Abstract
Objectives
Osteoarthritis is a painful joint disease responsible for walking impairment. Its quantitative assessment by gait analysis in mice may be a relevant and noninvasive strategy to assess the disease severity. In this study, we aimed to determine the severity of osteoarthritis at the tissular and gait levels in unilateral and bilateral posttraumatic murine osteoarthritis.
Methods
Twenty-four C57BL/6 male mice were randomly assigned to 3 groups (n = 8/group): controls, unilateral surgery, and bilateral surgery. Posttraumatic osteoarthritis was induced unilaterally or bilaterally by destabilization of the medial meniscus. Gait analysis was performed weekly with the CatWalkTM XT system until the 16th week after surgery. After animal sacrifices, histological and micro–computed tomographic assessment was performed.
Results
Operated knees showed a significant increase in the histological score compared with controls (P < 0.001). Calcified anterior medial meniscal bone volume was higher on the ipsilateral side after unilateral destabilization of the medial meniscus (P < 0.001) and on both sides after bilateral intervention (P < 0.01). One week after surgery, the mice mean speed decreased significantly in both operated groups (P < 0.001 and P < 0.05). In the unilateral group, a significant increase in the contralateral hind print area appeared from week 4 to week 16.
Conclusions
While bilateral destabilization of the medial meniscus induced no detectable gait modification except 1 week after surgery, unilateral model was responsible for a gait disturbance on the contralateral side. Further studies are needed to better define the place of the CatWalkTM in the evaluation of mouse models of osteoarthritis.
Introduction
Osteoarthritis (OA), the most prevalent joint disease, is a multifaceted inflammatory and degenerative disease that affects 10% of men and 18% of women aged above 60 years 1 and constitutes a worldwide leading cause of pain and disability. 2 OA is characterized by cartilage degradation, subchondral bone remodeling, osteophyte development, and synovial inflammation. 3 The knee is one of the most frequently affected joints, making knee OA responsible for more than 80% of the disease’s total burden.3,4 Knee OA implies walking disability due to pain and joint dysfunction (e.g., loss of range of motion) during loading and joint motion.4,5 Human OA development is usually slow over many years and variable from one subject to another, leading to the difficulty of longitudinal studies. To overcome this time-related issue, animal models that generate OA in a few weeks are useful. 6 While numerous mouse models are used to assess the functional consequences of OA,7-9 very few faithfully reproduce the human OA pathogenic mechanisms that dampen their pathomimicry level and limit the relevance of their human translatability. Indeed, chemical models with intra-articular agent injection, such as collagenase or sodium mono-iodoacetate, provide painful and fast end-stage OA. They are particularly used to study anti-inflammatory properties and efficiency of analgesic drugs.10,11 Some specific mouse strains are also used to investigate OA development, 6 for example, STR/ort mice develop OA spontaneously early in life and C57BL/6 are predisposed to age-related OA.12,13 Surgically induced models, such as the destabilization of the medial meniscus (DMM) or anterior cruciate ligament transection (ACLT), generate knee joint instability responsible for progressive articular degeneration mimicking human posttraumatic OA. 14 Although in human knee OA both knees are involved in more than 75% of the cases, 15 bilateral mouse models of OA seem to have been rarely used. 16
Walking disturbances are one of the main consequences of pain associated with OA. These walking disorders are responsible for patient’s decreased mobility and physical activities, leading to restriction of participation. 17 As one of the main symptoms of knee OA is walking disturbance, this parameter should be taken into account in the evaluation of OA in animal models or preclinical tests of disease-modifying OA drugs (DMOADs). Indeed, walking assessment can be an accurate predictive tool to evaluate the potential effectiveness of a therapy in humans, using common clinical criteria rather than radio-histological criteria, because symptoms do not often correlate to the degree of the disease progression on imaging. 18 To assess rodent walking behavior, several gait analysis systems have been developed using either freely walking methods or treadmill systems. Treadmills exhibit the advantage of controlling velocity to reduce speed variability between mice, but they prevent natural gait and the ability for the animal to adapt walking velocity. 19 On the contrary, freely walking systems allow assessment of natural gait, including natural speed velocity which may be modified with pain. Freely walking systems appear relevant to assess OA consequences on natural gait behavior. 19 Gait analysis systems may provide a noninvasive method of joint function assessment in murine posttraumatic OA, which could help scientists evaluate in vivo knee function and preclinical efficiency of DMOAD, with criteria closer to those used in human clinical practice.
In this study, we aimed to measure, in parallel analyses, gait disturbances, tissular changes, and micro–computed tomodensitometric alterations of knee joints in murine posttraumatic OA models, with the ultimate goal of determining whether gait disturbance could be a noninvasive and longitudinal method to assess posttraumatic OA. To answer this question, we have evaluated gait disturbance in mice after unilateral and bilateral DMM as a model of posttraumatic OA, using the CatWalkTM XT system. This system allows the evaluation of free-moving and weightbearing walking. To confirm the OA onset in unilateral and bilateral DMM in mice, we have also evaluated histological and micro–computed tomography (µCT) joint changes in both models.
Methods
Animals
All the animal experiments were performed according to the European directive 2010/63/EU governing ethic and regulations in animal experiments. These experiments have been approved under the project N°2015033111162660. Mice were housed in the animal facility of Nantes Faculty of Medicine (Agreement No. C44015). Our present work adheres to the ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines 2.0 20 (Table S1).
Surgical Induction of Posttraumatic OA
Twenty-four 10-week-old male C57BL/6 mice, weighing 25.3 ± 1.3 g, were obtained from Janvier Labs (Saint-Berthevin, France). All animals had reached full skeletal maturity at the time of the study. After 1 week of acclimatization, mice were randomly assigned to 3 groups (n = 8 per group): a control group with nonoperated animals, a group with a unilateral DMM (Uni DMM), and a group with a bilateral DMM (Bi DMM). DMM surgery was performed on anesthetized mice according to previously published description.21,22 Briefly, a 3-mm longitudinal incision from the patella to the tibial plateau was performed at the right knee (Uni DMM group) or both knees (Bi DMM group). Then, the joint capsule was incised medially near the patella. The patella was dislocated laterally and the medial menisco-tibial ligament was sectioned with #11 blade. After surgery, mice were monitored daily for the first week and weekly at the time of gait analysis for signs of physical distress.
Gait Analysis
Gait analysis was performed using the CatWalkTM XT gait analysis system (Noldus Information Technology, The Netherlands). 23 Before surgery and gait recording, mice were pretrained to cross the 42-cm walkway for 2 weeks. 7 Animals were individually allowed to walk freely on an 8-cm-wide corridor walkway with a glass floor in a dark compartment. Gait was recorded according to a video-based system. Upon contact with the glass plate, the paw reflected the green light to a video camera below (100 Hz), which recorded the entire run. Then, gait was recorded for each group before surgery (baseline) and every week for 16 weeks after OA induction. Gait analysis experiments were conducted blindly by the same investigator. The scheme of the study is illustrated in Figure 1 . The hind and the front paws were assessed for data analysis. Gait parameters were acquired with the CatWalkTM XT according to the method previously described by our lab. 24 Briefly, the criteria for compliant runs were a run duration between 1 and 5 seconds and a maximum speed variation of 80%. Runs with backing, sniffing, cleaning, or long stops were also discarded during the acquisition phase by the investigator. Five compliant runs were necessary for statistical analysis. In most cases, the first 5 or 6 runs produced the 5 compliant runs. We studied the following parameters based on their frequent use in rodent gait analysis recommendations19,25: velocity, paw print area, paw print intensity, stance phase, swing phase, swing speed, and duty cycle (the stance phase as a percentage of the step cycle: stance / [stance + swing] × 100).
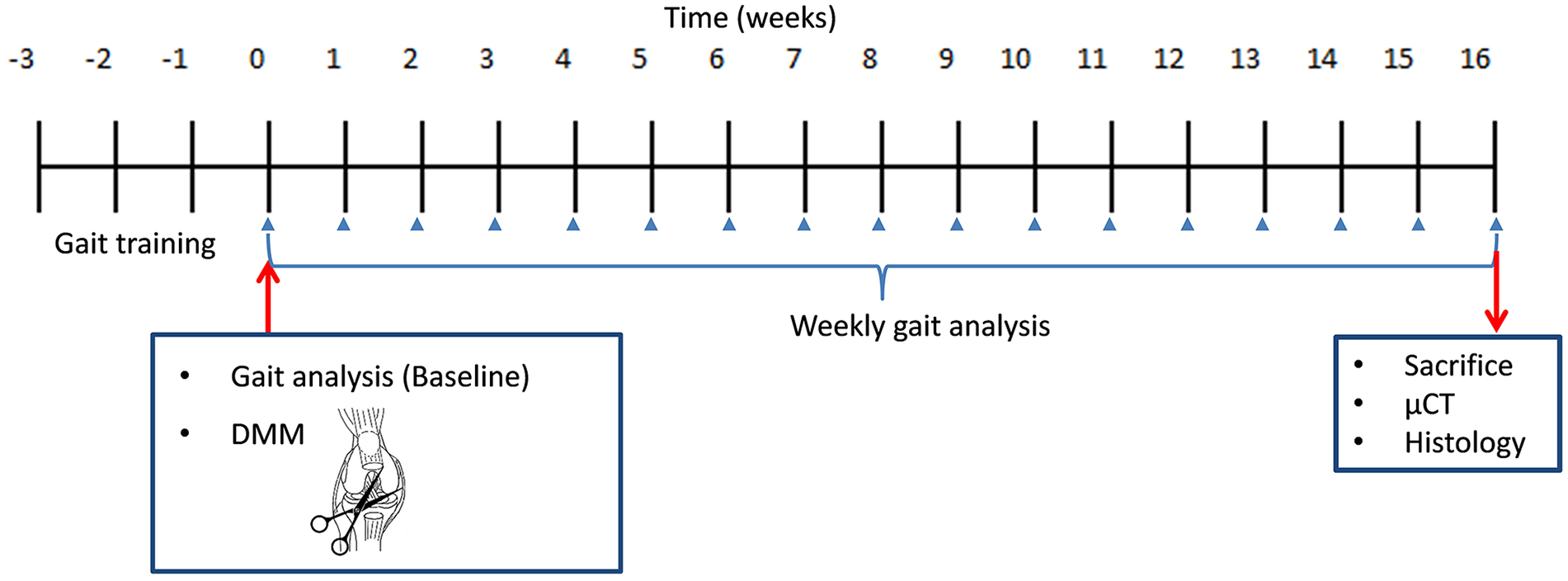
Experimental scheme of gait analysis and articular assessment. After 1 week of acclimatization, mice were randomly assigned to 3 groups: controls, unilateral DMM, and bilateral DMM. Mice were pretrained to cross the walkway for 2 weeks. Then, DMM surgery was performed. After 1 week of recovery, gait was recorded weekly for 16 weeks. At the end of the protocol, mice were euthanized and both knee joints were analyzed with micro–computed tomography (µCT) followed by histological scoring. DMM = destabilization of the medial meniscus.
µCT Assessment
At the end of the gait analysis protocol, mice were euthanized and both knee joints were analyzed with µCT according to Bouxsein et al.’s 26 guidelines. Joints were fixed during 48 hours in a 4% solution of paraformaldehyde and then placed in 70° ethanol. Joints were scanned with a Micro Computer Tomograph Skyscan 1272® (Bruker, Germany). The µCT acquisitions have been performed at a resolution of 5 µm without filters. For the acquisitions, we used a voltage of 50 kV and an intensity of 200 µA, with a rotation angle of 0.45°, an exposure time of 620 ms and a frame averaging 2, and a mean scan duration of 17 minutes 50 seconds. The images obtained during the acquisition were processed with NRecon® software (Bruker) to obtain a 3-dimensional reconstruction of the different samples. The reconstructions of each sample were then spatially oriented identically, using Dataviewer® software (Bruker), so that they could be compared to each other. In addition, the Dataviewer® software makes it possible to obtain images of the samples in the sagittal, coronal, and transverse planes, which constitute the starting point for the selection of the area of interest. Images allowing an overview of each sample were produced with the CTVox® software (Bruker). Then, the final selection of the area of interest was carried out using the CTAnalyser® software (Bruker) (Fig. S1). According to Shin et al., 27 we have focused on the subchondral bone of the tibial epiphysis and the anterior medial meniscus area to assess bone volume (mm3), subchondral bone thickness (mm), and trabecular bone thickness (mm). The anterior medial meniscus has been isolated after 3 different successive stages (Fig. S2) on a coronal plane by 2 different operators (M.M. and B.M.): (1) exclusion of the femur, the tibia, and the patella; (2) selection of the remaining medial structures; and (3) isolation of the anterior part of the medial meniscus.
Histology
After µCT assessment, knee joints were dissected, skin and muscle were removed, and knee joints were placed in 0.5 M (pH 7.4) EDTA solution at 4 °C for 2 weeks for decalcification. Then, samples were dehydrated in a graded series from 70° to 100° of ethanol solution before paraffin embedding. Paraffin sections of 5 µm were stained with safranin O-fast green with an automaton, Varistain Gemini S (Thermo scientific, USA). The severity of OA was evaluated using a scoring system adapted from the Osteoarthritis Research Society International (OARSI) as previously described ( Table 1 ). 22 The 4 quadrants of the joints, the medial femoral condyle, the lateral femoral condyle, the medial tibial plateau, and the lateral tibial plateau, were analyzed, and scores were assessed by calculating the total sum of the quadrants. Scoring was performed blindly and independently by 2 different trained readers (A.F.C. and B.M.) on at least 2 or 3 levels, from the anterior area to the mid-weightbearing region of the joint for each sample to obtain enough interpretable quadrants.
Adapted OARSI Grading System Used to Evaluate OA Severity.
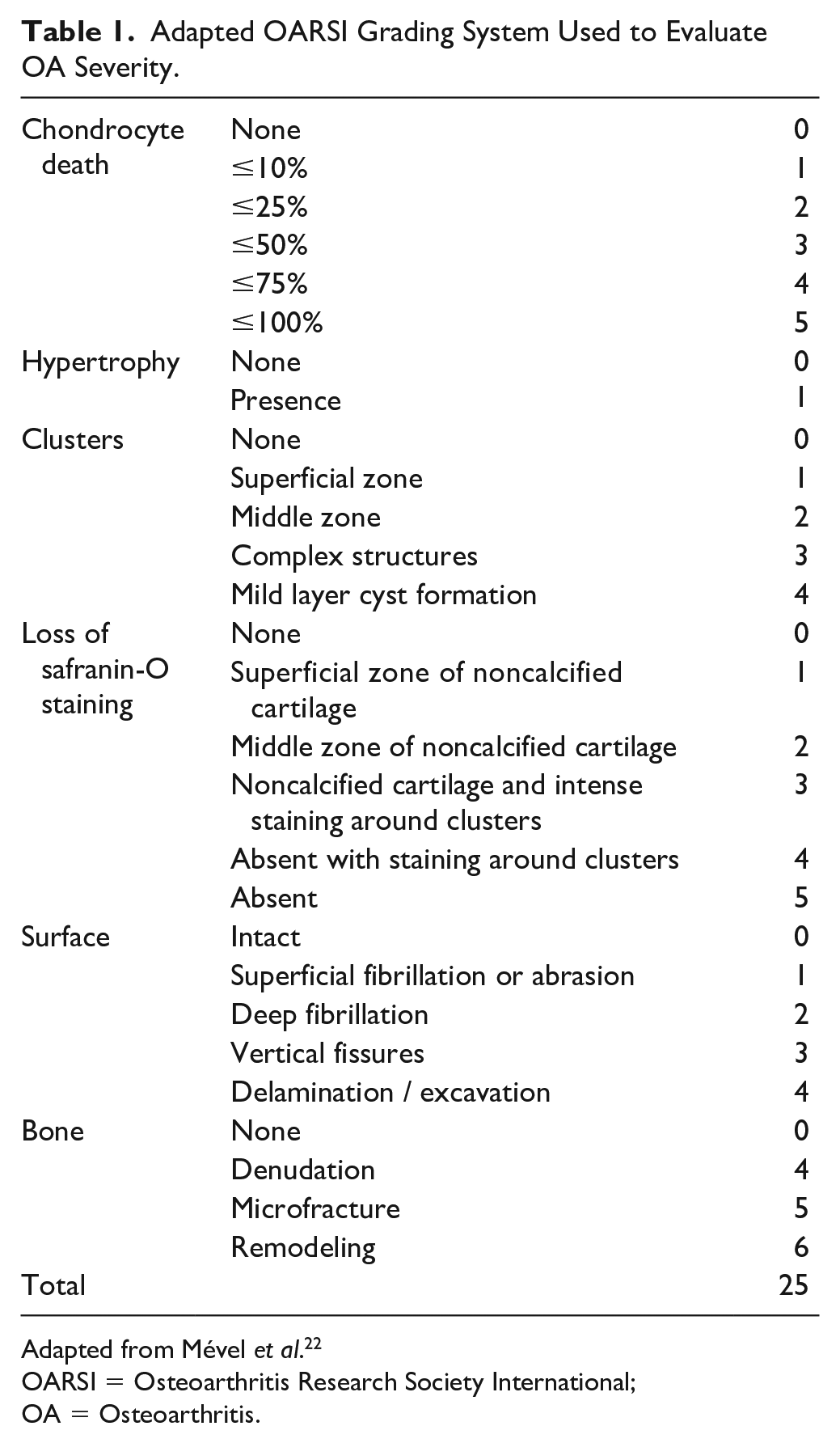
Adapted from Mével et al. 22
OARSI = Osteoarthritis Research Society International; OA = Osteoarthritis.
Statistical Analysis
Statistical analyses were performed with the SPSS 23.0® software (Chicago, IL). Results are expressed as mean (SEM). We used repeated-measures analysis of variance (ANOVA) followed by Bonferroni corrections to compare longitudinal changes in each gait parameter within each group. We also calculated the area under the curve for the parameter “paw print area” in each group. To compare histological and µCT scores among groups, Kruskal-Wallis tests followed by Dunn tests were used. The significant difference was determined at P < 0.05. Graphics and figures were performed with GraphPad Prism 7.0 ( GraphPad Software, San Diego, CA).
Results
Mice recovered quickly after surgery and healed in less than a week, which enabled us to start the gait analysis protocol at 1 week after surgery for each group. All groups were comparable concerning body weight (controls = 24.88 ± 1.55 g; unilateral DMM = 25.38 ± 1.19 g; bilateral DMM = 25.75 ± 1.28 g; P = 0.31).
Histology
To assess OA following unilateral and bilateral DMM, we performed histological analyses of knee joints 16 weeks after surgery. Our data (
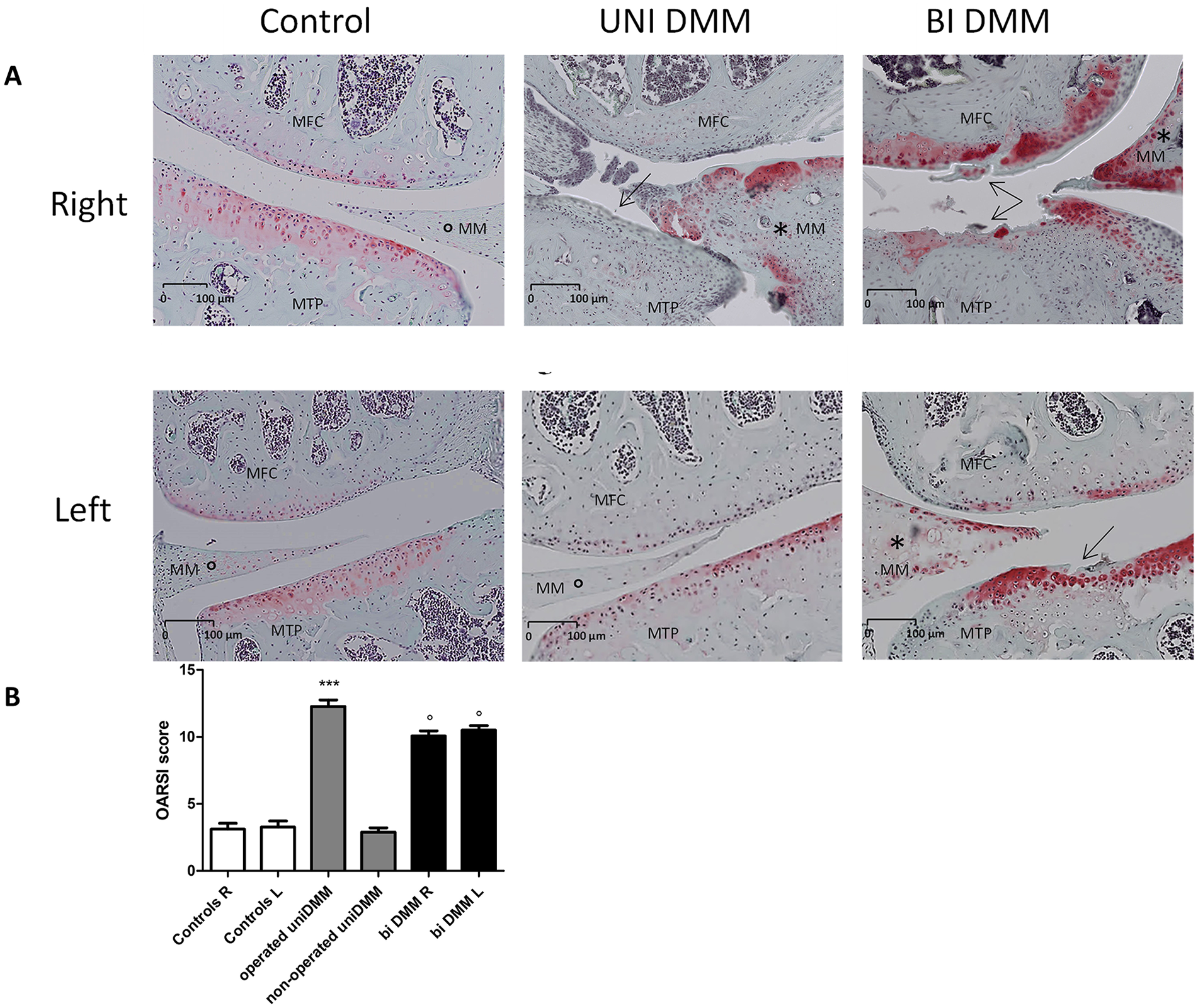
Histological analyses of knee joints after surgical OA induction: (
µCT Assessment
To further document the joint tissue damage induced by the unilateral and bilateral DMM, we performed µCT analyses on subchondral bone. Three-dimensional images of coronal sections of the joints showed morphological modification of right meniscus in the unilateral DMM (
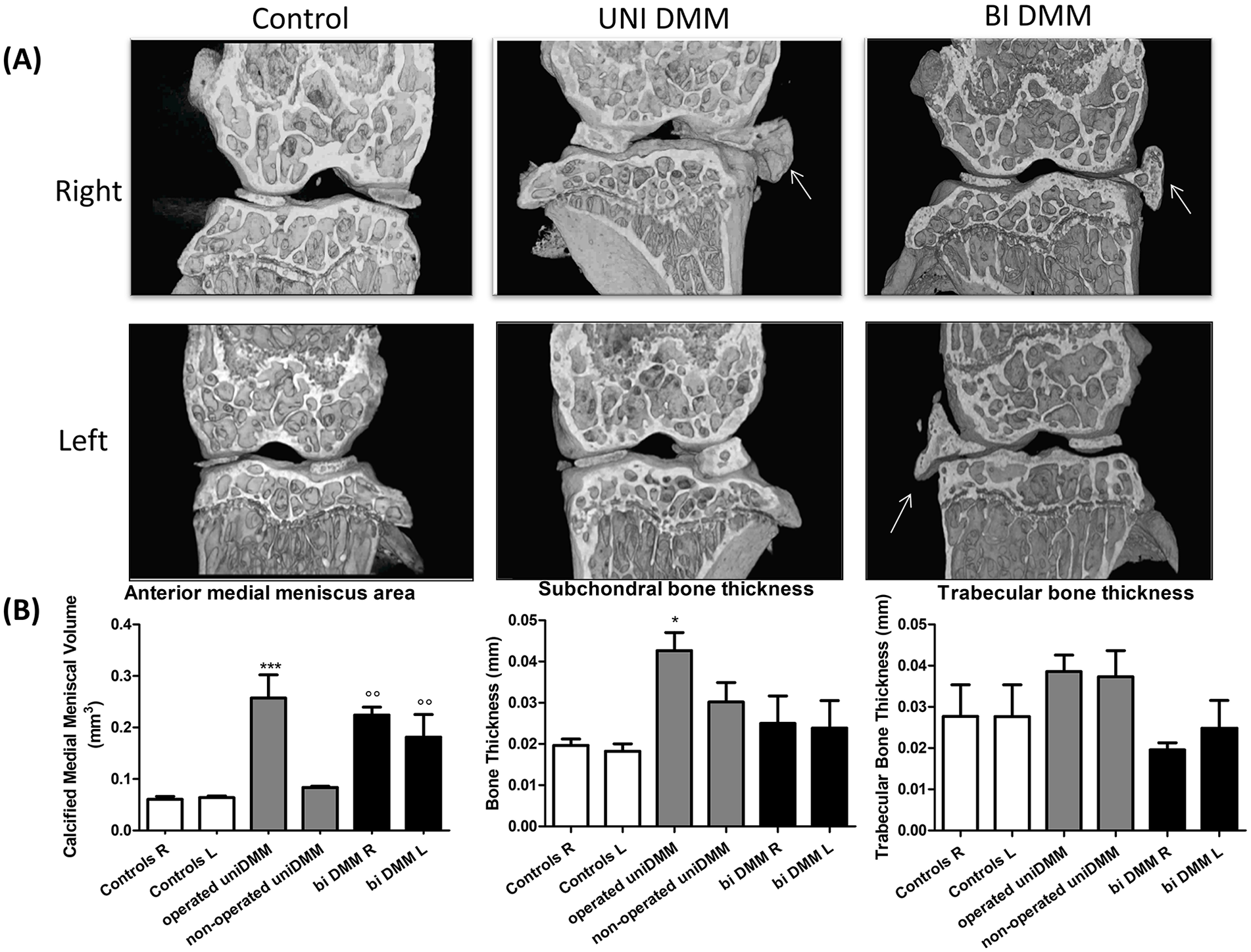
Micro–computed tomography (µCT) analyses of knee joints after surgical OA induction: (
Gait Analysis
After having confirmed that unilateral and bilateral DMM-induced knee joint damage attributed to the development of OA, we sought to compare the walking disturbance of controls and OA-affected mice. The mean speeds (velocity) are significantly inferior to the control group in both unilateral and bilateral DMM groups in the first week after surgery (P < 0.001 and P < 0.05, respectively) and recovered similar values to the control group as early as the second week until the 16th week (
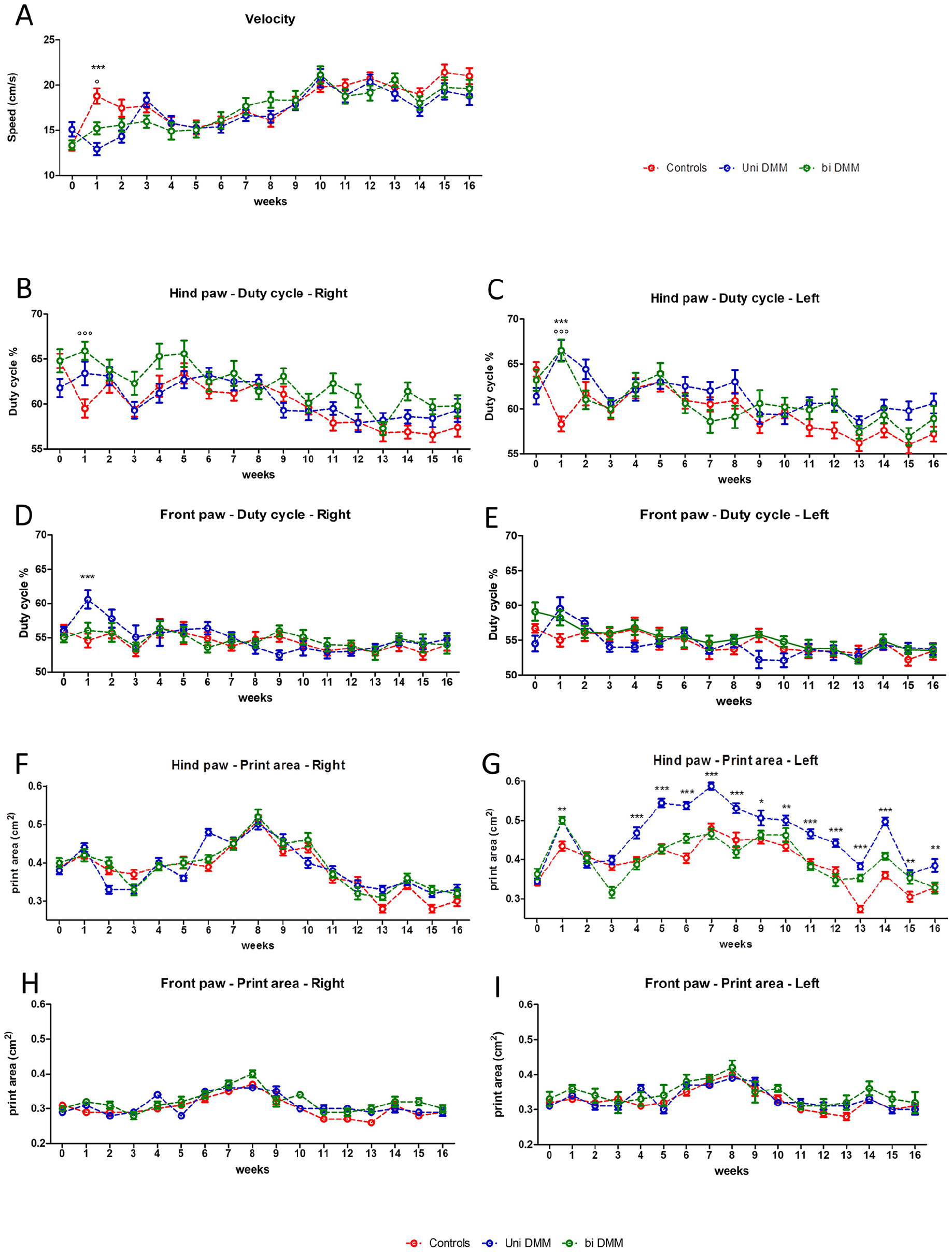
Gait parameter changes in mice following surgical OA induction. Velocity (
Comparison of the Areas Under the Curves Between Groups for the Hind Paw Print Areas (One-Way ANOVA Followed by a Bonferroni Post Hoc Test).
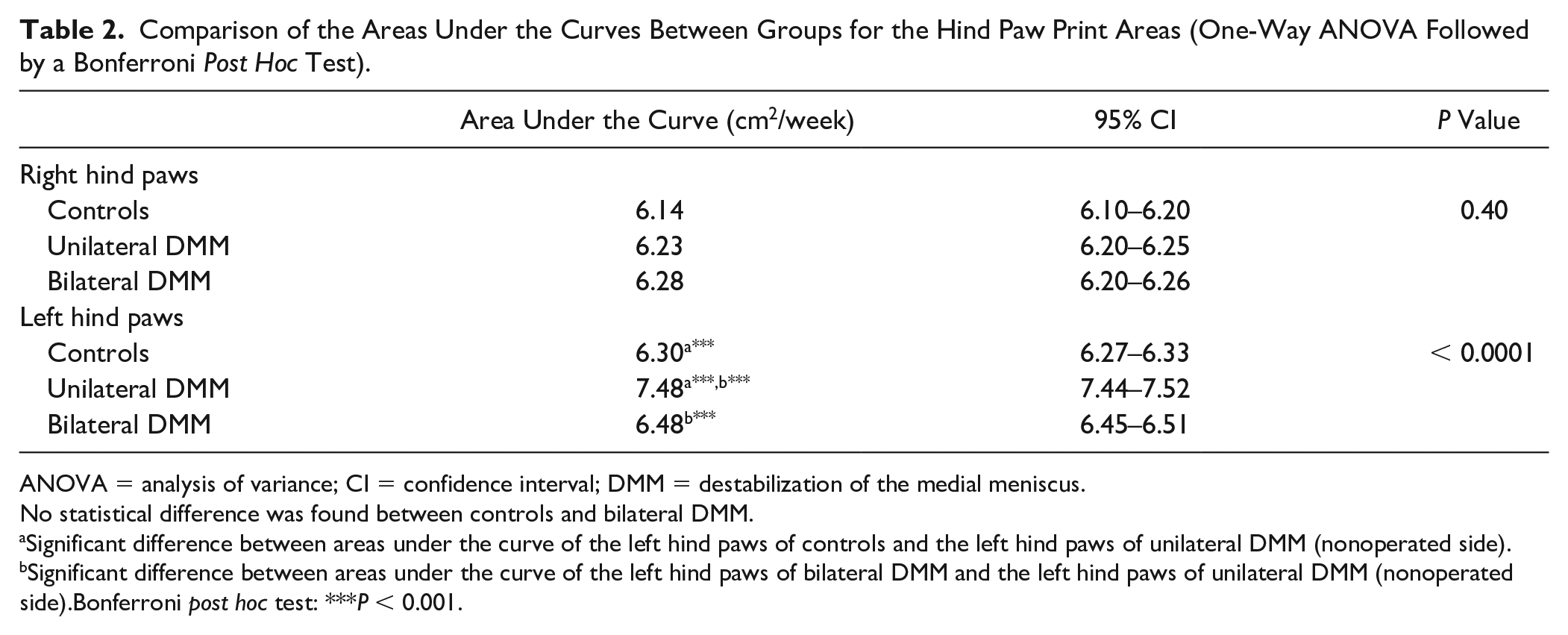
ANOVA = analysis of variance; CI = confidence interval; DMM = destabilization of the medial meniscus.
No statistical difference was found between controls and bilateral DMM.
Significant difference between areas under the curve of the left hind paws of controls and the left hind paws of unilateral DMM (nonoperated side).
Significant difference between areas under the curve of the left hind paws of bilateral DMM and the left hind paws of unilateral DMM (nonoperated side).Bonferroni post hoc test: ***P < 0.001.
Discussion
OA is the most prevalent degenerative joint disease in which cardinal symptoms are joint pain and functional disability.1,2 Therefore, OA strongly impacts patients’ mobility in daily living. While animal models of OA have been widely used to study the pathophysiology and progression of joint damage, only very few studies have focused their attention on the OA-associated gait disturbance. However, in preclinical studies, it appears to be of major interest to assess functional parameters that are common criteria to those used to judge the effectiveness of a treatment in humans. This study aimed to evaluate in a parallel manner the consequences of a unilateral and a bilateral medial meniscus destabilization on gait parameters and tissular changes in mice. Indeed, while in human pathology both sides are often affected by OA in about 80% of cases,16,28 few studies have investigated the bilateral model of OA in mice.16,25 Therefore, to reproduce more accurately posttraumatic human OA pathogenesis, we have chosen to use a surgical OA induction model, such as DMM, ACLT, or medial meniscus transection (MMT), which is known to induce a slower progressive OA than chemical models.21,29,30
Histological analyses showed a significant increase in the OARSI score in the right knee of the unilateral DMM group and the right and left knees of the bilateral DMM group. These results are consistent with previous studies showing a significant increase in the OARSI score in DMM.21,29,31 Interestingly, the OARSI score appears similar in both groups, and bilateral DMM did not increase the OARSI score, indicating that cartilage degradation and OA are not aggravated when both joints are affected. As expected from literature findings, 27 unilateral and bilateral DMM exhibited higher calcified meniscal volume in the operated knees compared with controls or nonoperated sides. Surprisingly, we found an increase in the subchondral bone thickness only in unilateral DMM, whereas no difference was noted in bilateral DMM. Moreover, in both models, no significant difference was measured concerning trabecular bone thickness. This is particularly astonishing because DMM mice could more easily favor the nonoperated knee when walking, and that in human knee OA no obvious radiological difference between the bilateral and unilateral forms seems to exist. 28
Using gait analysis, we wanted to assess gait disturbances that could be induced by unilateral or bilateral DMM. Gait analysis allows the assessment of walking impairment, which is one of the main symptoms in human OA and a major therapeutic issue. This technique also allows to perform a longitudinal follow-up with repeated measurements over time and without sacrificing animals at each time point. However, results should be taken with caution because investigated gait disturbance parameters could vary from one study to another. For example, velocity is not always assessed in rodent models because some systems such as DigiGaitTM (Mouse Specifics, Cambridge, MA) or TreadscanTM (CleverSys, Reston, VA) use treadmills with constant speed to get rid of speed variation.19,25 These systems may be of interest to eliminate aberrant results and variability due to mouse behavior issues. However, setting apart speed variation may also constitute a limit because natural velocity is not taken into account and may induce gait variations due to inappropriate speed. Therefore, walkways such as CatWalkTM XT or GaitScanTM allowing free walking appear as interesting tools to perform gait analysis. Moreover, a comparison between CatWalkTM XT and DigiGaitTM seemed to be in favor of the use of CatWalkTM XT, due to its technical advantages in data analysis with high-quality images. 32 In our study, speed variations appeared only during the first week after surgery, and that could be explained by postoperative phenomena such as knee pain or knee swelling despite an apparent skin healing. This speed parameter is not systematically taken into account, notably because of the disparate data from one study to another. On the one hand, some studies on ACLT rat models found no speed variation in a daily analysis during a short period after surgery or a monthly analysis performed up to 6 months.30,33 On the other hand, in other studies using DMM models and CatWalkTM XT gait analysis, the velocity was not provided.8,29,31,34 In a systemic mouse model of OA with a collagen IX deficiency, using a custom gait arena, Allen et al. 35 observed a decrease in the velocity compared with controls. At only 1 week after surgery, we have noted an increase in the hind paw duty cycle on the DMM-operated side and on the ipsilateral front paw in the unilateral group and on both sides at the hind paws in the bilateral group, which implies a relative prolonged stance phase and/or a reduced swing phase for each walking cycle, even if these 2 parameters are not significantly modified taken independently. Thus, it may be coherently associated with the reduction in speed during this same period. Yet, results regarding the duty cycle seemed variable among the previous studies. Indeed, using CatWalkTM XT, Ferland et al. 30 found no duty cycle modification in their ACLT group during the 26 days of analysis, whereas Muramatsu et al. 29 and Zhu et al. 36 observed a decrease in the duty cycle ratio at different time points after DMM induction in mice. Moreover, Kloefkorn et al., 37 with a custom gait arena, found no duty cycle modification in the first 6 weeks after OA induction by MMT in rats. In the present work, we noted no difference regarding the swing and stance phases between the 3 groups. Once again, both parameters are variably modified according to the studies. On the one hand, Ferland et al. 30 exhibited no modification of the swing phase without mentioning the stance phase, and on the other hand, Muramatsu et al. 29 reported shortening of the stance phase and elongation of the swing phase. Allen et al. found a reduction of the ipsilateral stance phase, 23 days after surgery in MMT mice, but did not mention results concerning the swing phase. 37 In our unilateral DMM group, we showed an increase in the footprint area of the nonsurgical side, whereas bilateral DMM mice presented similar footprint areas to control mice from the beginning to the end of the study. Poulet et al. 38 reported similar results in a mechanical loading model of OA, where they found only contralateral gait modifications, especially an increase in the footprint area. This is of particular interest because they obtained similar results with a different OA model and the use of a treadmill system of gait analysis, suggesting that contralateral footprint areas may be a gait parameter of interest to study in unilateral OA. However, these findings may be astonishing especially because no other modification was found on the contralateral side and the front paws. This may result from a lack of power in the study due to the low number of mice, which do not allow to show a modification of the operated side compared with controls. However, the intragroup analysis (Fig. S4) showed a difference between the 2 legs in the unilateral DMM group, from the first week to the end of the study. Moreover, on an observational point of view (Fig. S4), delta between left and right paw print area seemed to globally increase until week 7 and then seemed stabilized. We can assume that this absence of obvious changes from the seventh week (week 14 excepted) may result from adaptation phenomena despite an increase in OA severity. Unfortunately, this contralateral parameter is not always mentioned in OA surgical models using CatWalkTM XT for gait analysis.25,30,33 In this study, the bilateral DMM group exhibited fewer gait modifications than the unilateral DMM group. This closer gait behavior observed between the control and bilateral group could be due to an inability to compensate with a healthy side, leading to a more symmetrical walking in the bilateral DMM group. This observation may be correlated to the increase in subchondral bone thickness, which was observed only in unilateral DMM, whereas no difference was noted in bilateral DMM. In human gait analysis, Creaby et al. 39 showed that in patients with knee OA, those with unilateral knee pain and either unilateral or bilateral structural medial knee OA demonstrate disturbances in knee biomechanics while walking. Interestingly, patients with bilateral pain and structural disease exhibited symmetrical walking, close to healthy controls. The conclusion was that gait disturbance was associated with the presence of unilateral pain and not with structural changes. This strongly suggests that pain in OA mice likely deserves to be assessed in parallel to gait modification to determine whether both parameters may be correlated.
According to our results and those of the literature, gait analysis systems allow highlighting some gait disturbances in surgically induced OA animal models. However, as already mentioned, high variability and heterogeneity between studies are observed.8,25,29,34,37 Hypotheses may be drawn to explain such variability. First, results may depend on the animal model chosen; mice and rats may have a different behavior after a similar intervention. Second, while DMM, ACLT, or MMT seems to provide comparable histological and radiological OA lesions, we cannot rule out the possibility that rodents present different gait disturbance. Third, the different types of gait analysis systems used appear as a potential issue to compare results from different studies. Indeed, we have already noticed that a free walking system would not provide the same results as a treadmill system which imposed the speed. 32 Besides, the reproducibility between 2 different systems using both treadmills appears poor in murine model. 40 Finally, variability in results could also be the consequence of the gait analysis protocol. Although some studies use similar murine models and identical gait analysis systems, the delay and the frequency of analyses may vary from a daily analysis for a short period to a weekly or monthly analysis of up to 6 months after surgery. These variabilities in the timeline of analysis may also have an impact on animal behavior due to differences in gait training. Moreover, the progression of the OA is compared at a different time of development.29-31,33 From our point of view, due to the problems of comparability, studies should provide control groups with standardized gait analysis protocol to limit misinterpretations.
Our study has several limitations. First, we did not use the sham surgery group, which might have been interesting to explain the gait modifications observed in the first week after surgery. Second, CatWalk can provide a lot of parameters, more than 30 per paw, and we have chosen to restrain our analyses to 7 of them, so theoretically, we may have missed significant changes in mice. Yet, we have chosen the most commonly used and recommended CatWalk parameters 25 to allow us comparisons with previous studies. Third, it has previously been shown that some of the gait modifications observed in mice could be related to pain. This parameter, not assessed in this study, deserves to be further investigated in the pain-related model of murine OA. Fourth, our results showed a highly significant increase in the speed in the control group, and then in all groups over the 16 weeks of the protocol. Although we had performed a 2-week training period for the 3 groups before the beginning of the walking assessment, we cannot exclude a learning effect in our mice. We could therefore discuss the interest of a longer training period to better prevent this effect. Finally, our findings may also be limited due to the relatively low number of mice, which may have been responsible for a lack of power. However, considering the logistical complexity of CatWalk analysis, an increase in the number of animals would have made the study complicated to perform in practice.
This study shows that both the unilateral and the bilateral DMM mouse models provided histological and radiological OA lesions. Bilateral DMM exhibited no gait modification excepted at an early time point after surgery; it could be due either to a better gait adaptation in case of symmetrical OA or to an impossibility to privilege an unpainful side. Unilateral DMM was responsible for a gait disturbance, mainly on the contralateral side, and restricted to few parameters. These changes were relatively stable according to time, which prevents the longitudinal assessment of unilateral OA with gait analysis. Further studies are needed to better define the place of the CatWalk in the evaluation of mouse models of OA.
Supplemental Material
sj-pdf-5-car-10.1177_19476035211053821 – Supplemental material for Posttraumatic Osteoarthritis Damage in Mice: From Histological and Micro–Computed Tomodensitometric Changes to Gait Disturbance
Supplemental material, sj-pdf-5-car-10.1177_19476035211053821 for Posttraumatic Osteoarthritis Damage in Mice: From Histological and Micro–Computed Tomodensitometric Changes to Gait Disturbance by Alban Fouasson-Chailloux, Marc Dauty, Benoit Bodic, Martial Masson, Yves Maugars, Benoit Metayer, Joëlle Veziers, Julie Lesoeur, François Rannou, Jérôme Guicheux and Claire Vinatier in CARTILAGE
Supplemental Material
sj-pptx-1-car-10.1177_19476035211053821 – Supplemental material for Posttraumatic Osteoarthritis Damage in Mice: From Histological and Micro–Computed Tomodensitometric Changes to Gait Disturbance
Supplemental material, sj-pptx-1-car-10.1177_19476035211053821 for Posttraumatic Osteoarthritis Damage in Mice: From Histological and Micro–Computed Tomodensitometric Changes to Gait Disturbance by Alban Fouasson-Chailloux, Marc Dauty, Benoit Bodic, Martial Masson, Yves Maugars, Benoit Metayer, Joëlle Veziers, Julie Lesoeur, François Rannou, Jérôme Guicheux and Claire Vinatier in CARTILAGE
Supplemental Material
sj-pptx-2-car-10.1177_19476035211053821 – Supplemental material for Posttraumatic Osteoarthritis Damage in Mice: From Histological and Micro–Computed Tomodensitometric Changes to Gait Disturbance
Supplemental material, sj-pptx-2-car-10.1177_19476035211053821 for Posttraumatic Osteoarthritis Damage in Mice: From Histological and Micro–Computed Tomodensitometric Changes to Gait Disturbance by Alban Fouasson-Chailloux, Marc Dauty, Benoit Bodic, Martial Masson, Yves Maugars, Benoit Metayer, Joëlle Veziers, Julie Lesoeur, François Rannou, Jérôme Guicheux and Claire Vinatier in CARTILAGE
Supplemental Material
sj-pptx-3-car-10.1177_19476035211053821 – Supplemental material for Posttraumatic Osteoarthritis Damage in Mice: From Histological and Micro–Computed Tomodensitometric Changes to Gait Disturbance
Supplemental material, sj-pptx-3-car-10.1177_19476035211053821 for Posttraumatic Osteoarthritis Damage in Mice: From Histological and Micro–Computed Tomodensitometric Changes to Gait Disturbance by Alban Fouasson-Chailloux, Marc Dauty, Benoit Bodic, Martial Masson, Yves Maugars, Benoit Metayer, Joëlle Veziers, Julie Lesoeur, François Rannou, Jérôme Guicheux and Claire Vinatier in CARTILAGE
Supplemental Material
sj-pptx-4-car-10.1177_19476035211053821 – Supplemental material for Posttraumatic Osteoarthritis Damage in Mice: From Histological and Micro–Computed Tomodensitometric Changes to Gait Disturbance
Supplemental material, sj-pptx-4-car-10.1177_19476035211053821 for Posttraumatic Osteoarthritis Damage in Mice: From Histological and Micro–Computed Tomodensitometric Changes to Gait Disturbance by Alban Fouasson-Chailloux, Marc Dauty, Benoit Bodic, Martial Masson, Yves Maugars, Benoit Metayer, Joëlle Veziers, Julie Lesoeur, François Rannou, Jérôme Guicheux and Claire Vinatier in CARTILAGE
Footnotes
Supplemental Material
Acknowledgments and Funding
The authors thank the SFR Bonamy animal facility “UTE Medecine” Nantes University. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the French network “Research on OsteoArthritis Disease” (ROAD network), the ANR projects KLOTHOA and PPAROA, and “Agence de la Biomedecine” project 18GREFFE012.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All the animal experiments were performed according to the European directive 2010/63/EU governing ethic and regulations in animal experiments. These experiments have been approved under the project N°2015033111162660. This work adheres to the ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines 2.0.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
