Abstract
Introduction
This study develops assays to quantify collagen subtypes and crosslinks with liquid chromatography-mass spectrometry (LC-MS) and characterizes the cartilages in the Yucatan minipig.
Methods
For collagen subtyping, liquid chromatography-tandem mass spectrometry (LC-MS/MS) analysis was performed on tissues digested in trypsin. For collagen crosslinks, LC-MS analysis was performed on hydrolysates. Samples were also examined histologically and with bottom-up proteomics. Ten cartilages (femoral condyle, femoral head, facet joint, floating rib, true rib, auricular cartilage, annulus fibrosus, 2 meniscus locations, and temporomandibular joint disc) were analyzed.
Results
The collagen subtyping assay quantified collagen types I and II. The collagen crosslinks assay quantified mature and immature crosslinks. Collagen subtyping revealed that collagen type I predominates in fibrocartilages and collagen type II in hyaline cartilages, as expected. Elastic cartilage and fibrocartilages had more mature collagen crosslink profiles than hyaline cartilages. Bottom-up proteomics revealed a spectrum of ratios between collagen types I and II, and quantified 42 proteins, including 24 collagen alpha-chains and 12 minor collagen types.
Discussion
The novel assays developed in this work are sensitive, inexpensive, and use a low operator time relative to other collagen analysis methods. Unlike the current collagen assays, these assays quantify collagen subtypes and crosslinks without an antibody-based approach or lengthy chromatography. They apply to any collagenous tissue, with broad applications in tissue characterization and tissue engineering. For example, a novel finding of this work was the presence of a large quantity of collagen type III in the white-white knee meniscus and a spectrum of hyaline and fibrous cartilages.
Introduction
Collagen is the most abundant protein in the mammalian body and is the foundation of the extracellular matrix (ECM) of connective tissues. 1 Collagen is better described as a superfamily of proteins consisting of 28 currently discovered subtypes, each of which is a trimer consisting of 3 alpha-chains that associate into supercoiled triple helices. 2 The main fibrillar collagen subtypes, collagen types I and II, make up a large majority of collagens found in different tissues all over the body. 1 A primary barrier to determining the collagen composition of biological tissues is that high-throughput and low-cost methods for collagen subtype quantification remain a major challenge. 1 The traditional method of collagen quantification, the hydroxyproline assay, does not discriminate among collagen subtypes. 3 Other methods, such as enzyme-linked immunosorbent assay (ELISA), can be prohibitively expensive or unavailable depending on the animal species or collagen subtype. 1 Some imaging methods, such as immunohistochemistry, can be used to visualize different collagen subtypes, but these are nonquantitative. 1
In this work, we introduce a liquid chromatography-tandem mass spectrometry (LC-MS/MS) assay that uses multiple reaction monitoring 4 to quantify collagen types I and II. This assay is inexpensive compared with antibody-based methods and applicable to collagenous tissues from human, bovine, porcine, and murine sources. Enzymatic collagen crosslinks, such as the mature trivalent crosslink pyridinoline (PYR) and the immature divalent crosslink dihydroxylysinonorleucine (DHLNL), play important roles in the mechanical properties of collagenous tissues.5,6 Recent work has introduced the use of diamond hydride chromatography for collagen crosslink quantification, 7 and, in this experiment, this chromatography method is used on an LC-MS with high sensitivity and specificity. As a demonstration of these 2 novel assays, 10 cartilages of the Yucatan minipig are analyzed. This animal model was selected because it is a well-recognized model for cartilage repair studies.8,9
Cartilages are classified into 1 of 3 categories: hyaline cartilage, fibrocartilage, and elastic cartilage, based on the composition of the tissue’s ECM. Traditionally, cartilages are classified by the types of fibrillar proteins within the tissue. Hyaline cartilage, found on the ends of bones (articular cartilage) and in the ribs, nose, and trachea, is mostly collagen type II by dry weight (DW). 10 Fibrocartilage, found in the knee meniscus, temporomandibular joint (TMJ), and annulus fibrosus, has an ECM that contains mostly collagen type I by DW. 11 Elastic cartilage, found in the auricle of the ear and in the epiglottis, has an ECM that contains both collagen types I and II, as well as a substantial portion of elastin. 12
Previous tissue engineering studies have shown that it is possible to engineer a spectrum of hyaline and fibrous cartilages, based on the ratio of collagen types I and II 13 , that is, more hyaline cartilage having more collagen type II and less collagen type I, and more fibrous cartilage having more collagen type I and less collagen type II. While there has been evidence of higher collagen type II in the white-white portion of the knee meniscus compared with the red-red meniscus, 14 it is not known whether a wide spectrum of collagen ratios exist in native cartilages. Prior work has shown that femoral condylar cartilage contains higher PYR crosslinks compared with knee meniscus in the immature knee joint, 5 but it is unknown whether this trend holds true for other cartilages or for mature joints.
The objectives of this work are to develop high-throughput LC-MS assays to analyze collagen subtypes and crosslinks, to use these assays to characterize 10 cartilages of the Yucatan minipig, and to use bottom-up proteomics to quantitatively characterize the proteome of these minipig cartilages. The hypotheses of this work are that: (1) the LC-MS/MS collagen subtyping assay can quantify marker peptides of collagen types I and II in a specific and sensitive manner, and (2) the LC-MS collagen crosslinks assay can quantify PYR, DHLNL, and hydroxyproline. Using minipig cartilages as examples, it is expected that: (1) the novel collagen subtyping assay will show a majority of collagen type I in fibrocartilages and a majority of collagen type II in hyaline cartilages, (2) the collagen crosslinks assay will show different crosslink ratios in hyaline cartilages than fibrocartilages, and (3) bottom-up proteomics will reveal different ratios of collagen types I and II in different cartilages within the traditional hyaline and fibrous classifications, revealing a spectrum of cartilage tissues.
Methods
LC-MS/MS for Collagen Subtype Quantification
A Waters ACQUITY UPLC I-Class core system coupled to a Waters Quattro Premier XE triple quadrupole mass spectrometer was used for the collagen subtype LC-MS/MS assay. For all liquid chromatography in this study, solvent A was 0.1% (v/v) formic acid in water and solvent B was 0.1% (v/v) formic acid in acetonitrile. Liquid chromatography gradient settings were as follows: initial, 3% B; 2 minutes, 40% B; 3 minutes, 97% B; 4 minutes, 3% B; 5 minutes, 3% B; total run time, 5 minutes. An ACQUITY UPLC BEH C18 column was used with a flow rate of 300 μl/min for reverse-phase separation. Target peptides of collagen were selected under the following criteria: susceptible to digestion from trypsin; not found in any other protein aside from 1 specific collagen subtype; found in human, bovine, porcine, and murine tissues; and mass between 800 and 1800 Da. Briefly, PeptideRank was used for in silico prediction of tryptic peptides for collagen subtypes with the best ionization characteristics.
15
Peptides were checked with NCBI BLASTp to ensure uniqueness and identical sequence across human, bovine, porcine, and murine proteomes. The set of determined peptides is summarized in
Mass Spectrometry Settings for Target Analytes.
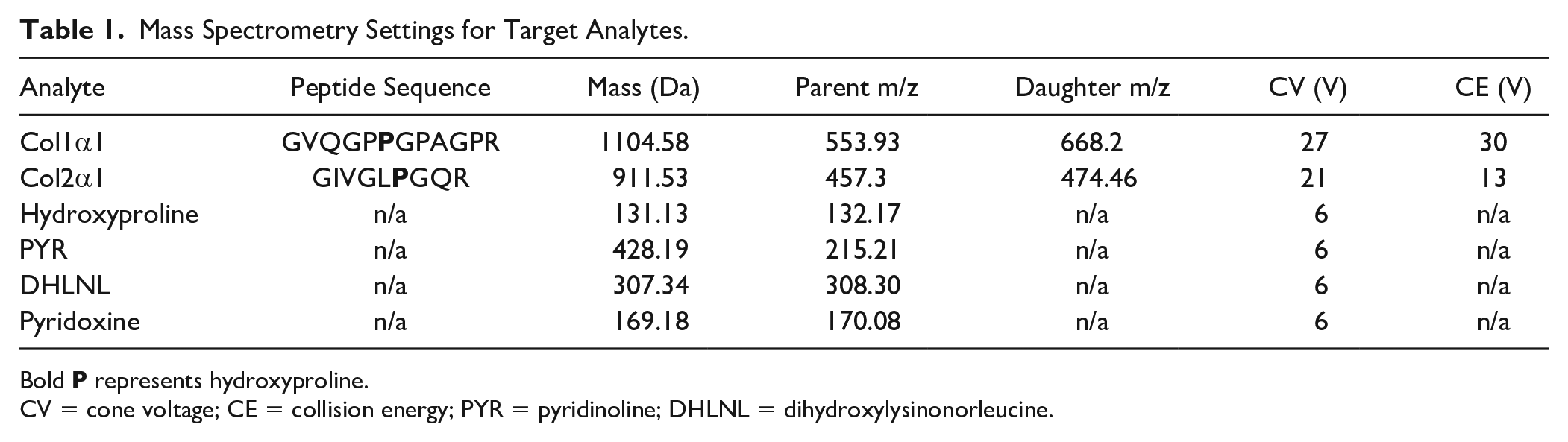
Bold
CV = cone voltage; CE = collision energy; PYR = pyridinoline; DHLNL = dihydroxylysinonorleucine.
Quantification of Collagen Crosslinks and Hydroxyproline
Liquid chromatography gradient settings were as follows: initial, 90% B; 1 minute, 90% B; 2 minutes, 20% B; 4 minutes, 20% B; 5 minutes, 90% B; 10 minutes, 90% B; total run time, 10 minutes. A Cogent Diamond Hydride 2.o HPLC column was used with a flow rate of 400 μl/min for aqueous normal phase separation, coupled to a Waters ACQUITY QDa mass spectrometer. PYR standard was ordered from BOC Sciences, DHLNL standard was ordered from Santa Cruz Biotechnology, and hydroxyproline (OHP) and pyridoxine (PDX) standards were ordered from Sigma Aldrich. Quantification methods were developed by diluting PYR, DHLNL, OHP, and PDX standards to 1 μg/ml in solvent A and then optimizing CV to ±1 V on repeated 5 μl injections. Standard curves were prepared by 3x serial dilution of a 1 μg/ml mix of PYR, DHLNL, and PDX and 10 μg/ml of OHP. PDX was used as an internal standard. LOD and LOQ were calculated as described above.
Bottom-up Proteomics
For all bottom-up proteomics samples, the same peptide digest used for the collagen I and II LC-MS/MS assay was used. For each tissue, digests from all 7 animals were combined into 1 representative sample. The peptide digests were subjected to LC-MS/MS analysis using a Thermo Fischer Scientific UltiMate 3000 RSLC system coupled on-line to a Thermo Fischer Scientific Orbitrap Fusion Lumos mass spectrometer. Liquid chromatography gradient was as follows: 4% to 25% solvent B over 57 minutes. A 50 cm × 75 μm I.D. Acclaim® PepMap RSLC column was used at a flow rate of 300 nl/min for reverse-phase separation. Each cycle consisted of 1 full Fourier transform scan mass spectrum (375-1500 m/z, resolution of 60,000 at m/z 400) followed by 15 data-dependent MS/MS acquired in the linear ion trap with collision-induced dissociation with normalized CE of 25%. Target ions already selected for MS/MS were dynamically excluded for 30 seconds. Identification and label-free quantification were carried out using MaxQuant, as previously described. 16 Briefly, raw files were searched using MaxQuant (v. 1.6.0.16) against a FASTA containing the sus scrofa proteome obtained from the SwissProt open-source database (version from December 2020) along with sus scrofa collagen proteins from TrEMBL. The first search peptide tolerance was set to 20 ppm, with main search peptide tolerance set to 4.5 ppm. The protein, peptide, and peptide spectrum match level false discovery rates were all 1% as determined by a target-decoy approach. For quantification, intensities were determined as the full peak volume over the retention time profile. The degree of uniqueness required for peptides to be included in quantification was “Unique plus razor peptides.” The resulting label-free quantification (LFQ) values calculated through MaxQuant were used for comparing protein relative abundance among different samples. The proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository with the data set identifier PXD025482.
Data Analysis and Statistical Analysis
For collagen types I and II and for collagen crosslinks, MassLynx v4.1 software with QuanLynx was used to calculate area-under-curve measurements for all MRM peaks for interpolation into the standard curve. The resulting ng/ml result was multiplied by the sample volume and appropriate dilution factor to quantify the analyte quantity in the sample. Any analyte quantity below the LOQ was set to zero. For crosslinks, the resulting mass of each crosslink was normalized to DW or the amount of hydroxyproline in the hydrolysate. For peptides, the peptide mass was converted to protein mass. For collagen type II, the peptide mass (Mpep) is multiplied by the mass ratio of the alpha-chain to peptide (k) to calculate the mass of collagen type II in the digest (MCol2):
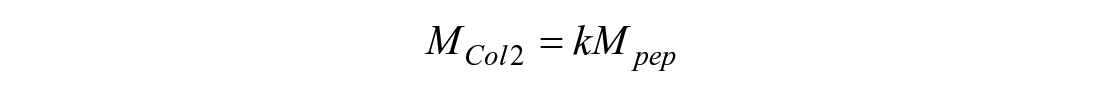
For collagen type I, protein mass is calculated based on the quantification of the col1α1 peptide; because collagen type I is a heterotrimer which has 2 col1α1 chains and 1 col1α2 chain, the mass may be calculated, assuming the molar ratio of col1α1 to col1α2 to be 2:1. Here, k is the mass ratio of a col2α1 molecule to the col2α1 target peptide, and A is the molar ratio of 1 col1α2 molecule to 2 col1α1 molecules as calculated by their amino acid sequences and posttranslational modifications:
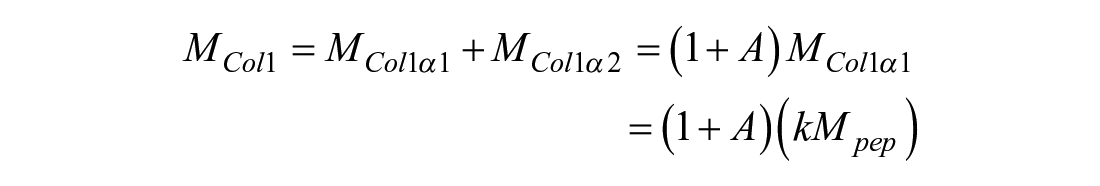
For collagen type II, k = 105.09. For collagen type I, k = 86.01 and A = 0.49. Molecular weights of the collagen proteins were calculated after cleavage of the N-terminal propeptides. Protein masses were normalized to sample DW.
Statistical analysis was performed in JMP Pro 14. Post hoc Tukey tests were used after performing 1-way analyses of variance (ANOVAs). Statistical significance (P < 0.05) is indicated with a connecting-letters report on all bar graphs; bars not sharing the same letter are significantly different from each other. All graphs were generated in GraphPad Prism 8.
Sample Preparation
Ten cartilage tissues from the Yucatan minipig were analyzed: knee meniscus (both red-red portion and white-white portion separately), TMJ disc (center region), annulus fibrosus of the intervertebral disc (L5/S1 or L6/S1 joint), auricular (ear) cartilage, femoral head cartilage, femoral condyle cartilage, costal cartilages of the true rib (rib 1) and floating rib (rib 14), and facet joint cartilage (
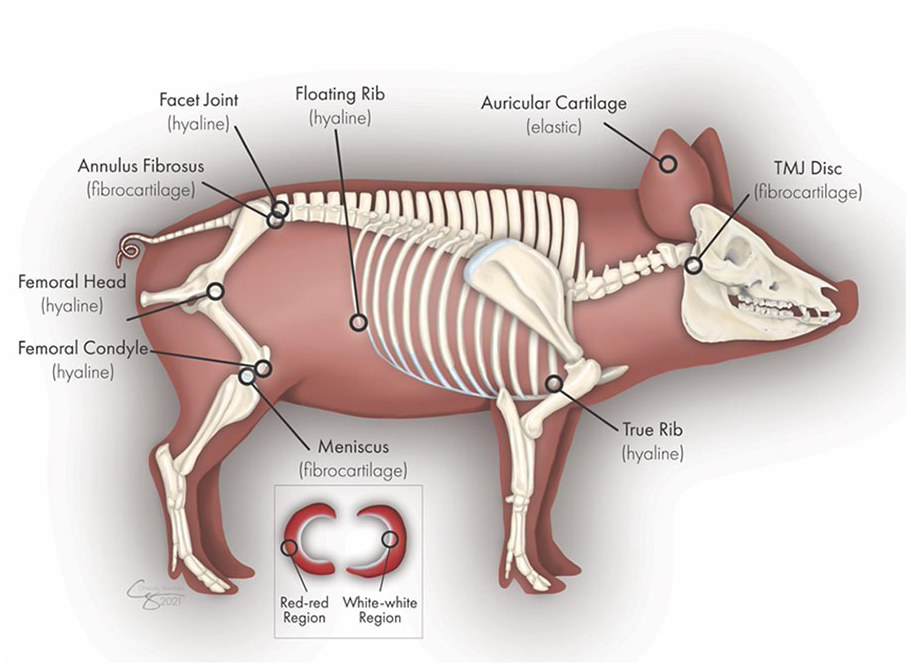
The locations of the different cartilages harvested from the Yucatan minipig. Illustration by Chrisoula Skouritakis. TMJ = temporomandibular joint.
For collagen subtyping and bottom-up proteomics, samples were washed twice in 10 mM ammonium citrate and twice in 50 mM ammonium bicarbonate, and then mass spectrometry–grade trypsin was added in a 1:20 (w/w) ratio of trypsin to sample DW, and samples were digested at 65°C in 200 μl of 50 mM ammonium bicarbonate. After digestion, samples were filtered through 100 kDa molecular weight cut-off centrifugal filters and diluted 1:1 in 0.1% formic acid, yielding a transparent, colorless peptide digest solution. For the crosslinks assay, biopsy samples were submerged in 1 mM NaOH containing NaBH4 (1% weight to sample weight) for 2 hours at room temperature, washed overnight in ultrapure water, and then hydrolyzed in 6N HCl at 105°C for 18 hours in a heat block. HCl was evaporated, and samples were resuspended in 400 μl of 0.1% formic acid, then filtered through 100 kDa molecular weight cut-off centrifugal filters, and diluted 20:1 in 0.1% formic acid, yielding a transparent, colorless hydrolysate.
Results
LC-MS Assays for Collagen Subtypes and Crosslinks
The determined parameters required for running the LC-MS/MS assays for collagen subtype and crosslink quantification are displayed in
Collagens in Minipig Cartilages
Collagen types I and II were quantified in 10 tissues via LC-MS/MS high-throughput quantification (
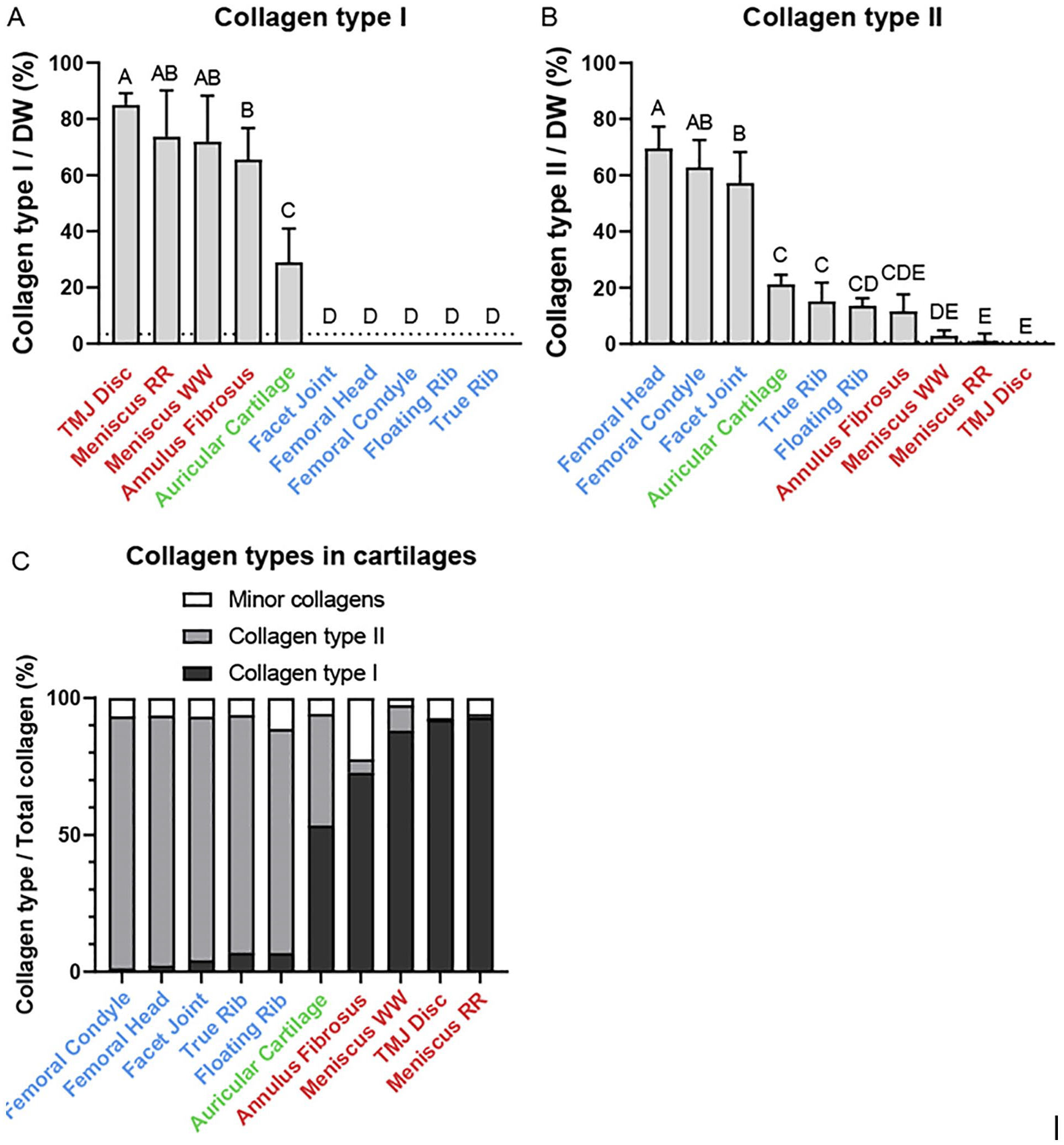
Collagen subtype quantification in the cartilages of the Yucatan minipig. (
Cartilage Crosslinks
The mature (PYR) and immature (DHLNL) crosslink contents of the cartilages are depicted in
Figure 3
. Normalized to collagen as measured by hydroxyproline content, the highest PYR content was found in the auricular cartilage (62.0 ± 13.0 mmol/mol), while most fibrocartilages such as the red-red meniscus (19.6 ± 6.6 mmol/mol) had higher mean PYR than hyaline cartilage such as the femoral condyle (8.1 ± 2.2 mmol/mol). Normalized to collagen, there were no significant differences in DHLNL content among all cartilage types. Normalized to dry weight, all elastic and fibrocartilages except for annulus fibrosus have significantly more PYR than hyaline cartilages. For example, red-red meniscus has the highest PYR/DW at 2420 ± 568 ng/mg, whereas true rib cartilage has the lowest PYR/DW at 609 ± 381 ng/mg. A similar pattern was found with DHLNL normalized to dry weight, where most fibrocartilages had higher mean DHLNL/DW than hyaline cartilages, with annulus fibrosus being an exception. For example, TMJ disc (692 ± 163 ng/mg) had significantly more DHLNL/DW than true rib (242 ± 106 ng/mg), The “maturity ratio,” defined as the molar ratio of PYR to DHLNL (
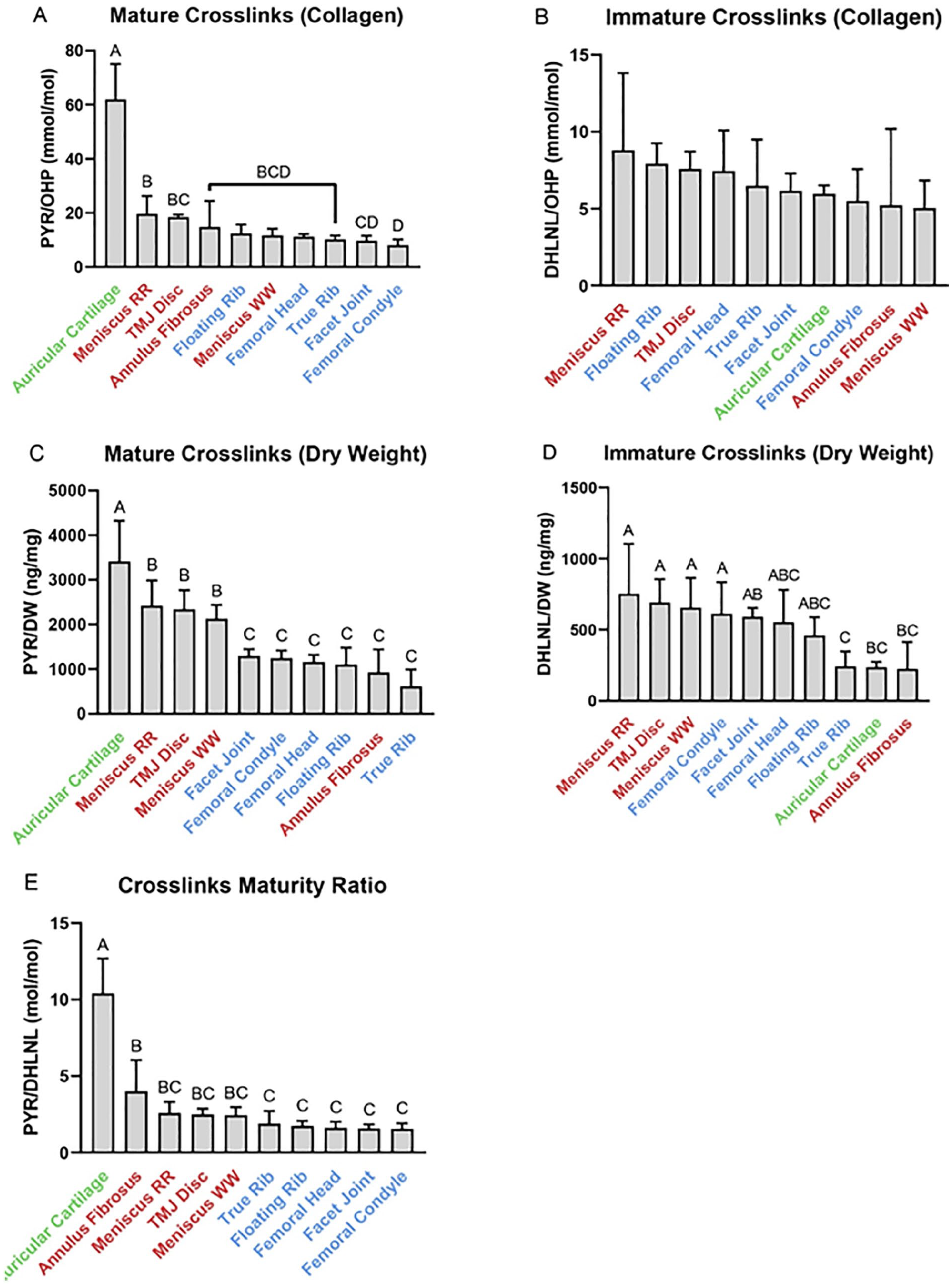
Crosslink quantification in the cartilages of the Yucatan minipig. (
Bottom-up Proteomics Analysis
A full list of relative proportions of all quantified proteins with greater than 0.1% protein per total protein can be found in
Bottom-up Proteomics Results on Yucatan Minipig Cartilages.
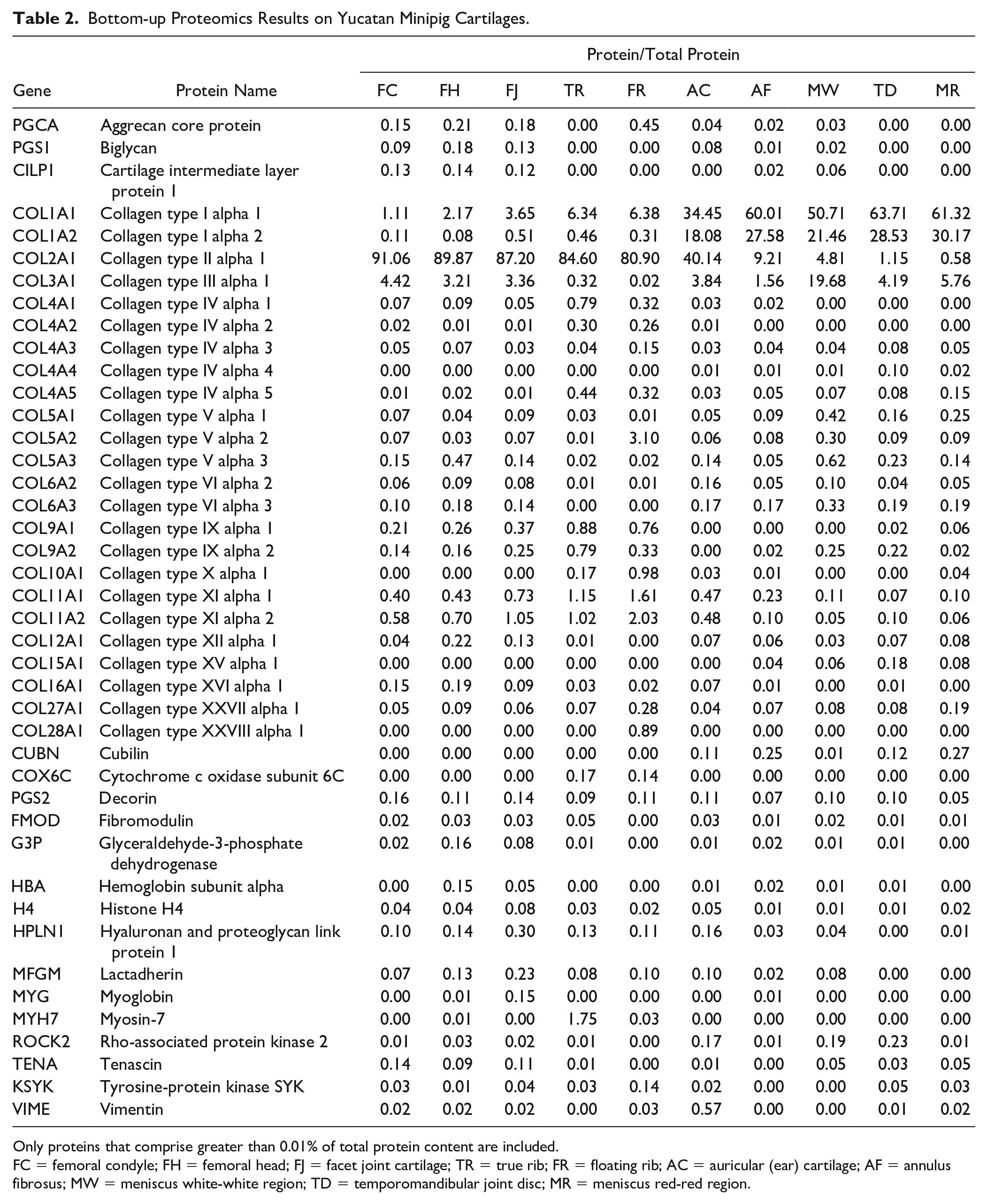
Only proteins that comprise greater than 0.01% of total protein content are included.
FC = femoral condyle; FH = femoral head; FJ = facet joint cartilage; TR = true rib; FR = floating rib; AC = auricular (ear) cartilage; AF = annulus fibrosus; MW = meniscus white-white region; TD = temporomandibular joint disc; MR = meniscus red-red region.
Histology
Histological stains on all cartilages are shown in Figure 4 . H&E histology was used to visualize cells and ECM. Fibrocartilages, which stained more heavily with eosin than hyaline, and elastic cartilages showed striated patterns of anisotropic collagen organization. Histological results were consistent with the literature in terms of the morphology and relative staining intensities; for example, hyaline and elastic cartilages appeared more isotropic, with less strong eosin staining and stronger hematoxylin staining due to higher quantities of glycosaminoglycans (GAGs) in the tissues. 18 The true rib morphology had a fragmented appearance in areas of high calcification. PR staining, similar to H&E staining, stained more intensely for collagen in the fibrocartilages and appeared fibrous and anisotropic. PR staining intensity was lower in the nonfibrous cartilages, and there was less organization of collagen fibers.
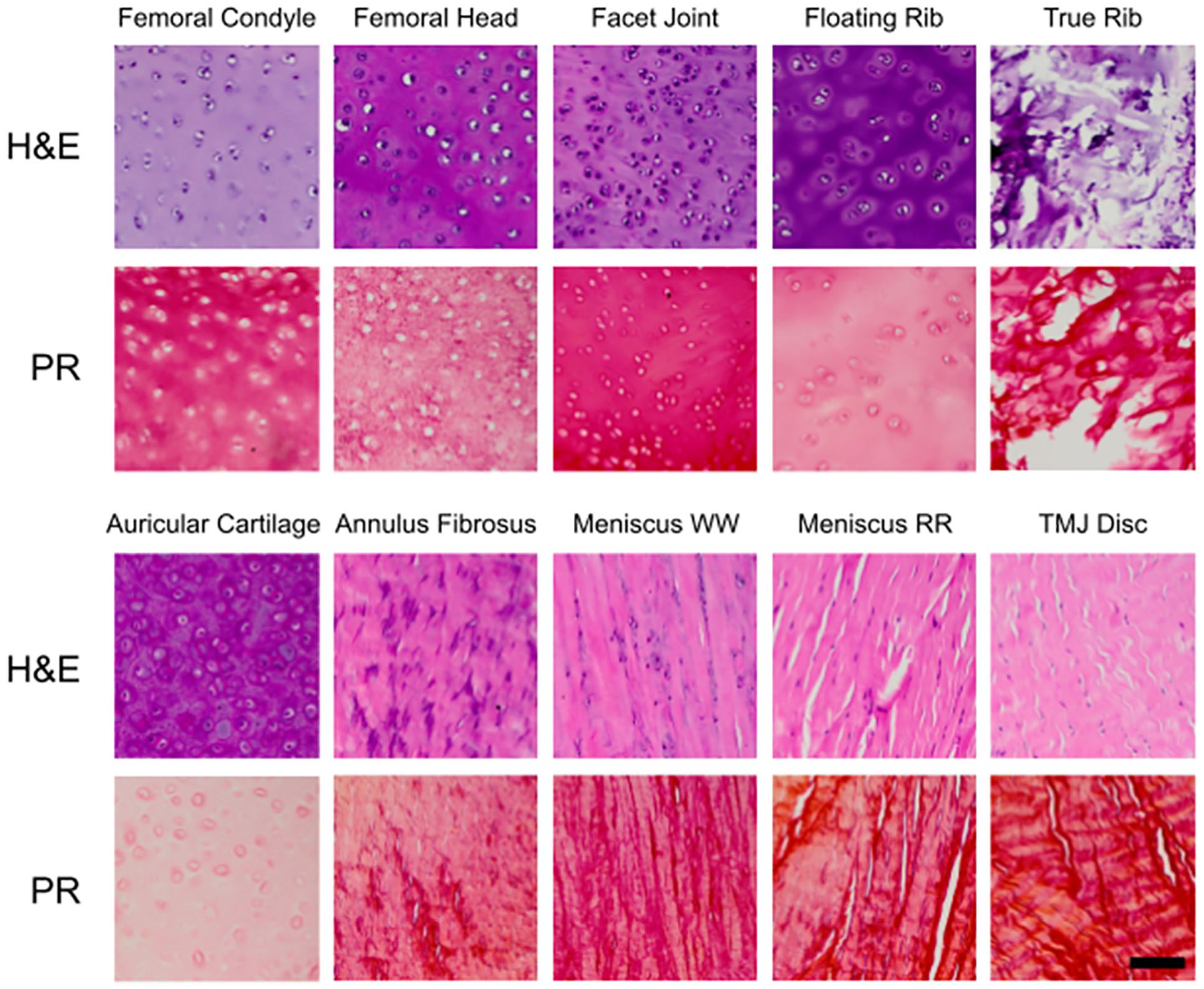
Histological stains on minipig cartilages. H&E, hematoxylin and eosin; PR, picrosirius red; TMJ = temporomandibular joint. Scale bar, 100 μm.
Discussion
In this experiment, we developed and optimized 2 LC-MS assays for quantification of collagen subtypes and collagen crosslinks, and confirmed the hypotheses that LC-MS can be used as a sensitive, specific, quantification method for collagen subtypes and crosslinks in biological tissues. With the parameters detailed in
To demonstrate these novel tools, 10 different cartilages of the Yucatan minipig were analyzed with the collagen subtype and crosslink quantification assays. The collagen subtyping assay accurately showed a majority of collagen type I in fibrocartilages and a majority of collagen type II in hyaline cartilages, and the crosslinks assay showed that the collagen of hyaline cartilages contains more crosslinks than the collagen of fibrocartilages. A bottom-up proteomics analysis was also performed, and the proteomes, quantified in
Determining collagen content is a vital step for characterization of biological tissues and products. The currently practiced methods for collagen quantification are described in
Comparison of Collagen Assays.
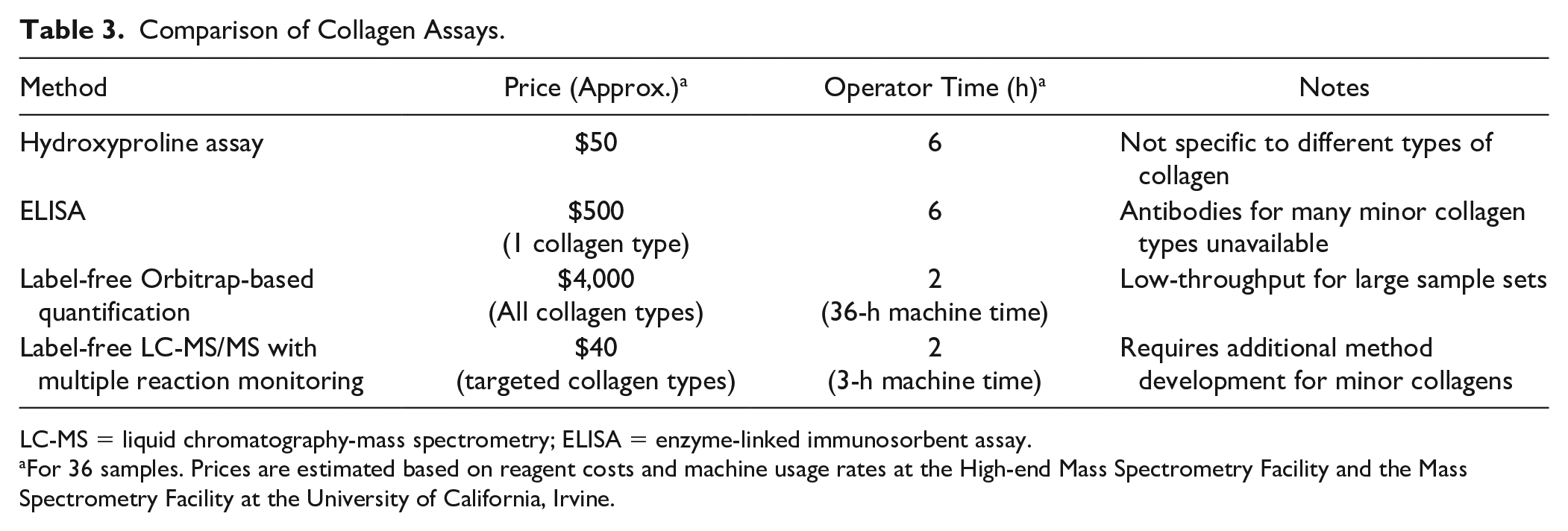
LC-MS = liquid chromatography-mass spectrometry; ELISA = enzyme-linked immunosorbent assay.
For 36 samples. Prices are estimated based on reagent costs and machine usage rates at the High-end Mass Spectrometry Facility and the Mass Spectrometry Facility at the University of California, Irvine.
While collagen crosslink quantification assays have recently seen improvements in the development of silica hydride–based chromatography and mass spectrometry, 7 the assay in this study features an improvement in using a 10-minute chromatography gradient for a low assay runtime compared with commonly used crosslinks assays (30-45 minutes). This collagen crosslink assay, like the collagen subtype quantification assay, is applicable to collagenous tissues all over the body. A novel aspect of this work is the usage of a maturity ratio for cartilages. While other groups have looked at the ratio of immature and mature crosslinks in collagenous tissues, 6 this is the first study that compares this ratio across different types of cartilage. This study showed that fibrocartilages have a greater maturity ratio than hyaline cartilages. Whether or not this is due to rates of collagen turnover in these cartilages, mechanical demands of the tissue, or other factors is yet to be determined.
The bottom-up proteomics method in this study provided quantitative evidence that a spectrum of hyaline and fibrocartilages exists in the Yucatan minipig, shown in the ratio of collagen type I to collagen type II. In hyaline cartilage, the ratio of collagen type II to collagen type I varies from about 74:1 in femoral condyle to about 12:1 in floating rib. In fibrocartilage, the ratio of collagen type I to collagen type II ranges from over 150:1 in the red-red meniscus to less than 10:1 in the annulus fibrosus. Auricular cartilage, the only elastic cartilage in this data set, contains roughly the same amount of collagen types I and II. This wide array of ratios suggests the existence of a spectrum of fibrous and hyaline cartilage rather than discrete categories.
A spectrum of cartilage properties also appears not only in the collagen ratios but also in the collagen crosslinks. As shown in
When damage to hyaline articular cartilage results in fibrocartilage formation, via bleeding either from subchondral bone or from microfracture procedures, a fibrocartilage repair tissue is formed in the defect that contains both collagen types I and II, 31 which fails mechanically in the long term. 32 The failure of this fibrocartilage repair tissue may be in part due to the aforementioned role of collagen II in dissipating compressive forces; if the ECM of the repair tissue is of the wrong composition, it may not stand up to the large, repeated forces withstood by hyaline articular cartilage. While current methods for repair of cartilage defects are unsuccessful in the long term, tissue engineering is a promising strategy to generate biomimetic cartilage implants for cartilage regeneration. 33
Because different types of collagen are implicated in the loading capabilities of cartilage tissues, engineered cartilages must be designed to match the ECM of the cartilage that they are designed to replace. The bottom-up proteomics analysis of the cartilages in this study can be used to define gold standard ratios of collagen types I and II in engineered cartilages. Tissue engineering studies using the self-assembling process have engineered a spectrum of fibrous and hyaline neocartilages by varying the time of 3-dimensional aggregate culture, 13 although collagen types I and II were not quantified. Other studies have used different ratios of cell sources (e.g., meniscus fibroblasts and articular chondrocytes) to achieve different ratios of collagen types. 34 Additional studies to fine-tune these techniques and generate specific collagen type I:II ratios must be completed to replace different cartilages around the body. While the minipig is a promising animal model for cartilage repair8,9 and has been used in attempts at cartilage regeneration using tissue-engineered implants, 35 a thorough characterization on the hyaline-fibrocartilage spectrum of human cartilage must also be completed to determine engineering specifications for tissue-engineered cartilage in humans because interspecies differences in collagen content have been noted. 36 In addition, the Yucatan minipigs in this study were all skeletally mature. Because the collagen and crosslink content of cartilage changes with age and disease,37,38 additional characterization of cartilages at different ages will be needed for a diverse patient population.
The relative proportion of collagen types I and II in the knee meniscus is supported by conflicting literature. An early study claimed that collagen of the porcine meniscus is completely type I,
39
but a subsequent study claimed that the white-white portion of the bovine meniscus is 60% collagen type II and 40% collagen type I.
14
This idea that the white-white meniscus contains abundant collagen type II, and, thus, is reminiscent of hyaline articular cartilage, has become accepted in the meniscus field.33,40 A more recent proteomics analysis on various cartilages reveals over a 12-fold ratio of collagen type I to collagen type II in the human knee meniscus,
41
although the region was not specified. The bottom-up proteomics analysis in our study revealed a 15-fold ratio of collagen type I to type II in the white-white region and a greater than 150-fold ratio in the red-red region. While there is indeed a stark difference in this ratio between the 2 regions, these data, in conjunction with the histological staining (
In this study, we developed LC-MS assays to quantify collagen subtypes and crosslinks in biological tissues. These assays are inexpensive, require a low operator time, and can be modified to quantify minor collagen types and their crosslinks. As a demonstration of these assays, they were applied to the cartilages of the Yucatan minipig, a promising animal model for cartilage repair. Collagen types I and II, along with the crosslinks PYR and DHLNL, were quantified in hyaline, elastic, and fibrocartilages. Bottom-up proteomics revealed a spectrum of collagen ratios in these tissues, thus showing the existence of a hyaline-fibrocartilage spectrum in native cartilage. This is one of many potential ways that the high-throughput collagen subtyping developed in this work can be used for characterization of collagenous tissues, informing best practices for tissue engineers and surgeons as they develop treatment strategies.
Footnotes
Acknowledgments and Funding
The authors would like to acknowledge Benjamin Katz and Felix Grun at the UCI Mass Spectrometry Facility for guidance on the collagen subtype and crosslink LC-MS quantification methods. The authors would like to acknowledge Clinton Yu at the UCI High-end Mass Spectrometry Facility for running samples on the Orbitrap mass spectrometer and assistance with database searching and MaxQuant analysis. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by NIH grant nos. R01DE015038, R01AR071457, and R01AR067821.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kyriacos A. Athanasiou and Jerry C. Hu are scientific consultants at Cartilage Inc.
Ethical Approval
Ethical approval was not sought for this study because the tissue samples used in this study were excised from pigs that were previously culled for purposes unrelated to this work.
Availability of Data
The proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository with the data set identifier PXD025482. The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
