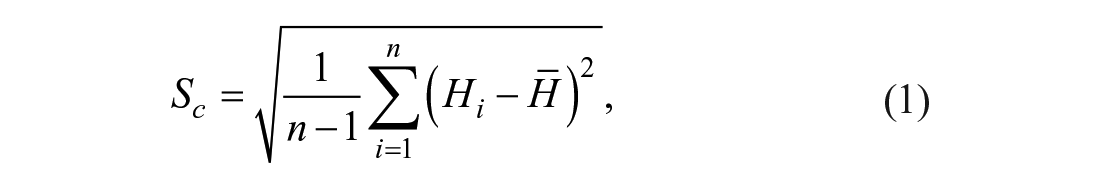Abstract
Objective
Accurate analysis to quantify cartilage morphology is critical for evaluating degenerative conditions in osteoarthritis (OA). Three-dimensional (3D) optical scanning provides 3D data for the entire cartilage surface; however, there is no consensus on how to quantify it. Our purpose was to validate a 3D method for evaluating spatiotemporal alterations in degenerative cartilages in a rabbit OA model by analyzing their curvatures at various stages of progression.
Design
Twelve rabbits underwent anterior cruciate ligament transection (ACLT) unilaterally and were divided into 4 groups: 4 weeks control, 4 weeks OA, 8 weeks control, and 8 weeks OA. 3D scanning, India ink staining, and histological assessments were performed in all groups. In 3D curvature visualization, the surfaces of the condyles were divided into 8 areas. The standard deviations (SD) of mean curvatures from all vertices of condylar surfaces and subareas were calculated.
Results
Regarding the site of OA change, curvature analysis was consistent with India ink scoring. The SD of mean curvature correlated strongly with the India ink Osteoarthritis Research Society International (OARSI) score. In curvature histograms, the curvature distribution in OA was more scattered than in control. Of the 8 areas, significant OA progression in the posterolateral part of the lateral condyle (L-PL) was observed at 4 weeks. The histology result was consistent with the 3D evaluation in terms of representative section.
Conclusions
This study demonstrated that 3D scanning with curvature analysis can quantify the severity of cartilage degeneration objectively. Furthermore, the L-PL was found to be the initial area where OA degeneration occurred in the rabbit ACLT model.
Introduction
Osteoarthritis (OA) is the most common cause of disability in the elderly. 1 The main gross morphologic characteristics are narrowing of the joint space width, sclerosis of the subchondral bone, and the presence of osteophytes. 2 Epidemiological and genetic studies of OA showed that many pre-OA disease statuses can be improved. 3 However, early diagnosis and treatment of OA is still a challenge due to the complexity and length of the disease process that is influenced by mechanical, biological, and structural factors. 4
Accurate analysis of cartilage morphology is critical for evaluating the degenerative conditions in OA. Conventionally, histology and India ink staining are the main tools to assess the health of articular cartilage in animal models. However, histology is invasive, time-consuming, and represents only two-dimensional (2D) data on cross-sections; India ink staining is a semi-quantitative macroscopic examination, and the images acquired are also 2D. Recently, magnetic resonance imaging (MRI) and computed tomography (CT) have provided three-dimensional (3D) imaging to assess cartilage surface characteristics, such as roughness, which is a marker of early OA.5,6 However, due to the partial volume artifact in MRI and CT, it is difficult to extract cartilage profiles perfectly. Arthroscopic methods rely on direct visual inspection without invading the joint surface, but they are subjective evaluations by scoring systems such as the International Cartilage Repair Society (ICRS) system, 7 and they lack objectivity and do not provide stereoscopic information to the surgeon.
3D optical scanning technology has evolved over the last several years and can capture millions of points per second to create point cloud data. 8 The process works by a detector traversing an object and collecting data on its shape and texture. 9 The resolution of 3D scans can be up to 18 μm, which is equal to or greater than that of MRI and CT. More importantly, 3D scanning does not have a partial volume artifact, because the surface evaluation does not involve the separation of the cartilage layer. Surface curvature analysis, which is mainly used in earth science and mechanical engineering,10-13 quantitatively evaluates how uneven a surface is and is sensitive to abrupt variations in elevation. Applying curvature analysis to 3D optical scanning data of joint surfaces may enable objective and quantitative evaluation of degenerated cartilage.
Our hypothesis is that quantitative evaluation of cartilage degeneration is possible by automatic calculation of the curvature distribution on the articular cartilage surface derived from a high-resolution 3D optical scanning data. We demonstrate the utility of this 3D method to detect cartilage degeneration in a rabbit OA model during different stages of OA and to indicate subareas where OA degeneration occurs first.
Methods
Animals and Surgical Procedures
All animal procedures used in this study were approved by the Institutional Animal Care and Use Committee of Hokkaido University (approval number: 19-0141). Twelve male Japanese white rabbits (20 weeks old, 3.03.5 kg, 24 knees), which were obtained from Sankyo Labo Service Corporation (Tokyo, Japan), were divided equally into 4 groups: 4w control, 4w OA, 8w control, and 8w OA group. Six knees/group were considered sufficient to determine group differences. Anterior cruciate ligament transection (ACLT)-induced instability OA modeling was performed on left knees as previously shown 14 ; this procedure induces cartilage lesions similar to those observed in humans with OA. The right knees were used as controls without any operation. At 4 weeks or 8 weeks after surgery, the rabbits were euthanatized by intravenous injection of overdosed pentobarbital.
3D Scanning Procedures
Femurs were collected and fixed in 4% formaldehyde for 48 hours and then removed to phosphate buffered saline (PBS). A total of 24 scans from 24 samples were acquired using a 3D structured light scanner (Steinbichler Comet 5, Neubeuern, Germany), which includes two parts, an 11 megapixel camera (resolution, 18 μm) and a white light fringe projector. The projector is used to project structured light patterns on the object to be scanned, and the camera is used to acquire images of the object under structured lighting. 15 The femurs were put on a rotation table, and the scanner remained static while the femurs were turning. Therefore, detailed surface information was obtained from different views. These data were then merged to create a 3D model (triangle mesh model) by COMETplus software (Steinbichler). The time of image acquisition for one sample was less than 5 minutes. This process was performed by an experienced operator.
Roughness Evaluation by Curvature Analysis
The curvature is intuitively interpreted as the deviation between a curve and a straight line in 2D, or between a surface and a plane in 3D. 13 In 2D, the curvature κ of a curve at any point is defined as the reciprocal of the osculating circle radius r at that point. In 3D, the curvature value of one point changes depending on the changes of direction of the normal plane at the point on the surface, but what we need are principal curvatures, which are the maximum curvature (κ1) and minimum curvature (κ2) of that point. Mean curvature (H) is defined as the mean value of the principal curvatures, H = (κ1+κ2)/2. To determine the mean curvature (H) in the triangle mesh model of a cartilage surface, we used algebraic point set surfaces (APSS) to fit a higher order algebraic surface on the triangle mesh model, 16 then calculated mean curvature (H) values of each vertex on the surface by MeshLab software. 17 This approach locally fits an algebraic sphere in the moving least-squares (MLS) manner to the set of vertices included in the local neighborhood of a mesh vertex. The local neighborhood itself is a small spherical area centered at the vertex with a radius so-called the filter scale. The curvature of the fitted algebraic sphere can be intuitively controlled by the filter scale values by the user. The higher the filter scale value, the larger the neighborhood of the vertex for which the sphere is fitted and the curvature is calculated. 18 If the scale is too large, areas of high sampling density will be smoothed excessively, while numerical instabilities occur in sparsely sampled regions if the scale is too small. 19 We tested several filter scale settings and finally used a scale of 2 mm, which depicted textures of cartilage surfaces accurately, such as convex and concave areas. The sensitivity of correlation coefficient in following correlation analysis to filter scale was also evaluated. The results presented that the filter scale had little influence on correlation coefficient within certain limits (Suppl. Table S1).
According to previous studies,11,12 we defined the roughness index (Sc) based on the standard deviation (SD) of mean curvature (H) from all vertices of a condylar surface 3D triangle mesh model at the given scale. Sc is defined as
where n is the number of vertices of the surface.
For curvature analysis of compartments, we divided the cartilage surface into 8 areas in the 3D curvature visualization, including anterolateral part of lateral condyle (L-AL), anteromedial part of lateral condyle (L-AM), posterolateral part of lateral condyle (L-PL), posteromedial part of lateral condyle (L-PM), anterolateral part of medial condyle (M-AL), anteromedial part of medial condyle (M-AM), posterolateral part of medial condyle (M-PL), and posteromedial part of medial condyle (M-PM). In 3D meshes, medial and lateral condyles have 4 edges, respectively. First, we found the midpoint of each side, then connected the dots to partition the medial and lateral condyle into 4 parts, respectively. Finally, each part was chosen individually, and curvature analysis was performed as above.
Macroscopic Cartilage Assessment
Femoral condyles were covered with a solution of India ink in PBS (1:5). Excess ink solution was gently absorbed by a gauze that was premoistened with PBS. Subsequently, all samples were photographed and assessed using the Osteoarthritis Research Society International (OARSI) grading system. 20 The score of one sample is the sum of medial and lateral condyles, ranging from 2 to 14, and was acquired by two independent and blinded observers. The scores obtained from the two observers were averaged. Correlation analysis was performed between India ink scores and curvature analysis to validate the accuracy of 3D scanning.
Histological Assessment
Followed by 3D scanning and India ink, the distal femurs were decalcified with 10% ethylenediaminetetraacetic acid (EDTA) and then embedded in paraffin. A 5-μm-thick sagittal section was cut from the most severe lesions. Sections were stained with Safranin O-fast green to detect the presence of proteoglycan. The severity of cartilage degeneration was assessed semi-quantitatively using the OARSI scoring system. 20 The scores were also determined by two independent blinded observers and averaged. Correlation analysis was performed between histology scores and curvature analysis to show the relationship between microscopic and macroscopic methods.
Statistical Analysis
The data are presented as means with 95% confidence intervals (CIs). Data were considered independently with the assumption of a Gaussian distribution. Statistical significance was determined by one-way analysis of variance with Tukey’s post hoc test. Correlations were determined by Spearman’s rank correlation coefficient. P values smaller than 0.05 were considered statistically significant. All analyses were performed using GraphPad Prism 6 (version 6.02, CA, USA).
Results
3D Evaluation and India Ink Staining
In terms of the location of OA changes, 3D evaluation was consistent with India ink staining ( Fig. 1 ).
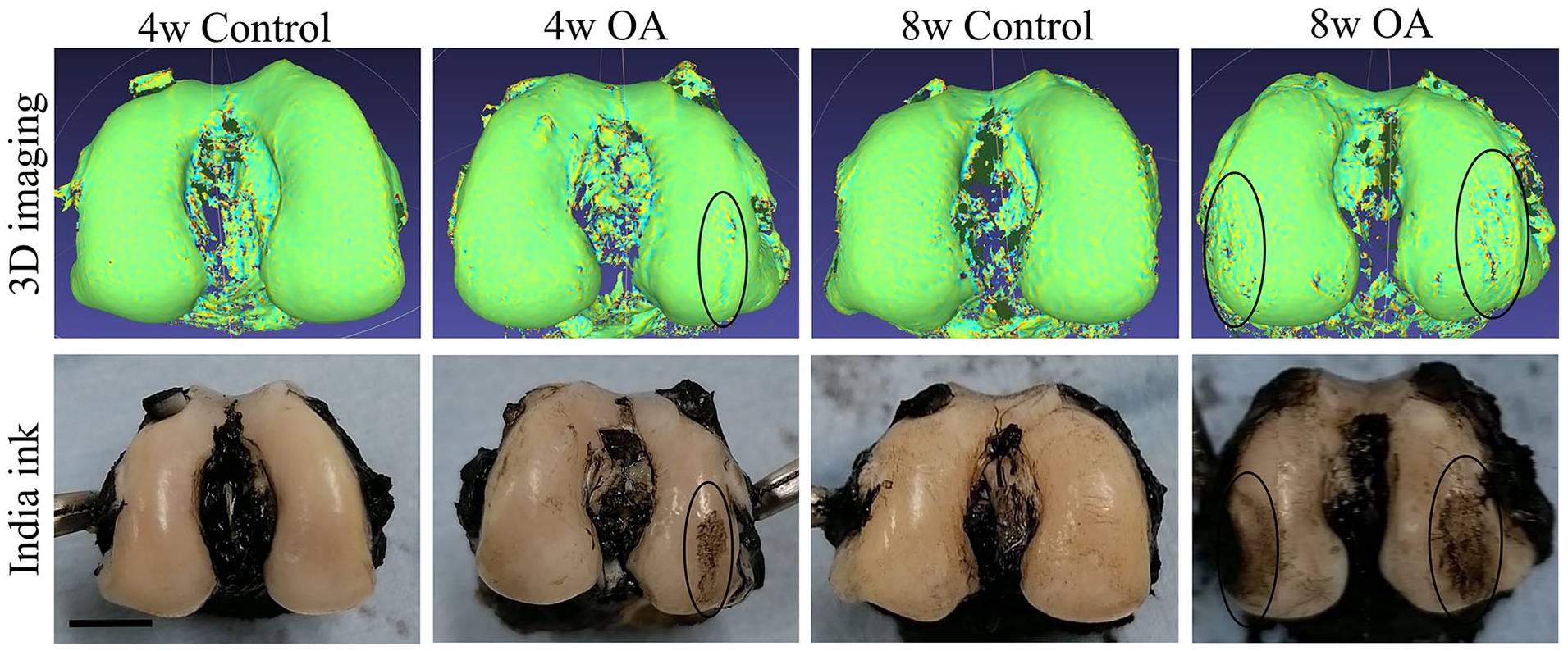
The pictures above show 3D visualization of the location of convex and concave areas of the cartilage. The areas in the circles show OA lesions in both 3D images and India ink photos. Scale bar is 5 mm. OA = osteoarthritis.
In the 3D curvature visualization (top row in
Fig. 2
), because the surface of rough areas becomes convex and concave, the color becomes warmer and colder accordingly. In the curvature histograms (bottom row in
Fig. 2
), the curvature distribution in OA groups was more scattered than in control groups. The India ink OARSI score shared the same tendency as the 3D evaluation of condyle cartilage surfaces (
Fig. 3A
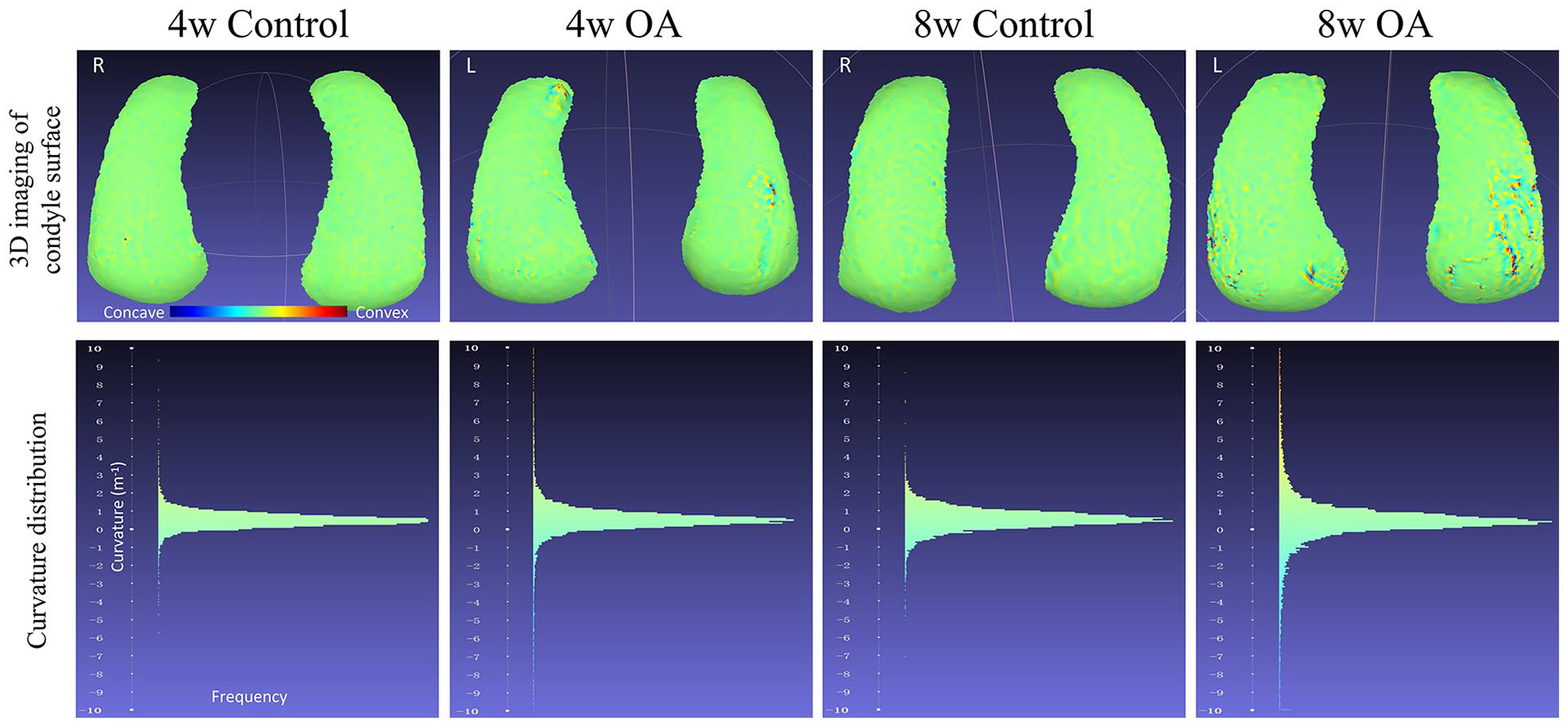
The lower pictures show the distribution of curvature values based on the 3D imaging above. The curvature distribution in the OA groups was more scattered than in control groups, and the amount of extreme curvature values in 4- and 8-week groups was also more than in control groups. OA = osteoarthritis.
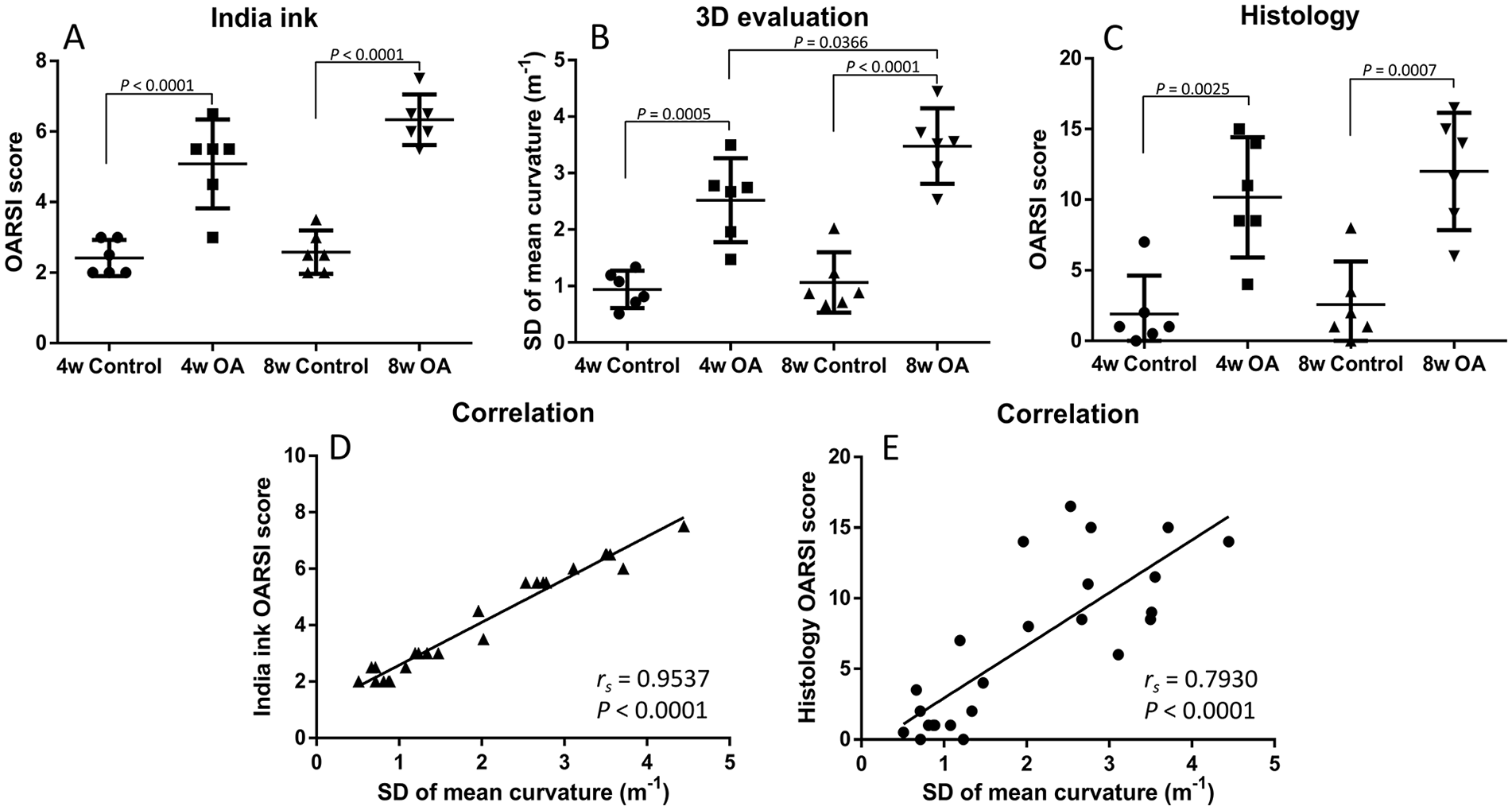
India ink score, histology score, and SD of mean curvature were increased with advanced OA (
To examine where OA degeneration occurred first, we divided condyles into 8 areas. ( Fig. 4 ). Of the 8 areas, significant OA progression in the posterolateral parts of lateral condyles (L-PL) was observed at 4 weeks ( Fig. 5 ). Over time, OA degeneration started to be observed in the anterolateral parts of medial condyles (M-AL), the posteromedial parts of medial condyles (M-PM), and the posterolateral parts of medial condyles (M-PL).
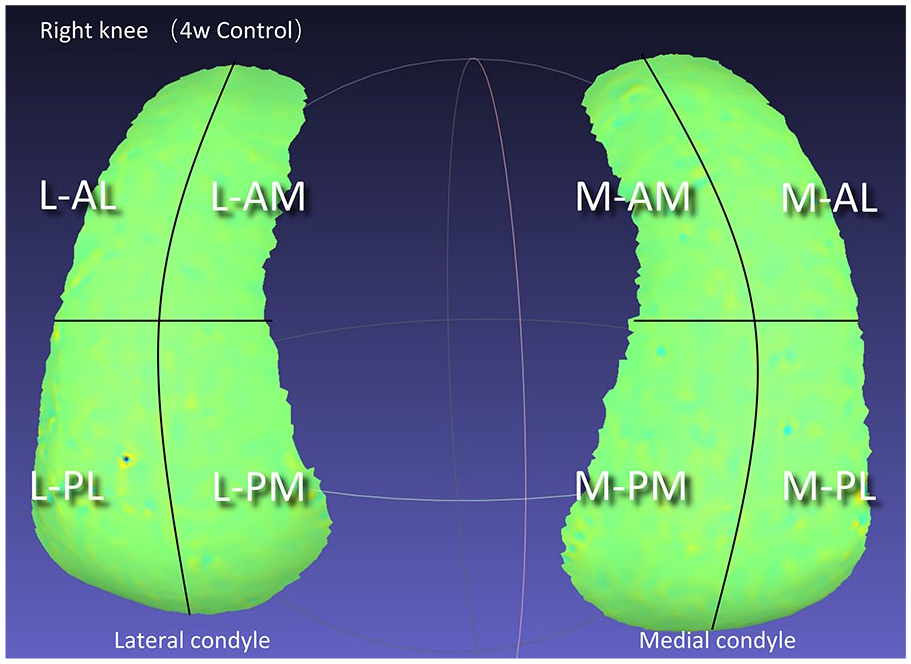
Analysis of sub-compartments. L = lateral condyle; AL = anterolateral part; AM = anteromedial part; M = medial condyle; PL = posterolateral part; PM = posteromedial part.
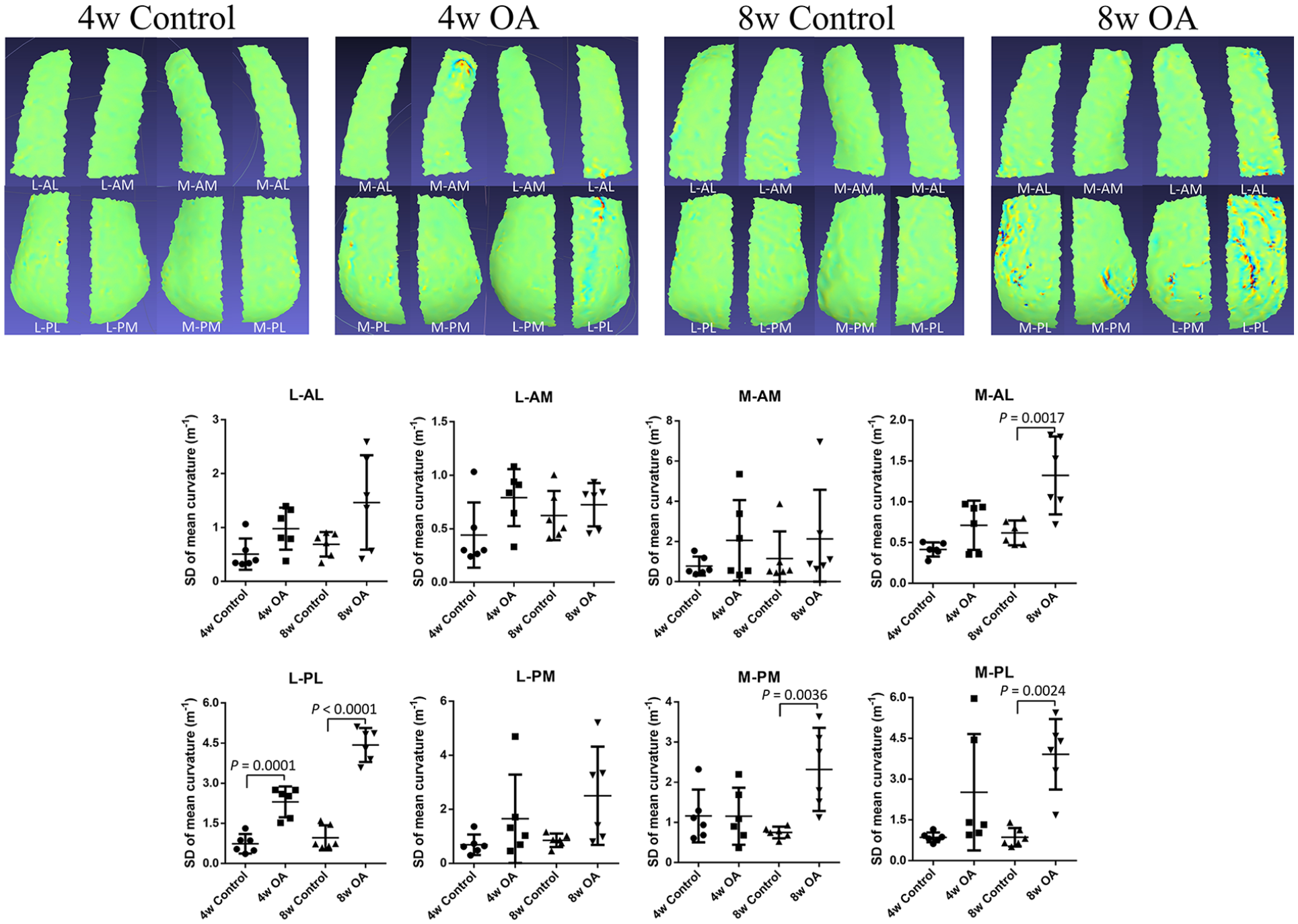
Condyle surfaces were divided into 8 areas (upper pictures). In very early OA, cartilage degeneration occurred in the L-PL area. In advanced OA, OA changes began to appear in M-AL, M-PM, and M-PL areas. OA = osteoarthritis; L = lateral condyle; PL = posterolateral part; M = medial condyle; AL = anterolateral part; PM = posteromedial part; PL = posterolateral part.
Histological Assessment
The Safranin-O staining OARSI scores were 1.92 [95% CI: −0.79-4.62], 10.17 [95% CI: 5.91-14.43], 2.58 [95% CI: −0.47-5.63], and 12.00 [95% CI: 7.84-16.16] (n = 6) in the 4w control, 4w OA, 8w control, and 8w OA groups, respectively. There were significant differences between 4w control and 4w OA, and 8w control and 8w OA groups ( Fig. 3C ). However, there was no significant difference between the 4w OA and 8w OA groups. The histology results were consistent with the 3D evaluation in terms of local areas ( Fig. 6 ). Correlation analysis showed that the OARSI score correlated with the SD of mean curvature, but not strongly (rs = 0.7930, P < 0.0001) ( Fig. 3E ).
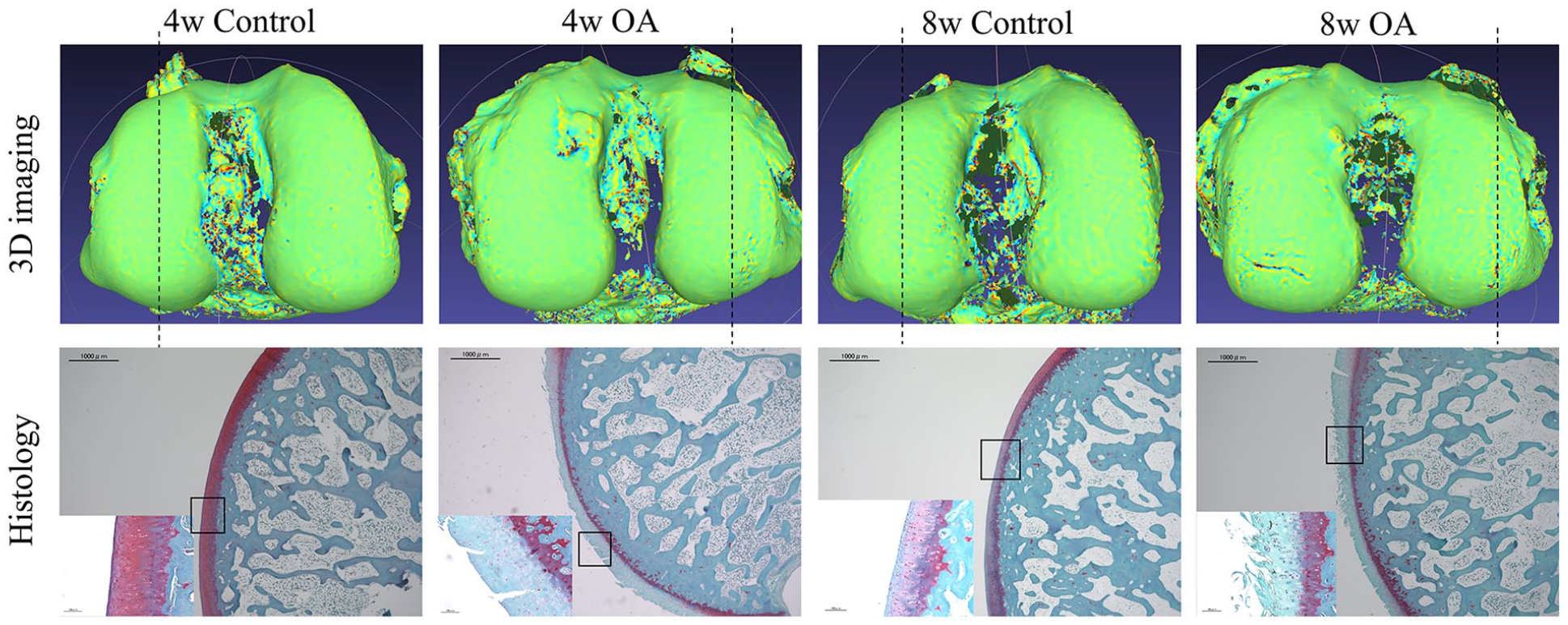
Representative 3D images and sagittal histological sections of condyles. The dotted line shows the location of sectioning. The 3D map reflects the degenerative changes well compared with the microscopic view in 4w and 8w OA groups, while the control cartilage shows a smooth surface in both 3D images and histological results. OA = osteoarthritis.
Discussion
In this study, we applied 3D scanning to observe cartilage morphology and curvature analysis to quantify the severity of OA changes. To analyze and identify the areas where OA degeneration occurred after the ACLT, surface morphological analysis was also performed by dividing the joint surface into several areas. Our results showed that the variation in mean curvature of the surface was proportional to the severity of cartilage degeneration assessed using other analytical techniques. In addition, the posterolateral part of the lateral condyle (L-PL) is the region that undergoes degenerative changes first in the rabbit ACLT model. These results indicate that 3D scanning and curvature analysis can be reproducible imaging modalities for quantifying OA severity and clarifying pathological mechanisms.
Comparing the conventional evaluation methods with curvature analysis, our results were highly consistent with those of India ink, which verified the accuracy of the 3D technique, whereas they were moderately correlated with the histology OARSI score. The OARSI score is composed of four components: staining, structure, chondrocyte density, and clusters, while 3D scanning is a method of quantifying macrostructural alterations on the joint surface. This difference is possibly why the SD of mean curvature was not strongly correlated with the histology OARSI score. While histological evaluation allows detailed observation of the structure of cartilage, 3D scanning allows structural and quantitative analysis of the entire cartilage surface.
India ink is a semi-quantitative morphological evaluation. The principle of this method is that ink particles are trapped in fissures on the cartilage. 21 Early OA stage cartilage sometimes undergoes thickening before tissue peeling, 22 which can be detected by curvature analysis instead of India ink staining. In addition, the 3D curvature visualization is rendered in a vector graphics format. Obtained 3D images can be magnified to show tiny surface texture changes without distortion compared with India ink. Moreover, the triangle mesh models obtained by 3D scanning can be uniformly divided into subareas. Hence, a 3D scan using curvature analysis has the advantage of not only quantitatively assessing cartilage degeneration but also allowing for subarea analysis of the damaged cartilage compared with India ink staining.
Subarea analysis revealed significant OA progression in the L-PL at 4 weeks. In addition, significant OA progression occurred in the M-AL, M-PM, and M-PL at 8 weeks. Previous studies have reported that femoral condylar cartilage injury causes degenerative changes evenly on both sides, or degenerative changes predominantly medially.2123-25 However, our results showed that OA changes first occurred on the lateral femoral condyle in the early stages and subsequently bilaterally. These results were possible because 3D curvature analysis can detect not only the fissure but thickening areas that cannot be examined by conventional methods. Li et al. 26 reported that the contact points shift both posteriorly and laterally on the surface of the tibial plateau after anterior cruciate ligament (ACL) injury in humans. Although there are differences between rabbits and humans, our result is consistent with previous reports showing that OA changes after ACLT are mainly concentrated on the posterolateral side of the femur. Ojanen et al. 27 demonstrated that cell morphology and cell deformation are altered in lateral femoral condyle cartilage of a rabbit ACLT model in the early stage, which may account for why L-PL is vulnerable to OA. As an early OA marker, the decreasing curvature value of subchondral bone was able to predict OA progression, which suggested flattening of convex shapes and increased concavity of concave shapes.28-30 Interestingly, Hunter et al. 28 revealed that the curvature changes of subchondral bone were observable in lateral femur at early stage after ACL injury in humans. Although the lesion sites of subchondral bone were not necessarily the sites of cartilage surface at early stage, the lateral condyle, especially L-PL, is likely the initial point of OA in rabbits and humans. To our knowledge, the present study is the first to focus on the lesions of rabbits in early OA by 3D scanning.
This study has several limitations. In optical scans, a frequent problem is the presence of noise on the surface of the scan. MLS-based methods are major point cloud denoising approaches. In this study, we used an APSS, which is an extension of an MLS-based method, to robustly generate a smooth surface. So, to some extent the noise will be decreased but not totally eliminated. Future work should focus on developing the quality of the input point set. Another limitation is that 3D scanning is currently an invasive method. Because the optical light cannot penetrate the joint, we have to open the joint to scan the surface. Although technical progression will be required, the combination of the arthroscope with 3D devices can minimize surgical intervention in the future. Nevertheless, our results demonstrated that the SD of the mean curvature of the knee cartilage surface as acquired by 3D scanning with a mathematical algorithm is in direct proportion to the severity of OA degeneration. This accurate and quantitative method is a promising approach for evaluating cartilage surfaces.
In conclusion, 3D scanning has high resolution and can capture the features of joint surfaces accurately in a time-saving way. This study demonstrated that 3D scanning with curvature analysis was able to quantify the severity of cartilage degeneration objectively in early stage OA, and that L-PL was the area where OA degeneration occurred first, which is useful for further analysis of the biochemical and biomechanical mechanisms of OA.
Supplemental Material
sj-docx-1-car-10.1177_19476035211059597 – Supplemental material for Quantification of Cartilage Surface Degeneration by Curvature Analysis Using 3D Scanning in a Rabbit Model
Supplemental material, sj-docx-1-car-10.1177_19476035211059597 for Quantification of Cartilage Surface Degeneration by Curvature Analysis Using 3D Scanning in a Rabbit Model by Dawei Liang, Tomohiro Onodera, Masanari Hamasaki, Ryosuke Hishimura, Kentaro Homan, Liang Xu, Yuan Tian, Satoshi Kanai and Norimasa Iwasaki in CARTILAGE
Footnotes
Supplemental Material
Author Contributions
N.I., S.K., and T.O. designed the study. D.L. performed the experiments, statistical analysis, and drafted the manuscript. M.H. and R.H. assisted with the acquisition of data. L.X., Y.T., and K.H. assisted with analysis and interpretation of data. All authors approved the version to be published.
Acknowledgments and Funding
The present study was supported by the project research funded by the Japanese Orthopaedic Association (#2017-4).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All experiments were performed according to a protocol approved by the Institutional Animal Care and Use Committee of the Graduate School of Medicine, Hokkaido University.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.