Abstract
Objective
Early intervention therapies targeting inflammation and cell death during the acute phase of cartilage injury have the potential to prevent posttraumatic osteoarthritis. The objective of this study was to investigate the effects of interleukin receptor antagonist protein (IRAP), hyaluronan (HA), dexamethasone (DEX), and mesenchymal stem cell (MSC) treatment on the expression of established genetic markers for matrix degradation, apoptosis, and inflammation in articular cartilage during the acute phase of injury.
Design
A custom impact device was used to create replicable injury ex vivo to intact porcine knee joint. One hour after impact, IRAP, HA, DEX, or MSCs was intra-articularly injected. At 8 hours postinjury, cartilage and meniscus samples were harvested for genetic expression analysis. Expression of miR-27b, miR-140, miR-125b, miR-16, miR-34a, miR-146a, miR-22, ADAMTS-4, ADAMTS-5, MMP-3, IL-1β, and TNF-α was analyzed by real-time polymerase chain reaction.
Results
At 8 hours postinjury, expression of ADAMTS-4, ADAMTS-5, MMP-3, IL-1β, and TNF-α in cartilage was significantly decreased in IRAP- and DEX-treated joints as compared to nontreated injured joints, whereas only IRAP upregulated expression of miR-140, miR-125b, miR-27b, miR-146a, and miR-22 in cartilage. HA and MSC treatments had no significant effects on catabolic and inflammatory gene expression in cartilage. However, HA treatment significantly upregulated expression of all miRNAs except miR-16. In addition, the treatments tested also exhibited significant influences on meniscus.
Conclusions
This study provides a valuable starting point for further research into potential targets for and efficacy of various early intervention strategies that may delay or prevent the progression of posttraumatic osteoarthritis after acute cartilage injury.
Introduction
Knee injury is the leading cause of posttraumatic osteoarthritis (PTOA) in young people and can occur as early as 10 years following a traumatic injury. 1 PTOA accounts for up to 5.6 million per year, or 12%, of the total cases of symptomatic OA in the United States. 1 The incidence of PTOA is expected to increase as the number of traumatic injuries affecting young adults rises from increased rates of participation in sports and tendency toward heavier players. 2 Current therapy for PTOA is similar to that of primary OA, with optimal management requiring a combination of nonpharmacological and pharmacological modalities. 3 The limitation of these modalities is that they focus on management of the disease and do not attempt to correct the underlying pathology, 4 resulting in continued disease progression. Unlike primary OA, PTOA has a known starting point of disease, namely, the injury event, which uniquely represents an opportunity to prevent or delay the onset of PTOA by providing proper treatments during the early phase of injury.
The acute phase of cartilage injury is characterized by increased inflammation and chondrocyte death within the first few hours of injury.5,6 The increase in inflammatory cytokines has been shown to promote production of catabolic enzymes and apoptotic activity.7,8 Recent studies have shown that “superhealer” mice, which exhibit decreased levels of inflammatory cytokines interleukin (IL)-1β and tumor necrosis factor (TNF)-α, were protected from loss of cartilage and the development of PTOA after acute cartilage injury.9,10 Therefore, early inhibition of inflammation could be a potential intervention strategy for the prevention of PTOA development after traumatic cartilage injury. 5
Several current clinical OA treatments have been shown to exhibit anti-inflammatory effects. For instance, in addition to immunosuppressive and chondroprotective characteristics of mesenchymal stem cells (MSCs), 11 intra-articular MSC injection in large animal OA models reduced joint inflammation.12,13 Previous studies have shown that intra-articular injection of interleukin receptor antagonist protein (IRAP), a naturally occurring anti-inflammatory cytokine, decreases catabolic activities and joint discomfort in established OA models 14 and reduces cartilage degeneration and synovial inflammation in a mouse articular cartilage fracture model.15,16 Intra-articular injections of dexamethasone (DEX), a potent anti-inflammatory corticosteroid,17,18 have been shown to prevent aggrecan degradation and decrease joint inflammation.19,20 High-molecular-weight hyaluronan (HA), the principal glycosaminoglycan in synovial fluid that is responsible for viscosity and lubrication within the joint, 21 has been used as an alternative for the treatment of knee OA pain with beneficial chondroprotective effects. 22 High-molecular-weight HA suppresses synovial membrane IL-1β production 23 and reduces matrix metalloproteinases (MMP)-13 production in IL-1-treated chondrocytes. 24 However, use of these current clinical OA treatments has yet to be evaluated as early intervention strategies to prevent cartilage degeneration and PTOA development after acute cartilage injury.
MicroRNAs (miRNAs) are 20- to 22-nucleotide, noncoding RNA molecules that function at the post-transcriptional level to negatively regulate gene expression in various cellular processes. 25 Dysregulation of several miRNAs has been associated with the development and progression of OA or its symptoms in regard to cartilage degeneration (miR-140, 26 125b, 27 miR-27b 8 ), apoptosis (miR-16, 28 miR-34a 7 ), and inflammation (miR-22, 29 miR-146a 30 ). Aggrecanase-mediated matrix degradation is one of the first processes expressed during the acute phase of cartilage injury, as well as early stage OA. 31 Aggrecanases ADAMTS-4 and ADAMTS-5 are directly regulated by miR-125b 27 and miR-140, 26 respectively. Expression of miR-146 was found to be downregulated in OA cartilage, 32 acting as a negative feedback regulator in the inflammatory response. 33 Downregulation of miR-27b expression by IL-1β treatment was found to correlate with upregulation of MMP-13 in human OA chondrocytes. 8 MicroRNA-22 negatively regulates the proteins BMP-7 and PPARA, which regulate IL-1β and MMP-13 expression. 29 Previous studies reported that expression of miR-16 and miR-34a was upregulated in human OA cartilage.32,34 Gene expression of MMPs and aggrecanases can be upregulated by miR-16, 34 while silencing of miR-34a reduced IL-1β-induced chondrocyte apoptosis. 7 Upregulation of miR-16 28 and miR-34a 35 has also been associated with increased cellular apoptosis. Although miRNAs related to cartilage degeneration, apoptosis, and inflammation could be treatment targets in PTOA, their expression profiles have not been elaborated in cartilage injury. Therefore, using the ex vivo intact joint injury model, 6 the objective of this study was to investigate the effect of IRAP, HA, DEX, and MSC treatments as early intervention strategies on the established genetic markers for matrix degradation, apoptosis, and inflammation in articular cartilage during the acute phase of injury.
Materials and Methods
Injury and Administration of Treatment
The procedure described previously was used to injure the joint.
6
In brief, porcine knees from 35- to 40-kg pigs were obtained from the University of Miami Department of Surgery Tissue Sharing program (Institutional Animal Care and Use Committee approved source) within 2 hours of death. Impact injury was caused by dropping a 10-kg weight one time from 1 m directly above the knee in extension using a custom impact device.
6
One hour after impact, 20 µg of recombinant human IRAP (Peprotech; Rocky Hill, NJ), 15 mg (MW 1.5-2.2 MDa) sodium hyaluronate (HA; Sigma, St. Louis, MO), 5 × 106 porcine MSCs, or 4 mg DEX (Sigma) in 1 mL of phosphate-buffered saline (PBS) was administered to injured knee via intra-articular injection (
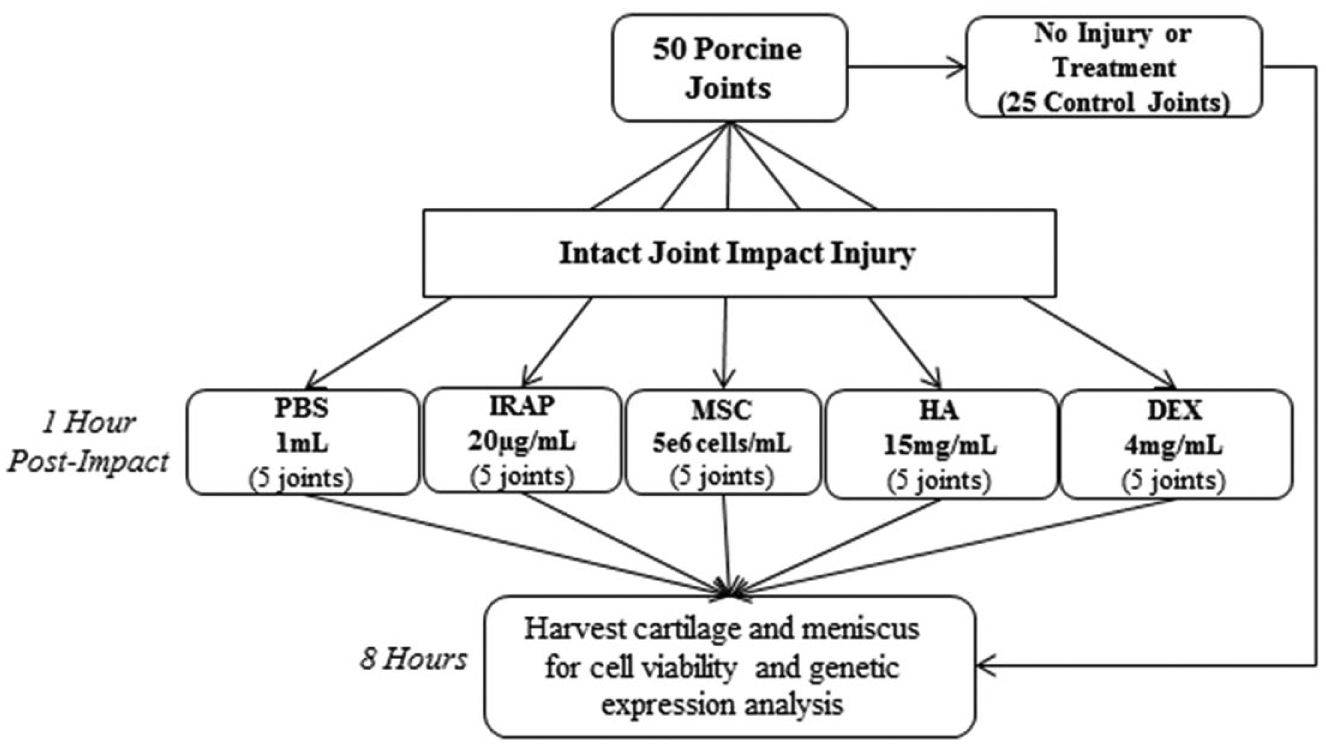
Schematic of study design and groups. PBS = phosphate-buffered saline; IRAP = interleukin receptor antagonist protein; MSC = mesenchymal stem cells; HA = hyaluronan; DEX = dexamethasone.
The contralateral joint of the same pig served as the control for each treatment and were administered 1 mL PBS in the same manner previously described. 6 Following the injection, the joint was repeatedly flexed and extended to disperse the suspension throughout the intra-articular space. At 8 hours after injury, cartilage samples from the impacted regions on femoral condyles and meniscus from both groups were harvested, flash-frozen, and stored at −80°C for analyses of miRNA and mRNA expression. Because the intact joint model used also allows us to examine other tissues in the joint, tissue samples were also harvested from the impact regions of menisci and examined for the mRNA and miRNA expression at 8 hours after injury
Analysis of Chondrocyte Viability
Cartilage samples from control and treatment joints were stained for cell viability at the time of harvest as previously described. 6 In brief, after exposing the joint capsule medially and laterally, the contact regions of the medial and lateral femoral condyle with the tibial plateau were identified with the knee being positioned in the impact condition. Two 6-mm cylindrical samples covering the contact region of each condyle were punched from the control and treatment joints. A 100-µm-thick full-depth sample was prepared and incubated in PBS containing 1 µmol/L ethidium homodimer-1 and 1 µmol/L calcein AM from the Live/Dead Viability/Cytotoxicity Kit (Invitrogen, Carlsbad, CA). Staining was visualized on an inverted fluorescent microscope with 495 nm/515 nm excitation/emission for calcein (live cells) and 495 nm/635 nm excitation/emission for ethidium homodimer (dead cells).
Image analysis was conducted to determine the number of live and dead. 6 A defined region of interest was identified (1.11 mm wide × 0.88 mm deep at the center of the sample, measured from the articular surface) from each image taken through a 10× objective lens utilizing MATLAB R2014a (MathWorks Natick, MA). The resulting images were analyzed with ImageJ software and the cell viability (= the number of live cells/the total number of cells × 100%) was calculated.
Analyses of mRNA and miRNA Expression
Established genetic markers for matrix degradation, apoptosis, and inflammation were used for treatment evaluation, including ADAMTS-4, ADAMTS-5, MMP-3, IL-1β, TNF-α, miR-140, miR-125b, miR-22, miR-34a, miR-16, miR-27b, and miR-146a. These specific genes were chosen because they are known to be associated with OA development and/or its symptoms.7,8,26-30
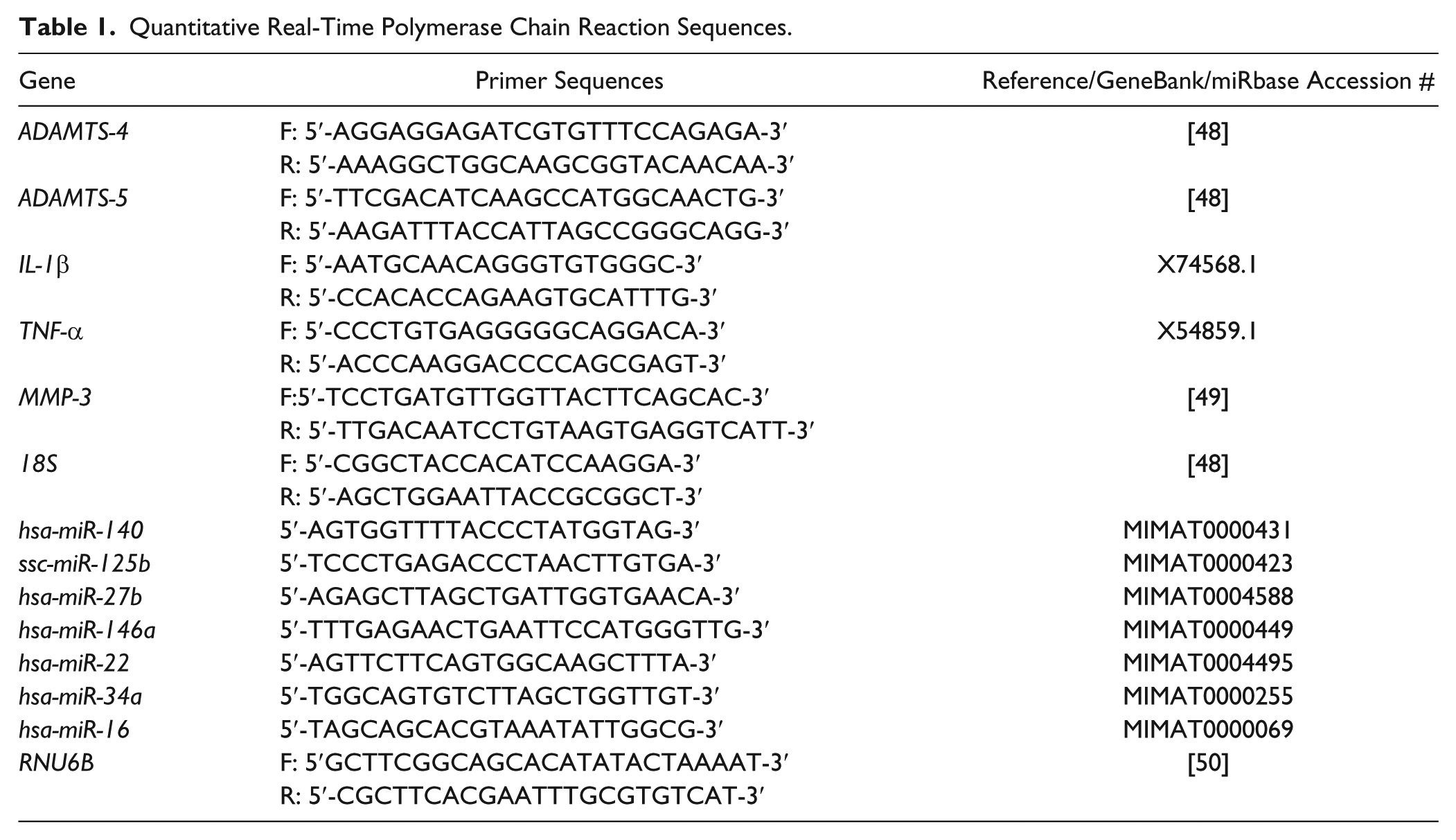
Cartilage tissue adjacent to the areas used for cell viability staining was processed for analyses of mRNA and miRNA expression. 6 For meniscus, tissue samples from the impacted region were used for analyses. Total RNA was isolated using the guanidinium thiocyanate-phenol-chloroform extraction method. 39 For mRNA expression analyses, the High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Carlsbad, CA) was used to synthesize cDNA from 0.25 µg of total RNA following manufacturer’s instructions. For miRNA expression analyses, the qScript microRNA cDNA synthesis kit (Quanta Biosciences, Beverly, MA) was used to polyadenylate miRNAs and synthesize cDNA from 0.1 µg of total RNA following manufacturer’s instructions. Gene expression was analyzed using SteponePlus (Applied Biosystems) quantitative real-time polymerase chain reaction (PCR) system. Ribosomal 18S has been shown to be a stable internal control for gene expression analysis of articular cartilage, 40 while RNU6B has been used as an internal control for miRNA expression in previous cartilage studies.8,27 Thus, expression of ribosomal 18S and RNU6B were used as endogenous controls for mRNA and miRNA, respectively. Primer sequences are listed in Table 1 . Quantification of expression levels was determined by the 2−ΔΔCt method and normalized to expression levels of respective control leg. Stability of 18S and RNU6B in cartilage and meniscus after injury was examined by a comparison of cycle threshold (CT) number between control and injured samples.
Quantitative Real-Time Polymerase Chain Reaction Sequences.
Statistical Analysis
In this study, we analyzed 25 control joints, 5 injury joints without treatments, 5 IRAP-treated joints, 5 MSC-treated joints, 5 DEX-treated joints, and 5 HA-treated joints. Comparison of control, injury, and treatment groups at 8 hours postinjury was performed by a one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test (Graphpad Prism 6). Data are represented as mean ± standard error (SE). The P value <0.05 was considered significant.
Results
Chondrocyte Viability after Impact Injury and Treatment
At 8 hours postinjury, chondrocyte viability analysis showed significantly decreased viability in the injury group as compared to the control group (P = 0.034) (
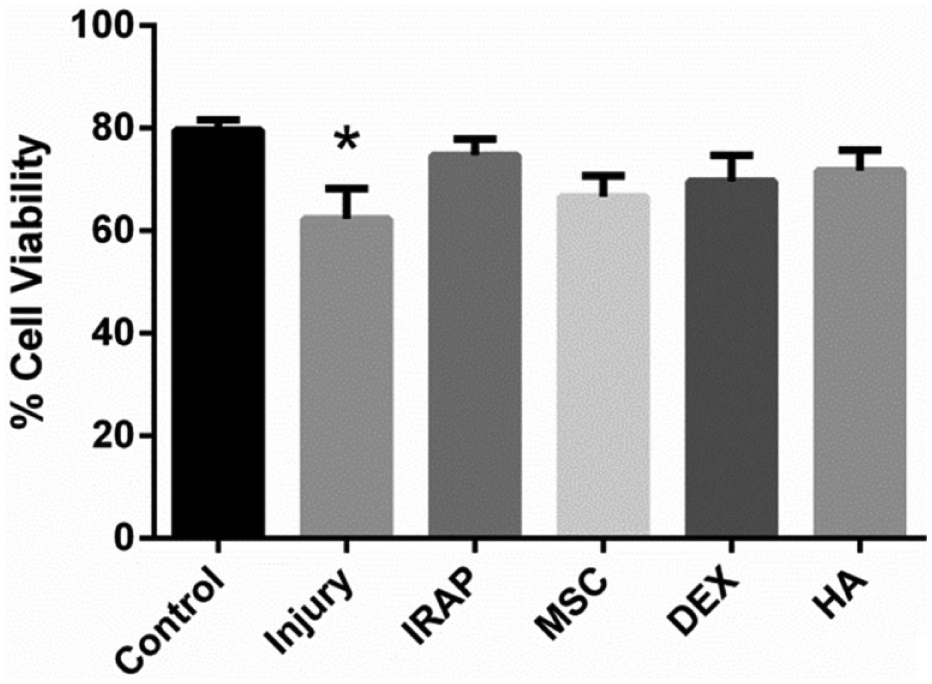
Quantified cell viability of cartilage at 8 hours postimpact and treatment. *Indicates significant to control P < 0.05.
Effects of the Treatments on mRNA and miRNA Expression in Articular Cartilage
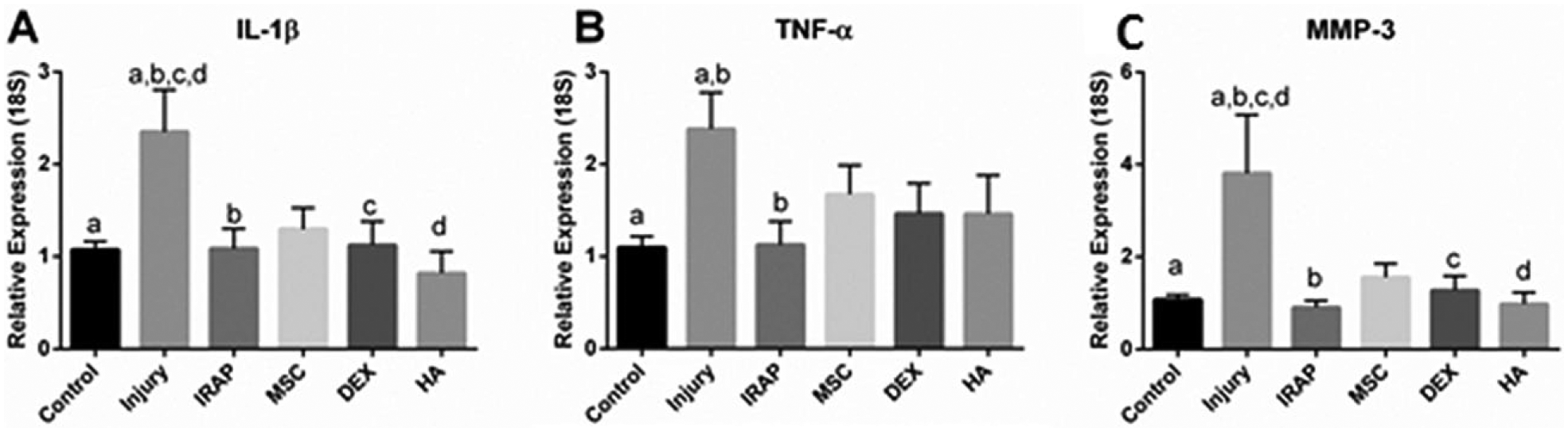
The IRAP and DEX treatments administered 1 hour after impact injury significantly prevented upregulation of catabolic enzymes (i.e., ADAMTS-4, ADAMTS-5, and MMP-3) and pro-inflammatory cytokines (i.e., IL-1β, TNF-α) in cartilage (
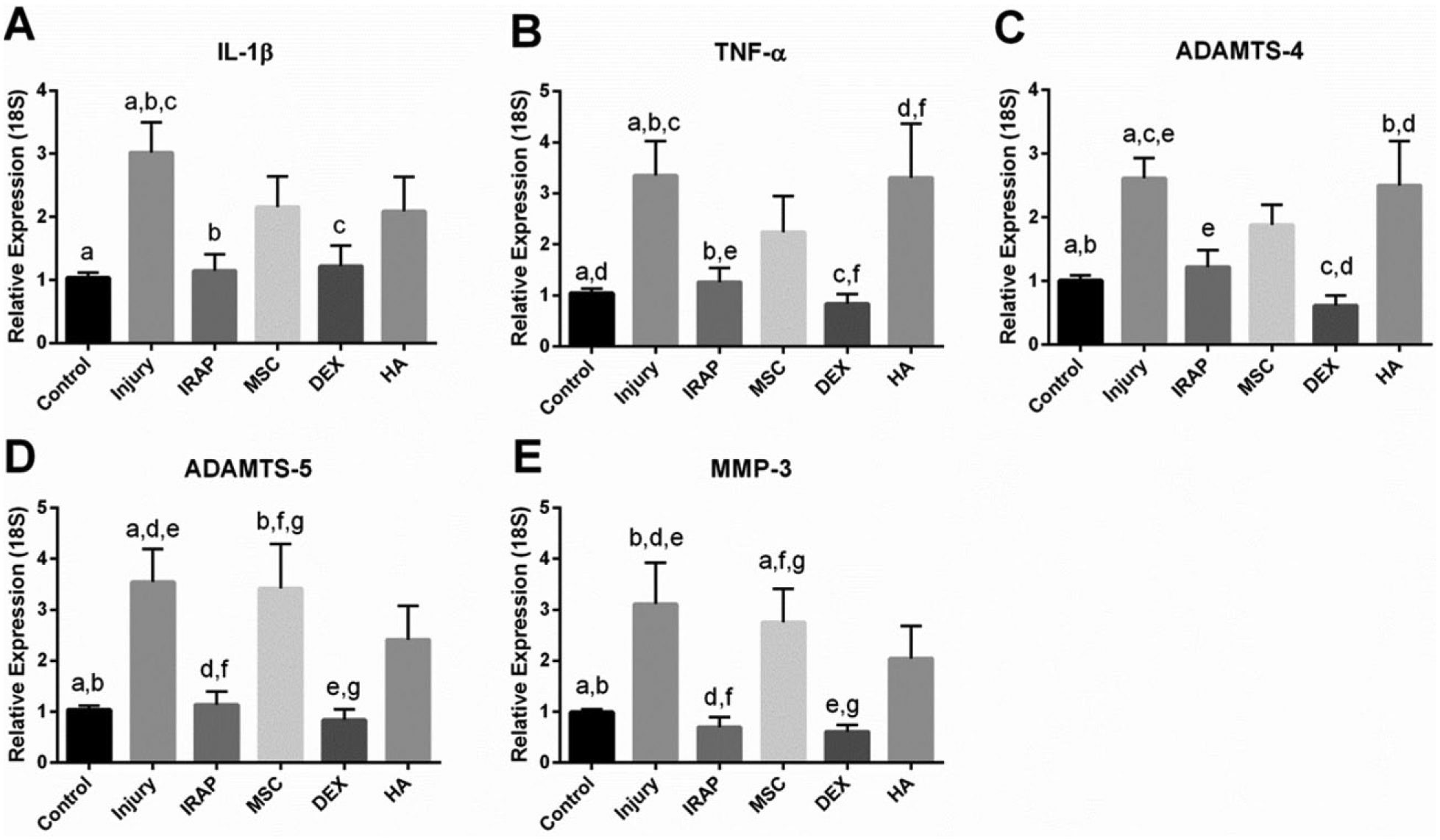
Relative expression of (
Expression of miR-140, miR125b, miR-27b, miR-22 in cartilage was significantly upregulated by the IRAP and HA treatments as compared to the control and injury groups (P < 0.001) (
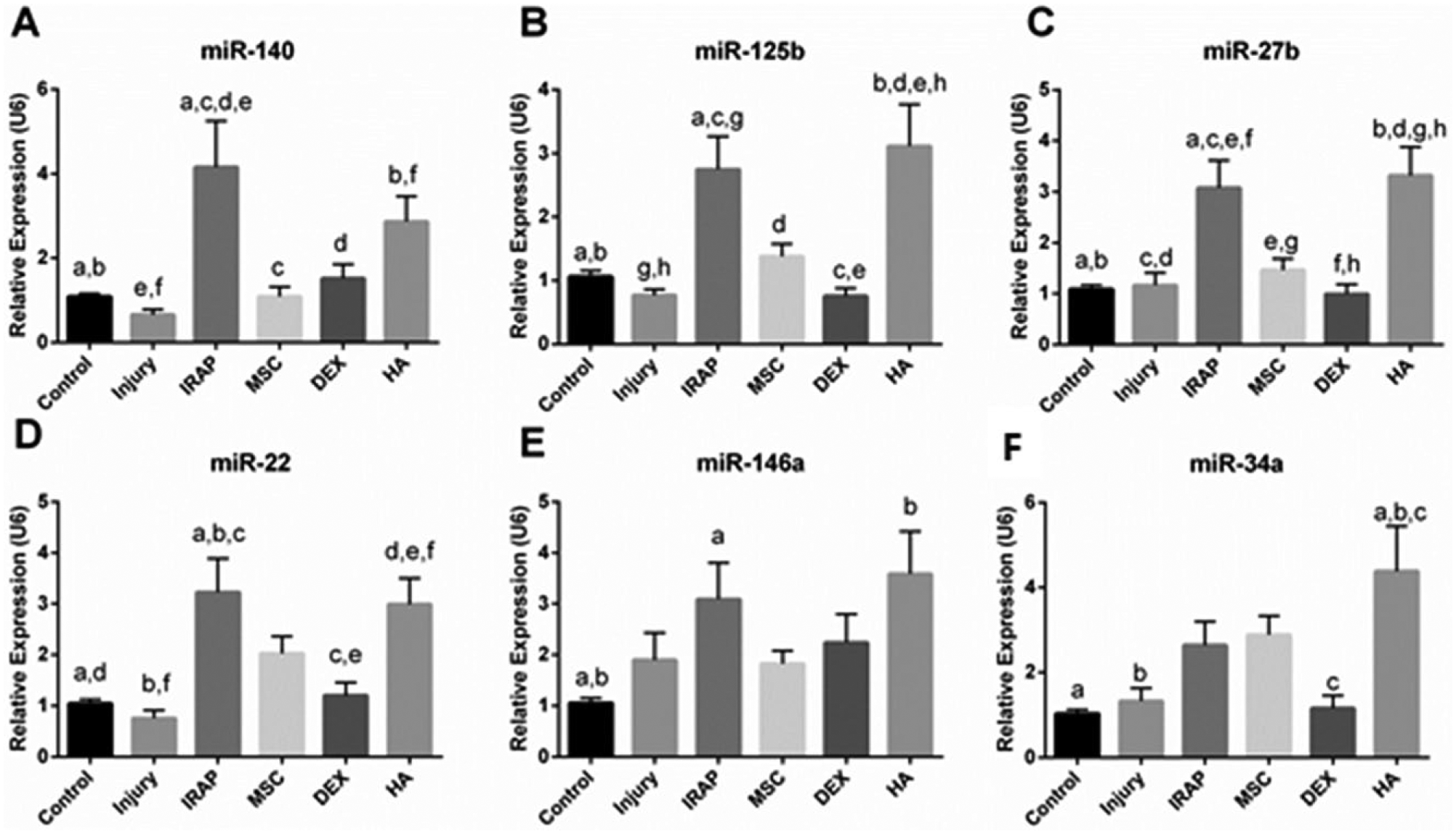
Relative expression of (
Effects of the Treatments on mRNA and miRNA Expression in Meniscus
After impact injury, expression of MMP-3, IL-1β, and TNF-α was significantly upregulated in menisci without treatments (P = 0.002, 0.001, and 0.02, respectively) (
Relative expression of (
The expression of miR-140 in menisci was significantly increased in the DEX treatment group (P < 0.001) as compared to the control and injury groups (
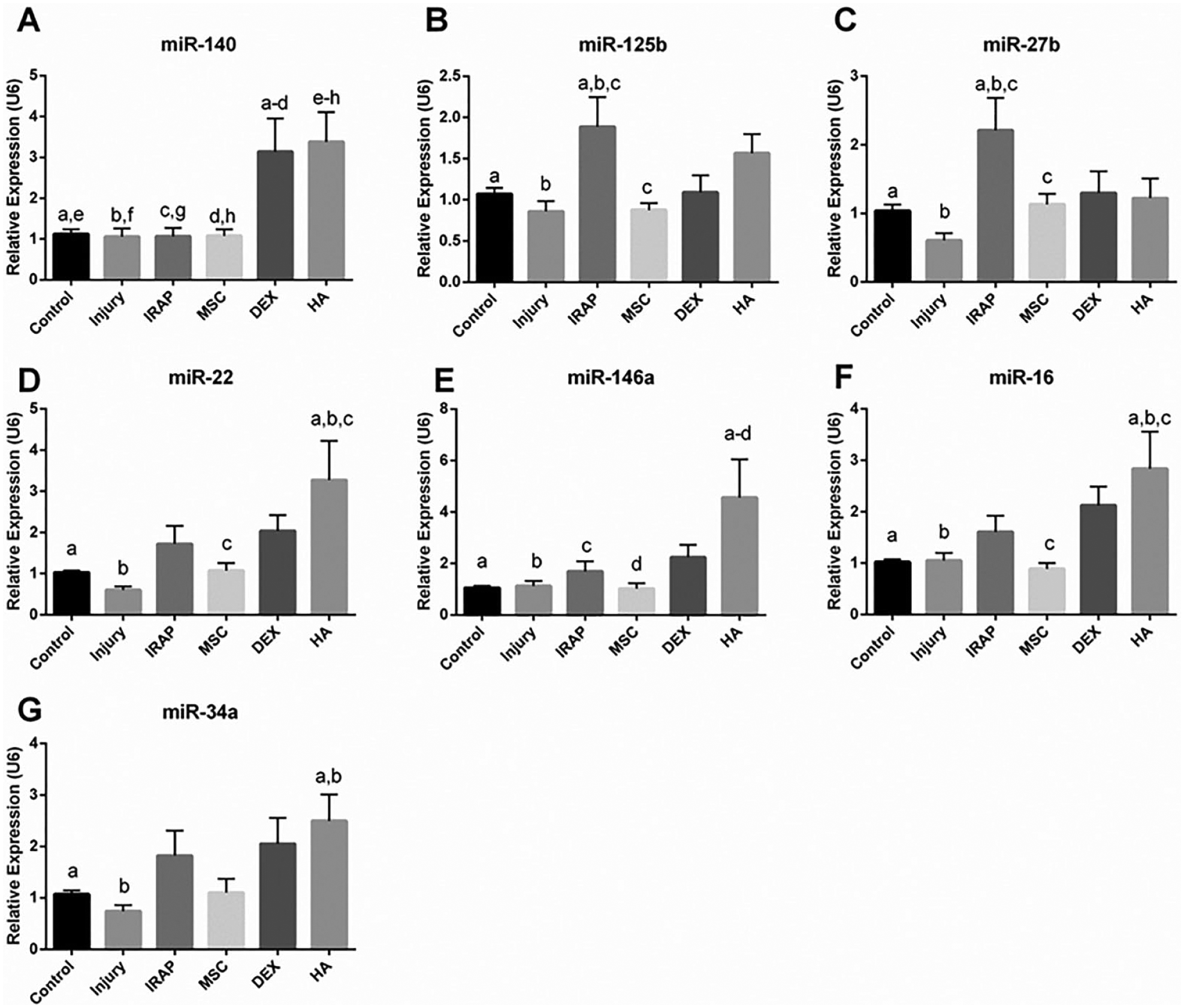
Relative expression of (
Discussion
The goal of this study was to evaluate the intra-articular injections of IRAP, MSC, HA, and DEX as potential early intervention strategies for acute cartilage injury by examining their effects on the expression of the miRNAs and mRNAs that are related to matrix degradation and inflammation. During the acute phase of cartilage injury, there is increased production of pro-inflammatory cytokines (i.e., IL-1β and TNF-α) within the tissue, 15 which may lead to apoptosis and extracellular matrix breakdown. In this study, the expression of inflammatory (IL-1β and TNF-α) and catabolic (ADAMTS-4, ADAMTS-5, and MMP-3) genes was upregulated in articular cartilage at 8 hours after injury. The intra-articular injections of IRAP and DEX given 1 hour after injury were able to prevent upregulation of inflammatory and catabolic gene expression in injured cartilage. This finding suggests that intra-articular injection of clinically used OA treatment has potential as an early intervention strategy for acute cartilage injury. The treatment groups were also found to exhibit different effects on the expression of mRNAs and miRNAs examined, indicating differences in effectiveness and molecular mechanism among the treatments evaluated in this study. This is the first study to report the effects of clinically used OA treatments on the expression of inflammatory and catabolic mRNAs and miRNAs at the acute phase of cartilage injury.
The intra-articular injection of IRAP prevented upregulation of aggrecanase gene expression (ADAMTS-4 and ADAMTS-5), while expression of miR-146a, miR125b, miR-27b, and miR-22 was promoted by the IRAP treatment. Since miR-125b and miR-140 regulate aggrecanases (ADAMTS-4 and ADAMTS-5),26,27 this result suggests that the IRAP treatment can downregulate the expression of ADAMTS-4 and ADAMTS-5 by upregulating the expression of miR-125b and miR-140. Additionally, since IL-1β-induced production of TNF-α was previously shown to be reduced by miR-146 overexpression, 32 upregulation of miR-146a expression by IRAP treatment suggests that IRAP may reduce TNF-α expression via upregulation of miR-146. Furthermore, previous studies have shown that overexpression of miR-27b downregulates expression of MMP-13 in human OA cartilage, 8 whereas overexpression of miR-22 exhibits an opposite effect. 29 Upregulation of miR-22 has also been associated with an upregulation of IL-1β. 29 Although miR-22 expression was upregulated by the IRAP treatment, since upregulation of IL-1β was not observed and MMP-13 expression was not detected in the IRAP group at 8 hours after injury, the effects of miR-22 may have been diminished by other factors, such as miR-27b. However, future in vivo studies are required to determine the long-term effects of IRAP treatment on acutely injured cartilage.
As a corticosteroid, DEX passes freely through the cell membrane and binds to an intracellular glucocorticoid receptor, which directly inhibits the upregulation of inflammatory cytokines at the transcriptional level.17,18 For this reason, the upregulation of aggrecanases and inflammatory cytokines may have been inhibited by the DEX treatment without modulating expression of the miRNAs examined in this study. While previous in vivo animal models of PTOA created visible cartilage lesions by fracture or drilling, administration of IRAP or DEX immediately after injury (<1 hour) effectively prevented the development of OA.15,20 Therefore, the inhibition of degenerative enzyme and inflammatory cytokine expression after IRAP and DEX treatment during the acute phase of injury supports the potential of early intervention to prevent OA development after cartilage injury. Nonetheless, the findings from IRAP and DEX with this injury model need to be confirmed and expanded upon in longitudinal in vivo studies. Additional studies with varying doses of treatments administered after injury are needed to determine optimal doses for long-term effect in vivo.
Intra-articular injection of high-molecular-weight (HA (>1 × 106 Da) is widely accepted as beneficial for viscosupplementation in OA joints or acutely injured cartilage. 22 HA has been shown to elicit a cellular response by binding to specific receptors, such as cluster determinant (CD)-44 and receptor for hyaluronate-mediated motility (RHAMM), which are expressed by many cells including synoviocytes and chondrocytes.41,42 Studies have shown that high-molecular-weight HA suppresses synovial membrane IL-1β production 23 and reduces MMP-13 production in IL-1-treated chondrocytes by acting on the CD-44 receptor. 24 It is theorized that high-molecular-weight HA binds to the CD-44–RHAMM complex and prevents receptor cross-linking and adverse cellular effects in chondrocytes, such as activation of the nuclear factor (NF)-κβ and p38 pathways.24,43 In this study, HA had no significant effects on the aggrecanase and inflammatory cytokine expression in injured cartilage, indicating that HA suppression of IL-1β by may not have taken effect at 8 hours after injury. It could be due to the slow transport of large molecules, like HA, into injured cartilage. However, HA significantly upregulated expression of miR-125b, miR-27b, miR-140, and miR-146a in cartilage, suggesting an attempt to downregulate the increased catabolic activity after injury.8,26,27,30 HA treatment also upregulated expression of miR-22 and miR-34a, which have been associated with an increase in IL-1β production 29 and apoptotic activity.7,44 While no significant decrease in cell viability was observed after HA treatment at 8 hours, future studies are still required to determine the long-term effects of the upregulation of miR-22 and miR-34a by HA treatment.
Previous in vivo studies have reported that the beneficial effects of MSC treatment were seen as early as day 2 of treatment after cartilage injury due to the production and release of anti-inflammatory cytokines. 11 However, no effects of MSCs were observed in this study. Potential factors affecting the outcome of the MSC treatment include ineffective number of cells administered or too short of a time point (8 hours) for the MSCs to produce anti-inflammatory proteins at an effective concentration. While the number of MSCs used for treatment in this study (5 × 106 cells) was based on the successful inhibition of OA progression, other animal models have reported beneficial therapeutic effects for OA using up to 107 cells 13 and as little as 106 cells, 45 each with end time points greater than 6 weeks. Therefore, further studies investigating the optimal dosage for MSC treatment as an early intervention strategy for acute cartilage injury are still required.
The intact joint model used in this study allows us to examine the response of meniscus to impact force. Expression of MMP-3, IL-1β, and TNF-α was significantly upregulated after impact to meniscus tissue, suggesting that meniscus also exhibited a similar injury response as cartilage. All treatments when administered 1 hour after injury tended to result in lower inflammatory cytokine and MMP-3 expression in meniscus at 8 hours after impact, indicating that meniscus can also benefit from the treatments for injured cartilage. It is known that meniscus and cartilage distribute loads differently under impact due to inherent structural and material property differences. 46 Slight differences observed in miRNA and mRNA expression between cartilage and meniscus could be due to different mechanical and genetic properties, which could result in different mechanobiological responses from cartilage and meniscus tissues.
A potential limitation of this study is the lack of dosing studies performed. While the dosage of each treatment used in this study was based on previous successful in vivo and clinical studies, future studies using an in vivo model are necessary to identify appropriate dosing and timing protocols for the administration of the early intervention treatments. This study also used the ex vivo model of cartilage injury, which is limited in its ability to perform long-term studies. Since the studies were conducted within the first 8 to 10 hours of death, minimum effect on articular and meniscal chondrocyte viability was expected due to lack of continuous nutrient supply. 47 Another limitation of this study is absence of a confirmation of gene expression data at protein level. Since this study was focused on the expression of miRNAs and their corresponding mRNAs in cartilage and meniscus tissues in response to treatments after acute injury, the mRNA expression was not confirmed at the protein level. Last, gene expression of other relevant knee joint tissues that may be affected in an acute knee injury and subsequent treatment, such as the synovial membrane and fat pad tissues, were not included in this study. However, further research is needed to determine the effect of early intervention strategies on these surrounding tissues.
Despite these limitations, the ex vivo injury and treatment model allowed for the comparison of 4 potential early intervention therapies for acute cartilage injury. Treatment with either IRAP or DEX prevented upregulation of pro-inflammatory cytokine and aggrecanase expressions in acutely injured cartilage at 8 hours. Additionally, each treatment elicited a unique response in miRNA and mRNA expression, suggesting that each treatment has its own specific mechanism of action. Furthermore, the data suggest that changes in miRNA expression caused by treatment may give insight into the potential long-term outcome of that treatment. Currently, there is no known treatment for the prevention of PTOA. This study provides a valuable starting point for further research into potential targets for and efficacy of various early intervention strategies that may delay or prevent the progression of PTOA after acute cartilage injury.
Footnotes
Acknowledgments and Funding
We would like to thank Dr. Joshua Hare in the Interdisciplinary Stem Cell Institute at the University of Miami Miller School of Medicine for providing stem cells. This study was supported by a gift donation from Paul DiMare to the University of Miami Department of Orthopedics Division of Sports Medicine.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study because tissue samples were obtained from the University of Miami Department of Surgery Tissue Sharing program and no live animals were involved in this study.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no live animals were involved in this study.
Supplemental Material:
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
