Abstract
Objective
Articular cartilage-derived progenitor cells (ACPCs) are a potential new cell source for cartilage repair. This study aims to characterize endogenous ACPCs from healthy and osteoarthritic (OA) cartilage, evaluate their potential for cartilage regeneration, and compare this to cartilage formation by chondrocytes.
Design
ACPCs were isolated from full-thickness healthy and OA human cartilage and separated from the total cell population by clonal growth after differential adhesion to fibronectin. ACPCs were characterized by growth kinetics, multilineage differentiation, and surface marker expression. Chondrogenic redifferentiation of ACPCs was compared with chondrocytes in pellet cultures. Pellets were assessed for cartilage-like matrix production by (immuno)histochemistry, quantitative analyses for glycosaminoglycans and DNA content, and expression of chondrogenic and hypertrophic genes.
Results
Healthy and OA ACPCs were successfully differentiated toward the adipogenic and chondrogenic lineage, but failed to produce calcified matrix when exposed to osteogenic induction media. Both ACPC populations met the criteria for cell surface marker expression of mesenchymal stromal cells (MSCs). Healthy ACPCs cultured in pellets deposited extracellular matrix containing proteoglycans and type II collagen, devoid of type I collagen. Gene expression of hypertrophic marker type X collagen was lower in healthy ACPC pellets compared with OA pellets.
Conclusions
This study provides further insight into the ACPC population in healthy and OA human articular cartilage. ACPCs show similarities to MSCs, yet do not produce calcified matrix under well-established osteogenic culture conditions. Due to extensive proliferative potential and chondrogenic capacity, ACPCs show potential for cartilage regeneration and possibly for clinical application, as a promising alternative to MSCs or chondrocytes.
Introduction
Cartilage defects larger than 2 cm2 are currently treated by transplantation of autologous culture-expanded chondrocytes.1,2 As chondrocytes are the resident cell type in cartilage, this cell type is the prime candidate for cartilage repair. Although long-term results are promising with good patient reported outcomes and radiographic signs of cartilage formation,1,3 drawbacks of the treatment remain. Extensive culture of chondrocytes for autologous administration induces dedifferentiation and loss of phenotype. 4 In addition, graft hypertrophy can lead to continued ailments and may necessitate further revision surgery. 5 These drawbacks can potentially be resolved by other cell sources. Mesenchymal stromal cells (MSCs) are frequently used 6 due to the relatively non-invasive methods for isolation, extensive culture-expansion potential,7,8 and efficient in vitro differentiation into chondrocytes producing cartilaginous tissue.9,10 However, the associated risk of MSCs differentiating into hypertrophic chondrocytes and subsequent endochondral ossification poses a challenge. 11 While chondrocytes and MSCs are impacting the way cartilage defects are treated, different cell sources overcoming potential limitations may further advance the quality of repair tissue, and hence possibly clinical outcomes, leaving a gap for improvement in cell-based cartilage tissue engineering.
Initially, a small portion of isolated articular chondrocytes was described to grow clonally and differentiate into several lineages. 12 Next, a distinct progenitor cell with stem cell-like characteristics was identified in the superficial zone, first in bovine 13 and later also in human cartilage. 14 This endogenous progenitor population is referred to as cartilage stem cells, mesenchymal or chondrogenic progenitor cells, or articular cartilage-derived progenitor cells (ACPCs).
Besides extensive culture expansion, 15 ACPCs are successful at producing neo-cartilage in vitro16,17 and in vivo.14,18 Several reports indicate that ACPCs have no tendency to differentiate into hypertrophic chondrocytes, unlike MSCs.19,20 These combined features give ACPCs a preference over chondrocytes and MSCs for cartilage regeneration. Furthermore, similar to chondrocytes, 21 pathological origin could influence ACPC performance. Indeed, OA-derived ACPCs were shown to possess chondrogenic properties, like healthy cartilage-derived ACPCs. 22 However, direct comparisons of chondrogenic potential of ACPCs from healthy and OA cartilage are limited.22,23 Direct comparison can provide insight in the pathophysiology of OA and the potential role of ACPCs in health and disease.
The current study aims to characterize and compare fibronectin-selected ACPCs from healthy and OA human cartilage. By direct comparison of ACPC populations to full-depth cartilage cell populations derived from the same donors, their potential for cartilage regeneration is investigated.
Methods
Tissue Collection
Macroscopically healthy cartilage (n = 6, age = 46-49) was isolated post-mortem from full-weight-bearing and non-weight-bearing locations of the knee (Department of Pathology, University Medical Center Utrecht). Osteoarthritic (OA) cartilage was obtained from redundant material from patients (n = 6, age = 55-79) undergoing total knee arthroplasty. Anonymous collection of this tissue was performed according to medical ethics regulations of the University Medical Center Utrecht and the guideline “good use of redundant tissue for research” of the Dutch Federation of Medical Research Societies. 24 Human MSCs (n = 6, age = 30-66) were derived from bone marrow from the iliac crest of patients receiving spondylodesis or total hip arthroplasty surgery after their informed consent and according to a protocol approved by the local medical ethical committee.
Cell Isolation and Expansion
Cartilage from all parts of the joint (weight-bearing and non-weight-bearing) was pooled for each donor. Cartilage pieces were digested in 0.2% (w/v) pronase (Sigma-Aldrich) in Dulbecco’s modified Eagle’s medium (DMEM; Gibco) with 1% (v/v) penicillin/streptomycin (pen/strep; 100 U/mL, 100 µg/mL; Gibco) for 2 hours at 37°C, followed by overnight digestion in 0.075% (w/v) type II collagenase (Worthington, Lakewood, NJ) in DMEM with 10% (v/v) heat-inactivated fetal bovine serum (FBS; Biowest) and 1% pen/strep under agitation. The population from here on referred to as “chondrocytes” is the total cell population isolated from cartilage, without any purification or selection. 14 Chondrocytes were expanded using chondrocyte expansion medium (DMEM, 10% FBS, 1% pen/strep). To isolate ACPCs, the digest was seeded at 500 cells/cm2 at 37°C on culture plastic pre-coated for 1 hour with fibronectin (1 µg/mL in PBS containing MgCl2 and CaCl2; Sigma-Aldrich) using serum-free medium (DMEM with 1% pen/strep). After 20 minutes, non-adhered cells were washed away and ACPC expansion medium was added (DMEM, 10% FBS, 1% pen/strep, 200 µM l-ascorbic acid 2-phosphate [ASAP; Sigma-Aldrich], and 5 ng/mL basic fibroblast growth factor [bFGF; PeproTech]). On day 6, colonies (>32 cells) were isolated using sterile glass cloning cylinders (Sigma-Aldrich). Collected colonies were pooled and ACPCs were further expanded on conventional tissue culture plastic with ACPC expansion medium.
The mononuclear fraction of bone marrow was separated by centrifugation using a Ficoll-Paque density gradient (GE Healthcare, The Netherlands) and plated using MSC expansion medium (Minimum Essential Media [αMEM; Gibco, The Netherlands], 10% FBS, 1% pen/strep, 200 µM ASAP, and 1 ng/mL bFGF). MSCs were expanded up to passage 4 before use in differentiation assays.
Flow Cytometry
Cells were washed in buffer (0.5% w/v bovine serum albumin [BSA], 2mM EDTA in PBS) and incubated with antibodies against CD49e, CD146, CD166 (Miltenyi Biotec), CD105 (Abcam), CD90, CD73, or a cocktail of markers (CD45, CD34, CD11b, CD79A, HLA-DR; all R&D Systems) according to the manufacturers’ instructions. Labeled cells were analyzed using a BD FACSCanto II or BD LSRFortessa (BD Biosciences, USA). Dead cells were excluded using 100 ng/mL 4′,6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich) or 1 µg/mL 7-Aminoactinomycin D (7AAD; Molecular Probes). Results were analyzed using FlowJo V10 data analysis software package (TreeStar, USA).
Colony Forming Unit Fibroblast Assay
Chondrocytes directly after isolation (passage 0) and ACPCs and chondrocytes at passages 2 and 4 were seeded at 50 and 100 cells per 6-well plate well in duplicates. Cells were cultured with corresponding expansion medium. After 7 days, colonies were fixed with 10% formalin and stained for 45 minutes using 0.05% (v/v) Crystal Violet (Sigma-Aldrich) in Milli-Q water and counted.
Multilineage Differentiation
For chondrogenic and hypertrophic differentiation, 2.5 × 105 ACPCs (passage 4) or chondrocytes (passage 2) were pelleted in 15 mL Falcon tubes by centrifugation at 320 g for 5 minutes. Pellets were cultured in chondrogenic medium (DMEM with 1% pen/strep, 2% (v/v) human serum albumin [HSA; Albuman, Sanquin Blood Supply Foundation], 1% insulin-transferrin-selenium-ethanolamine [ITS-X; Gibco], 0.1 µM dexamethasone, 0.2 mM ASAP, and 10 ng/mL transforming growth factor [TGF]-β1 [PeproTech]). After 21 days of chondrogenic differentiation, half of the pellets was fixed using 10% buffered formalin. The other half of the pellets was exposed to hypertrophic medium (DMEM with 1% pen/strep, 1% ITS-X, 1 nM dexamethasone, 200 µM ASAP, 10 mM β-glycerophosphate [BGP; Sigma-Aldrich], and 1 nM 3,3′,5-Triiodo-L-thyronine [Sigma-Aldrich]) for 7 additional days. After a total of 28 days, the hypertrophic-treated pellets were fixed with 10% formalin and processed for histology.
For osteogenic and adipogenic differentiation, ACPCs (passage 4) and chondrocytes (passage 2) were seeded in 24-well plate wells using corresponding expansion media. Upon subconfluency, monolayers were treated with osteogenic medium (αMEM with 10% FBS, 1% pen/strep, 200 µM ASAP, 10 mM BGP, and 10 nM dexamethasone [Sigma-Aldrich]) or adipogenic medium (αMEM with 10% FBS, 1% pen/strep, 1 µM dexamethasone, 0.5 mM 3-Isobutyl-1-methylxanthine [IBMX; Sigma-Aldrich], 0.2 mM indomethacin [Sigma-Aldrich], and 1.72 µM insulin [Sigma-Aldrich]) for 21 days. After 21 days, monolayers were fixed with 10% formalin and stained for calcium deposits by 40 mM alizarin red S in demineralized water (pH 4.1; Sigma-Aldrich) or intracellular lipid vacuoles by 7.3 mM oil red O (Sigma-Aldrich) in 60% isopropanol. For all assays, MSCs were ran in parallel as positive controls.
Additional osteogenic differentiation was performed by expanding and differentiating ACPCs using several batches of FBS (Biowest and Gibco) and platelet lysate 25 (Sanquin Blood Supply Foundation). Also, monolayers were treated with osteogenic medium with 100 ng/mL recombinant human Bone Morphogenetic Protein-2 26 (InductOS) and cell pellets 14 were stimulated with osteogenic medium, both for 21 days. Results are shown in Supplementary Figure S1.
Pellet Redifferentiation Culture
For the redifferentiation cultures, 2.5 × 105 cells were pelleted in ultra-low attachment 96-well plate wells by centrifugation at 320 g for 5 minutes. Pellets were cultured for 28 days in redifferentiation medium (DMEM with 1% pen/strep, 2% HSA, 2% ITS-X, and 1% ASAP). Half of the pellets was supplemented with 10 ng/mL TGF-β1. Used medium was stored at −20°C for further analysis. Pellets were processed for biochemical analyses, gene expression, and (immuno)histochemistry.
Biochemical Analysis of Pellets
Pellets were digested overnight with papain (250 µg/mL papain [Sigma-Aldrich], 0.2 M NaH2PO4, 0.1M EDTA, 0.01M cysteine, pH 6) at 60°C. Deposition of sulphated glycosaminoglycans (GAGs) and release into the medium was measured by a dimethylmethylene blue assay (pH 3). The 525 / 595 nm absorbance ratio was measured using chondroitin-6-sulfate (Sigma-Aldrich) as a reference. DNA was quantified using a Quant-iT PicoGreen assay (Invitrogen) according the manufacturer’s instructions.
Histological Analysis of Pellets
Pellets were processed for histology by dehydration through graded ethanol steps, clearing in xylene, and embedding in paraffin. Five-µm sections were stained for proteoglycan production with 0.125% Safranin-O (Merck) counterstained with 0.4% fast green (Sigma-Aldrich) and Weigert’s hematoxylin (Clin-Tech). Collagen deposition was visualized by immunohistochemistry. Sections were blocked in 0.3% (v/v) hydrogen peroxide. Antigen retrieval for type I and II collagen was performed with 1 mg/mL pronase (Sigma-Aldrich) and 10 mg/mL hyaluronidase (Sigma-Aldrich) and for type X collagen with 1 mg/mL pepsin (Sigma-Aldrich) and 10 mg/mL hyaluronidase for 30 minutes at 37°C. Sections were blocked with 5% (w/v) BSA in PBS for 1 hour at room temperature and incubated with primary antibodies for type I collagen (EPR7785 [BioConnect], 1:400 in 5% PBS/BSA), type II collagen (II-II6B3 [DHSB], 1:100 in 5% PBS/BSA), and type X collagen (X53 [Quartett], 1:20 in 5% PBS/BSA) overnight at 4°C. Appropriate IgGs were used as isotype controls. Next, type I collagen sections were incubated with BrightVision Poly-HRP-Anti Rabbit (ImmunoLogic) and type II collagen sections were incubated with goat-anti-mouse IgG HRP-conjugated (DAKO, P0447; 1:100 in 5% PBS/BSA), both for 1 hour at room temperature. Type X collagen sections were incubated with biotinylated sheep-anti-mouse IgG (RPN1001V [GE Healthcare]) for 1 hour at room temperature, then with streptavidin-HRP for 1 hour at room temperature (DAKO, P0397; 1:1000 in 5% PBS/BSA). Next, all stainings were developed using 3,3’-diaminobenzidine (DAB, Sigma-Aldrich). Cell nuclei were counterstained with Mayer’s hematoxylin (Klinipath).
Gene Expression
Gene expression analysis was performed by real-time polymerase chain reaction (PCR). RNA was isolated from cells and pellets using TRIzol (Invitrogen) according to the manufacturer’s instructions. Total RNA (200-500 ng) was reverse-transcribed using the High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems). Real-time PCRs were performed using iTaq Universal SYBR Green Supermix (Bio-Rad) in the LightCycler 96 (Roche Diagnostics) according to the manufacturer’s instructions. Primers (Invitrogen) are listed in Table S1 . Relative gene expression was calculated using 18S as a housekeeping gene. Amplified PCR fragments extended over at least one exon border (except for 18S). The primer for detection of 2 splice variants of COL2A1 extended across exon 2 of the gene and results in amplification of splice variants IIa and IIb. PCR products were separated on 2% (w/v) agarose gel electrophoresis and stained with SYBR Safe (Invitrogen).
Statistical Analysis
Experiments for flow cytometry, colony-forming efficiency, and multilineage differentiation were performed with cells from 6 healthy and 6 OA cartilage donors, unless stated otherwise. Pellet redifferentiation culture was performed with cells from 3 donors and 3 technical replicates per donor. All data are expressed as mean ± standard deviation (SD). Data were analyzed using the GraphPad Prism 8 software package (GraphPad Software, United States). Normality was confirmed with a Shapiro-Wilk test (p>0.05). Groups were compared using an unpaired t test, 1- or 2-way analysis of variance (ANOVA) with Bonferroni post hoc test. Gene expression data were not normally distributed and therefore analyzed with a Mann-Whitney or Kruskall-Wallis test with Dunn’s post hoc test. A value of P < 0.05 was considered statistically significant.
Results
Cells Isolated from Human Cartilage Have a Chondrogenic Profile
All samples consistently contained cells highly expressing the cartilage-specific marker cartilage link protein (HAPLN1), while having low expression of the synovial-specific marker microfibril-associated glycoprotein-2
27
(MFAP5;
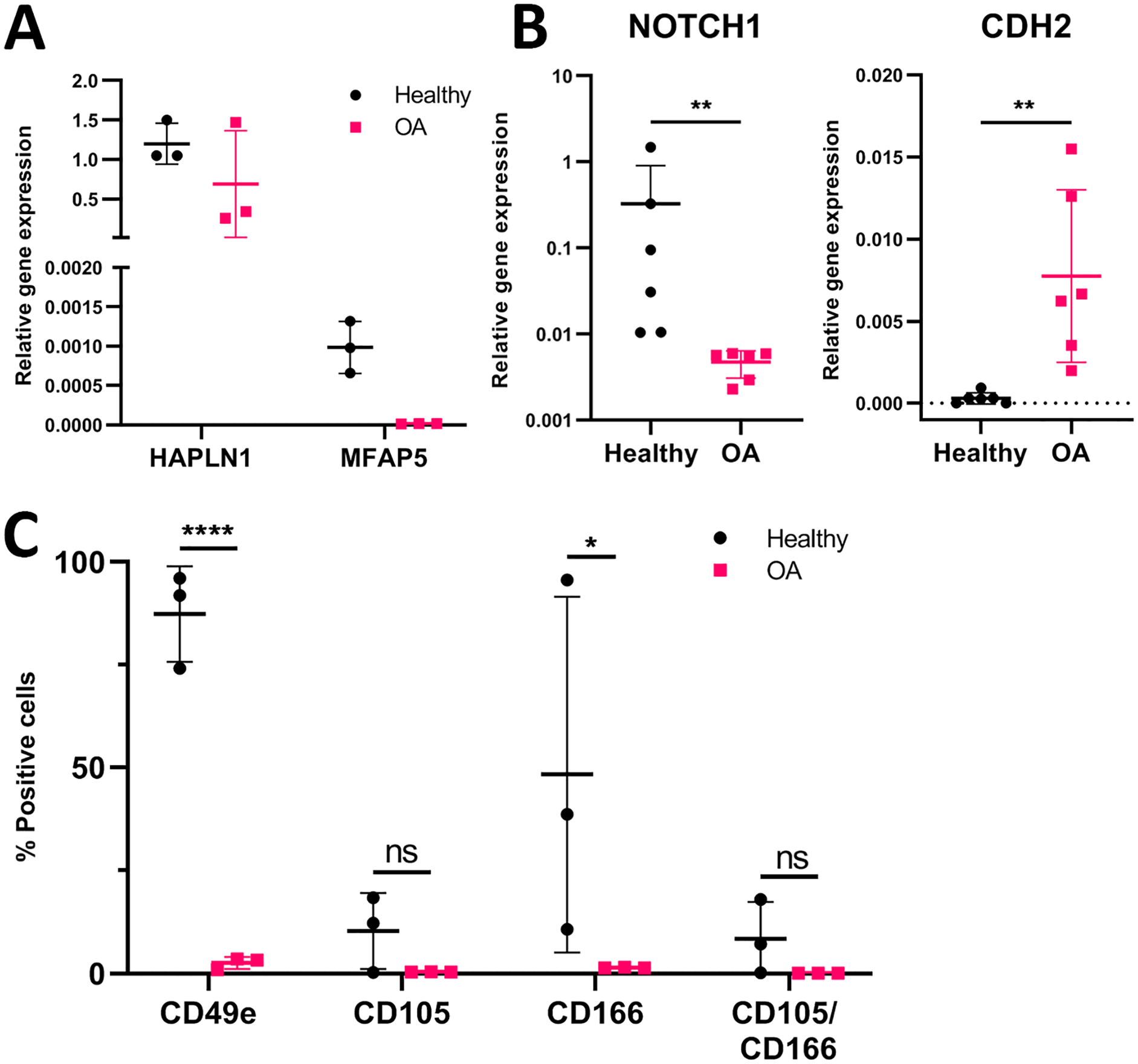
Characterization of full-depth cartilage cell populations. (
Expression of Progenitor-Specific Markers in Freshly Isolated Chondrocytes
Gene expression of freshly isolated chondrocytes revealed a significant difference in Notch receptor 1 (NOTCH1) between healthy and OA-derived cells (0.3274 ± 0.5821 vs. 0.0047 ± 0.0016; p = 0.0022;
Selection by Adhesion to Fibronectin Results in a Population with High Clonogenicity and Proliferative Capacity
Colony-forming efficiency of ACPCs at passage 2 and 4 was higher than of the full population (P < 0.0001 for all groups;
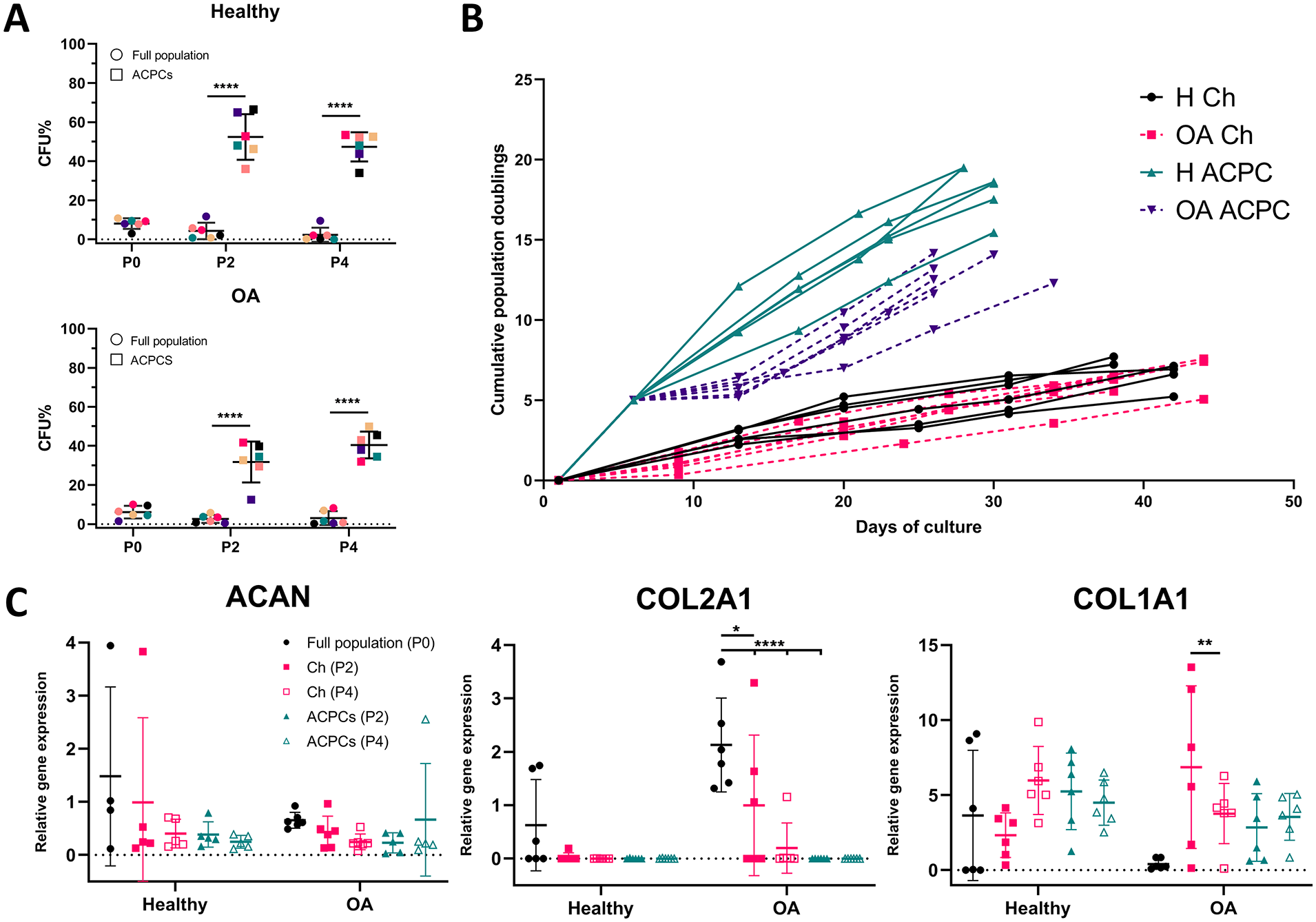
Culture expansion of articular cartilage-derived progenitor cells (ACPCs) versus non-selected chondrocytes. (
ACPCs Fail to Produce Mineralized Matrix upon Osteogenic and Hypertrophic Induction
All healthy and OA ACPCs differentiated into the chondrogenic and adipogenic lineage, indicated by Safranin-O and oil red O stainings (
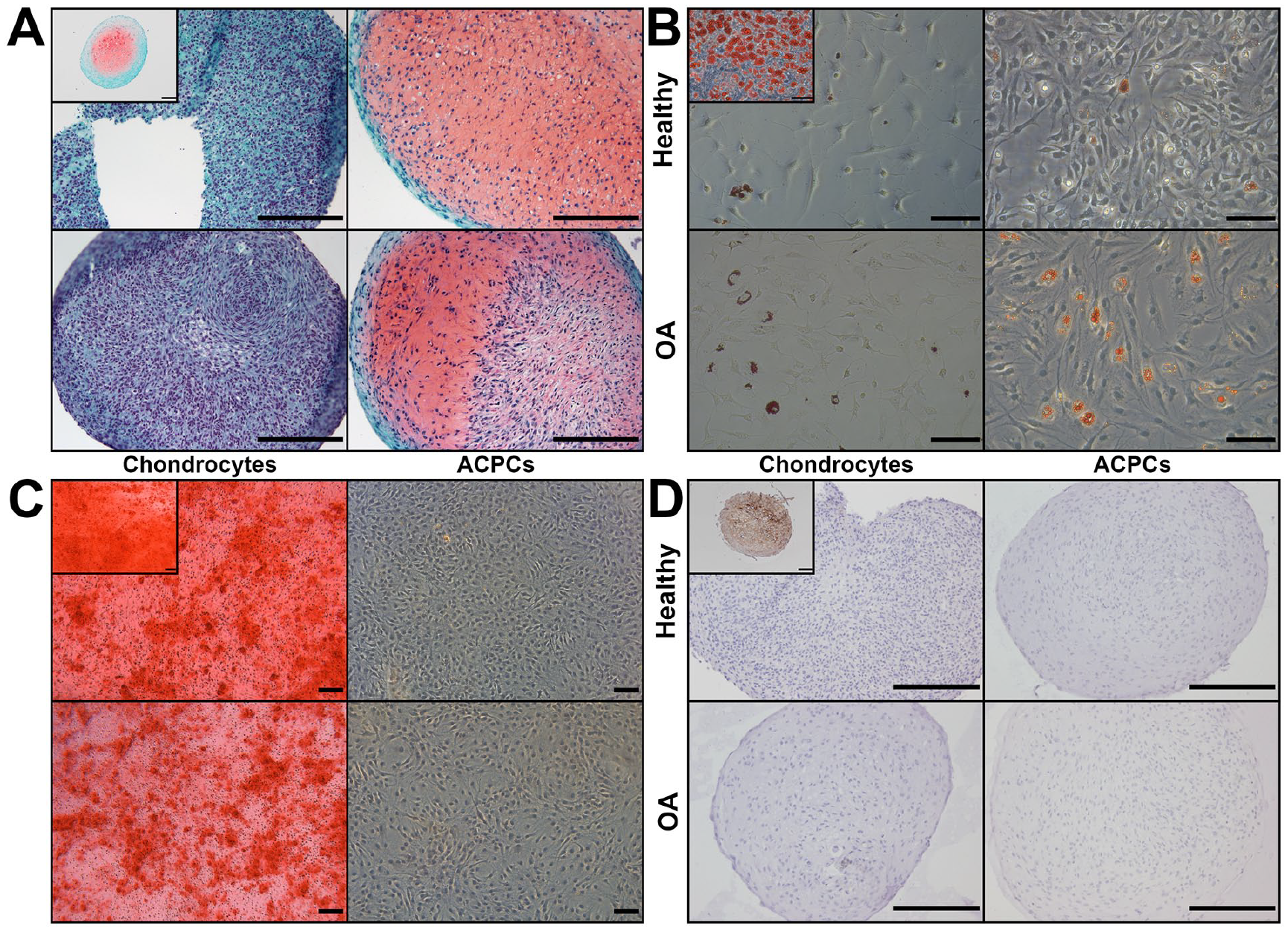
Differentiation of chondrocytes and articular cartilage-derived progenitor cells (ACPCs) into 4 lineages. (
Expression of Bone Marrow-Derived MSC Surface Markers in Culture-Expanded ACPCs
Expression of cell surface markers defined for bone marrow-derived MSCs in monolayer culture,
28
CD90, CD105, and CD73, was >95% in all populations. CD166-expression was >99% in all ACPC donors, but lower in OA chondrocytes (87.8% ± 13.6%) compared with healthy ACPCs (99.7% ± 0.1%; p = 0.0322) and OA ACPCs (99.8% ± 0.1%; p = 0.0315). Expression of CD146 was higher in OA chondrocytes (39.0% ± 8.1%) compared with healthy chondrocytes (22.2% ± 0.0%; p = 0.0322), healthy ACPCs (27.3% ± 15.2%; p = 0.0352), and OA ACPCs (20.9% ± 4.6%; p = 0.0003). Expression of several markers was tested using a cocktail containing antibodies against CD45, CD34, CD11b, CD79A, and HLA-DR. All cell types were <2% positive for this cocktail of markers (
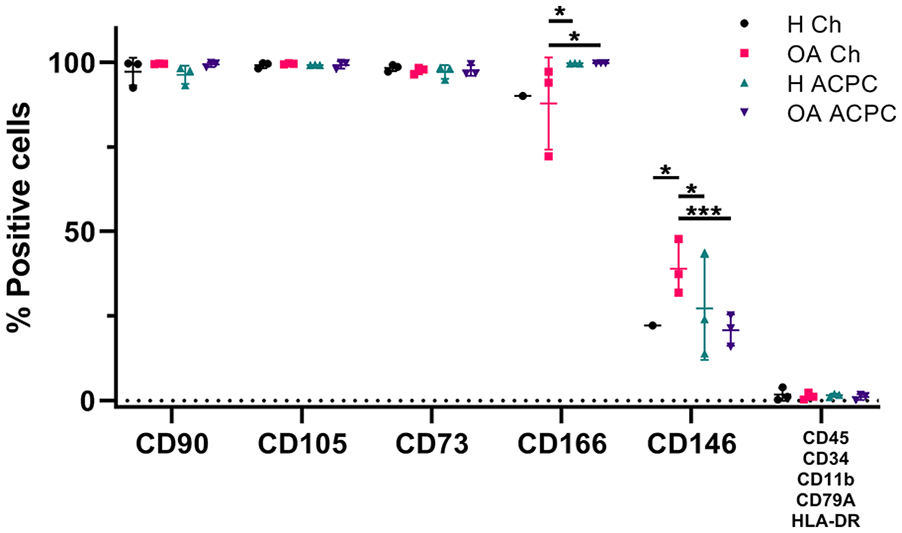
Cell surface marker expression by flow cytometry. Expression of CD90, CD105, and CD73 was >95% in all donors (n = 3). CD166 expression was lower in osteoarthritic (OA) chondrocytes compared with healthy and OA articular cartilage-derived progenitor cells (ACPCs). CD146 expression was higher in OA chondrocytes compared with the other cell types. All cell types were <2% positive for CD45, CD34, CD11b, CD79A, and HLA-DR. Three technical replicates per donor, each data point represents data of one donor. *P < 0.05. ***P < 0.001.
ACPC Pellets Produce Proteoglycans and Type II Collagen In Vitro
Chondrocytes and ACPC pellets stained positive for proteoglycans when stimulated with 10 ng/mL TGF-β1. When the redifferentiation medium was not supplemented with TGF-β1, pellets stained negative for proteoglycans (
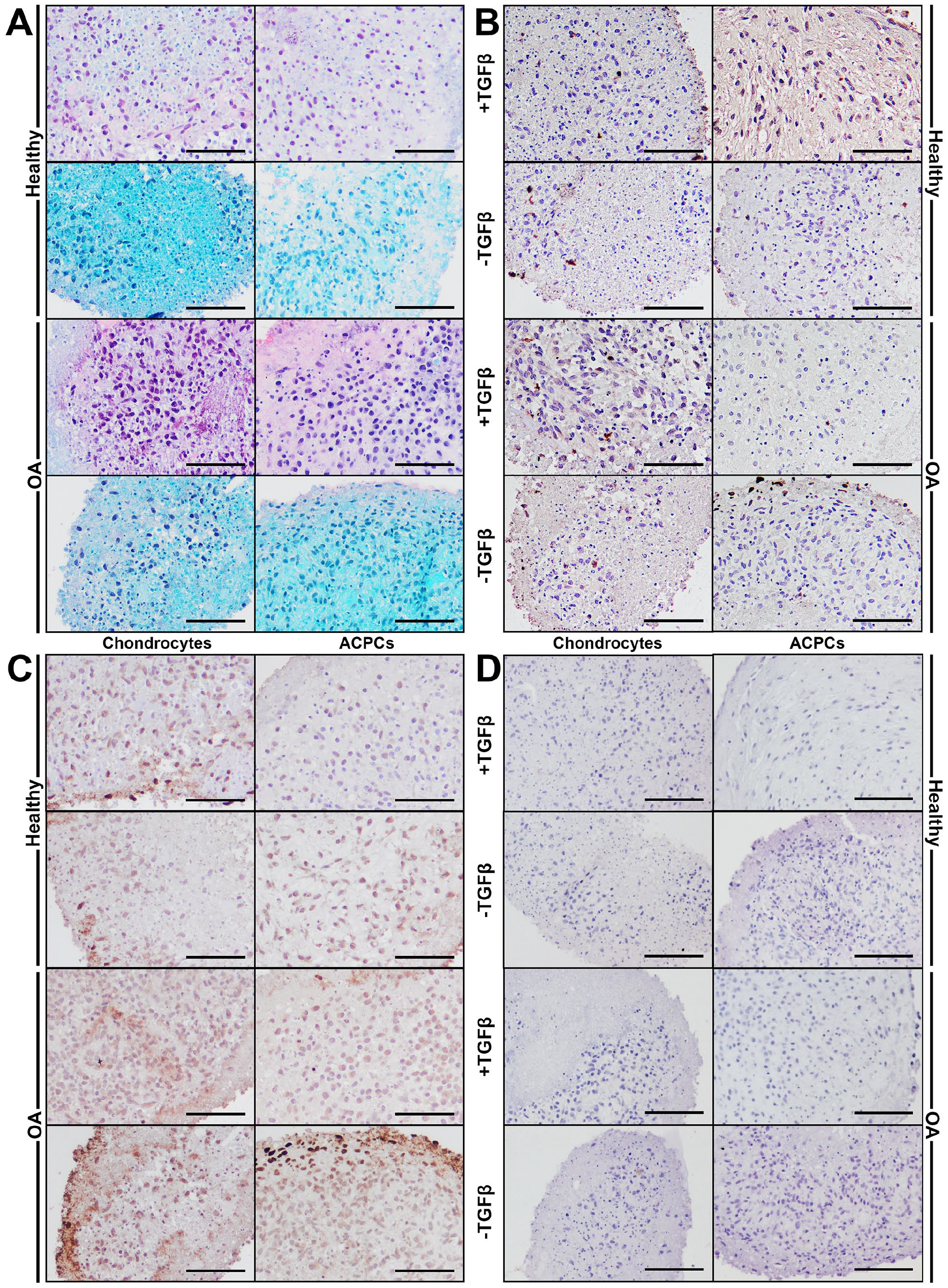
Histological staining of redifferentiated pellets. (
Healthy ACPC pellets cultured with TGF-β1 contained more GAGs compared with OA chondrocyte and OA ACPC pellets (1.1 ± 0.4 µg vs. 0.3 ± 0.3 µg and 0.3 ± 0.3 µg, p = 0.0115 and p = 0.0152, respectively;
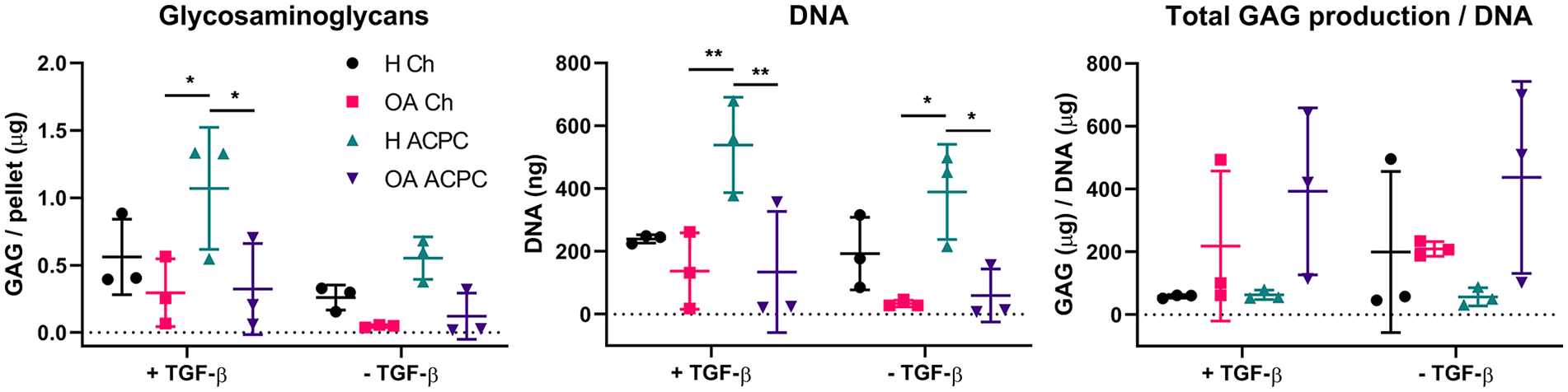
Glycosaminoglycan (GAG) and DNA content in redifferentiated pellets after 4 weeks. Quantification of GAGs shows significantly more GAGs produced in healthy articular cartilage-derived progenitor cells (ACPC) pellets cultured in the presence of transforming growth factor (TGF)-β1 (left panel). DNA quantification shows significantly more DNA present in healthy ACPC pellets, suggesting bigger pellet sizes (middle panel). Total GAG production corrected for DNA content reveals no differences between the experimental groups (right panel). N = 3 for all cell types. Three technical replicates per donor, each data point represents data of one donor. *P < 0.05. **P < 0.01.
Reduced Expression of Hypertrophic Marker Type X Collagen
Gene expression analysis was only performed on TGF-β1-treated pellets, as insufficient amounts of RNA could be isolated from non-TGF-β1-treated pellets. No difference was found between expression of chondrogenic genes aggrecan (ACAN), type II collagen (COL2A1), and SRY-box transcription factor 9 (SOX9) between the experimental groups (
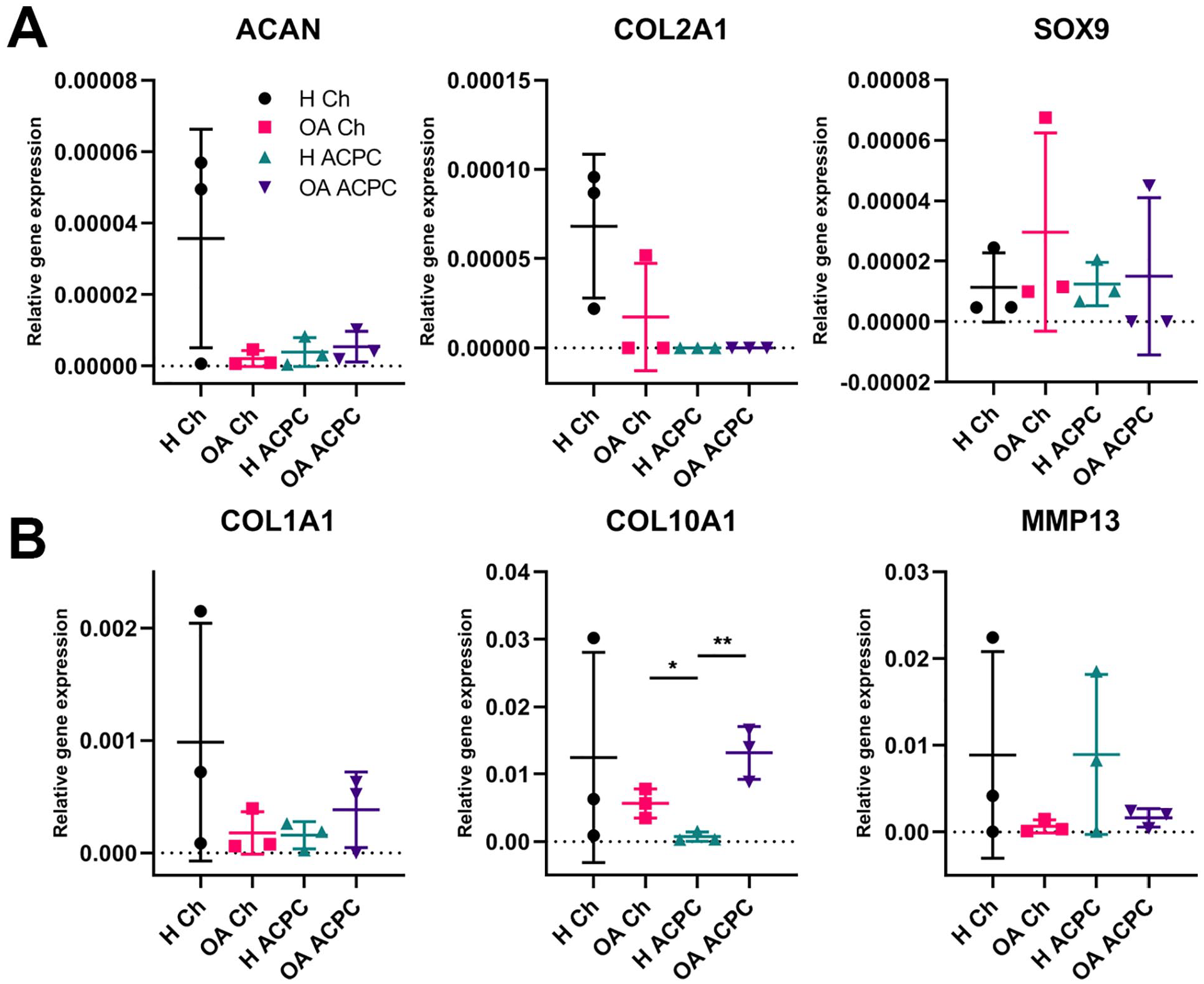
Gene expression of redifferentiated pellets. (
Discussion
The current study aimed to characterize human ACPCs from healthy and OA cartilage and determine their potential for cartilage regeneration. While fibronectin-selected progenitor populations have been described in healthy 14 and OA human cartilage,22,29 this study is the first to directly compare chondrogenic functionality of ACPCs from healthy and OA cartilage to chondrocytes derived from matching donors. The results confirm the presence of an ACPC population in human articular cartilage.14,22,23,29 Differential adhesion to fibronectin resulted in a cell population that was capable of clonal growth, extensive culture expansion, multilineage differentiation, and had a limited tendency to produce mineralized matrix and terminally differentiate into hypertrophic chondrocytes. As chondrogenic potential of chondrocytes can be dependent on the disease state of cartilage, 21 ACPCs derived from healthy and OA cartilage might provide as good candidates for cartilage repair. The current side-by-side comparison of healthy- with OA-derived ACPCs and donor-matched chondrocytes provides an overview of these cells’ potential for cartilage regeneration.
We investigated full-depth healthy and OA cell populations attempting to find correlations between previously reported progenitor markers and ACPC quantity. Expression of NOTCH1 was found to be higher in healthy cells compared with OA, confirming previous findings. 30 At the same time, CDH2 was significantly upregulated in OA cells. The cell-cell adhesion molecule N-cadherin is related to cellular condensation in early chondrogenesis during development and absent in differentiated cartilage.31,32 This might be a result of chondrocyte clustering in OA33,34 and the cells potentially obtaining a more premature chondrogenic phenotype. CDH2 in culture-expanded ACPCs was previously found to be higher in non-OA ACPCs compared with OA ACPCs, 35 indicating that this difference is lost upon selection for progenitors and culture expansion. Therefore, NOTCH1 and CDH2 might be used as markers to distinguish between healthy- and OA-derived cells.
Expression of surface markers CD49e (integrin-α5, part of the key fibronectin receptor) and CD166 was lost in the total population of OA cells compared with healthy cells, and no difference in CD105/CD166-double positive cells was found. These findings are in contrast to previous ones, 23 where a higher percentage of double positive cells was found in OA tissue. However, others found similar amounts 36 or more CD105/CD166-double positive cells in healthy cartilage versus OA. 37 The OA cartilage in the current study was obtained from end-stage OA patients and was not scored on OA severity. Severely degraded OA cartilage has lost most of its superficial layer and would subsequently also have lost superficial zone cells, which express CD49e 13 and CD166. 36 Separation based on CD49e-expression could lead to a population with improved chondrogenic potential, like healthy cartilage-derived ACPCs.
Our results show separation based on differential adhesion to fibronectin results in a population with enriched colony-forming efficiency and increased proliferative potential. Fibronectin-selected ACPCs were previously found to maintain telomerase activity and telomere length up to at least 22 population doublings,14,38 which is more than the number of population doublings reached in our study. When using the same expansion media to expand both cell types, population doublings in chondrocytes were limited compared with ACPCs with chondrocyte expansion medium, supporting the findings of higher cell yields of ACPCs. To add, culture time of chondrocytes decreased over passaging, indicating increasing cell size and possible dedifferentiation. On the contrary, OA-derived ACPCs lost mRNA expression of type II collagen upon culture expansion. Articular chondrocytes are known to dedifferentiate in monolayer expansion, 4 but regain their phenotype when exposed to appropriate culture conditions.12,39 Similarly, our ACPCs regained their differentiation potential and especially healthy cartilage-derived ACPCs were successful in producing type II collagen- and proteoglycan-rich matrix in vitro, while chondrocytes were less effective. Gene expression and protein deposition after the culture period of 4 weeks did not correspond, for which type II collagen is the most evident example. While H-ACPCs pellets cultured with TGF-β1 did not express the gene corresponding for type II collagen, immunohistochemistry revealed a slightly positive staining in these pellets. While discrepancies between protein and gene expression are regularly seen, 40 the chondrogenic response of ACPCs in this case might have been earlier than that of chondrocytes, resulting in differences in gene expression between the cell types at the 4-week evaluation point. Evaluation of gene expression throughout the culture period would give insight into the timing of the response. Furthermore, evaluation of individual clones of ACPCs would shed more light on cell performance and would allow for selection of populations with optimal characteristics, as was investigated before.14,38 Yet, for the purpose of the current study it would considerably delay expansion time to obtain sufficient amounts of cells for tissue engineering and limit clinical application.
Since there is a need for identification of unique markers for selecting ACPCs, we specifically looked into gene expression of type II collagen splice variants in order to investigate whether this marker could be used for discerning ACPCs and chondrocytes. Splice variant IIa is an established marker for juvenile chondrocytes or mesenchymal cells, while variant IIb is expressed by mature chondrocytes.41,42 Although ACPCs would be chondrogenic precursors and were expected to express the immature variant of type II collagen, no differences were found here between the cell types or passage numbers, a possible result of the cells being in the expansion phase rather than in redifferentiation and are not actively producing extracellular matrix.
ACPCs are generally referred to as MSC-like as they, besides holding multilineage differentiation potential, meet the surface marker criteria to identify MSCs. 28 In addition, there are some indications that ACPCs have similar anti-inflammatory properties as MSCs. 43 More than 95% of the ACPCs described here expressed MSC-markers CD90, CD105, CD73, and CD166, and expression of a panel of negative markers is <2%. Noteworthy, ACPCs were negative for HLA-DR, making these populations potentially interesting for allogeneic applications. Culture-expanded chondrocytes exhibit a similar pattern of surface marker expression. While the expression pattern of the ACPC populations investigated here are in line with previous reports,14,16,44 caution should be taken when drawing conclusions. Evaluating expressed surface markers straight after ACPC-selection from the total pool of cells is the only way to directly compare cell populations and avoid the effect of culture expansion on the expression profile.
Both ACPC populations were unable to produce mineralized matrix upon stimulation with various osteogenic differentiation media and protocols. While osteogenesis is generally confirmed in human ACPCs,14,22,29 indications of reduced osteogenic potential exist. Interestingly, consistent results have been reported on decreased or absent expression of hypertrophic chondrocyte marker type X collagen16,19,20,45 or early osteogenic marker alkaline phosphatase. 17 The differences in osteogenic differentiation potential between the populations investigated here and fibronectin-isolated ACPC populations described by others are remarkable. As ACPCs originate from the cartilage, the cells might be more primed toward the chondrogenic lineage rather than to differentiate into osteoblasts or continue toward terminal hypertrophic differentiation. Since others do report on osteogenic differentiation of ACPCs, minor differences in culture media composition might explain the discrepancies. Isolation and culture protocols should be conducted side by side to elucidate differences between ACPC populations. Bone marrow-derived MSCs are associated with the risk of hypertrophic cartilage formation, when cells either differentiate or deposited matrix is remodeled into bone.19,46 Because hypertrophy in autologous chondrocyte implantation (ACI) continues to be a challenge, 5 the reduced osteogenic drift of ACPCs holds great promise for these cells.
Healthy cartilage-derived ACPCs produce cartilage ECM in vitro containing proteoglycans and type II collagen, and are devoid of type I collagen. In addition, these healthy ACPC pellets had low expression of type X collagen mRNA. Cartilage harvest site and tissue quality can be important for eventual cartilage production. To obtain healthy ACPCs, we combined all load-bearing and non-load-bearing cartilage from healthy knee joints, while chondrocytes isolated for ACI procedures are generally from non-load-bearing areas. However, chondrocytes from macroscopically healthy, full-weight bearing cartilage were shown to produce more proteoglycans and type II collagen in vitro. 21 Separating sub-groups of ACPC populations based on the degree of weight-bearing might provide further insights into the physiological role of progenitors in cartilage homeostasis. While we have not investigated it in the current study, several studies report on an increased number of ACPCs in OA cartilage22,23,47,48 and numbers of ACPCs increasing after mechanical stimulation.49,50
ACPCs were used in a caprine model and had good lateral integration with the native cartilage, 14 showing potential for use in a 2-step cartilage repair procedure. Furthermore, a pilot study with 15 patients employing matrix-assisted autologous chondrocyte transplantation reported satisfactory histological and pain scoring 1 year after surgery. 18 ACPCs were expanded for a maximum of 3 weeks, substantially shorter than the expansion time needed for chondrocytes, which is generally 4 to 8 weeks,51-53 depending on growth speed and defect size. In spite of these promising early clinical results, direct comparisons between chondrocytes and ACPCs are necessary to identify advantages in length of culture expansion and quality of the repair tissue.
To conclude, ACPCs isolated here show potential for cartilage regeneration, possibly in an autologous approach replacing chondrocytes. The limited potential of these ACPC populations to produce mineralized matrix and absence of type X collagen protein and mRNA expression in healthy cartilage-derived ACPCs is promising. These observations combined with extensive in vitro expansion potential of ACPCs can have major implications for future cartilage repair treatments.
Supplemental Material
sj-docx-5-car-10.1177_19476035211059600 – Supplemental material for Progenitor Cells in Healthy and Osteoarthritic Human Cartilage Have Extensive Culture Expansion Capacity while Retaining Chondrogenic Properties
Supplemental material, sj-docx-5-car-10.1177_19476035211059600 for Progenitor Cells in Healthy and Osteoarthritic Human Cartilage Have Extensive Culture Expansion Capacity while Retaining Chondrogenic Properties by M. Rikkers, J.V. Korpershoek, R. Levato, J. Malda and L.A. Vonk in CARTILAGE
Supplemental Material
sj-tif-1-car-10.1177_19476035211059600 – Supplemental material for Progenitor Cells in Healthy and Osteoarthritic Human Cartilage Have Extensive Culture Expansion Capacity while Retaining Chondrogenic Properties
Supplemental material, sj-tif-1-car-10.1177_19476035211059600 for Progenitor Cells in Healthy and Osteoarthritic Human Cartilage Have Extensive Culture Expansion Capacity while Retaining Chondrogenic Properties by M. Rikkers, J.V. Korpershoek, R. Levato, J. Malda and L.A. Vonk in CARTILAGE
Supplemental Material
sj-tif-2-car-10.1177_19476035211059600 – Supplemental material for Progenitor Cells in Healthy and Osteoarthritic Human Cartilage Have Extensive Culture Expansion Capacity while Retaining Chondrogenic Properties
Supplemental material, sj-tif-2-car-10.1177_19476035211059600 for Progenitor Cells in Healthy and Osteoarthritic Human Cartilage Have Extensive Culture Expansion Capacity while Retaining Chondrogenic Properties by M. Rikkers, J.V. Korpershoek, R. Levato, J. Malda and L.A. Vonk in CARTILAGE
Supplemental Material
sj-tif-3-car-10.1177_19476035211059600 – Supplemental material for Progenitor Cells in Healthy and Osteoarthritic Human Cartilage Have Extensive Culture Expansion Capacity while Retaining Chondrogenic Properties
Supplemental material, sj-tif-3-car-10.1177_19476035211059600 for Progenitor Cells in Healthy and Osteoarthritic Human Cartilage Have Extensive Culture Expansion Capacity while Retaining Chondrogenic Properties by M. Rikkers, J.V. Korpershoek, R. Levato, J. Malda and L.A. Vonk in CARTILAGE
Supplemental Material
sj-tif-4-car-10.1177_19476035211059600 – Supplemental material for Progenitor Cells in Healthy and Osteoarthritic Human Cartilage Have Extensive Culture Expansion Capacity while Retaining Chondrogenic Properties
Supplemental material, sj-tif-4-car-10.1177_19476035211059600 for Progenitor Cells in Healthy and Osteoarthritic Human Cartilage Have Extensive Culture Expansion Capacity while Retaining Chondrogenic Properties by M. Rikkers, J.V. Korpershoek, R. Levato, J. Malda and L.A. Vonk in CARTILAGE
Footnotes
Supplemental Material
Acknowledgments and Funding
The antibody against type II collagen, developed by T. F. Linsenmayer, was obtained from the Developmental Studies Hybridoma Bank, created by the NICHD and maintained at The University of Iowa, Department of Biology, Iowa City, IA, United States.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the partners of Regenerative Medicine Crossing Borders (RegMed XB), a public-private partnership that uses regenerative medicine strategies to cure common chronic diseases. This collaboration project is financed by the Dutch Ministry of Economic Affairs by means of the PPP Allowance made available by the Top Sector Life Sciences & Health to stimulate public-private partnerships. The research was also supported by the Dutch Arthritis Foundation (LLP-12 and LLP-22). J.V.K. and L.V. acknowledge ZonMw (The Netherlands Organization for Health Research and Development) and the strategic theme “Regenerative Medicine & Stem Cells” of the University Medical Center Utrecht. R.L. acknowledges funding from the European Research Council (Grant Agreement No. 949806), and the Horizon 2020 Research and Innovation Program under the Grant Agreement No. 814444 (MEFISTO).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by our institutional review board.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
