Abstract
Objective
Although tissue engineering is a promising option for articular cartilage repair, it has been challenging to generate functional cartilaginous tissue. While the synthetic response of chondrocytes can be influenced by various means, most approaches treat chondrocytes as a homogeneous population that would respond similarly. However, isolated cells heterogeneously progress through the cell cycle, which can affect macromolecular biosynthesis. As it is possible to synchronize cells within discrete cell cycle phases, the purpose of this study was to investigate the effects of cell cycle synchronization on the chondrogenic potential of primary articular chondrocytes.
Design
Different methods of cell synchronization (serum starvation, thymidine, nocodazole, aphidicolin, and RO-3306) were tested for their ability to synchronize primary articular chondrocytes during the process of cell isolation. Cells (unsynchronized and synchronized) were then encapsulated in alginate gels, cultured for 4 weeks, and analyzed for their structural and biochemical properties.
Results
The double-thymidine method yielded the highest level of cell purity, with cells synchronized in S phase. While the cells started to lose synchronization after 24 hours, tissue constructs developed from initially S phase synchronized cells had significantly higher glycosaminoglycan and collagen II amounts than those developed using unsynchronized cells.
Conclusions
Initial synchronization led to long-term changes in cartilaginous tissue formation. This effect was postulated to be due to the rapid auto-induction of TGF-βs by actively dividing S phase cells, thereby stimulating chondrogenesis. Cell synchronization methods may also be applied in conjunction with redifferentiation methods to improve the chondrogenic potential of dedifferentiated or diseased chondrocytes.
Introduction
Damage to the articular cartilage tissue is a common occurrence caused by a range of factors including acute injury
1
and deterioration by disease,2,3 which often leads to joint pain, loss of joint mobility, and the progressive destruction of tissue. Current strategies for articular cartilage repair, such as autologous osteochondral transfer and cell transplantation, aim to replace the damaged tissue with a repair tissue that has the same biochemical, mechanical, and functional properties as native articular cartilage. Autologous osteochondral transfer involves the transplantation of healthy cartilage plugs from non-load-bearing joint regions into the defect site.
4
Cell transplantation (e.g., autologous chondrocyte implantation and matrix-induced autologous chondrocyte implantation) involves isolating cells from non-load-bearing regions in the joint,
While tissue engineering methods have been shown to generate cartilaginous tissues
Thus, the purpose of this study was to (1) develop methods to synchronize primary articular chondrocytes
Methods
Cell Isolation
Articular cartilage was harvested from the metacarpophalangeal joints of cows (<18 months old) obtained from a local slaughterhouse (Beverly Creek Farms, Milton, Ontario). The harvested cartilage tissues (<5 mm × 5 mm × 1 mm) were placed into a 100-mm petri dish containing complete media consisting of Ham’s F12 supplemented with 10% fetal bovine serum (FBS) and 1% antibiotics/antimitotics. Cells were isolated by enzymatic digestion of the cartilage tissues with 0.5% w/v protease in Ham’s F12 for 1 hour followed by 0.15% w/v collagenase A in Ham’s F12 with 10% FBS for 12 hours at 37°C.
Cell Synchronization In Situ
Cell cycle synchronization of chondrocytes was achieved by serum starvation (G0/G1 phase) 25 or by applying commonly used cell cycle arresting agents, specifically, thymidine (S phase), 26 nocodazole (G2 phase), 27 aphidicolin (S phase), 28 or RO-3306 (G2 phase), 29 during the process of cell isolation.
Serum Starvation
The serum starvation method involved cell isolation in the absence of FBS by enzymatic digestion of the cartilage tissue with 0.5% w/v protease in Ham’s F12 for 1 hour followed by 0.15% w/v collagenase A in Ham’s F12 for 12 hours at 37°C.
Double Thymidine, Thymidine-Nocodazole, and Thymidine-RO-3306 Methods
The double thymidine, thymidine-nocodazole, and thymidine-RO-3306 methods involved first blocking the cells resident within the harvested tissues with 20 mL complete media containing 10 mM thymidine for 24 hours at 37°C, followed by a wash with 5 mL Ham’s F12 media. Cells were then released from the thymidine block in 20 mL Ham’s F12 media at 37°C for 8 hours (for double thymidine method), 3 hours (for thymidine-nocodazole method), or 5 hours (for thymidine-RO-3306 method). The media was then aspirated and replaced with 20 mL of 0.5% w/v protease in Ham’s F12 for 1 hour at 37°C. This was followed by a wash with 5 mL Ham’s F12 media and a second block in 20 mL Ham’s F12 media containing 10% FBS, 0.15% w/v collagenase A, and either 10 mM thymidine (double thymidine method), 100 ng/mL nocodazole (thymidine-nocodazole method), or 10 µM RO-3306 (thymidine-RO-3306 method) for 12 hours at 37°C.
Aphidicolin Method
The aphidicolin method involved serum starvation by incubating cartilage tissues with 20 mL Ham’s F12 media for 48 hours at 37°C, followed by incubation in 0.5% w/v protease in Ham’s F12 for 1 hour at 37°C. This was followed by a wash with Ham’s F12 media, and a subsequent 24-hour treatment with Ham’s F12 media containing 10% FBS, 0.15% w/v collagenase A, and 5 µg/mL aphidicolin.
Cell Encapsulation
To maintain the phenotype of chondrocytes in culture (synchronized or unsynchronized), isolated cells were encapsulated in alginate beads that were created by first mixing a high-density cell suspension of chondrocytes (20 × 106 cells/mL) in equal parts with a sterile 1.25% alginic acid solution (20 mM HEPES, 150 mM sodium chloride, pH 7.4) at room temperature. Cell-encapsulated alginate beads were then created by dropping 30 µL of the cell-alginate solution in a calcium chloride bath (102 mM CaCl2 containing 10 mM HEPES, pH 7.4). After 10 minutes, polymerized cell-encapsulated beads were extracted using forceps, placed into individual wells of a 24-well plate containing 1 mL Ham’s F12 media supplemented with 10% FBS and 100 µg/mL ascorbic acid, and cultivated at 37°C with 5% CO2 and 95% humidity. The culture media was changed every 48 hours and cell-encapsulated bead constructs were cultured for a maximum of 4 weeks.
Evaluation of Cell Viability and Cell Cycle Phase
Cell Recovery and Viability
Cells were recovered from encapsulation in order to assess cell viability and degree of cell cycle synchronization. Harvested cell-encapsulated alginate beads were dissociated at 37°C under light agitation (120 rpm) in a calcium chelating solution (55 mM EDTA in 1× PBS) for 15 minutes. Cell viability was then determined using the trypan blue exclusion method. 30
Assessment of Cell Cycle Phase Synchronization
Cell cycle phase synchrony was determined by flow cytometric analysis and gating of cells in the target cell cycle phase. Briefly, dissociated cells were pelleted by centrifugation (700 ×
Analysis of Bead Constructs After Long-Term Culture
Biochemical Analysis
After 2 weeks and 4 weeks of cultivation, cell-encapsulated bead constructs were first lyophilized overnight and then digested by papain (40 µg/mL in 20 mM ammonium acetate, 1 mM EDTA, and 2 mM dithiothreitol) for 72 hours at 65°C. Biochemical assays were then performed on the papain digests to determine cellularity and extracellular matrix accumulation.15,31,32 DNA content was determined by the PicoGreen assay. 33 Proteoglycan content was determined by measuring the amount of sulfated GAG using the 1,9-dimethylmethylene blue (DMMB) dye binding assay.34,35 Collagen content was determined by hydrolyzing the papain digested samples in 6 N hydrochloric acid for 18 hours at 110°C and measuring the hydroxyproline content of the hydrolyzed samples using the chloramine-T/Ehrlich’s reagent assay. 36 Collagen content was estimated as 10 times the hydroxyproline content. 37
Histological and Immunohistochemical Evaluation
After 2 weeks and 4 weeks of cultivation, representative bead constructs were fixed in 4% paraformaldehyde for 24 hours. Fixed beads were then dehydrated in graded ethanol solutions and embedded in paraffin at 65°C. Sections were cut at 8 µm thickness and mounted on Superfrost slides. Sections were deparaffinized and rehydrated, and then stained with safranin-O with fast green counterstain or picrosirius red with hematoxylin counterstain for the visualization of GAG and collagen distribution, respectively. 15
For collagen I and collagen II immunostaining, rehydrated sections were treated with a solution of 3.9 kU/mL pepsin in 0.5% acetic acid for 30 minutes at 37°C, followed by 2 washes with PBS. The sections were then blocked with a solution of 1% bovine serum albumin (BSA) in PBS for 30 minutes at room temperature. After blocking, sections were incubated with an anti-collagen I (Abcam, ab90395) or anti-collagen II (Developmental Studies Hybridoma Bank, CIIC1) primary antibody (1:100 dilution in PBS containing 1% BSA) overnight at 4°C. Two washes with PBS were performed to remove the excess primary antibody, followed by staining with Texas red-labelled goat anti-mouse secondary antibody (Abcam, ab6787, 1:200 dilution in PBS containing 1% BSA) for 2 hours at room temperature. Three washes with PBS were performed, followed by mounting the slides with coverslips using Vectashield mounting medium containing DAPI.
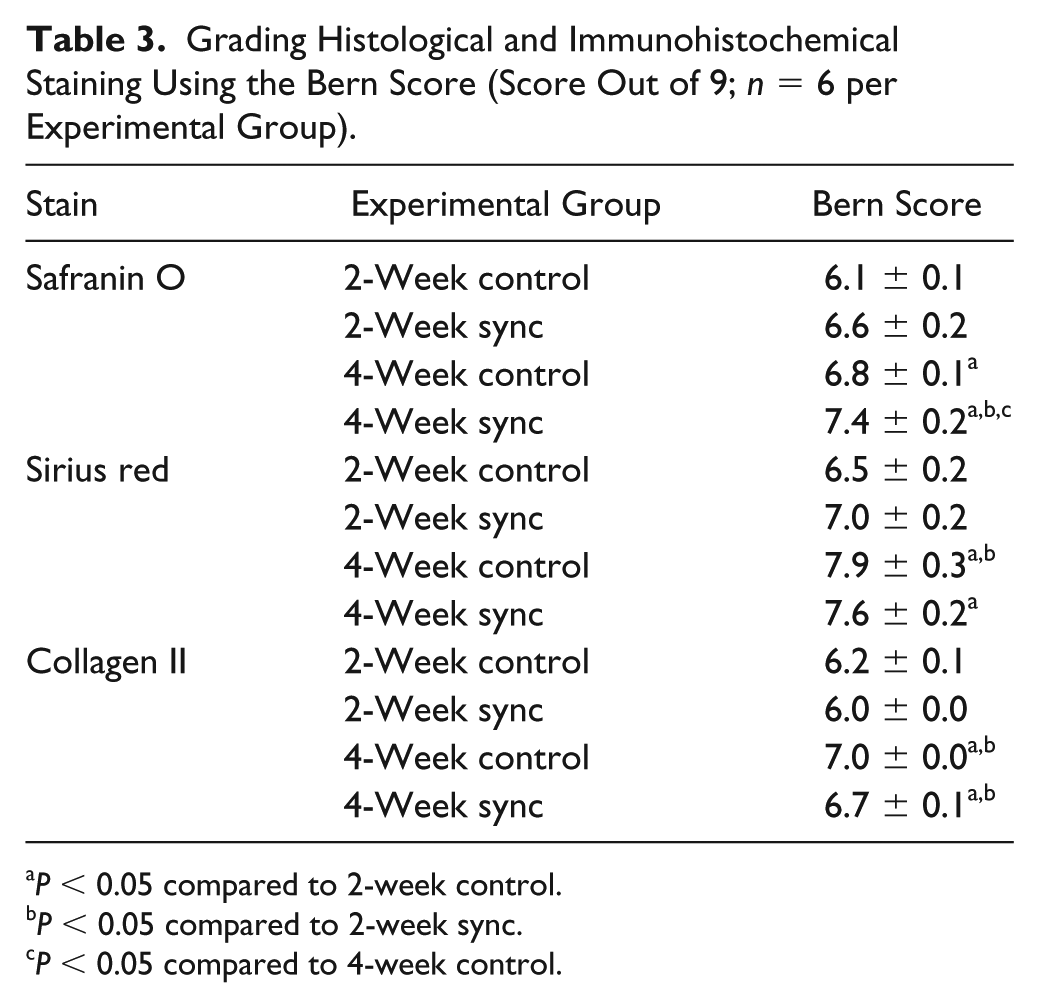
Safranin O, picrosirius red, and collagen II staining were graded using the Bern Score, 38 according to 3 scoring categories: (1) stain uniformity and intensity, (2) cell distance, and (3) cell morphology (each scoring category with a score out of 3 for a total score of 9). The images were scored by 2 independent observers.
Statistical Analyses
All results were expressed as the mean with the standard error of the mean. Statistical differences were analyzed using one-way ANOVA and Tukey’s post hoc testing, and significance was associated with
Results
Cell Cycle Synchronization of Primary Articular Chondrocytes
Primary articular chondrocytes were synchronized into different phases of the cell cycle using various chemical synchronizing agents during the process of cell isolation from harvested tissue. The best method was determined by analyzing the proportion of cell population synchronized within the phase of interest as well as viability of the synchronized cell population.
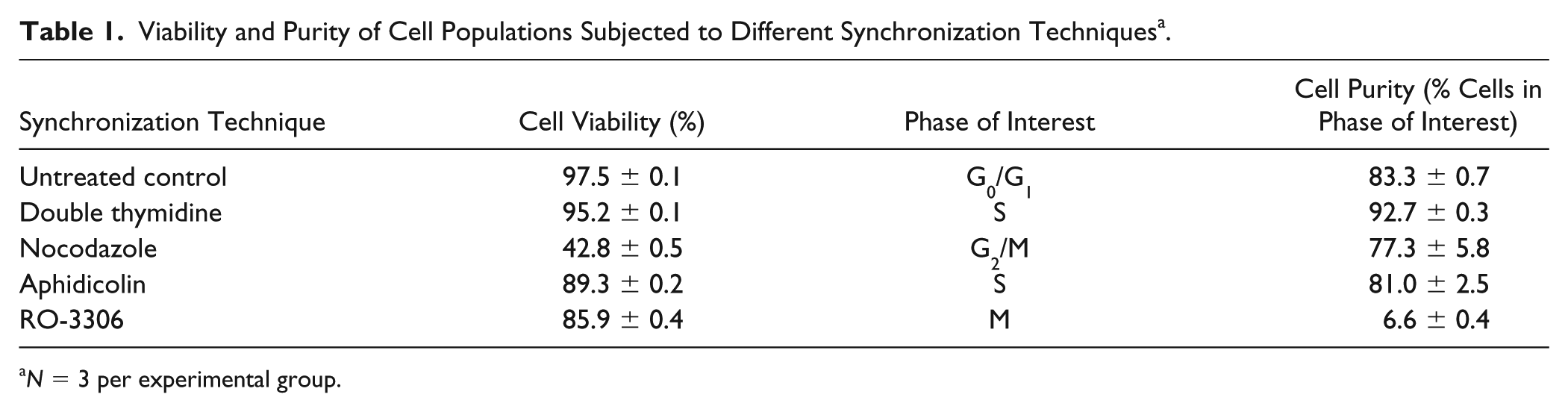
The double thymidine method yielded the highest percentage of cells synchronized within S phase, while maintaining a high cell viability ( Table 1 ). However, all other synchronization methods investigated (nocodazole, RO-3306, and aphidicolin) resulted in decreased cell viability (<90%) and/or a lack of highly synchronized cell populations (<90% within the cell cycle phase of interest) ( Table 1 ). For these reasons, the double thymidine method was chosen to synchronize primary articular chondrocytes for all subsequent studies, and the other synchronization methods were not investigated further.
Viability and Purity of Cell Populations Subjected to Different Synchronization Techniques a .
To assess the quality and longevity of the synchronization process using the double thymidine method, cell populations were recovered from encapsulation (via calcium chelation) and analyzed by flow cytometry at 12-hour intervals for up to 48 hours after synchronization ( Fig. 1A and B , Table 2 ). Initially, the synchronized cells were predominantly resident within the S phase (~93%). However, tracking of the cell cycle phase (at 12-hour increments post encapsulation) showed that while the cells proceeded to traverse through the cell cycle, synchronization of the population appeared to wane over time. A visible trailing peak of cells appeared to initiate at 24 hours post encapsulation, and with subsequent time in culture, this peak became visibly larger as evidenced by the broadening width of the histogram. At 48 hours post encapsulation, the percentage of cells resident within S phase declined to ~82%.
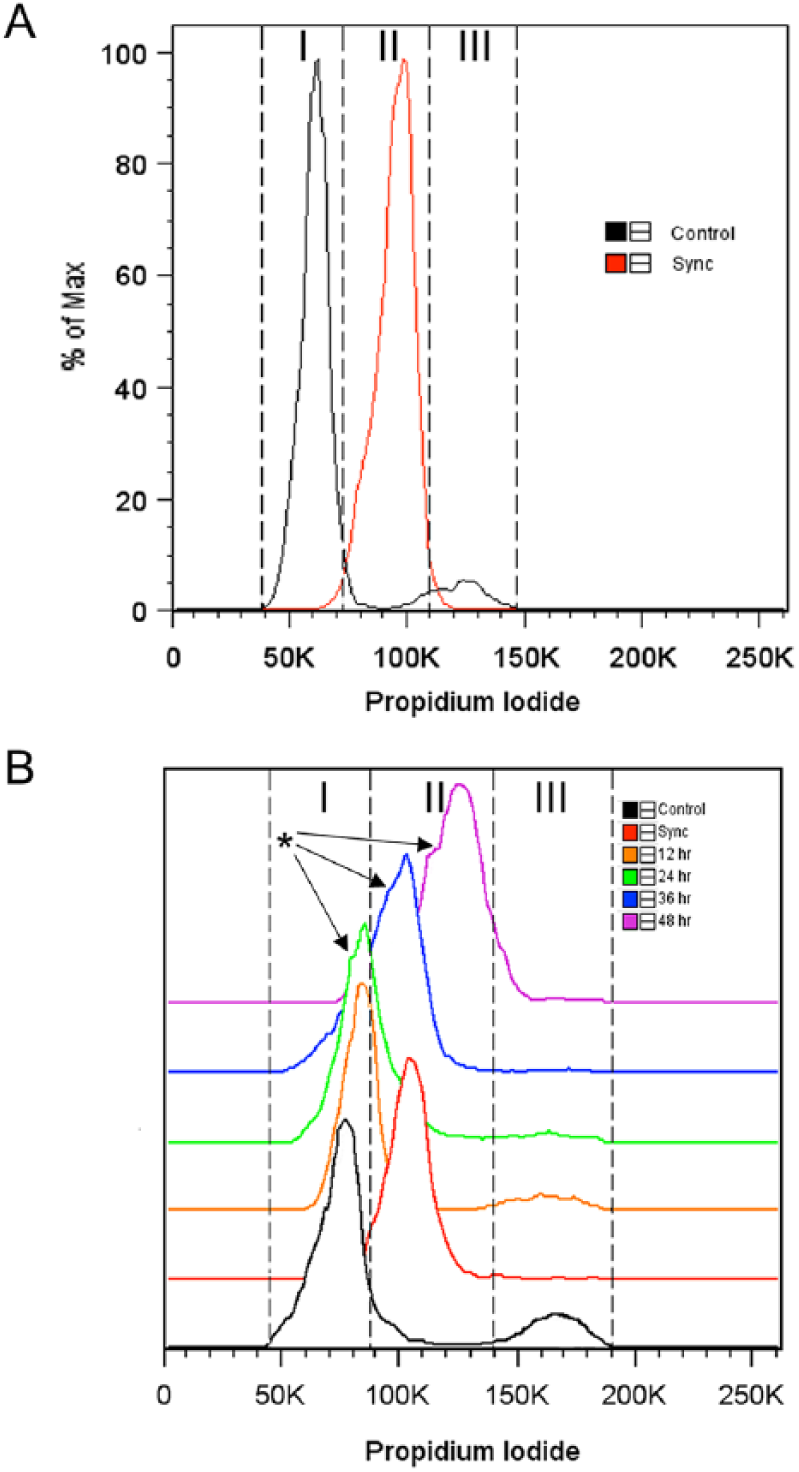
Cell cycle synchronization of primary articular chondrocytes. (
Percentage of Cells within Each Phase of the Cell Cycle at Different Time Points After Synchronization a .
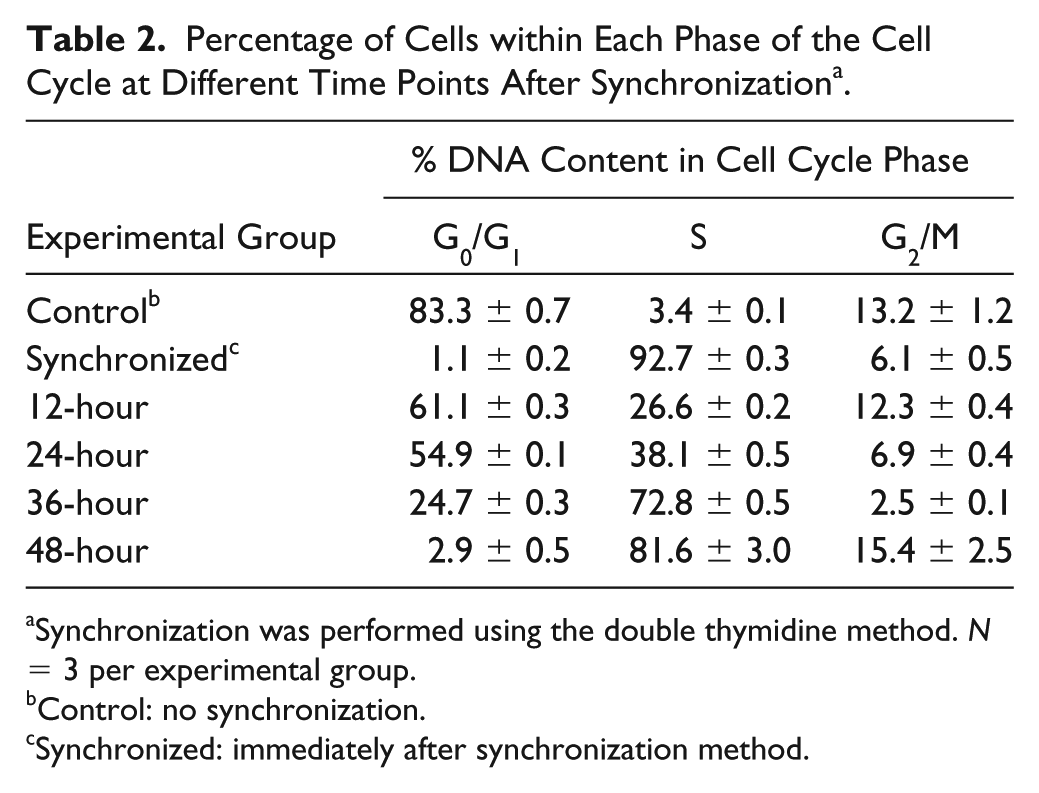
Synchronization was performed using the double thymidine method.
Control: no synchronization.
Synchronized: immediately after synchronization method.
Effect of Cell Cycle Synchronization on Chondrogenesis
The biochemical composition and histological structure of the extracellular matrix accumulated within the alginate beads developed from unsynchronized and synchronized chondrocytes were analyzed and compared after 2 weeks and 4 weeks of culture. Amounts of DNA, sulfated GAG, and collagen (including comparison of type I and II collagen) were evaluated as primary markers of cartilaginous tissue synthesis.
While there were no significant differences in the biochemical composition between unsynchronized control and synchronized groups after 2 weeks of culture, the DNA, GAG, and collagen contents were significantly increased in the synchronized constructs compared with the unsynchronized controls after 4 weeks (
Fig. 2A
-
C
,
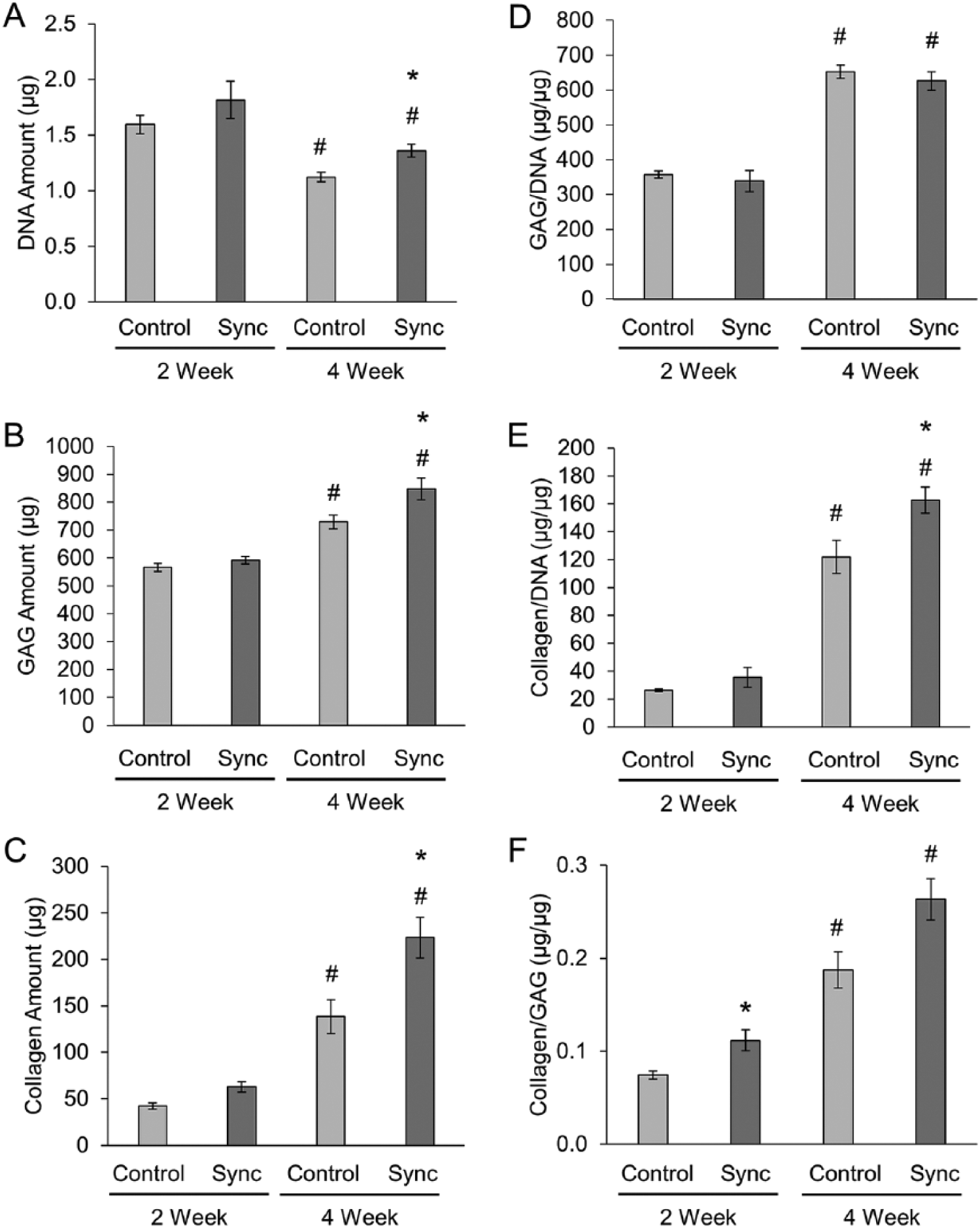
Biochemical properties of engineered cartilage constructs. (
Histological staining for sulfated proteoglycans and collagen displayed similar results. Safranin-O staining showed more accumulation of GAG in the extracellular matrix for the synchronized groups compared with the unsynchronized controls at both the 2-week and 4-week time points ( Fig. 3 , Table 3 ). Synchronized constructs generally appeared more cellular and showed increased presence of doublets, indicating active cellular division and increased formation of lacunae after longer times in culture (4 weeks compared with 2 weeks). Sirius red staining for collagen also showed similar results with increased collagen accumulation in the synchronized groups compared with the unsynchronized controls and increasing accumulation with time in culture ( Fig. 4 , Table 3 ). Immunohistochemical staining indicated that the collagen content within the accumulated extracellular matrix was primarily collagen II with negligible amounts of collagen I ( Fig. 5 , Table 3 ), thereby indicating a maintenance of chondrogenic phenotype irrespective of whether or not the cells were initially synchronized.
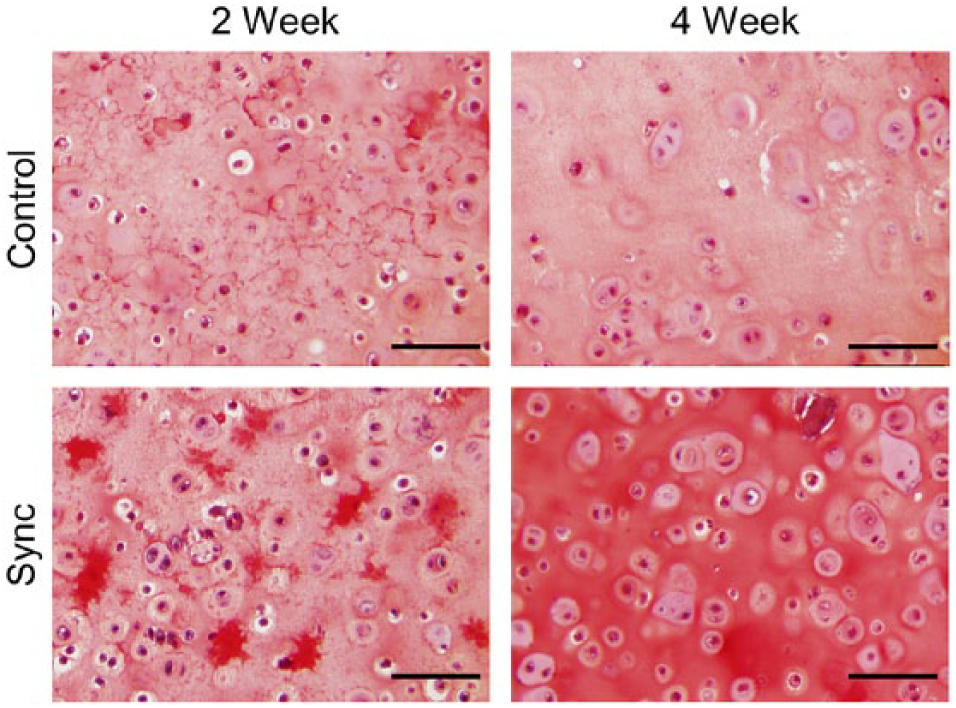
Representative microscopy images showing safranin-O staining of tissue constructs. Cartilage stained in red, nuclei stained in black, and background stained in bluish green. Scale bar = 100 µm.
Grading Histological and Immunohistochemical Staining Using the Bern Score (Score Out of 9;
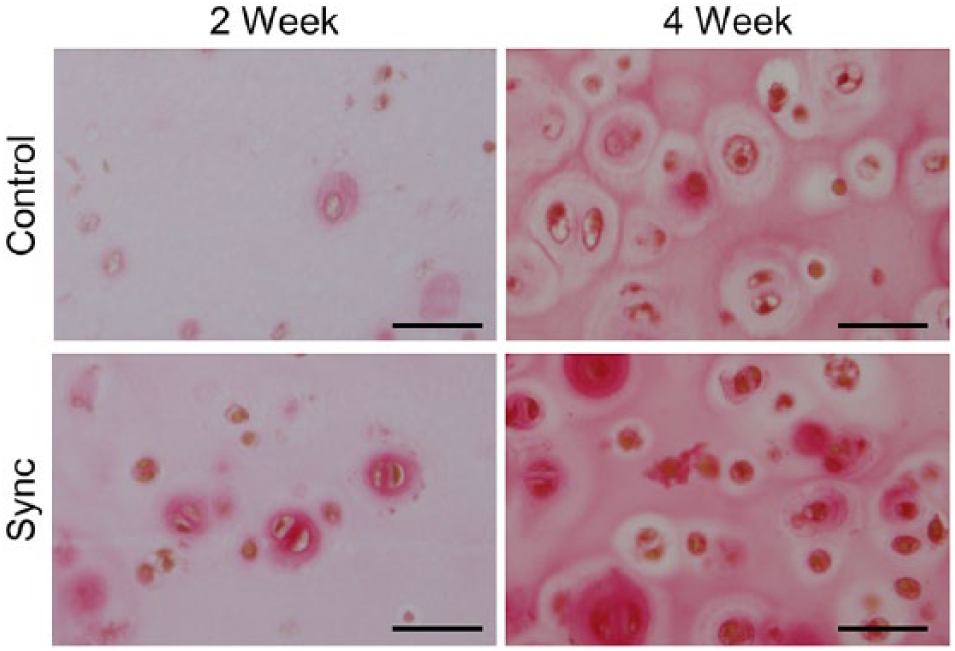
Representative microscopy images showing sirius red staining of tissue constructs. Collagen stained in red, nuclei stained in brown, and background stained in pale yellow. Scale bar = 100 µm.
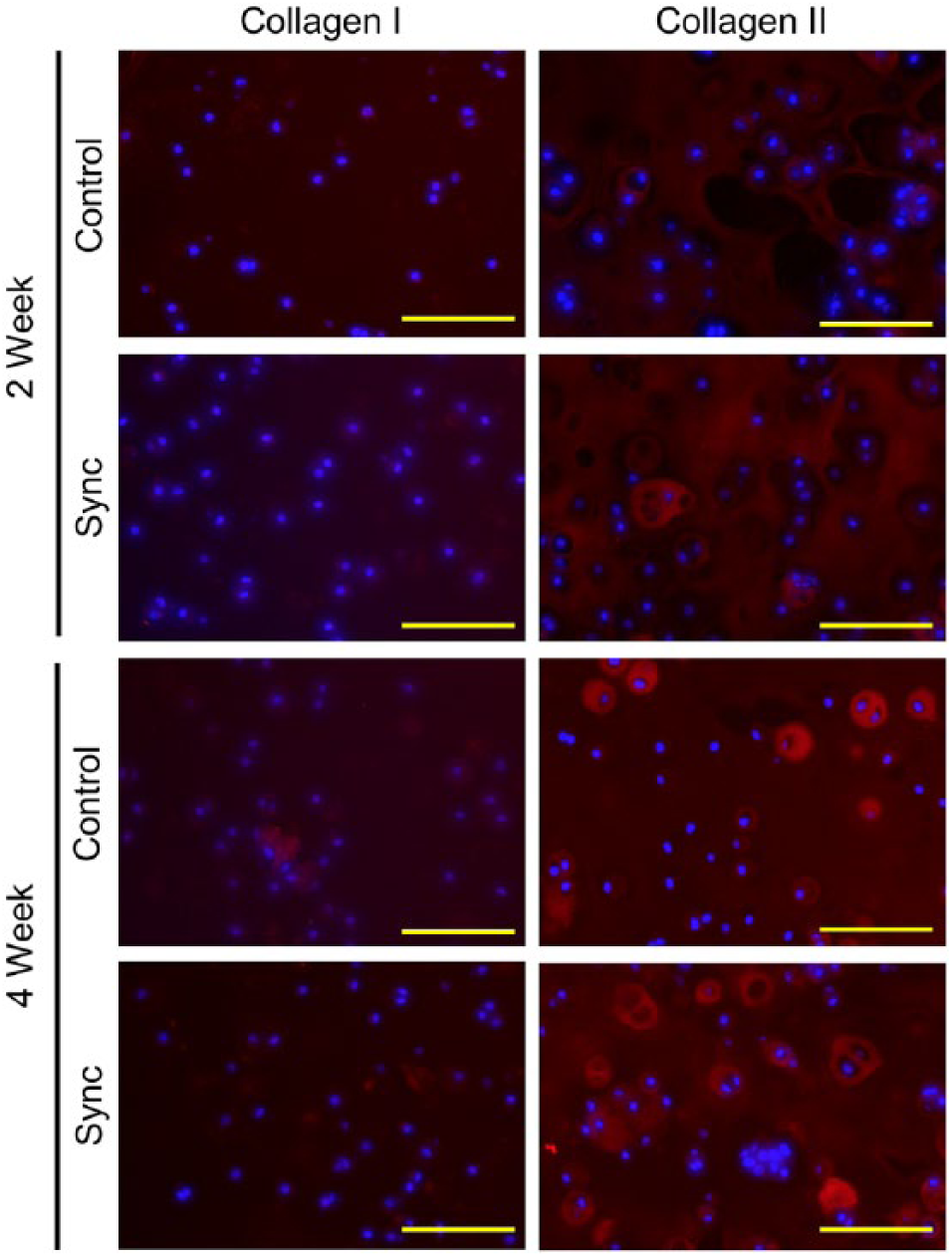
Immunohistochemical staining of collagen I and collagen II in tissue constructs. Collagen I and collagen II stained in red, and nuclei stained in blue. Scale bar = 100 µm.
Discussion
As articular cartilage has a minimal capacity to repair itself, tissue engineering offers an alternative treatment option as tissue-engineered cartilage. However, it has been challenging to improve the deposition of cartilaginous extracellular matrix by the cells and, in turn, generate tissue with functional mechanical properties. Various studies have been performed to increase the deposition of cartilaginous extracellular matrix by articular chondrocytes, including growth factor stimulation,9,39 bioreactor cultivation,16,40 and/or application of mechanical stimuli.9-16 These approaches, however, tend to assume that the cultivated cells are a homogeneous population and hence would respond similarly to the imposed treatment(s). While there are several sources of inhomogeneity within the cell population, cells can also have varying properties due to their progression through the cell cycle. Similarly, it has been shown that cells synchronized within discrete phases of the cell cycle can respond more efficiently to biological treatments.23,25,41 As such, we hypothesized that pre-synchronization of isolated cells would facilitate improved tissue formation
Cell cycle phase synchronization can be achieved by physical or chemical methods. 17 Physical methods (e.g., gradient centrifugation,42,43 membrane elution,44,45 fluorescence-activated cell sorting46,47) involve selection of cells based on their physical properties (size or density) and typically only result in the synchronization of a fraction of cells within the population. Alternatively, chemical synchronization methods rely on the reversible blocking of cells at a cell cycle phase by either deprivation or addition of chemical agents in the culture media during monolayer culture. Previous studies have achieved synchronization at different cell cycle phases by serum starvation (G1/G0) 25 or treatment with lovastatin (G1 phase), 48 aphidicolin or thymidine (S phase),26,28 and nocodazole or RO-3306 (G2/M phase).27,29 In this study, chemical synchronization methods were utilized due to their ability to yield higher levels of synchrony compared to physical methods. 48 However, traditional chemical synchronization methods cannot be directly translated to chondrocytes as these cells are known to dedifferentiate in monolayer culture. 24 For this reason, we adapted and evaluated different chemical synchronization techniques (serum starvation, thymidine, nocodazole, aphidicolin, and RO-3306) applied during the process of cell isolation from harvested cartilage. Among the evaluated techniques, the double thymidine method was found to yield the highest degree of cell synchrony without eliciting significant loss in chondrocyte viability. The other chemical agents investigated resulted in decreased cell viability and/or a lack of highly synchronized cell populations (within any phase) and were not investigated further.
Primary articular chondrocytes initially synchronized in S phase (by double thymidine method), and cultivated in alginate beads, led to the increased accumulation of cartilaginous extracellular matrix without observable changes in phenotype. Interestingly, this effect was sustained over long-term culture (4 weeks) even though the cells started to lose their synchrony at ~24 hours post encapsulation. The long-term effect on chondrogenesis, despite the cells losing their initial synchrony, may be attributed to the prolonged duration of the chondrocytes within S phase. Preliminary studies have shown that chondrocytes synchronized to S phase (in monolayer) are more responsive to growth factors compared with other phases of the cell cycle, resulting in enhanced chondrogenic tissue formation.
49
Chondrocytes express different transforming growth factor-β1 (TGF-β1) receptor systems depending on the cell cycle phase.
50
In particular, S phase synchronized cells express the lowest affinity TGF-β1 receptor (
The use of cell cycle synchronization to improve chondrogenic potential may also potentially be beneficial to improve the chondrogenic potential of passaged or osteoarthritic (OA) chondrocytes. In addition to the generally low biosynthetic rates of passaged and OA chondrocytes, these cells suffer from a loss of the characteristic chondrogenic phenotypic (e.g., dedifferentiation), thereby also requiring redifferentiation steps in order to produce cartilaginous tissue
Conclusions
In this study, we investigated the effects of cell cycle synchronization of primary articular chondrocytes on their potential to generate cartilaginous tissue
Footnotes
Acknowledgments and Funding
The authors gratefully acknowledge Andjela Bajic for histological and immunohistochemical scoring. The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study because the tissue samples used in this study were purchased from a local abattoir.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because the tissue samples used in this study were purchased from a local abattoir.
