Abstract
Objective:
The knowledge about functions of caspases, usually associated with cell death and inflammation, keeps expanding also regarding cartilage. Active caspases are present in the growth plate, and caspase inhibition in limb-derived chondroblasts altered the expression of osteogenesis-related genes. Caspase inhibitors were reported to reduce the severity of cartilage lesions in osteoarthritis (OA), and caspase-3 might represent a promising biomarker for OA prognosis. The objective of this investigation was to decipher the transcriptomic regulation of caspase inhibition in chondrogenic cells.
Design:
Limb-derived chondroblasts were cultured in the presence of 2 different inhibitors: Z-VAD-FMK (FMK) and Q-VD-OPH (OPH). A whole transcriptome RNA sequencing was performed as the key analysis.
Results:
The analysis revealed a statistically significant increase in the expression of 252 genes in the FMK samples and 163 genes in the OPH samples compared with controls. Conversely, there was a significant decrease in the expression of 290 genes in the FMK group and 188 in the OPH group. Among the top up- and downregulated genes (more than 10 times changed), almost half of them were associated with OA. Both inhibitors displayed the highest upregulation of the inflammatory chemokine Ccl5, the most downregulated gene was the one for mannose receptors Mrc1.
Conclusions:
The obtained datasets pointed to a significant impact of caspase inhibition on the expression of several chondro-/osteogenesis-related markers in an in vitro model of endochondral ossification. Notably, the list of these genes included some encoding for factors associated with cartilage/bone pathologies such as OA.
Introduction
Functions of caspases are traditionally linked to inflammation and cell death. Knowledge about these processes and the underlying mechanisms progressed during the last decade. 1 This trend applies to cartilage as well. Chondrocyte death by apoptosis and associations with cartilage matrix degradation were reported during cartilage development and homeostasis. 2 Notably, strong correlations exist between chondrocyte apoptosis and pathogenesis, including osteoarthritis (OA). 3 Recently, also other types of cell death such as autophagy were investigated in connection with OA. 4 Notably, the biomarkers of chondrocyte apoptosis and autophagy in OA include cysteine proteases (caspases). 5
Due to the key role of caspases in cell death and inflammation, their pathways have been investigated particularly in pathological processes, and also with respect to therapeutic approaches.6,7 Broad-spectrum caspase inhibitors, small cell-permeable molecules which bind caspase active site and irreversibly block caspase activity, belong to the attractive targets. 6 Caspase inhibitors contain carboxyterminal group: the classical Z-VAD-FMK (FMK) caspase inhibitor includes fluoromethyl ketone, whereas the more effective “new generation” Q-VD-OPH (OPH) inhibitor includes O-phenoxy group.8,9
The therapeutic effects of a broad spectrum of caspase inhibitors were examined in experimentally induced OA in rabbits. In this context, injections of general caspase inhibitor reduced the severity of cartilage lesions in the knee joint. 10 Beneficial effects of intraarticular caspase inhibition therapy were also observed in the case of experimental osteochondral injury. 11 Inhibition of chondrocyte cell death was further performed using the general inhibitor in porcine cartilage explants, 12 or in the case of induced non-apoptotic cell death in the pre-chondrogenic ATDC5 cell line. 13 More recently, the effects of caspase inhibition on cell survival rate were examined in rabbit articular chondrocytes 14 and TMJ chondrocytes. 15 General caspase inhibitor was tested also in engineered cartilage analogues to reduce cell death 16 related to posttraumatic OA. Caspase inhibitors were shown to reduce the severity of cartilage lesions in experimental OA 10 ; however, the specific modifications of gene expression were not investigated.
As apparent from the literature, general caspase inhibitors have been used to prevent cell death and thus to protect chondrocytes and cartilage. However, there is a lack of evidence about their effects beyond apoptosis 1 despite the first reports are emerging.17-19
Therefore, the objective of this investigation was to decipher the transcriptomic regulation of caspase inhibition in chondrogenic cells. General caspase inhibition followed by whole-genome RNA sequencing was performed to test this hypothesis. The effects of Z-VAD-FMK, as the most commonly used broad-spectrum inhibitor, and Q-VD-OPH, as a “new generation” inhibitor, 7 were investigated in parallel.
Material and Methods
Micromass Cultures
Cells for micromass cultures were obtained from mouse forelimbs at embryonic day (E) 12. Fresh post mortem limbs were collected in PSA buffer (Puck’s Saline A liquid) and cut into pieces by a needle. Pieces of tissue were incubated for 1 to 2 hours at 37 °C with Dispase I (Gibco, final activity 1 U/mL) solved in PSA buffer with 10% fetal bovine serum (FBS; Sigma-Aldrich). Dispase I was inactivated by adding culture medium, cells were passed through a 40-µm cell strainer (Corning), centrifuged (100 g/5 minutes), washed with fresh medium, and counted. Subsequently, cell suspension at concentration of 2 × 107/mL were spotted in 10 µL drops on the culture plate and left in 5% CO2/37 °C to adhere. After 60 minutes, spots were covered by a fresh culture medium. Culture medium which supports chondrogenic differentiation was composed of DMEM (Sigma-Aldrich) and Nutrient Mixture F12 (Sigma-Aldrich) in proportion 2:3, 10% FBS (Sigma Aldrich), penicillin/streptomycin (Sigma-Aldrich, final concentration 100 U/mL and 100 µg/mL), L-glutamine (Sigma-Aldrich, final concentration 2 mM), β-glycerol phosphate (Sigma-Aldrich, final concentration 10 mM), and ascorbic acid (Sigma-Aldrich, final concentration 50 µg/mL). Cells were cultured without treatment overnight to adhere to the surface. For caspase inhibition, pharmacological inhibitors Z-VAD-FMK (FMK001, R&D Systems) and Q-VD-OPH (OPH001, R&D Systems) were applied to the micromass cultures at a concentration of 100 µM, according to the manufacturer’s recommendation and previous studies. 18 In the controls, DMSO (dimethyl sulfoxide) as an inhibitor vehicle was added and the control micromass cultures were run in parallel. The medium with caspase inhibitors or DMSO was changed every second day up to 7 days of cultivation. The experiments were performed in 3 biological replicates.
RNA Isolation and RNA Sequencing
The cultured cells were harvested using 350 µL RLT lysis buffer (Qiagen) with β-mercaptoethanol (Sigma-Aldrich). RNeasy Mini Kit (Qiagen) with included DNase I treatment was used for RNA isolation. Isolated RNA of triplicates of samples treated with FMK, OPH, and DMSO was sent to Lexogen GmbH, Campus Vienna Biocenter 5, Wien, for QuantSeq analysis, where the samples were processed and sequenced according to the company's standard operating process. Briefly, the library for sequencing was constructed using QuantSeq 3′mRNA-Seq Library Prep kit FWD for Illumina (Lexogen) and Lexogen i5 6 nt Unique Dual Indexing Add-on kit (Lexogen). Single read RNA-Seq (1 × 75) was conducted using an Illumina NextSeq 500 system (Illumina).
Analysis of RNA-Seq Data
Data analysis including demultiplexing, read quality control, trimming and filtering procedures, mapping and read counting, and differential expression analysis was performed by Lexogen GmbH using their standard pipeline for QuantSeq-FWD data. Briefly, quality control of reads was performed using FastQC, 20 RSeQC, 21 and MultiQC 22 packages. Samples were trimmed using cutadapt tool 23 and aligned to the reference genome using STAR aligner. 24 R package DeSeq2 25 was used for exploration and differential expression of the count data.
Enrichment Analysis
Significantly differentially expressed genes (Foldchange > 2, Padj < 0.01) were analyzed by MouseMine warehouse. 26 The pathway enrichments of Reactome 27 were visualized using Cytoscape and Cytoscape 28 plugins ClueGO 29 and CluePedia. 30 Differentially expressed genes were further analyzed by Singular Enrichment Analysis (SEA) for GO terms enrichment by using agriGO. 31
Results
Transcriptomic Analysis of Caspase-Inhibited Primary Chondroblasts
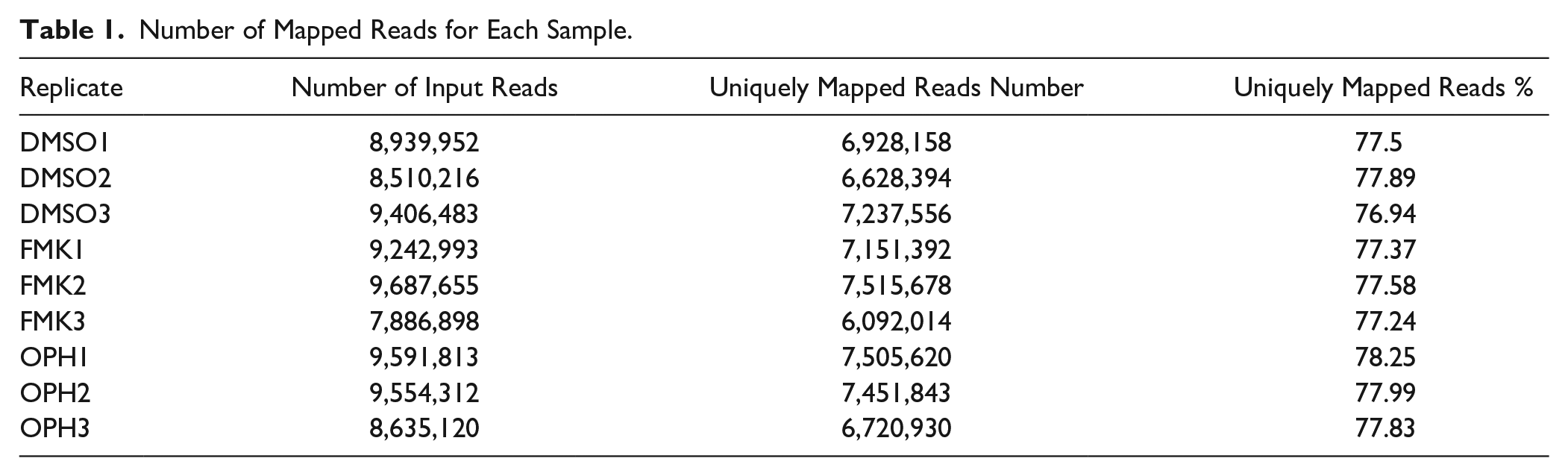
Mouse limb–derived primary chondroblasts were separately treated with 2 different caspase inhibitors (FMK and OPH) and compared with control (DMSO). A total of 81,455,442 high-quality reads were obtained for 9 samples with an average of 9,050,605 reads for each sample ( Table 1 ). The reads of each sample were mapped to the Mus musculus genome sequence when 78% of the reads were mapped to the genome sequence. A total of 13,633 genes from 52,550 annotated genes were identified; among these, 542 genes were significantly differentially expressed (Foldchange > 2, Padj < 0.01) in FMK samples and 351 in OPH samples compared with control ( Fig. 1 , Supplementary file 1). RNA sequencing analysis of transcriptome revealed a statistically significant increase in expression of 252 genes in FMK samples and 163 genes in OPH samples ( Fig. 1 , Supplementary File 1). Statistically significant downregulated were 290 genes in FMK samples and 188 genes in OPH samples. Focused on the genes that were common between the 2 inhibitors, 130 were upregulated and 153 were downregulated ( Fig. 1 ). The genes upregulated only in FMK samples were the most enriched for processes such as “response to stimulus,” “response to stress,” “response to organic substance,” whereas genes upregulated in OPH samples were the most enriched for processes such as “apoptotic process” or “response to stress.” The downregulated genes were the most enriched for processes like “cell surface receptor signaling pathway,” “cellular response to stimulus,” “cell communication,” and only in FMK samples (Supplementary File 2).
Number of Mapped Reads for Each Sample.
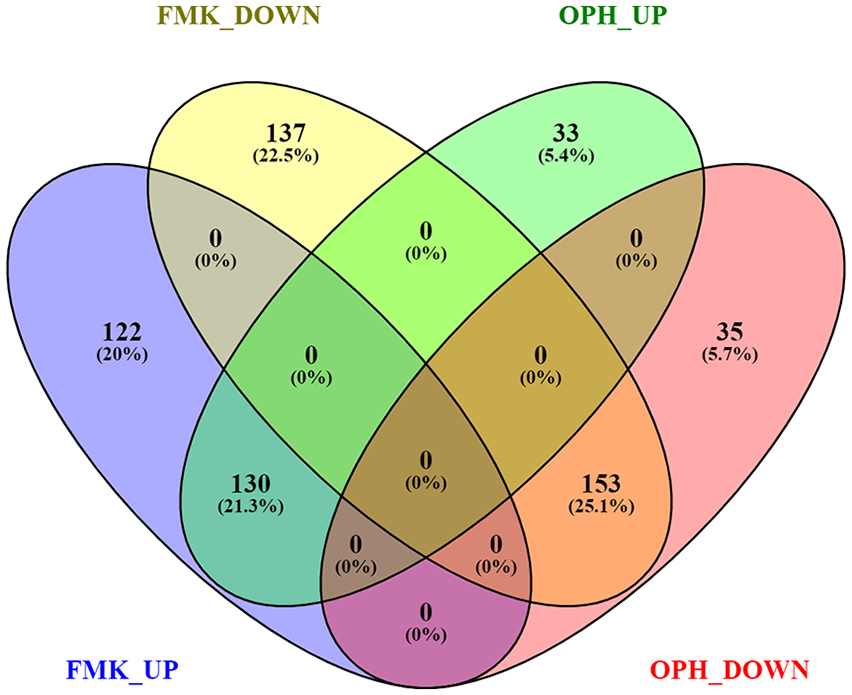
Overlap between genes differentially regulated after FMK and OPH inhibitor treatment. FMK UP = genes upregulated after FMK inhibitor; FMK DOWN = genes downregulated after FMK inhibitor; OPH UP = genes upregulated after OPH inhibitor; OPH DOWN = genes downregulated after OPH inhibitor. ≥2-fold difference and P < 0.01 were used as cutoffs. Visualization was done using Venny 2.1 (https://bioinfogp.cnb.csic.es/tools/venny/index.html).
The top 20 upregulated and downregulated genes for each inhibitor are shown in Table 2 . The majority of these genes were related to OA. After FMK as well as OPH inhibition the top (over 50 times) upregulation was applied for the pro-inflammatory cytokine Ccl5. To the others highly expressed genes connected with OA, Cxcl10, Ifi44, Isg15, Irf7, Mx1, and Cxcl9 were upregulated in the top 20 for both inhibitors. The most downregulated OA-related genes were Laptm5 in FMK inhibitor and Mrc1 in OPH inhibitor. Upregulated genes were the most enriched for GO components like “cell,” “cell part,” “intracellular,” or “nucleus.” Downregulated genes were connected with terms such as “plasma membrane,” “cell periphery,” “plasma membrane part,” or “membrane” (Supplementary File 2).
Top 20 Significantly Upregulated and Downregulated Genes in FMK and OPH Samples.
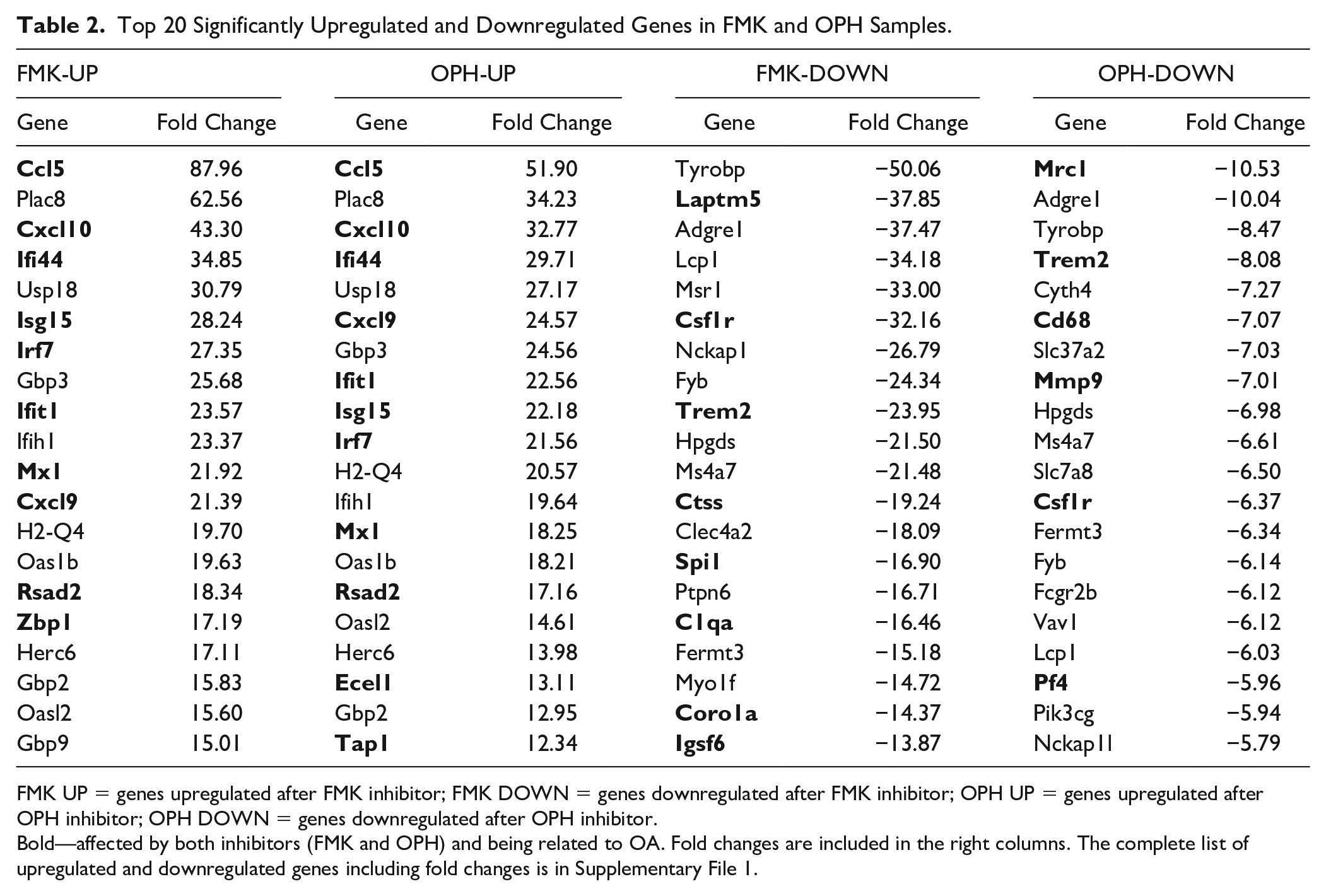
FMK UP = genes upregulated after FMK inhibitor; FMK DOWN = genes downregulated after FMK inhibitor; OPH UP = genes upregulated after OPH inhibitor; OPH DOWN = genes downregulated after OPH inhibitor.
Bold—affected by both inhibitors (FMK and OPH) and being related to OA. Fold changes are included in the right columns. The complete list of upregulated and downregulated genes including fold changes is in Supplementary File 1.
Networks displaying impacted reactome pathways for both general caspase inhibitors compared with control are shown in Figures 2 to 5 where the impact on the immune system is emphasized. In the case of upregulated genes in the FMK ( Fig. 2 ) and OPH ( Fig. 3 ) samples, the affected reactome pathways included the cell-cell communications based on cytokine signaling mediated by interleukins (e.g., IL3, IL5) as well as interferons (IFN). The alterations in interferon-signaling were linked also with ubiquitin-like proteins and the corresponding mechanisms including damage/repair responses. The impact on the innate immune system was apparent also in the spectrum of downregulated genes associated with triggering of the complement system and activating signals in natural killer cells as well as neutrophil degranulation as shown for FMK ( Fig. 4 ) and OPH ( Fig. 5 ) groups.
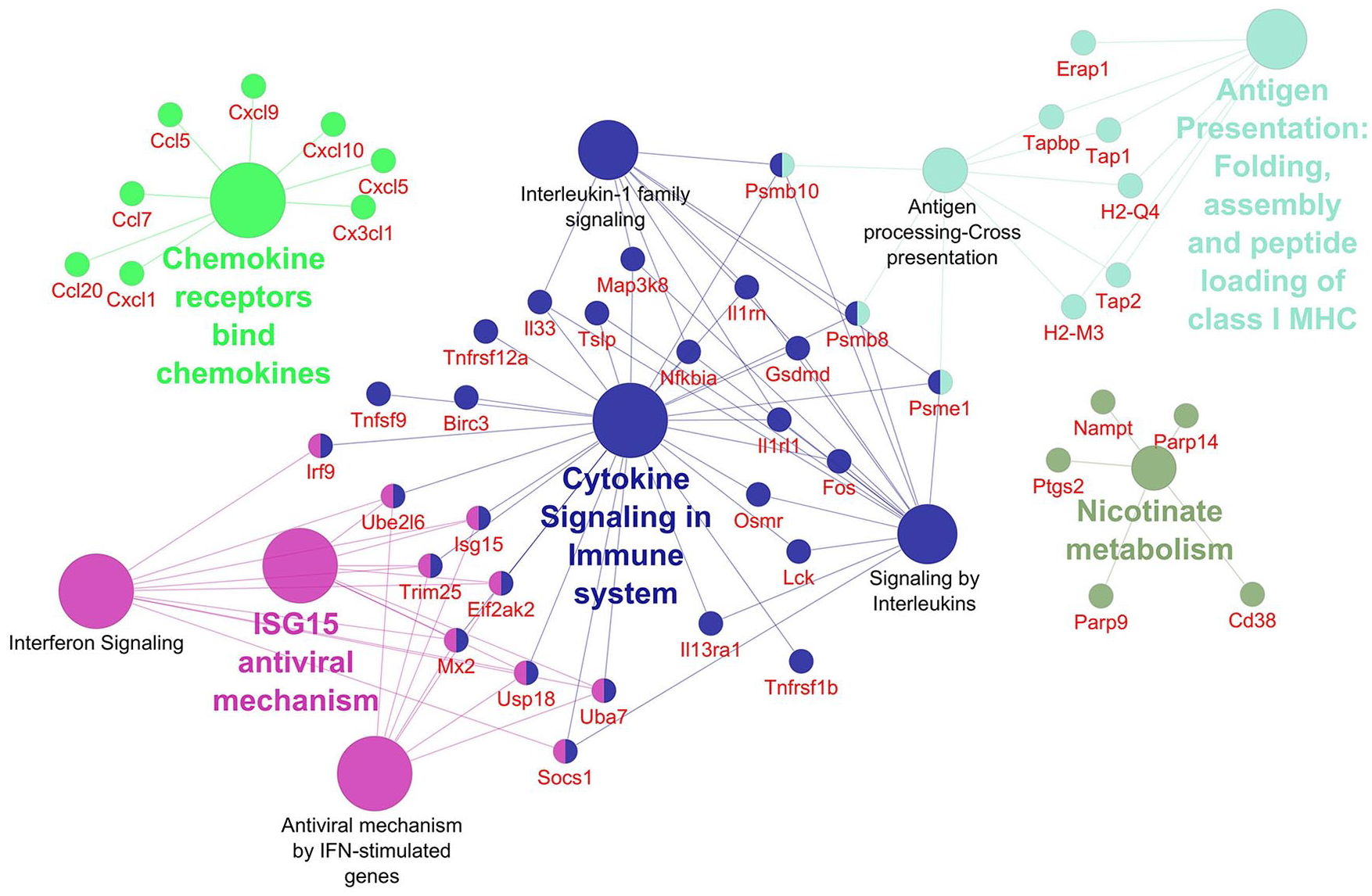
Pathways significantly enriched with upregulated transcripts after inhibition using FMK inhibitor. Significantly enriched Reactome pathways 27 (P < 0.01) and related significantly upregulated transcripts (Foldchange > 2, Padj < 0.01) after caspase inhibition using FMK inhibitor. The network was created using Cytoscape 28 and Cytoscape plugins ClueGO 29 and CluePedia. 30
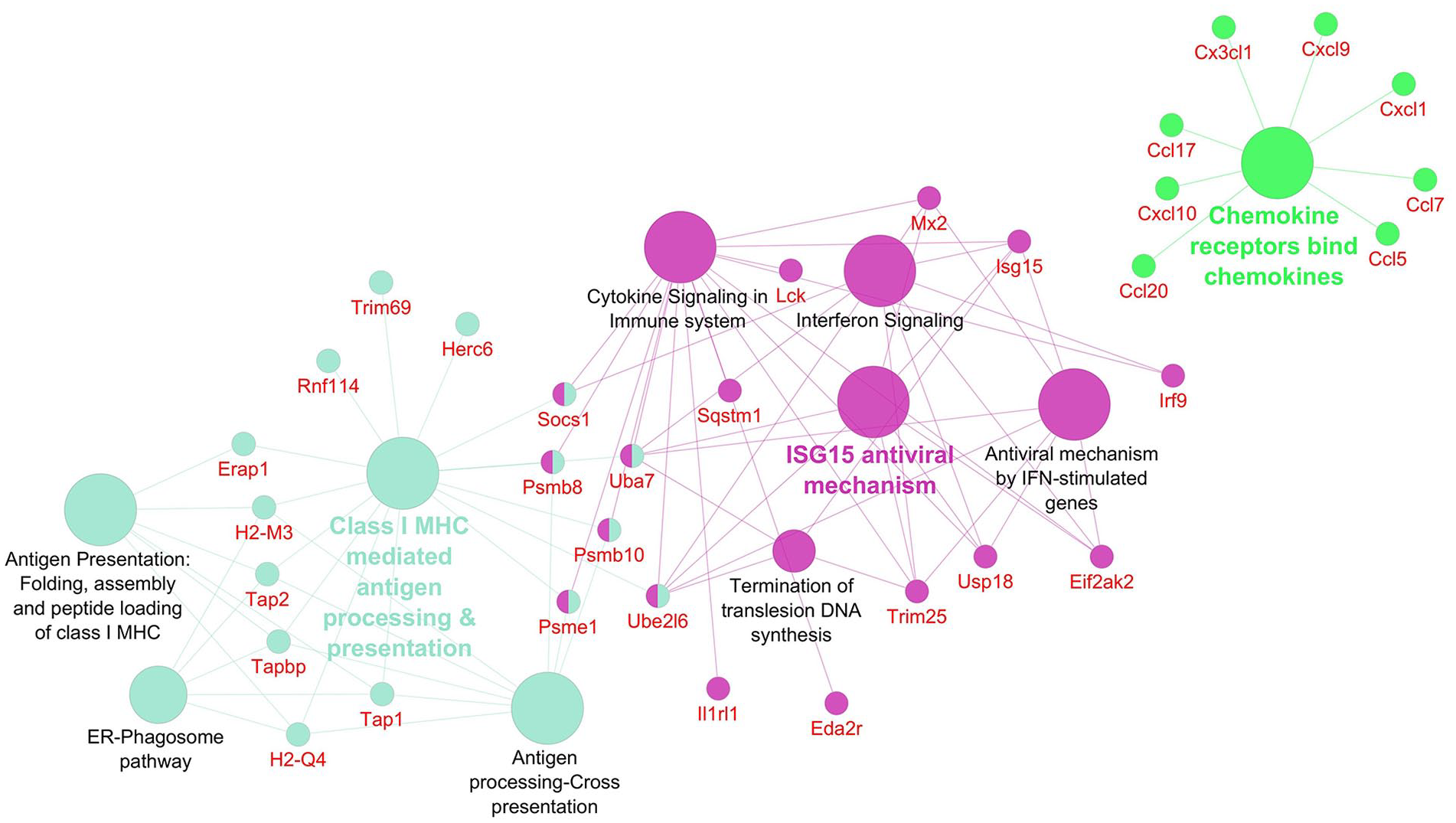
Pathways significantly enriched with upregulated transcripts after inhibition using OPH inhibitor. Significantly enriched Reactome pathways 27 (P < 0.01) and related significantly upregulated transcripts (Foldchange > 2, Padj < 0.01) after caspase inhibition using OPH inhibitor. The network was created using Cytoscape 28 and Cytoscape plugins ClueGO 29 and CluePedia. 30
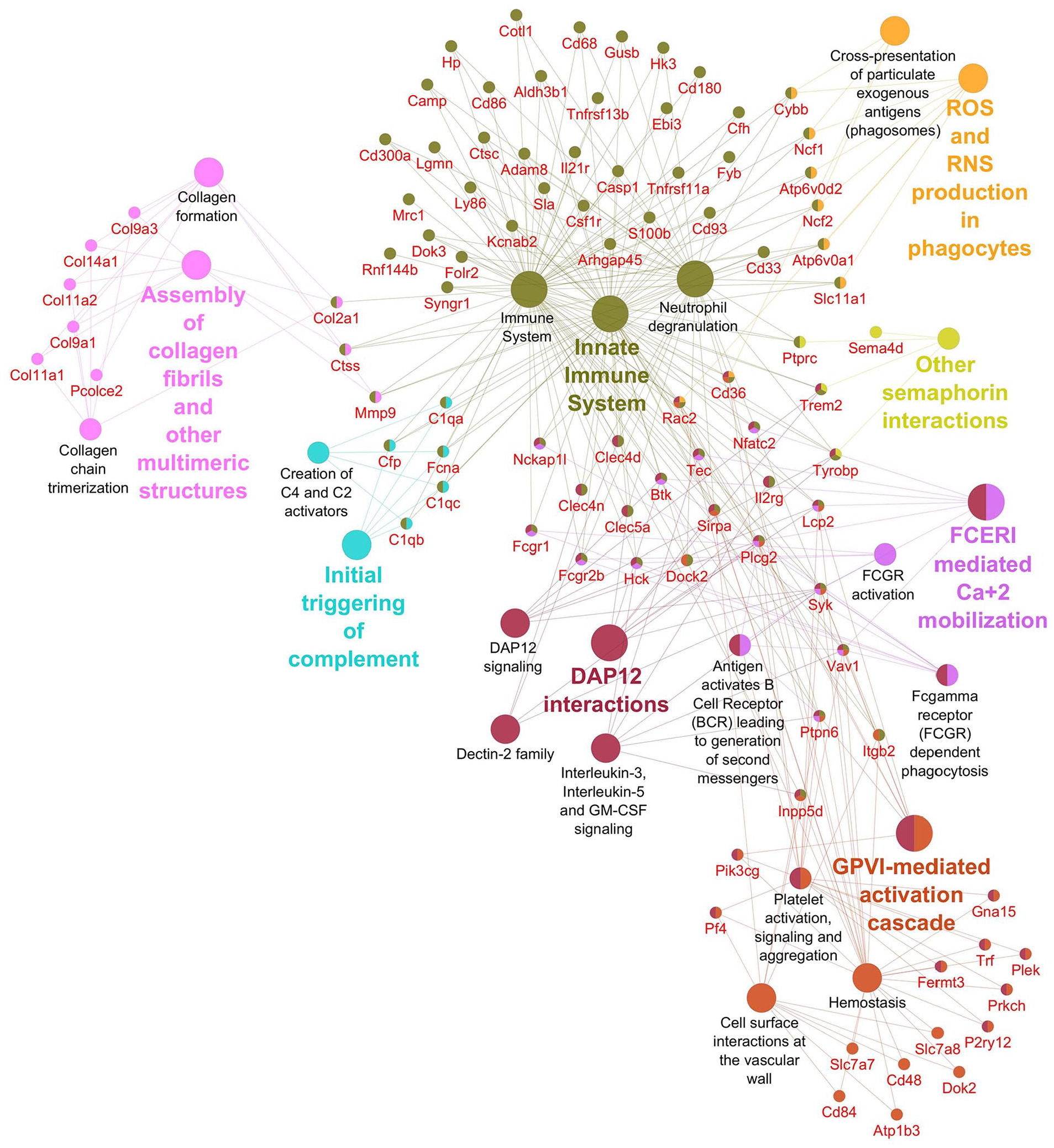
Pathways significantly enriched with downregulated transcripts after inhibition using FMK inhibitor. Significantly enriched Reactome pathways 27 (P < 0.01) and related significantly downregulated transcripts (Foldchange > 2, Padj < 0.01) after caspase inhibition using FMK inhibitor. The network was created using Cytoscape 28 and Cytoscape plugins ClueGO 29 and CluePedia. 30
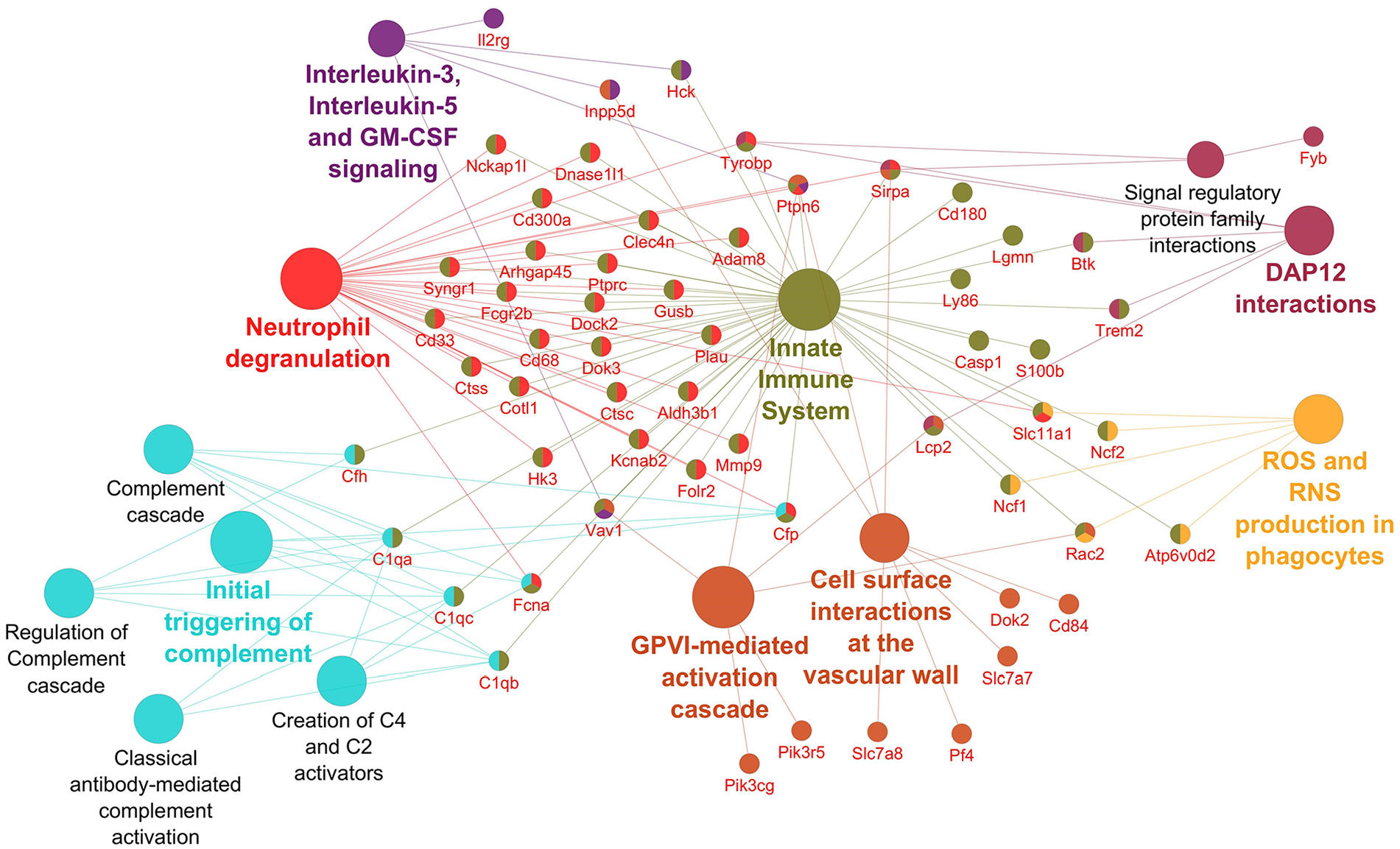
Pathways significantly enriched with downregulated transcripts after inhibition using OPH inhibitor. Significantly enriched Reactome pathways 27 (P < 0.01) and related significantly downregulated transcripts (Foldchange > 2, Padj < 0.01) after caspase inhibition using OPH inhibitor. The network was created using Cytoscape 28 and Cytoscape plugins ClueGO 29 and CluePedia. 30
Taken together, both inhibitors caused massive changes in homeostasis and the immune system factors. Moreover, OPH inhibitor affects the cell cycle and adaptive immune system, especially upregulates transcripts related to DNA damage checkpoints and antigen processing and presentation, respectively. FMK inhibitor additionally caused the downregulation of transcripts associated with extracellular matrix organization, in particular collagen formation and degradation.
Datasets generated and analyzed during the current study are available in the GEO repository under the GEO accession number GSE164835.
Discussion
In this investigation, the impact of caspase inhibition on the transcriptome profile of primary chondrogenic cultures was evaluated. Micromass system is a primary cell culture providing 3D microenvironment to follow chondrogenesis in vitro and as such is the most commonly used model.32,33
Two different general caspase inhibitors, Z-VAD-FMK (FMK) and Q-VD-OPH (OPH), were used to achieve caspase inhibition in the cultures. The main reason for using these 2 most common commercially available general caspase inhibitors comes from the fact that they do not display the same efficiency in inhibiting individual caspases. Moreover, in the case of FMK, other cysteine proteases (cathepsins) may be affected along with caspases. 7 The results from both types of inhibitions are provided in parallel among the data presented here, but only those OA-related molecules being significantly impacted by both inhibitors are discussed. Notably, almost half of them are somehow associated also with osteoarthritis.
The top upregulated gene for both inhibitors (FMK and OPH) was Ccl5. Ccls have the ability to induce the release of enzymes associated with cartilage damage and immune response.34,35 Elevated levels of Ccl5 have been identified in leukocytes and synovial tissue of patients with rheumatoid arthritis, reactive arthritis, as well as osteoarthritis.36,37 In human synovial fibroblasts, Ccl5 was demonstrated to promote IL6 production,38,39 a pro-inflammatory cytokine with a confirmed clinical diagnosis of OA. 40 The upregulation of Ccl5 after caspase inhibition is in agreement with complementary results from the RNA-Seq analysis, where other Ccls (namely, Ccl2, Ccl7, Ccl20) were also upregulated. Ccl2 was shown to mediate monocyte recruitment followed by inflammation and cartilage destruction. 39 Ccl7 has been detected in synovial tissue of OA patients. 36 Regarding Ccl20, its concentration within the synovial fluid correlates with the severity of OA. 41 Notably, intraarticular application of caspase inhibitors resulted in a significant reduction of cartilage degradation 10 supporting interplay of pro- and anti-osteoarthritic factors in vivo.
Among the top 20 genes upregulated by caspase inhibitors, there were several interferon-related molecules, such as Ifi44, Isg15, Irf7, Ifit1, and Mx1. Ifi44 (interferon-induced protein 44) gene encodes a protein able to aggregate to form microtubular structures. A study focused on the phenotype of osteoarthritis suggested that Ifi44 was downregulated in damaged cartilage. 42 Isg15 (interferon-stimulated gene 15) acts both as an extracellular cytokine and intracellularly, as a protein modifier. Recently, Isg15 as well as Ifit1 (interferon-induced protein with tetratricopeptide repeats 1) were identified as hub nodes in protein-protein interactions behind the effects of 1,25(OH)2D3 on primary cultures of chondrocytes from patients with osteoarthritis. 43 Irf7 (interferon regulatory factor 7) was associated with several disorders44,45 and was investigated also in synoviocyte innate responses. 46 Additionally, Irf7 was among differentially expressed genes in a gene expression profiling study related to osteoarthritis. 47
Mrc1 (mannose-receptor-C-type1), an anti-inflammatory marker, was the top downregulated gene in OPH and among the top 5 in FMK treated samples. MRC-mediating signaling is involved in collagen reorganization and lysosomal degradation of the cartilage. 48 MRC expressing macrophages have recently been considered as a potential target for cartilage regeneration. 49 Additionally, there is a significant association of Mrc1 with COMP (cartilage oligomeric matrix protein) being a biomarker of OA. 50 Recently, osteoarthritis-associated basic calcium phosphate (BCP) crystals have been shown to downregulate the expression of Mrc1. 51 Notably, treatment with BCP upregulated mRNA levels of Cxcl9 and Cxcl10. This association is in agreement with our observation when Mrc1 downregulation was accompanied by a significant upregulation of Cxcl10 (within top 3) and Cxcl9 in caspase inhibited samples.
The top 20 genes downregulated by caspase inhibition included also other genes being associated with cartilage homeostasis and OA disorders. Laptm5 (lysosomal-associated protein transmembrane 5) was earlier reported as one of the molecules affected in methylation analyses in patients with OA52,53 and in a microarray study related to the severity of OA. 54 Trem2 (triggering receptor expressed on myeloid cells) was investigated with respect to systemic inflammation related also to osteoarthritis. 55 Csf1r (colony-stimulating factor 1 receptor) was earlier examined in osteoarthritis patients in relation to the modulation of inflammatory reaction. 56 Csf1r was recently shown to positively correlate with Cd68 expression. 57 This is in agreement with our observation; Cd68 expression also decreased in the inhibited samples. Notably, a decrease in synovial CD68-positive macrophages represents one of the biomarkers of the effectiveness of arthritis treatment. 58
Mmp9 (matrix metalloproteinase 9), also appearing as a potential biomarker, was included in the top 10 downregulated genes after caspase inhibition. High levels of Mmp9 were detected in OA patients and might thus take part in the cartilage destruction in OA.59,60 Additionally, miR-targeted Mmp9 caused a reduction of articular cartilage degradation. 61
The analysis of interactions among impacted genes/proteins pointed to the major effects of caspase inhibition within the immune system components. The effect was associated not only with cytokine signaling but also with the regulation of IFN-γ signaling and neutrophils. The role of the immune system in the progression of osteoarthritis 62 has also been investigated in the case of other types of arthritis, such as rheumatoid arthritis. 63 IFN-γ plays role in IL-1β driven matrix metalloproteinases synthesis resulting in the downregulation of MMP-1 and MMP-3 production in vitro. 64 Besides this, IFN-γ was found to induce a dose-dependent decrease in cell proliferation and proteoglycan synthesis in cultured human chondrocytes. 65 Recently, the interest in the immunogenic properties of chondrocytes was stimulated by their possible implication in chondrocyte transplantations for therapeutic purposes. 66 The other components of the reactome impacted by both inhibitors included molecules involved in signal transduction, metabolism, and transport of small molecules. MAPK signalization, upregulated in both groups, was described as regulating the degradation of cartilage ECM. 67 Furthermore, PECAM-1 also decreased in both investigated groups. PECAM-1 inhibition was suggested for clinical treatment of rheumatoid arthritis. 68 The direct impact on cartilage differentiation can be connected also to a decrease of SNARE disinhibition participating in lysosomal degradation of ECM. v-SNARE proteins have potential roles in lysosomal exocytosis in hypertrophic chondrocytes. 69
The results from the present research provide a comprehensive view of the impact of general caspase inhibition on the transcriptional profile in primary chondrogenic cells. As a consequence of caspases inhibitions, a large number of genes were up- or downregulated. Additionally, impacts of the 2 most common caspase inhibitors (FMK and OPH) were compared and the overlapping spectrum of genes, as well as differences, were illustrated.
Although in vitro approaches were applied and a classical model of chondrogenesis was challenged, the results also indicated possible fragile points in chondrocyte homeostasis impacted by caspase inhibition. The obtained data thus offer a solid background for further extrapolations and complementary investigations toward chondrocyte differentiation, maintenance, and dysregulations. With this respect, the evaluation emphasized expression modifications of genes associated with osteoarthritis.
Research Data
sj-xlsx-1-car-10.1177_19476035211044823 – Supplemental material for General Caspase Inhibition in Primary Chondrogenic Cultures Impacts Their Transcription Profile Including Osteoarthritis-Related Factors
Supplemental material, sj-xlsx-1-car-10.1177_19476035211044823 for General Caspase Inhibition in Primary Chondrogenic Cultures Impacts Their Transcription Profile Including Osteoarthritis-Related Factors by Barbora Vesela, Martina Zapletalova, Eva Svandova, Alice Ramesova, Jaroslav Doubek, Hervé Lesot and Eva Matalova in CARTILAGE
Research Data
sj-xlsx-2-car-10.1177_19476035211044823 – Supplemental material for General Caspase Inhibition in Primary Chondrogenic Cultures Impacts Their Transcription Profile Including Osteoarthritis-Related Factors
Supplemental material, sj-xlsx-2-car-10.1177_19476035211044823 for General Caspase Inhibition in Primary Chondrogenic Cultures Impacts Their Transcription Profile Including Osteoarthritis-Related Factors by Barbora Vesela, Martina Zapletalova, Eva Svandova, Alice Ramesova, Jaroslav Doubek, Hervé Lesot and Eva Matalova in CARTILAGE
Footnotes
Acknowledgments and Funding
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study because the mouse samples were obtained post mortem in agreement with the recent legislative in the Czech Republic, law 359/2012 Sb., where according paragraph 3, part t) there is no specific requirement for approvals in case when organ/tissue samples are collected post mortem.
Informed Consent
Informed consent was not sought for the present study because only animal samples were used.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
