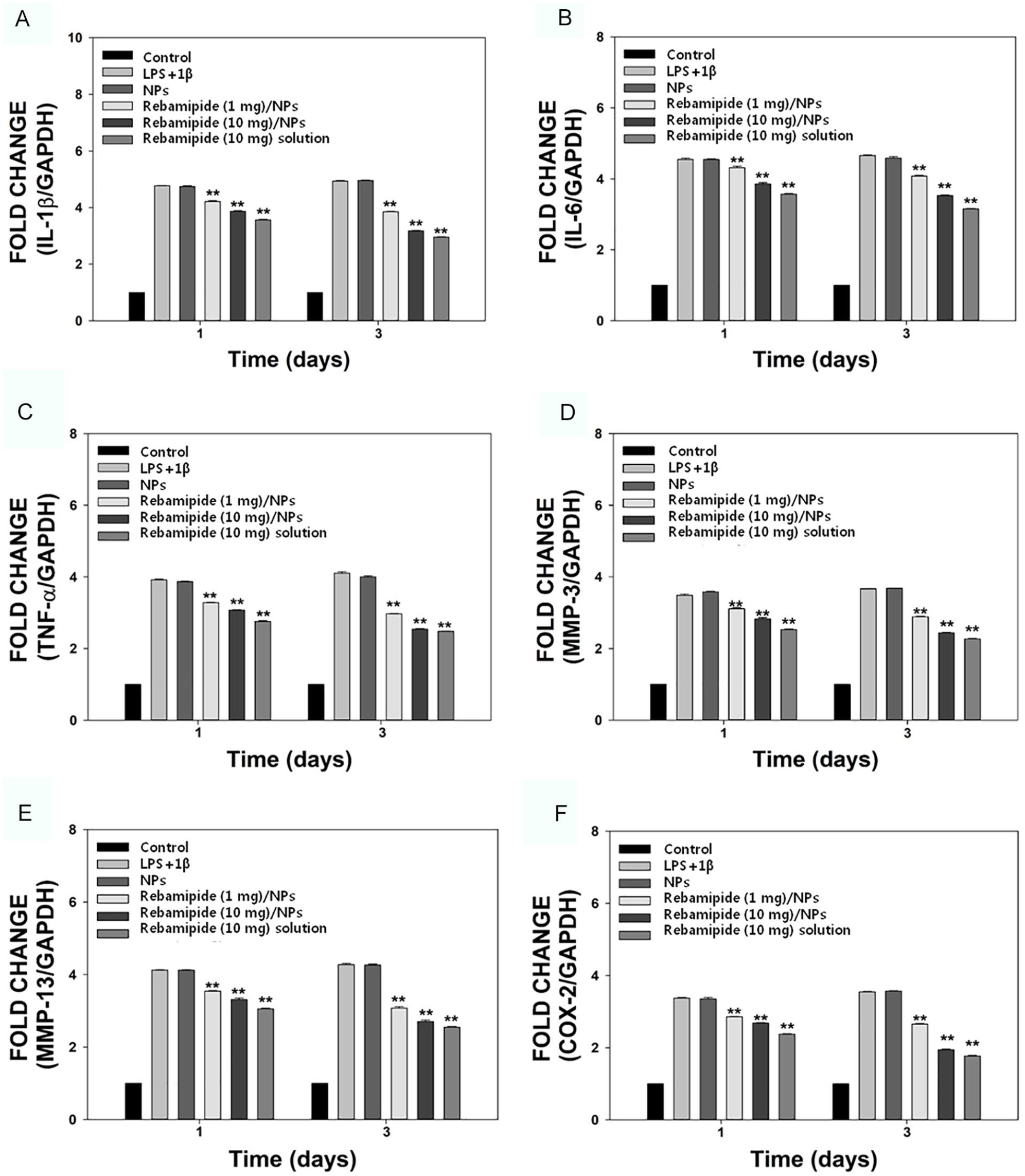
The mRNA expression levels of pro-inflammatory component, including (
Changes in Pro-Inflammatory Factors in Vivo
Four and 8 weeks after the administration of the NPs with or without rebamipide treatment for MIA-induced OA in rats, we performed real-time PCR to analyze the changes in the mRNA levels of the pro-inflammatory elements (IL-1β, IL-6, TNF-α, MMP-3, MMP-13, and COX-2) using whole blood samples. As shown in Figure 6 and Supplementary Table S2, the mRNA levels of pro-inflammatory components in the NPs-only group improved in time-dependent manner, similar to those in the MIA-induced group. At 4 weeks, the levels of the 6 genes in the rats treated with the rebamipide/NPs at both rebamipide concentrations decreased greatly compared to those in the rats treated with NPs. Moreover, differences in the mRNA levels of IL-1β, IL-6, TNF-α, MMP-3, MMP-13, and COX-2 were substantially reduced in the whole blood of rats between the oral rebamipide-treated group and the NP-treated group, with similar reduction in difference observed between the rebamipide solution-treated group and the NP-treated group. At 8 weeks, the IL-1β, IL-6, TNF-α, MMP-3, MMP-13, and COX-2 gene levels were markedly reduced in the rebamipide/NP-treated group in a time-dependent manner, whereas they increased slightly in both the rebamipide-treated group and rebamipide solution-treated group in a time-dependent manner. In particular, at 8 weeks after MIA injection, the mRNA expression levels of IL-6 were increased by approximately 3.7 times compared to the control. While after the intra-articular injection of rebamipide (10 mg)/NPs, the mRNA expression levels of IL-6 were increased by approximately 2.5 times. This indicated that the mRNA expression level of IL-6 between MIA only and rebamipide (10 mg)/NPs injection groups was reduced by approximately 34%. In the case of the mRNA expression level of TNF-α, it was increased by approximately 3.5 times compared to those of control at 8 weeks after MIA injection. While after the intra-articular injection of remaipide (10 mg)/NPs, the mRNA expression level of TNF-α was increased by approximately 2.4 times, which was reduced by approximately 31% compared to those of MIA injection only.
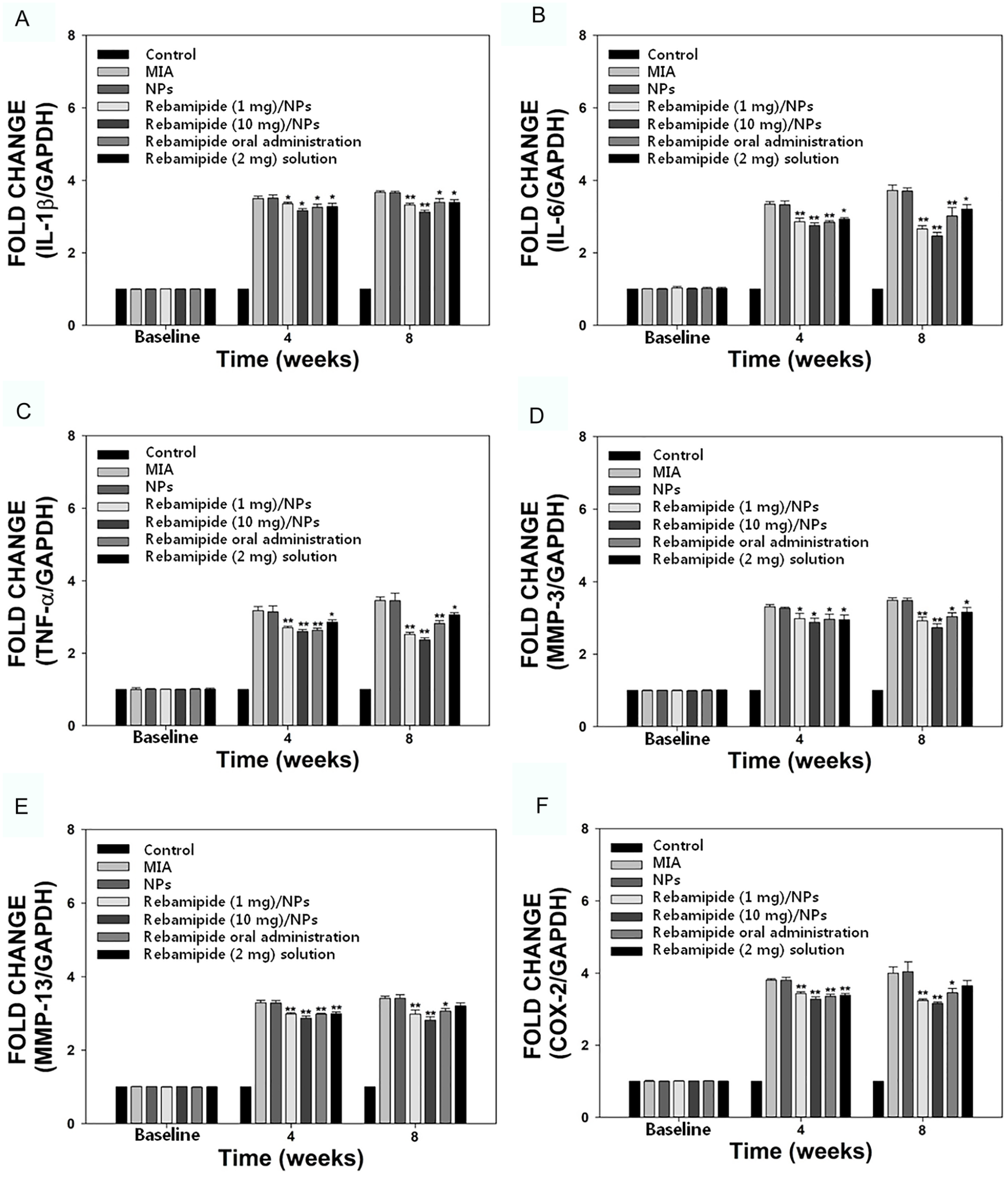
The mRNA expression levels of pro-inflammatory components, including (
Gross, Radiologic, and Histological Determinations
Eight weeks after treatment with rebamipide oral administration, rebamipide solution, rebamipide (1 mg)/NPs or rebamipide (10 mg)/NP injections, gross and radiological examinations were conducted to confirm cartilage alterations and amelioration of OA progression. As described in Figure 7A, rats treated with only MIA or with MIA and NPs displayed severe joint surface irregularities, space narrowing, articular cartilage erosions, subchondral bone sclerosis, and femoral condyle demolition. The rats in the oral rebamipide administration or rebamipide solution injection groups showed irregular surfaces and erosions of cartilage compared to the rats in the MIA-only or MIA + NP treatment groups. Intra-articular treatment with NPs containing rebamipide (1 or 10 mg) in rats with MIA-promoted OA remarkably decreased the erratic cartilage surface and subchondral sediment. In particular, the rebamipide (10 mg)/NP-injected group exhibited an even articular surface, which was analogous to that seen in the control group. Gross examinations were also performed to further demonstrate cartilage transformation during the progression of OA ( Fig. 7B ). MIA-only and MIA with NP injection groups revealed whole spaces of cartilage destruction with irregular surface fibrillation. Treatment with oral rebamipide or rebamipide solution resulted in partial degenerative changes. Treatment of OA with rebamipide/NP resulted in less degradation of cartilage surfaces compared to the rebamipide oral administration or rebamipide solution treatments. The injection of rebamipide (10 mg)/NPs in joints with OA resulted in the lowest degradation of cartilage surfaces compared to the other treatment groups.
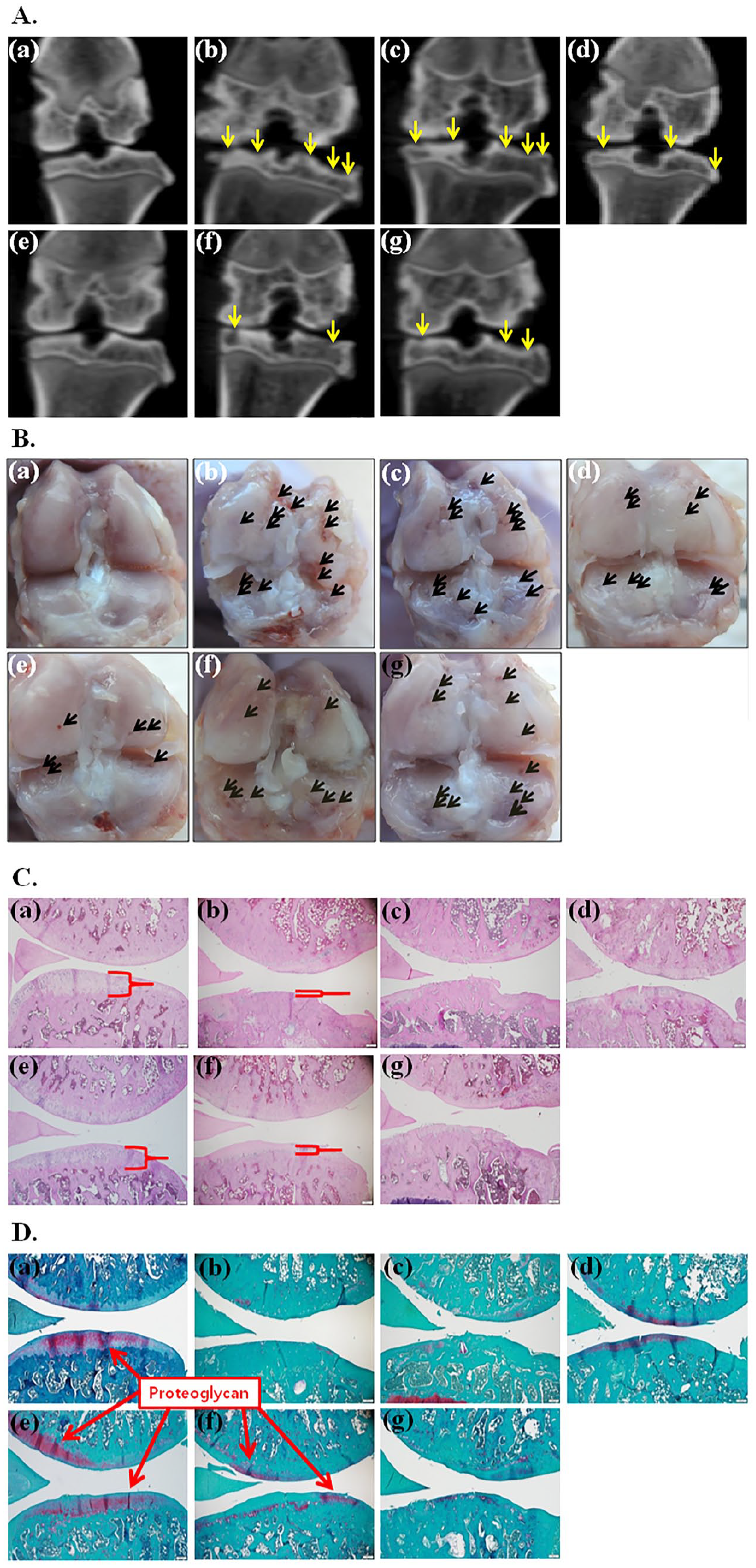
(
The results of H&E staining showed a clear alteration in the cartilage lesion between all rebamipide treatment groups (Fig. 7C). MIA-only and NP-treated groups showed roughness and destruction of the cartilage and meniscus. The rebamipide solution-injected group and the orally administered rebamipide group showed surface roughness, but mild cartilage destruction compared with the MIA-only and NP groups. Rats treated with NPs with different rebamipide concentrations (1 or 10 mg) exhibited little cartilage breakdown and surface roughness. Owing to the anti-inflammatory activity of the rebamipide released from the NPs, the rebamipide (10 mg)/NP-treated group displayed very little surface roughness as well as a minimum degree of cartilage destruction among the treated groups. In H&E staining, the cytoplasm and nucleus are mainly stained. In the control and rebamipide (10 mg)/NPs groups, the cytoplasms of the cartilage were intact and purple nuclei were visible, whereas in the MIA group, the cytoplasms and nuclei of the cartilage were hardly visible and in rebamipide oral administration group, the cytoplasms reduced and nuclei were almost invisible. The results of safranin-O staining allowed the determination of proteoglycan content during the progression of OA. In the MIA-only and NP treatment groups, a loss of proteoglycan to the deep area of the cartilage layers, subchondral bone exposure, and erosion and fibrillation of cartilage were observed (Fig. 7D). The rebamipide solution-injected group or rebamipide orally administered group showed a loss of proteoglycan in the superficial and intermediate layers of the cartilage layer. A slight loss of proteoglycan in the superficial zone of cartilage layers was observed in the rebamipide (1 mg)/NP group. However, rebamipide (10 mg)/NP-treated group showed a minor loss of proteoglycan in the superficial area of the cartilage layer, and most of the proteoglycan in the cartilage was retained. Osteoarthritis Research Society International (OARSI) scoring was conducted to further confirm the
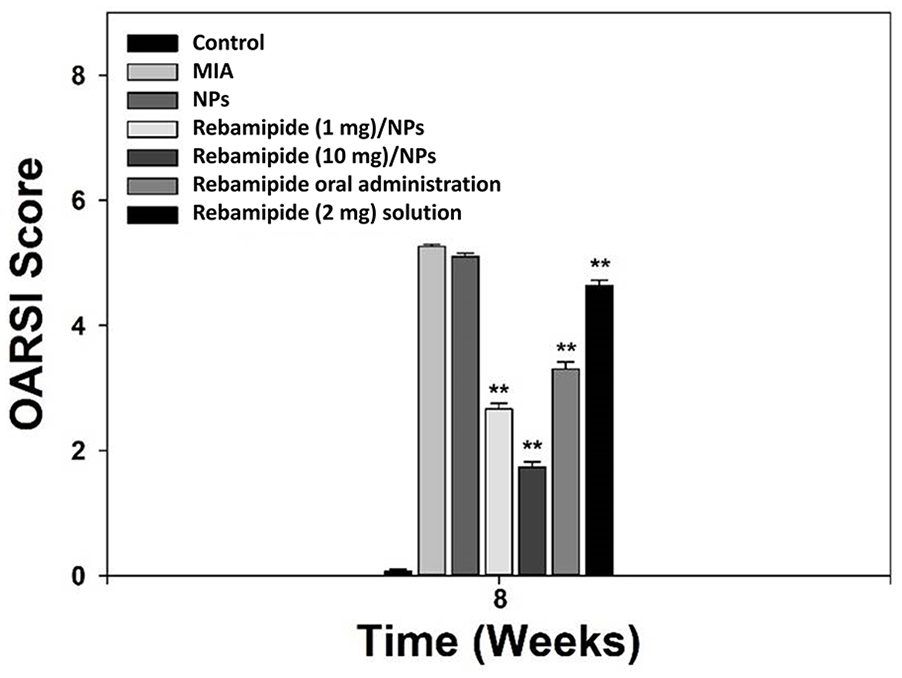
Osteoarthritis Research Society International scores of the rat knee joints during at the ninth week of monosodium iodoacetate injection (eighth week of rebamipide treatment). The score was measured by averaging the values of ‘4 rats belonging to each of the 7 groups. MIA = monosodium iodoacetate; NPs = nanoparticles; OARSI = Osteoarthritis Research Society International.
Discussion
OA initiates cartilage damage, causes synovial inflammation and sclerosis of subchondral bones, and damages bones resulting in the formation of osteophytes. 28 Disease-modifying OA drugs (DMOADs) have been developed to inhibit the progression of OA by targeting each process. Phase 3 studies on inducible NO synthase inhibitors, gene therapy, and existing possible DMOADs, such as diacerein, calcitonin, and doxycycline, are being conducted, and drugs that inhibit various mechanisms that affect the development of OA are being studied. 29 Oxidative stress plays a critical role in the development and progression of OA, and drugs that inhibit oxidative stress are fundamental DMOADs. Rebamipide is an oxygen radical scavenger that inhibits oxidative stress and osteoclastogenesis, and is considered a drug that can be used as a DMOAD. 10 In this study, rebamipide-loaded mPEG-PDLLA and PLGA NPs released rebamipide in a sustained manner for up to approximately 2 months without cytotoxic effects against synoviocytes. In addition, rebamipide released from NPs in the joint cavity significantly reduced the mRNA levels of pro-inflammatory cytokines in the blood and inhibited the progression of OA by protecting against cartilage degradation. In particular, a single intra-articular injection of rebamipide (10 mg)/NPs was more effective locally and systemically in inhibiting OA progression than daily rebamipide consumption.
Both mPEG-PDLLA and PLGA are NPs that have been used for cancer treatment, with tested efficacy and stability. When rebamipide was loaded onto the NPs of these 2 substances, the product maintained the nanosize and reduced inflammation in synoviocytes and chondrocytes without cytotoxicity. The MIA + NP-treated group showed no significant elevation in pro-inflammatory cytokines in the early or late stage compared to the MIA-treated group; therefore, the mPEG-PDLLA and PLGA NPs did not worsen arthritis. In addition, approximately 60% of rebamipide was released from the mPEG-PDLLA and PLGA NPs in 2 days, and rebamipide was gradually released continuously, which may suppress the elevation of IL-1β and TNF-α levels in synoviocytes. Although evaluated
OA progresses by increasing ROS levels due to oxidative stress caused by several factors, such as aging. ROS increase the secretion of pro-inflammatory cytokines, such as IL-1 and TNF-α, in chondrocytes and increase the expression of enzymes related to the progression of OA, such as MMP-1 and COX-2.
28
IL-1 and TNF-α further activate the cells to promote the secretion of other inflammatory or catabolic enzymes, such as NO synthase-2, COX-2, MMP-1, MMP-3, and MMP-13.
30
IL-1 is a powerful inducer of cartilage degradation and induces the mRNA expression of disease-relevant proteases, such as MMP13 and ADAMTS5.
31
TNF-α upregulates MMP gene expression and induces cartilage degeneration.
32
In
Since rebamipide has been developed as a gastrointestinal protective drug, it is unclear whether rebamipide has the role of inhibiting joint inflammation and OA progression after intra-articular injection
In conclusions, the sustained drug delivery system involving rebamipide-loaded mPEG-PDLLA and PLGA NPs suppressed the mRNA levels of pro-inflammatory elements in inflamed synoviocytes stimulated by LPS and IL-1. A single intra-articular injection of rebamipide/NPs also suppressed inflammatory reactions and appeared to mitigate cartilage degradation in MIA-induced OA. Considering the actual treatment dosage and the degree of anti-inflammatory effect, rebamipide/NP injection appeared to be more effective in the treatment of OA than the intra-articular injection or daily oral administration of rebamipide alone. The preliminary results of this pilot study support further adequately powered
Supplemental Material
sj-docx-1-car-10.1177_19476035211069250 – Supplemental material for Intra-Articular Injection of Rebamipide-Loaded Nanoparticles Attenuate Disease Progression and Joint Destruction in Osteoarthritis Rat Model: A Pilot Study
Supplemental material, sj-docx-1-car-10.1177_19476035211069250 for Intra-Articular Injection of Rebamipide-Loaded Nanoparticles Attenuate Disease Progression and Joint Destruction in Osteoarthritis Rat Model: A Pilot Study by Sung Eun Kim, Sung Jae Choi, Kyeongsoon Park, Hak-Jun Kim, Gwan Gyu Song and Jae Hyun Jung in CARTILAGE
Footnotes
Acknowledgments and Funding
We would like to thank Nano-based Disease Control Institute for assistance of the experiment.
The research was supported by a Korea University Ansan Hospital Grant (grant number: O1904051) and the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (grant number: 2018R1D1A1B07050687).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Animal Care and Use Committee of the Korea University Medical Center (KOREA-2018-0112).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
