Abstract
Objective:
To explore the effect of shifting in vitro culture conditions regarding cellular passage and onset of loading within matrix-associated bovine articular chondrocytes cultured under free-swelling and/or dynamical loading conditions on general chondrocyte maturation.
Methods:
Primary or passage 3 bovine chondrocytes were seeded in fibrin-polyurethane scaffolds. Constructs were cultured either free-swelling for 2 or 4 weeks, under direct mechanical loading for 2 or 4 weeks, or free swelling for 2 weeks followed by 2 weeks of loading. Samples were collected for glycosaminoglycan (GAG) quantification, mRNA expression of chondrogenic genes, immunohistochemistry, and histology.
Results:
Mechanical loading generally stimulated GAG synthesis, up-regulated chondrogenic genes, and improved the accumulation of matrix in cell-laden constructs when compared with free-swelling controls. Primary chondrocytes underwent more effective cartilage maturation when compared with passaged chondrocytes. Constructs of primary chondrocytes that were initially free-swelling followed by 2 weeks of mechanical load (delayed) had overall highest GAG with strongest responsiveness to load regarding matrix synthesis. Constructs that experienced the delayed loading regime also demonstrated most favorable chondrogenic gene expression profiles in both primary and third passage cells. Furthermore, most intense matrix staining and immunostaining of collagen type II and aggrecan were visualized in these constructs.
Conclusions:
Primary chondrocytes were more effective than passage 3 chondrocytes when chondrogenesis was concerned. The most efficient chondrogenesis resulted from primary articular chondrocytes, which were initially free-swelling followed by a standardized loading protocol.
Introduction
Mature cartilage holds a limited intrinsic capacity for self-regeneration on injury. Cell-based repair strategies have been considered a valuable treatment option. For an effective coverage of large-diameter cartilage defects, autologous chondrocyte implantation has evolved as a favored surgical technique.1,2 However, despite generally favorable outcomes, there is still limited knowledge about the influence of the cellular phenotype and external cues, such as the mechanical environment, on neo-cartilage formation. Following a cartilage biopsy, articular chondrocytes have been shown to rapidly dedifferentiate during in vitro proliferating procedures, which are required to achieve sufficient cell numbers. 3 Novel techniques, such as matrix-assisted chondrocyte implantation, have been introduced to reduce cellular dedifferentiation and also improve transplant handling via three-dimensional (3-D) cell culture conditions. 4
Yet it has been shown that static, free-swelling culture does not result in a significant redifferentiation of articular chondrocytes in 3-D culture.3,5 As mechanical stimulation is one of the major regulators known to maintain in situ cartilage integrity and function, various experimental models have shown that physiological mechanical stimulation results in chondrocyte maturation in vitro. 6 Bioreactors are capable of reproducing a joint-like surrounding by providing compression, shear, and fluid flow. 7 Scaffold-cultured articular chondrocytes have been shown to respond to such subjected loading by up-regulation of matrix products that are typical for hyaline cartilage. 6 Consequently, recent tissue-engineered products for cartilage repair have focused on bioreactor use to precondition chondrocytes prior to reimplantation. 8 Moreover, in vitro bioreactor systems can give valuable information on the potential behavior of a cell–scaffold construct by providing simulated in vivo surroundings. Still optimal mechanical stimulation protocols, with regard to achieving optimal cartilage maturation, have not been established. Optimal conditions during bioreactor tissue engineering of articular chondrocytes are those that would finally result in de novo tissue that completely resembles hyaline cartilage. It is still unclear which implant composition and culture conditions provide the optimal cellular response, and extracellular matrix gene expression profile, to potentially result in an optimal in vivo performance.
A favorable cell-matrix quality has to be produced prior to implantation, because it is known that implant quality is closely related to the resulting clinical outcome.9,10 Therefore, this study sought to improve our understanding of differing chondrocyte culture conditions to explore which configuration may provide best initial values. Using a controlled in vitro bioreactor environment, the single and combined impact of chondrocyte passage and the timing of initiation of mechanical stimulus on the chondrogenic response of chondrocyte–scaffold constructs was investigated.
Materials and Methods
Design
The experiment was repeated three times under exactly the same conditions; for each run, one calf fetlock joint was used. Following biopsy and after in vitro processing, the cells were seeded within 3-D scaffolds to divide into passage 0 (P0) or passage 3 (P3) constructs. Then the cell-matrix constructs were either cultured under free-swelling conditions (F) or under loaded surroundings within a bioreactor (L) to divide into 10 different experimental groups ( Table 1 ).
Experimental Groups
There were a total of 10 different groups divided by passage number prior to matrix seeding, according to loading protocol and culture duration. P = passage (number stands for passages); F = free-swelling culture; L = dynamic loading within bioreactor; F2L2 = free-swelling culture for 2 weeks followed by 2 weeks of loading within bioreactor.
Chondrocytes Isolation and Subculture
Full-thickness articular cartilage was dissected aseptically from three fetlock joints of 3- to 4-month-old calves directly after slaughter; chondrocytes were isolated by subsequent pronase (0.1%) and collagenase II (600 U/mL) digestions of cartilage fragments. 11 Primary chondrocytes were seeded into cell flasks at a density of 1.0 × 104 cells/cm2 and cultured in Dulbecco’s modified Eagle’s medium (DMEM) containing 10% fetal bovine serum (FBS). When chondrocytes were about 85% to 90% confluent, cells were detached using collagenase II (300 U/mL) and trypsin (0.5 mg/mL) and seeded at 1.5 × 104 cells/cm2 for subcultures. Medium was changed every second day.
Polyurethane Scaffolds and Cell Seeding
Cylindrical (diameter 8 mm × height 4 mm) porous polyurethane (PU) scaffolds were prepared as previously described. 12 Before cell seeding, the scaffolds were evacuated in the presence of DMEM containing 10% FBS under a vacuum for 1 hour, in order to wet the hydrophobic polymer.
Primary (P0) or third passage (P3) chondrocytes (4 × 106 cells/scaffold) were suspended in fibrinogen solution (Baxter Biosurgery, Vienna, Austria) and then mixed with thrombin solution (Baxter Biosurgery) before seeding. The final concentrations of the fibrin gel were 17 mg/mL fibrinogen and 0.5 U/mL thrombin. Constructs were incubated for 1 hour (37 °C, 5% CO2, 95% humidity) to permit fibrin gel formation before adding growth medium consisting of DMEM containing 10% FBS, 50 µg/mL ascorbic acid, 40 µg/mL L-proline, nonessential amino acids (Gibco, Carlsbad, CA), and 500 U/mL aprotinin (Sigma, St Louis, MO).
Bioreactor and Loading Protocol
Mechanical conditioning of cell–scaffold composites was performed using a bioreactor system, which had been designed to approach the loading and motion characteristics of natural joints.7,13 A commercially available ceramic hip ball (32 mm in diameter) was pressed onto the cell-seeded scaffold. Surface motion was generated by oscillation of the ball around an axis perpendicular to the scaffold axis. Simultaneously, dynamic compression was applied along the cylindrical axis of the construct. The whole apparatus is placed in an incubator (37 °C, 5% CO2 and 85% humidity).
After 1 day of preculture, all cell–scaffold constructs were transferred into bioreactor sample holders to provide with identical experimental surroundings for all groups. Each experiment with either P0 or P3 chondrocytes included five groups of cell–scaffold constructs. For loading experiments, chondrocyte-PU constructs were subjected to a standardized loading protocol within the bioreactor to simulate the articular milieu as described before 14 : preload 0.2 mm (5% of scaffold height), dynamic sinusoidal oscillation 0.4 ± 0.2 mm (5% to 15% of scaffold height) at 1 Hz, surface shear at ±25° and 1 Hz in phase difference for 1 hour twice a day, with 8 hours rest in between loadings, on every second day. During the non-loading periods, the constructs were kept free-swelling identical to the free-swelling groups, without any contact with the ball. Medium was changed every second day and collected for the analysis of glycosaminoglycan (GAG) content. On termination of the experiment every cell-matrix construct was cut vertically into 2 equal halves for further analysis.
Biochemical Assay for GAG/DNA Quantification
Cell–scaffold constructs were digested with proteinase K (0.5 mg/mL) at 56 °C overnight for further DNA and GAG assay. The DNA content was measured by spectrofluorometry using Hoechst 33258 dye solution against a calf thymus DNA standard curve. The amount of sulfated GAG was measured with a dimethylmethylene blue (DMMB) color reagent against bovine chondroitin sulfate standard. Conditioned culture medium was also analyzed for GAG content to determine the amount of proteoglycan released from the scaffolds into the medium.
Gene Expression Analysis
Chondrocyte–scaffold constructs were homogenized using the Tissue-Lyser system (Qiagen, Retsch, Germany), and total RNA was extracted with TRI Reagent (Molecular Research Cerner, Cincinnati, OH) according to the manufacturer’s specifications using the modified precipitation method with a high salt precipitation solution (Molecular Research Center). Reverse transcription was performed with TaqMan reverse transcription reagents (Applied Biosystems, Foster City, CA) using random hexamer primers and 1 µg of total RNA sample. Polymerase chain reaction (PCR) was performed on a 7500 Real-Time PCR system (Applied Biosystems). Oligonucleotide primers and TaqMan Probes ( Table 2 ; all from Microsynth, Balgach, Switzerland) were designed with Primer Express Oligo Design software, version 1.5/2.0 (Applied Biosystems). Probes were labeled with the reporter dye molecule FAM (6-carboxyfluorescein) at the 5′ end and with the quencher dye TAMRA (6-carboxy-N,N,N′,N′-tetramethylrhodamine) at the 3′ end. The nucleotide sequences were taken from the GenBank database. To exclude amplification of genomic DNA, the probe or one of the primers was selected to overlap an exon–exon junction. The 18S rRNA (from Applied Biosystems) was measured as endogenous control. The PCR was carried out with TaqMan Gene Expression master mix (Applied Biosystems), 900 nM primers (forward and reverse), 250 nM TaqMan probe, and 2 µL of cDNA sample in a total reaction volume of 20 µL. Relative quantification of target mRNA was performed according to the comparative C t method with 18S rRNA as the endogenous control. 15 Data were normalized to the expression levels at day 0 of construct culture.
Sequences of Oligonucleotide Primers and Probes Used for Real-Time PCR
Probes were labeled with the reporter dye molecule FAM (6-carboxyfluorescein) at the 5′ end and with the quencher dye TAMRA (6-carboxy-N,N,N′,N′-tetramethylrhodamine) at the 3′ end.
Histology and Immunohistochemistry
Cell–scaffold constructs were fixed in 70% methanol and incubated in 5% sucrose–phosphate-buffered saline PBS solution overnight at 4 °C before microtoming at 14 µm serial cryosections. Sections were stained with toluidine blue to visualize the cell morphology and extracellular matrix accumulation. The deposition of aggrecan and type I and II collagen was identified according to immunohistochemical analysis. The immunohistochemistry (IHC) staining was performed by using the Vectastain Elite ABC Kit (Vector Laboratories). Horse serum was used for blocking non-specific sites. The sections were probed with primary antibodies against collagen type I (Sigma-Aldrich, Saint Louis, MO), collagen type II, and aggrecan (Developmental Studies Hybridoma Bank, Iowa City, IA). The primary antibody was applied for 30 minutes followed by the biotinylated secondary horse anti-mouse antibody and then incubation with avidin–biotin–peroxidase complex for 30 minutes at room temperature. Color was developed using 3-3′-diaminobenzidine (DAB) monomer. Bovine cartilage tissue sections were employed as positive controls. For negative controls the primary antibody was replaced by phosphate-buffered saline.
Statistical Analysis
Statistical analysis was performed using the software package SPSS (Version 18; SPSS Inc, Chicago, IL). All data were tested for normal distribution using the Kolmogorov–Smirnov test. Then, data were compared using t tests or Mann–Whitney U and Wilcoxon signed rank tests. Group data were compared using one-way analysis of variance or Kruskal–Wallis analysis. Unless otherwise stated, descriptive results were demonstrated as the mean ± standard deviation. The significance level was defined at P < 0.05 or indicated a trend at 0.05 < P < 0.1 for all tests. The significance within the graphs is noted by an asterisk connecting the statistical comparison.
Results
GAG, DNA synthesis
Differences in the DNA content were noted neither between the different treatment groups nor between different culture times (data not shown). However, the accumulation of GAG in primary cell-laden scaffolds (P0) increased significantly after free-swelling culture for 2 weeks followed by loading for 2 weeks (P0 F2L2) when compared with F4 constructs (170.22 ± 54.10 µg vs. 92.15 ± 24.65 µg) (P < 0.05). The release of GAG from loaded scaffolds into the medium increased significantly after loading directly for 2 weeks (P0 L2) or 4 weeks (P0 L4), respectively (P0 L2: 951.36 ± 262.27 µg; P0 L4: 1542.69 ± 266.13 µg), when compared with the respective free-swelling control groups P0 F2 (418.96 ± 156.64 µg) or P0 F4 (721.98 ± 183.05 µg). Furthermore, the release of GAG increased in a loading time-dependent manner, significantly increasing GAG production in P0 L4 compared with the P0 L2 group (P < 0.01). A two-fold higher level of GAG in the culture medium of the P0 F2L2 group compared with the P0 F4 group was also noted ( Fig. 1A ).
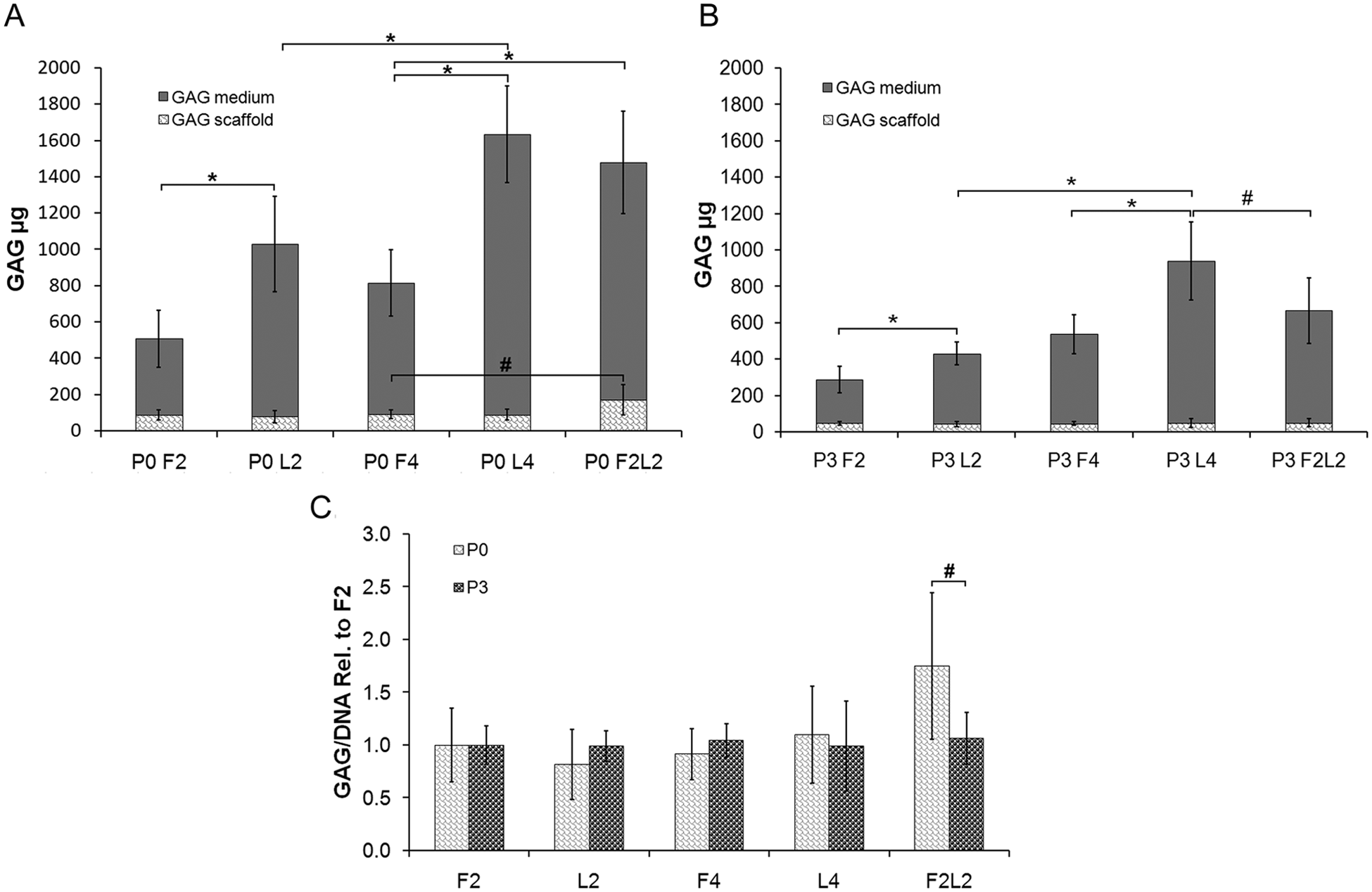
Mean medium and scaffold glycosaminoglycan (GAG) content in micrograms among the different primary (P0) (
Among P3 chondrocytes, no significant difference in GAG accumulation in the scaffold was found between unloaded and loaded groups ( Fig. 1B ). Whereas the GAG content reached 170.22 ± 54.10 µg in P0 F2L2 cells, it only remained 52.11 ± 20.82 µg in P3 cells, resulting in GAG/DNA ratios of ~7 for the P0 and ~3 for the P3 chondrocytes (data not shown). However, the amount of GAG released into the medium during 2 or 4 weeks of loading was markedly raised compared with the free-swelling constructs (P < 0.01), with a nearly three-fold higher release of GAG within the L4 group compared with the L2 group (P < 0.01). Interestingly, the F2L2 group retained a higher proportion of GAG in the scaffold than the immediately loaded (L4) constructs (P < 0.05) ( Fig. 1B ).
When comparing P0 with P3 scaffolds (normalized to F2 scaffolds), there was a significantly higher GAG/DNA ratio among P0 F2L2 constructs (P < 0.05), whereas within the remaining compositions no significant difference could be detected ( Fig. 1C ).
Gene Expression
In P0 cells, the gene expression of collagen type II was significantly up-regulated after loading for 4 weeks (P0 L4) or free-swelling followed by loading (P0 F2L2) compared with the free-swelling control group (P < 0.01) ( Fig. 2A ). The level of aggrecan mRNA was significantly higher in both P0 L4 and P0 F2L2 groups than in free-swelling constructs (P < 0.01). Moreover, the loading of F2L2 stimulated the chondrocytes to express more aggrecan than P0 L4 (P < 0.01). Any kind of loading mode significantly raised the cartilage oligomeric matrix protein (COMP) gene expression, either loading for 2 weeks (P < 0.01) or 4 weeks (P < 0.05) or free-swelling followed by loading (P < 0.01). However, superficial zone protein (SZP) mRNA was significantly elevated only in the F2L2 group compared with the free-swelling culture for 4 weeks (P < 0.05).
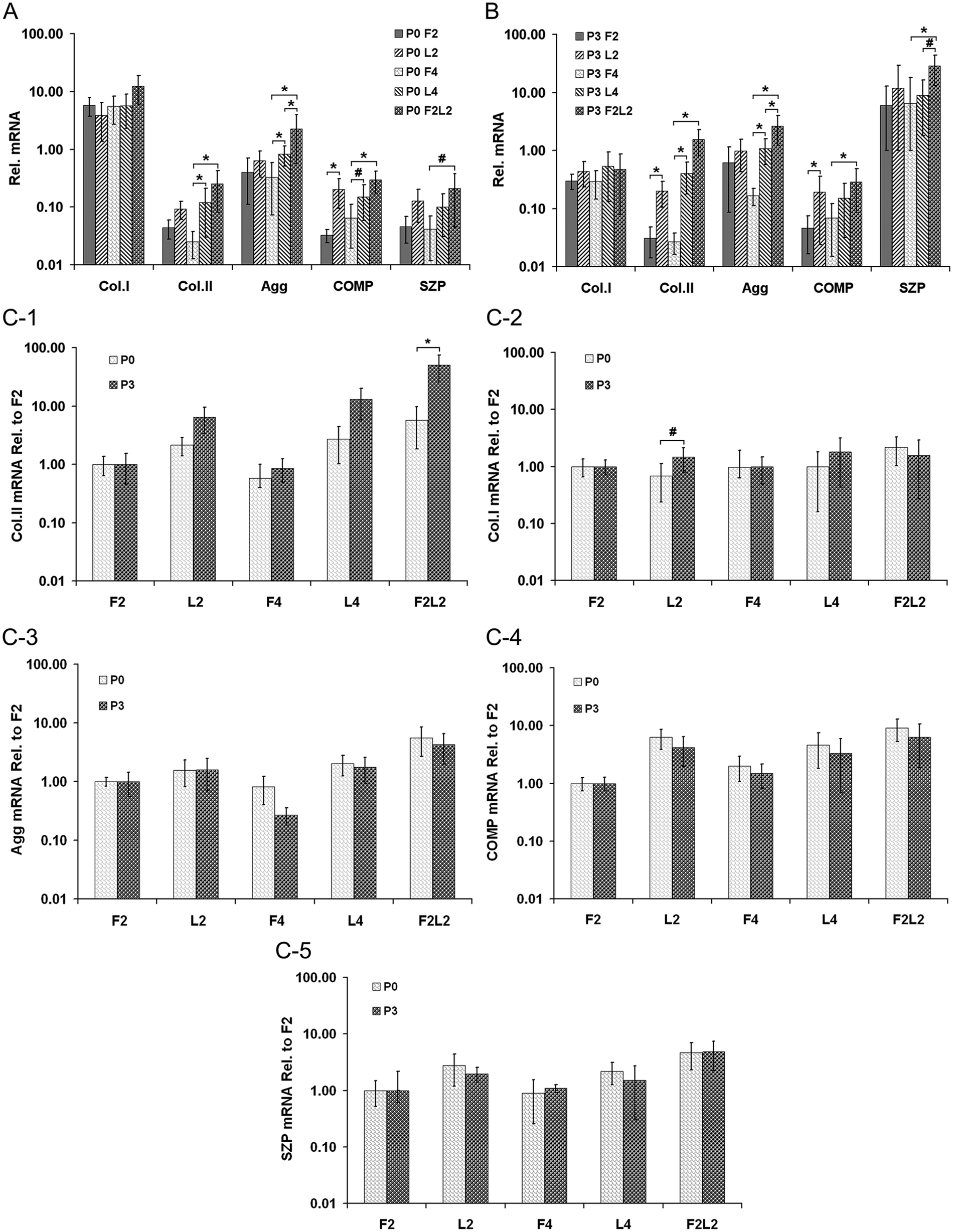
Relative mRNA expression among the different primary (P0) (
In P3 cell-laden constructs ( Fig. 2B ), different durations of direct loading, such as L2 or L4, significantly increased the gene expression of collagen type II compared with the respective free-swelling group (P < 0.01). Furthermore, the P3 F2L2 construct showed a 51-fold higher mRNA level of collagen type II than the control group (P < 0.01). The gene expression of aggrecan was significantly up-regulated after loading for 4 weeks (P3 L4) or free-swelling followed by loading (P3 F2L2) (P < 0.01); moreover, the latter loading regime significantly enhanced the production of aggrecan mRNA compared with the former (P < 0.01). In addition, higher mRNA levels of COMP were found in both P3 L2 and F2L2 constructs compared with free-swelling controls (P < 0.01). SZP mRNA level in the P3 F2L2 construct was significantly elevated (P < 0.01) and also higher than in the P3 L4 composite (P < 0.05). However, collagen type I expression level was not significantly influenced by loading in either P0 or P3 cell groups at all time points ( Fig. 2A and B ).
When comparing P0 with P3 scaffolds (normalized to F2 scaffolds), there was a significantly higher expression for collagen type II within P3 F2L2 scaffolds (P < 0.01) and a significantly higher collagen I expression among P3 L2 constructs (P < 0.05). Comparison among the remaining groups was without a significant difference ( Fig. 2C ).
Histology, Immunohistochemistry
Sections of primary cell constructs were stained with toluidine blue (
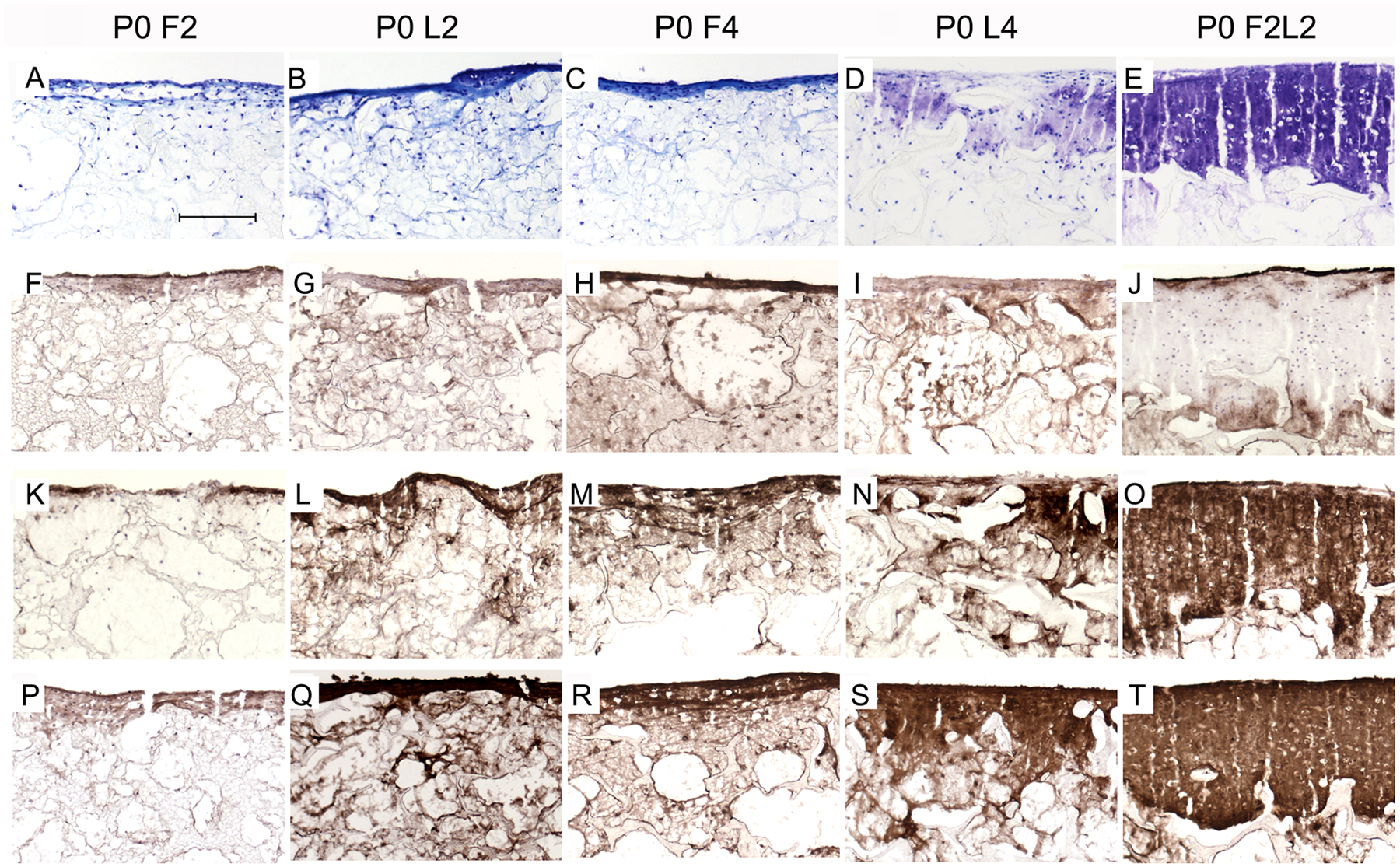
Representative cryo-sections from the different primary (P0) chondrocyte–scaffold constructs at 14/28 days postculture under free-swelling (F), dynamic loading within bioreactor (L), or combined conditions (FL). Toluidine blue staining (
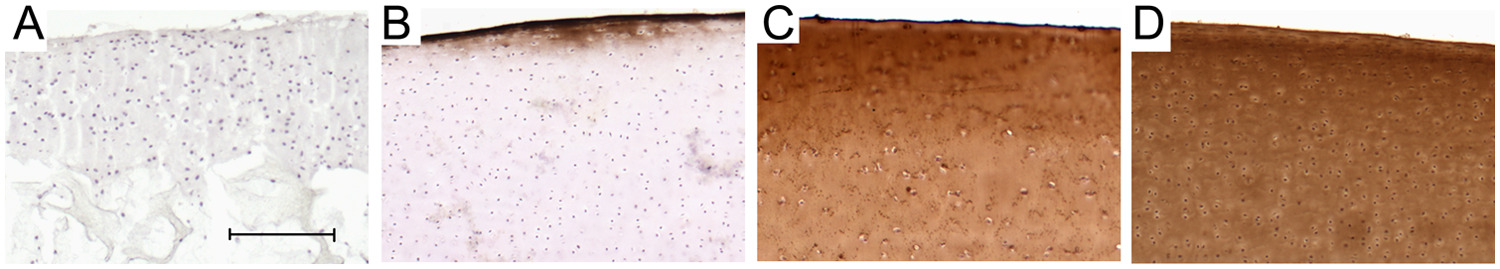
Representative cryo-sections of controls. Negative control from P0 F2L2 construct as representative section showed no staining throughout the cell–scaffold construct (
Histological analysis revealed different matrix patterns within P3 chondrocyte–scaffold constructs (
Fig. 5
). In sections from the P3 L2 composite, slightly more intense staining was detected on the fringes of the scaffold with less dispersed cells, in contrast to the barely stained matrix with more evenly distributed rounded cells through most parts of the construct from the P3 F2 group. After loading for 4 weeks (P3 L4), the accumulation of matrix was more pronounced at the surface of constructs. Interestingly, ECM staining was less rich and intense at the top layer of constructs in the P3 F2L2 group compared with those of the P3 L4 group (
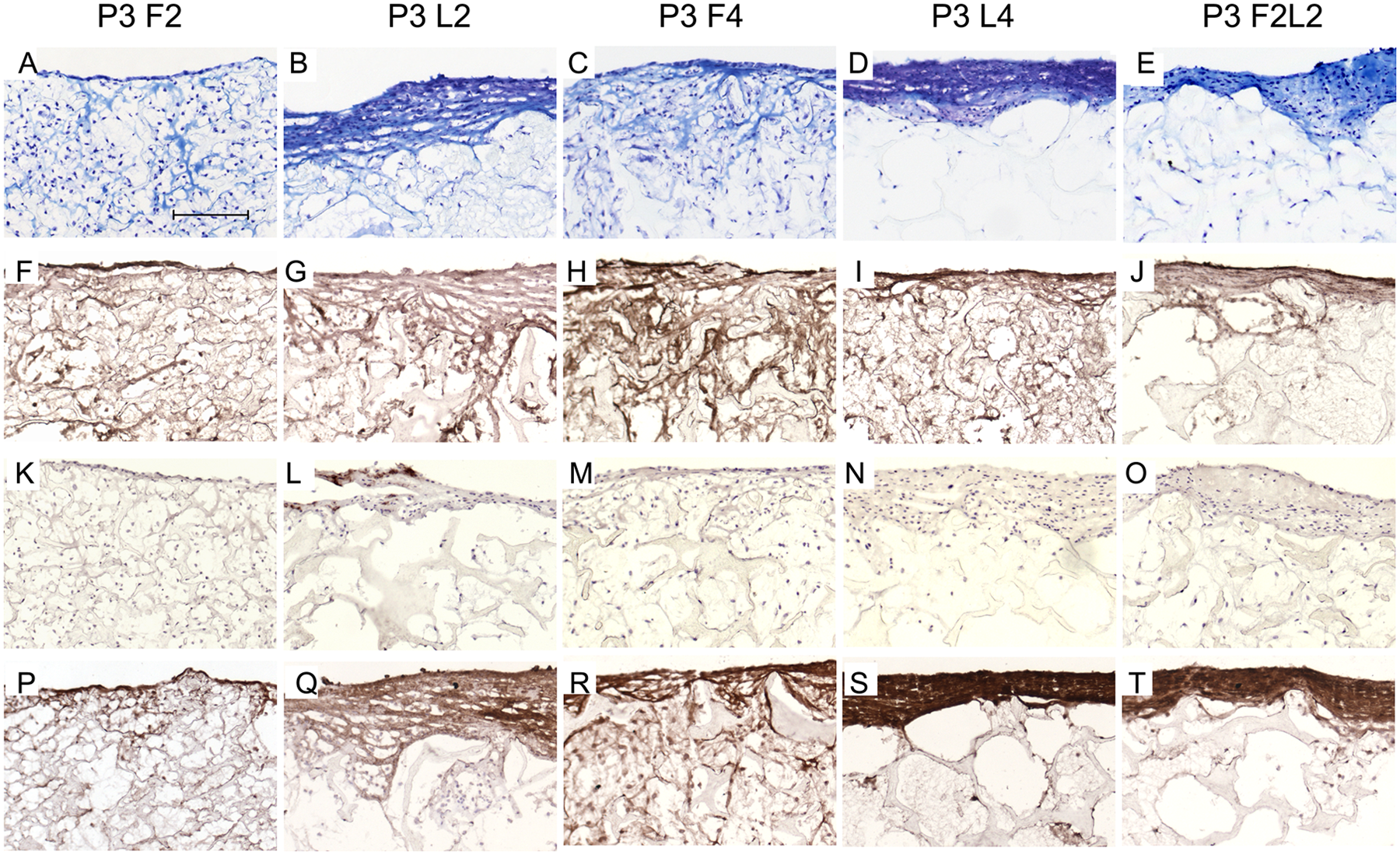
Representative cryo-sections from the different passage 3 (P3) chondrocyte–scaffold constructs at 14/28 days postculture under free-swelling (F), dynamic loading within bioreactor (L), or combined conditions (FL). Toluidine blue staining (
Comparing P0 and P3 cell–PU composites, ECM was considerably assembled in P0 compared with P3 cell-laden composites from the F2L2 groups ( Figs. 3E and 5E ). Moreover, loading increased the synthesis of cartilage-specific collagen type II in P0 more than in P3 L2 and L4 products ( Figs. 3L and 5L and Figs. 3N and 5N ). Of importance, the F2L2 P0 cell–scaffold constructs displayed a more pronounced immunostaining of collagen type II and aggrecan in comparison to P3 F2L2 group ( Fig. 3O and 3T and Fig. 5O and 5T ).
Discussion
The results of this study were produced using bovine articular chondrocytes cultured within a matrix that is not in clinical use for chondrocyte transplantation. Yet PU matrices have recently been successfully applied in human patients to replace defective meniscus tissue. 16 The findings of this study indicate that unpassaged articular chondrocytes generally undergo superior cartilage maturation within mechanical bioreactor surroundings when compared with chondrocytes that have been previously passaged. Furthermore, the mechanically induced chondrogenic response is much stronger when the articular chondrocytes have been precultured under free-swelling conditions within 3-D surroundings prior to the application of mechanical load. In addition, chondrocyte maturation increased with prolonged culture duration.
It has been previously reported that biomechanical stimulation promotes chondrogenesis within 3-D cultured chondrocytes over free-swelling control.6,17 In agreement with previous results,18,19 primary chondrocytes cultured in 3-D conditions showed highest GAG retention per scaffold in P0 F2L2 groups. The synthesis of GAG in primary cells was obviously increased by loading, especially in the F2L2 constructs, where the GAG/DNA ratio in P0 chondrocytes was about 7.0, whereas the ratio of GAG/DNA was around 3.0 in P3 cells, suggesting that, as expected, primary cells showed better responsiveness regarding matrix synthesis than passaged cells. One reason might be that the P3 chondrocytes were dedifferentiated and thus had a reduced capacity of anabolic responsiveness to mechanical stimuli. Also, Das et al. 20 demonstrated that passaged chondrocytes showed greatly decreased positive responses to loading and increased expression of MMPs compared with primary cells.
Consistent with previous studies, cartilaginous matrix gene expression was up-regulated in response to dynamic loading.21,22 PCR results revealed that chondrogenic gene markers, including collagen type II, aggrecan, COMP, and SZP (also known as proteoglycan-4 or lubricin), were mostly up-regulated in both loaded primary and P3 cell–PU constructs, whereas the F2L2 groups demonstrated most favorable chondrogenic gene expression. A previous work reported by Nicodemus and Bryant emphasized the type and timing of dynamic loading dramatically influenced ECM and MMP gene expression and to a lesser degree matrix deposition. 23 Although the group suggested that early application of immediately intermittent loading does stimulate both anabolic (i.e., deposition of collagen type II) and catabolic activities, and chondrocytes are less sensitive to loading after 1 week of free-swelling culture, the studies from our group had revealed pre-incubation of 6 days in constructs followed by the application of a short-term (3 days) surface motion is sufficient for an up-regulation of cartilage-related genes.6,24 In this study, constructs were precultured in free-swelling culture for 2 weeks prior to mechanical loading for 2 weeks, and an up-regulation of chondrogenic genes would thus confirm the previously reported results. It is likely that a preexisting extracellular matrix may facilitate the transduction of mechanical stimuli in PU scaffolds. Therefore, the pre-incubation of cells in 3-D culture milieus for a relatively longer period, in order to achieve more extracellular matrix prior to loading, might further improve the outcome of long-time loading experiments. Thus, our findings corroborate that free-swelling culture followed by loading may be the most favorable regime for cartilage maturation.
In both P0 and P3 cell–scaffold constructs, the unchanged expression of collagen type I in combination with the significant increase in expression of collagen type II and aggrecan indicated that the applied loading may either contribute to the prevention of cell dedifferentiation or promote cell redifferentiation during long-term culture. It has been shown that loading results in a mild collagen type 1 expression, which may be related to the fact that expression of collagen type I should be regarded as a sign of cellular activity rather than an exclusive marker of dedifferentiation. Moreover, mechanical load has previously been reported to elicit a temporary collagen type I up-regulation with down-regulation during the further course of dynamic in vitro culture. Furthermore, an up-regulated collagen type I expression may also reflect to some extent the activation of resident sleeping precursor or stem cell within cartilage biopsies.14,25 Besides, unlike more classic markers such as collagen type II, COMP is a sensitive marker that appears early in primary chondrocyte-derived chondrogenesis and is drastically reduced at the mRNA level within 6 weeks of monolayer culture, whereas collagen type II remains detectable for several months, 26 which potentially makes COMP a better candidate to provide early insight into the possible outcome of cartilaginous tissue formation in vitro. In this study, COMP mRNA was also clearly promoted after dynamic loading, which parallels previous investigations. 27 Consistent with previous findings that applied surface motion for short time periods promoted the SZP gene expression, 24 improved expression of SZP, especially in F2L2 groups, was observed under the influence of long-term complex loading. As SZP is known to contribute to boundary lubrication, 28 the regulated SZP synthesis may impart functional lubrication at sites responsive to mechanical stimuli.
In agreement with our PCR findings and GAG quantification, the histological and IHC results also illustrated the enhanced accumulation of cartilage matrix molecules (i.e., collagen type II and aggrecan) and weaker collagen type I staining in cell-laden constructs of the loaded groups, especially in the P0 F2L2 constructs. Also, in cell-laden constructs more intense toluidine blue staining of ECM was noticeable at the edge areas of loaded constructs compared with controls, confirming that the applied mechanical stimuli are able to enhance matrix accumulation. More intense matrix staining and cellularity at the respective scaffold edges was seen among all our scaffolds. This phenomenon has been seen previously and has been termed edging effect. Scaffold-seeded cells have a tendency to move or be more viable at the construct edges, which is most likely related to an improved nutrient supply at this position even though our loading regimen also includes fluid flow. The edging effect was seen not only in our previous bioreactor experiments but also in histology sections from many other working groups.6,14,29 It is a drawback of in vitro tissue engineering and has to be clearly addressed during future experiments to possibly solve this problem. In addition, the aggrecan immunostaining was more pronounced in loaded groups, particularly in P3 F2L2 groups. The immunoreactivity of collagen type II, the classic chondrogenic marker, was not markedly improved even after mechanical loading. We also noticed from IHC results that the significant differences in mRNA expression of collagen type II were not translated into detectable differences in protein deposition of P3 cells between groups. One possible reason is that the antibody may be not sufficiently sensitive to distinguish differences in collagen II protein deposition at low levels, especially comparing to the more sensitive real-time PCR method, which can amplify even a few mRNA copies; it may also be related to the fact that the protein production of collagen type II is a slow process.
On the basis of immunostaining of collagen type II, as well as GAG biosynthesis, in P3 chondrocyte-laden constructs compared with that of primary cell–PU constructs, it can be suggested that primary chondrocytes are more responsive to loading than P3 cells. This is also consistent with a previous study where the use of passaged cells led to a compromised tissue quality with decreased biochemical content and mechanical properties. 30 It has also been previously shown that the cell growth rate and viability or the expression of COL2/COL1 decreased and apoptotic cells increased as the chondrocyte was subcultured from P2 to P5, indicating chondrocyte dedifferentiation. Also, engineered cartilage in in vitro polymer scaffold or in vivo mice with P1 chondrocytes showed higher GAGs and type II collagen, whereas tissues engineered in vitro and in vivo with P5 cells lost chondrocyte morphology or cartilage-specific extracellular matrices. The frequency of chondrocyte passage is an important factor clearly affecting the quality of tissue-engineered cartilage constructs that have been cultured using chondrocytes. 31 Moreover, supporting the fact that bioreactor tissue engineering mimicking joint movement can produce similar results as animal experiments, it was additionally reported in one previous study by Salzmann and colleagues that linear mixed regression exposed that with increasing passage the expression of differentiation targets decreased in vitro and in vivo. 5
Practical chondrocyte transplantation techniques face an inherent limitation of transplanting a tissue-engineered cell–scaffold construct containing subcultured chondrocytes with inferior quality. Autologous chondrocyte implantation techniques commonly apply passaged (passage 3-5) chondrocytes, which are placed under a water-tight periosteum or under/within a 3-D membrane.4,32 -34 These isolated cells without any surrounding matrix for protection or even matrix promotion are exposed to the harsh intra-articular environment in the very early postoperative phase, even though patients are partial or non–weight bearing for defined time points. Often, patients are experiencing rehabilitation by use of continuous passive motion (CPM) apparatus, exposing the transplant area to at least shearing forces. According to our data, instant mechanical stimulation is improving chondrogenic markers such as GAG or collagen type II over unloaded control, supporting the utility of currently applied rehabilitation schemes. 34 Chondrocytes that are placed within the matrix and subsequently mechanically stimulated generate an improved chondrogenic response when compared with free-swelling control. Yet this response is dramatically increasing when the chondrocytes are initially enabled to generate a mild surrounding matrix and are intermittently exposed to dynamic surroundings. This information parallels previous reports.5,35 Even though it has to be reminded that in general static (and non-chemical enhanced) free-swelling culture of matrix-surrounded chondrocytes does not strongly enhance chondrogenesis. 36 Albrecht and colleagues recently reported that scaffold characteristics and culture conditions highly influenced gene expression among four different industrial transplant types (MACI, HyalograftC, CaReS, and Novocart 3D), attesting that simple culture parameters may have profound impact on the tissue regeneration following matrix-associated chondrocyte transplantation. 37 Among these products, in vitro 3-D culturing conditions are maintained for 2 days (Novocart 3D), 1 week (MACI), or 2 weeks (Hyalograft, Cares) on re-implantation. Chondrocytes are usually passaged with the exception of Novocart 3D. Albrecht reported, with partially heterogenous results, that the Cares implant (2 weeks precultivation) generally resulted in the overall best gene expression pattern. Particularly, when comparing with a short precultivation product (Novocart 3D), these in vitro data compare very well with data from our study and might become even more interesting when the transplants are mechanically stimulated by and within the patient.
Cartilage maturation occurs through several phases after implantation of grafts. 38 Generally, the first 4 weeks after transplantation surgery are dedicated to strictly restricting motion for 18 hours followed by continuous passive motion of the knee. Typically weight-bearing and progressive return to normal daily activity will begin after the first month. 39 In this study, the culture mode of free-swelling culture followed by mechanical loading may simulate the process of cartilage maturation after implantation of engineered constructs. Therefore, our study strongly indicates that initial in vitro free-swelling culture followed by dynamic loading (simulating in vivo load and motion) may be most favorable for chondrogenesis. Future studies including matrices that are in clinical use for chondrocyte transplantation as well as human cellular material are required to underline these preliminary data.
Footnotes
Acknowledgments and Funding
We are grateful to Robert Peter, AO Research Institute, for excellent technical assistance with cell culture and biochemical analysis; to Andrea Oswald, AO Research Institute, for his assistance in immunohistochemistry; and to Patrick Trüssel for his support in histology. This work was supported by the AO Research Fund of AO Foundation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This was an in vitro experiment therefore no ethical approval is required.
